Abstract
We aimed to identify clinicopathological differences and factors affecting survival outcomes of stage T2a and T2b gallbladder cancer (GBC) and validate the oncological benefits of regional lymphadenectomy and hepatic resection in these patients. This single-center study enrolled patients who were diagnosed with pathologically confirmed T2 GBC and underwent curative resection between January 1995 and December 2017. Eighty-two patients with T2a and 50 with T2b GBCs were identified, and clinical information was retrospectively collected from medical records and analyzed. Five-year overall survival rates were 96.8% and 80.7% in T2a and T2b groups, respectively (P = .007). Three- and 5-year survival rates among all patients with T2 GBC without and with lymph node metastasis were 97.2% and 94.4% and 81.3% and 81.3%, respectively (P = .029). There was no difference in survival rates between the 2 groups according to whether hepatic resection was performed (P = .320). However, in the T2b group, those who underwent hepatic resection demonstrated a better survival rate than those who did not (P = .029). The T2b group had more multiple recurrence patterns than the T2a group, and the lymph nodes were the most common site in both groups. Multivariate analysis revealed that lymph node metastasis, vascular invasion, and tumor location were significant independent prognostic factors. Hepatic resection was not always necessary in patients with peritoneal-side GBC. Considering clinicopathological features and recurrence patterns, a systematic treatment plan, including radical resection and adjuvant treatment, should be established for hepatic-side GBC.
Introduction
Gallbladder cancer (GBC) is known to have a poor prognosis, and curative radical resection is the gold standard treatment. 1,2 Tumor invasion depth and lymph node metastasis are factors affecting the prognosis of GBC 3 ; therefore, proper hepatic resection and lymphadenectomy are considered to be essential surgical procedures. Cancers of the gallbladder are staged according to their depth of invasion into the gallbladder wall and extent of spread to surrounding structures and lymph nodes. Several studies have reported that T1 GBC has a good prognosis with simple cholecystectomy alone. 4,5 Gallbladder cancers with T2 and above are still referred for radical cholecystectomy including liver parenchyma resection and regional lymphadenectomy. Recently, stage T2 GBC has been subdivided into T2a (peritoneal side) and T2b (hepatic side), according to tumor location in the 8th Edition of the American Joint Committee on Cancer (AJCC). This change in classification was based on the results of an international multicenter study that demonstrated worse survival outcomes in patients with hepatic-side tumor. Since the release of the new classification system, although there have been reports describing differences in the oncological prognosis of T2a and T2b GBC, insufficient consensus regarding the appropriate treatment strategy remains. 6
The purpose of this study was to identify clinicopathological differences and factors affecting the survival outcome of T2a and T2b GBC. In addition, we aimed to validate the oncological benefit of regional lymphadenectomy and hepatic resection in patients with T2a and T2b GBC, respectively.
Materials and Methods
Patients
This single-institution study enrolled patients who were diagnosed with pathologically confirmed T2 GBC and underwent curative resection between January 1995 and December 2017. Inclusion criteria were resection with curative intent, and pathologically confirmed adenocarcinoma and T2 staging. Classification was conducted according to AJCC 8th edition. The location of tumor was evaluated based on histological examination by the 2 experienced pathologists at our center. Tumors invading at least part of hepatic aspect of the gallbladder wall were classified as T2b tumors.
Eighty-two patients with T2a and 50 with T2b GBC were identified, and clinical information was retrospectively collected from medical records and analyzed, including age, sex, body mass index, chief complaint(s), comorbidity, operation type, operation time, tumor size, histological grade, resection margin, lymph node metastasis, vascular invasion, perineural invasion, and adjuvant chemotherapy. Our study was approved by the Korea University Guro Hospital institutional ethical board (approval no.2019GR0260). In consideration of age and comorbidity, multidisciplinary treatment was used to decide whether to perform chemotherapy.
Gemcitabine alone or gemcitabine plus cisplatin was used as adjuvant chemotherapy regimen. In principle, extended radical cholecystectomy is performed. This procedure consists of cholecystectomy, partial hepatectomy of the gallbladder fossa, and regional lymphadenectomy. Regional lymph nodes including the cystic duct, pericholedochal, retroportal, and common and proper hepatic artery lymph node groups were dissected.
Extrahepatic bile duct resection was not performed routinely and was performed selectively when the cystic duct margin was positive on frozen biopsy or the tumor was bulky and inflammation was severe. Twelve patients revealed T2 GBC confirmed by final biopsy report after simple cholecystectomy. However, patients and their caregivers do not want to perform additional surgery including the hepatectomy and lymphadenectomy due to concern about general condition deterioration and fear of repeated wide surgery. Two of 12 patients who underwent simple cholecystectomy showed intraoperative lymph node enlargement, so lymph node dissection without hepatectomy was performed. Recurrence patterns and survival outcomes were compared between those with T2a and T2b GBC. The site of recurrence was determined using radiographic imaging during the follow-up periods.
Statistical Analysis
Data that were normally distributed are expressed as mean (standard deviation), while data that were not normally distributed are expressed as median (range). Continuous variables were compared using the Student t test if normally distributed; otherwise, the Mann-Whitney U test was used. Categorical variables were compared using the χ2 test. Cancer-specific overall survival (OS) was measured from the date of definitive surgery to that of the last follow-up or death from cancer. Overall survival was estimated using the Kaplan-Meier method and compared using log-rank tests; P < .05 was considered to be statistically significant. Multivariate models were manually built using a forward strategy. Statistical analysis was performed using SPSS version 22.0 (IBM Corporation, Armonk, New York) for Windows (Microsoft Corporation, Redmond, Washington).
Results
Clinicopathological Characteristics of Patients With T2 GBC
During the study period, 132 consecutive patients with T2 GBC underwent curative resection. The male to female ratio was 28:54 in the T2a group and 20:30 in the T2b group. Asymptom is most common cause of visiting the hospital in T2a group, and abdominal pain was the most common cause in the T2b group. Hepatic resection was performed in 74 (90.2%) among the 82 T2a group patients and 46 (92.0%) among the 50 T2b group patients. Regional lymphadenectomy was performed in 74 (90.2%) patients among the T2a group and 48 (98.0%) among the T2b group. Lymph node metastasis was found in 14 (17.1%) patients in the T2a group and 24 (48.0%) in the T2b group. There was a statistically significant difference between the 2 groups, with more detailed information provided in Table 1.
Clinicopathologic Characteristics of Patients With T2 Gallbladder Carcinoma.

Abbreviations: BMI, body mass index; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen.
Survival Outcomes According to Tumor Location, Lymph Node Dissection, and Hepatectomy
After a median follow-up period of 58.4 months (interquartile range, 38.1-132.2 months), for all 132 patients with T2 GBC, the 3-year and 5-year cancer-specific survival rates were 92.3% and 90.3%, retrospectively. The 3-year and 5-year OS rates of the T2a group were 96.8% and 96.8%, 80.7% and 80.7% in the T2b group, respectively (P = .007; Figure 1). The 3-year and 5-year survival rates were 97.2% and 94.4% among all patients with T2 GBC without lymph node metastasis, and 81.3% and 81.3% in those with lymph node metastasis, respectively; this difference in survival rate between the groups was statistically significant (P = .029). When each group was examined separately, there was no difference in survival rate in the T2a group; however, there was a significant difference in the T2b group (P = .048).
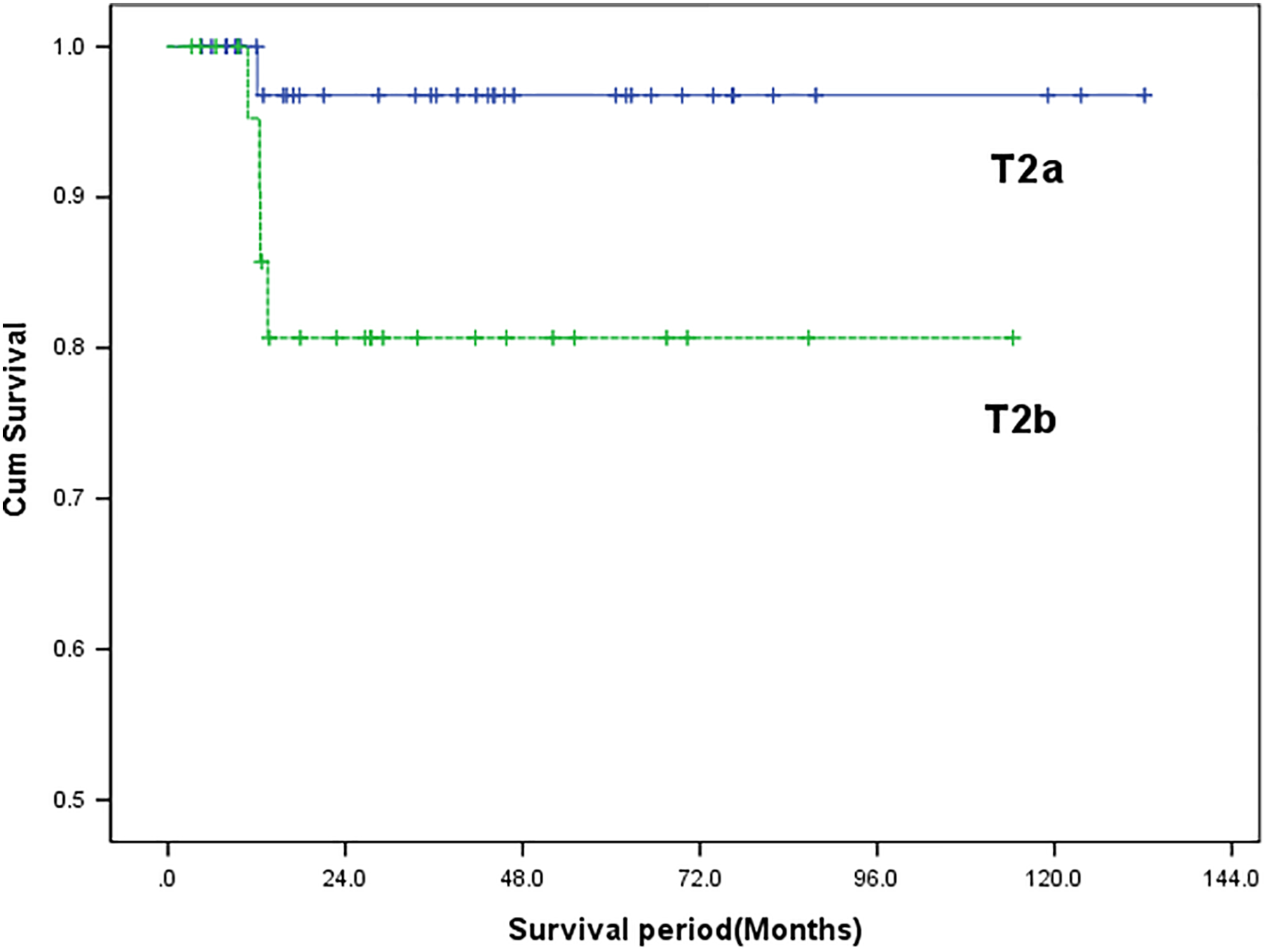
Overall survival rate in T2 GBC according to tumor location. Patients with GBC with T2a do achieve better overall survival than T2b (P = .007). GBC indicates gallbladder cancer.
There was no difference in survival rates according to whether regional lymphadenectomy was performed (P = .347) among all patients with T2 GBC, although when the T2a and T2b groups were examined separately, it was not possible to confirm differences in survival rate. When survival rates according to whether hepatic resection was performed were examined, there was no difference between the 2 groups (P = .320). However, in the T2b group, those who underwent hepatic resection had a better survival rate than those who did not (P = .029; Figure 2).

Overall survival rate in T2 GBC according to regional lymphadenectomy in T2a(A) and T2b(B) and according to hepatic resection in T2a(C) and T2b(D). GBC indicates gallbladder cancer.
Prognostic Factors Affecting Survival and Recurrence Pattern in Patients With T2 GBC
In univariate analysis, the following variables affecting the survival were statistically significant: carbohydrate antigen 19-9 (≥36.0 U/mL), lymph node metastasis, tumor location, vascular invasion, and perineural invasion. Additionally, multivariate analysis revealed that lymph node metastasis (hazard ratio [HR]: 2.56 [95% confidence interval, CI: 2.12-4.48], P = .011), vascular invasion (HR: 5.12 [95% CI: 4.48-222.78], P = .001), and tumor location (HR: 6.96 [95% CI: 1.21-39.89], P = .029) were significant independent prognostic factors. Hepatic resection, number of lymph nodes harvested, and lymph node dissection were not prognostic factors for survival in patients with T2 GBC (Table 2).
Risk Factor Analysis Affecting the Survival for Patients With T2 GBC.

Abbreviations: BMI, body mass index; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CI, confidence interval; HR, hazard ratio; GBC, gallbladder cancer.
Recurrence occurred in 6 (7.5%) of 82 patients in the T2a group and in 16 (32.0%) of 50 in the T2b group during the follow-up period. The T2b group had more multiple recurrence patterns than the T2a group, and the lymph nodes were the most common site in both groups. The difference in recurrence rate between the 2 groups was statistically significant (P = .002; Table 3).
Recurrence Site of Patients With T2 GBC.

Abbreviation: GBC, gallbladder cancer.
Discussion
It is difficult to determine a curative treatment strategy for GBC because of the characteristics of the advanced stage of disease at the time of the discovery. Therefore, early detection and appropriate surgical treatment are very important. After the introduction of radical cholecystectomy based on hepatic parenchymal resection with regional lymph node and gallbladder bed, this method became a first-line technique for GBC treatment. 7,8
Several studies have reported that even if lymph node dissection or hepatic resection is omitted in lower stage GBC, there is no difference in prognosis compared with that in patients in whom radical cholecystectomy is performed. 4,9 In particular, the routine application of radical cholecystectomy in T1 or T2 GBC has not yet been fully discussed. Recently, the AJCC staging system introduced a new model that divides T2 staging according to tumor location. The authors of the article that proposed the principle for this system identified that the difference in survival rate between the 2 groups was due to the degree of differences in vascular invasion, neural invasion, and nodal involvement depending on tumor location. 10 -12
In the present study, the 5-year survival rates of T2a and T2b GBC were 96.8% and 80.7%, respectively, and the validity of stratification between the 2 groups was confirmed. Moreover, similar to other studies, this study confirmed that there were differences in histological grading, nodal involvement, and perineural invasion between the 2 groups.
As clearly confirmed in other studies, T1 tumors are associated with good prognosis, with very low rates of vascular invasion and nodal and liver metastasis. 13,14 However, with T2 tumors, vascular invasion is increased because the subserosal layer contains many blood vessels. 2,15
Classical studies using staining methods have reported that the hepatic side of the gallbladder is drained by short cystic veins directly connecting with intrahepatic portal veins, while the peritoneal side has only 1 or 2 cystic veins terminating in the adjacent liver parenchyma. This may result in more vascular invasion in T2b than in T2a, which may have affected prognosis.
According to the definition of T2, the tumor itself is confined to the gallbladder wall, and its primary mode of spread outside the confines of the gallbladder for T2 GBC is lymphatic. In fact, as described in several reports, nodal metastasis is observed in 30% to 40% of T2 GBCs and, as such, lymphadenectomy is considered to be a critical treatment option for this disease entity. 16,17
Toge et al reported that lymphatic vessels are more dense on the hepatic side of the gallbladder than on the peritoneal side using immunohistochemistry on paraffin block sections. 8 Based on their results, the authors proposed a hypothesis that T2b has more nodal metastases than T2a. Our study also confirmed this finding, in that the incidence of regional lymph node metastasis in patients with T2a and T2b tumors was 17.1% and 52.0%, respectively. However, we could not confirm a correlation between lymphadenectomy and cancer prognosis. Considering the characteristics of T2 cancer, in which lymph node metastasis is relatively frequent, lymphadenectomy is a necessary procedure in terms of therapeutic and diagnostic value. Recent studies have reported that at least ≥6 lymph node dissections are necessary to properly evaluate the exact nodal stage. 18 -20 There is some research data on whether the distribution of positive lymph nodes affects tumor location and even oncologic outcome, but the authors understand that the more discussion and accumulation of result are still needed. 8,18 In the new AJCC staging system, lymph node metastasis is still assessed by the number only.
Hepatic metastasis of GBC has the following 3 routes: direct invasion of the liver from the gallbladder bed; micrometastasis of GBC near the gallbladder bed; and potential invasion by Glisson’s sheath connected with a hepatoduodenal ligament. 21 An important anatomical consideration is that the serosa along the liver edge is absent, and the perimuscular connective tissue at this interface is densely adherent to the liver. Because GBC directly implanted in the liver is, by definition, T3, the metastasis route through micrometastasis and Glisson’s sheath may play an important role in T2 GBC. Furthermore, the pathway through the above 2 routes occurs more easily when the tumor is located on the hepatic side rather than the peritoneal side because of anatomical structural features.
Park et al concluded that the hepatic resection of T2b GBC did not affect long-term survival. 22 In contrast, Lee et al showed that lymph node dissection without hepatectomy is an adequate treatment for T2a but not for T2b GBC. 23 In our study, hepatic resection and adjuvant chemotherapy were not found to be a factor affecting survival among all patients with T2 GBC. However, a significant difference in survival according to whether hepatic resection was performed was identified in T2b patients. Moreover, we observed that T2b exhibited a higher recurrence rate and more multiple recurrence patterns than T2a, suggesting that additional systemic treatment, may needed to prevent systemic recurrence, particularly in patients with T2b GBC. Further research is needed to determine which systemic treatment modality will affect.
The present study had several limitations, the first of which were its single-center retrospective design and the small number of patients over a long period of time. We could not conduct multivariate analysis in each subgroup because of the small number of patients. Further large-scale and randomized studies may be necessary to validate our conclusion.
Conclusion
Tumor location in patients with T2 GBC is an important factor affecting disease-specific survival. Hepatic resection is not always necessary in patients with peritoneal-side GBC. Considering clinicopathological features and recurrence patterns, a systematic treatment plan, including radical resection and adjuvant treatment, should be established for hepatic-side GBC.
Footnotes
Authors’ Note
W-.J.K. and W-.B.K. designed the study. W-.J.K., T-.W.L., P-.J.P., and S-.B.C. performed data acquisition and analysis. W-.J.K., W-.B.K., and S-.B.C. performed statistical analysis and data interpretation. W-.J.K. prepared the manuscript. All authors read and approved the final manuscript. Our study was approved by the Korea University Guro Hospital institutional ethical board (approval no.2019GR0260). All patients provided written informed consent prior to enrollment in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
