Abstract
Recent outbreaks of viral respiratory or resistant bacterial pathogens underscore the critical importance of infection prevention and control (IPC) measures in safeguarding healthcare professionals (HCPs), patients, and the environment against transmission of infectious agents. To identify the root causes of suboptimal IPC practices, such as low hand-hygiene (HH) compliance and improper personal protective equipment (PPE) use, we created the Applied Care Event Sequence Observation (ACESO) system. We used human factors engineering concepts to evaluate various observational tools developed for prior studies and create ACESO’s highly structured data-collection form and related electronic data-entry and compilation templates. The ACESO system streamlined the collection, entry, and initial analysis of granular data about patient-care sequences. Our initial findings revealed concerning trends, particularly regarding suboptimal HH compliance observed during HCP’s patient-care processes. Additionally, our observations revealed instances of HCPs touching various surfaces with potentially contaminated gloves, heightening the risk of cross-contamination within healthcare units.
Background
Recent outbreaks of viral respiratory or resistant bacterial pathogens highlight the importance of infection prevention and control (IPC) measures to prevent transmission of infectious agents to healthcare professionals (HCPs), patients, and the environment (Erasmus et al., 2010; Gidey et al., 2023). Poor compliance with safety standards is an expensive, often deadly problem affecting a myriad of industries worldwide. In healthcare, patient safety is further affected by an invisible enemy since infectious microorganisms cannot be seen with the naked eye (Pereira Da Silva, 2018; Sax & Clack, 2015). Moreover, whereas in other industries (e.g., manufacturing) PPE is the last line of engineering controls, in healthcare PPE and HH are often the first line of engineering controls against viral or bacterial pathogens (Salehi et al., 2019). Approximately 1 in 31 patients in U.S. hospitals develop at least one infection related to their care, resulting in an estimated annual cost ranging from $28.4 billion to $45 billion (Gidey et al., 2023). Although infection prevention programs have made progress, further measures are required to prevent healthcare-associated infections in all healthcare settings (Erasmus et al., 2010; Gidey et al., 2023; Ward et al., 2014).
Investigators have identified deficiencies, such as suboptimal hand-hygiene (HH) practices and inadequate personal protective equipment (PPE) selection and use, in HCPs’ integration of IPC guidelines into their patient care processes (Burdsall, 2017; Livorsi et al., 2018) . However, these studies have not identified the underlying causes affecting HCPs’ performance during patient care. Most studies have focused solely on HCPs behaviors to explain subpar compliance without factoring in system-related deficiencies (e.g., inadequate protocols). We applied human factors engineering (HFE) concepts to create a tool that would enable us to collect detailed information about HCPs’ processes of care so that we can better understand how HCPs integrate IPC practices into their care processes.
To effectively understand and evaluate HCPs’ performance during care activities, investigators must regard HCPs as target users and tailor prospective evaluation methods accordingly (Norman, 2013). Patient-care observations aligned with HFE methods are essential for identifying factors influencing HCPs during patient-care activities. Direct observations can provide insights that cannot be obtained through other research methods (Black, 1996). Data derived from observations can provide rich and detailed information about phenomena as they occur in the actual setting. Yet, observational studies often require prolonged data collection periods and generate data that is burdensome to quantify and analyze (Black, 1996). Moreover, healthcare settings present an extra layer of complexity for observers since patient-care activities vary substantially and are fast-paced.
Given the dearth of detailed information about patient care practices and the challenges in gathering this data, we developed the ACESO (Applied Care Event Sequence Observations) system. Our goal was to create a novel method for collecting granular observational data, allowing for a better understanding and evaluation of HCPs’ IPC practices during patient care. With ACESO, observers can collect clear and well-structured data, including task sequences, PPE selection and use, HH episodes and interactions with fomites. In addition to a data-collection template, the ACESO system includes electronic data entry and compilation templates, which can substantially streamline data analysis.
Methods
This work is part of a larger multidisciplinary study in which various investigators are assessing how HCPs integrate IPC practices into their patient-care processes. All findings reported here are based on HFE data collection and analysis methods. Findings related to other data collected will be reported in future articles. The study was reviewed and approved by the University of Iowa Institutional Review Board.
Study Design and Development Processes
Given the numerous variables that influence a subject in a complex system like healthcare, we utilized an HFE systems-theory based model to enhance our understanding of what the system provides to users (i.e., HCPs) as they conduct their tasks (Pereira Da Silva, 2018; Von Bertalanffy, 1968). Our goal was to examine the system’s attributes that could potentially hinder or enhance user performance (Rasmussen, 1983). To achieve this, we employed the Sociotechnical Systems Model for Safety Compliance, which consists of four driving factors: individual factors (e.g., HCP demographics), organizational factors (e.g., guidelines, protocols, education, and training), external stakeholders (e.g., regulatory agencies’ recommendations and regulations), and continuous feedback (e.g., communication methods and knowledge-sharing responsibilities) (Pereira Da Silva, 2018). These four key driving factors are likely to affect a given outcome (e.g., human performance) within the system’s framework (Pereira Da Silva, 2018). Using this model, we were able to identify and consider important elements that may influence HCPs’ performance while developing ACESO. For example, we used the World Health Organization’s My Five Moments for Hand Hygiene guidelines (i.e., external stakeholder), which healthcare organizations worldwide use to identify HH opportunities within patient care sequences and to assess HH compliance. This model structure enhanced our conceptual and development processes, enabling us to create a cohesive and therefore effective data collection and analysis system.
Additionally, we applied user-centered design and usability-testing techniques to assess various observational tools developed for different studies. Then, we undertook an iterative-design process to develop and refine ACESO, which included rapid-cycle piloting in intensive care units (ICUs). We also tested ACESO in a wider variety of scenarios by using it to document patient care sequences occurring during medical reality-TV shows (e.g., Lenox Hill, Extremis, and Diagnosis).
ACESO is composed of a well-defined data collection form for use during patient care observations, along with electronic data entry and compilation templates for analysis. The form allows trained observers to simultaneously record both gloved (marked with a circle) and ungloved touch points, including body sites and surfaces, as well as care-sequence steps such as HH episodes and PPE use. We define HH episodes as every instance an HCP performs HH within a care sequence event. A care sequence event begins when an HCP enters and ends when they exit the patient's room. HH performed immediately before the HCP enters or exits the patient room is also included. We organized patient-care activities under eight categories:
(A). Environment and Furnishings. This category refers to items belonging to the physical setting including furniture and equipment attached to the room. Example: curtains, bed, bedding, chairs, bedside tables, door handles and nobs, handrails, guardrails, and sink.
(B). Clean Equipment and Supplies. This category refers to clean materials, tools, and devices that are used for medical-related care including examination instruments. Example: thermometers, blood pressure cuffs, stethoscopes, and new dressings, bandages and syringes.
(C). Clean Patient Sites. This category refers to clean patient body areas including intact skin, clothing, and accessories. Example: bare skin, gowns, and underwear.
(D). Ingestible Items and Related Supplies. In addition to food and beverages and related supplies (i.e., dinnerware and related accessories), this category includes oral medications. Example: medicines, beverages, food, cups, and adult clothing protectors.
(E). Patient’s Personal Belongings. This category refers to items that belong to the patient and were not provided by the healthcare facility. Example: electronic devices, clothing and comfort items, and mobility aids.
(F). HCP Body Sites. This category refers to HCP body areas including intact skin, clothing, and accessories. Example: bare skin, scrubs, jewelry and watches, badges, and personal electronic devices.
(G). Contaminated Sites. This category refers to surfaces potentially contaminated with blood, secretions or excretions, as well as items that may come into contact with mucus membranes. Example: perineum, used adult incontinence briefs, used dressings and bandages, dental prostheses, and contact lenses.
(H). Surgical or percutaneous sites. This category refers to open areas on skin, incision sites, vascular and enteral access, catheter insertion sites, and needle sites. Example: wounds, exposed burns, central venous and intravenous access, implantable ports, and injection areas.
Additionally, we organized the layout of the data collection form and the data entry and compilation templates based on usability design principles, such as usability heuristics and natural mapping principles (Nielsen, 1994; Norman, 2013).
Participants and Data Collection
After developing and testing ACESO, we collected data at a large teaching hospital in the Midwestern United States from January 2023 to March 2024. We shadowed HCPs in the medical ICU (MICU), surgical and neurological ICU (SNICU), inpatient dialysis unit, and burn unit and documented their care processes using ACESO. Observers entered data into the ACESO electronic templates after each observation session.
Results
Our sample comprised 281 randomly selected care events, involving 278 registered nurses (RNs), two nurse practitioners (NPs), and one medical doctor (MD). Of the HCPs observed, 230 identified as female, while 51 identified as male. Throughout the 281 care events, HCPs performed a total of 3,362 steps, for an average 11.6 steps and a median of eight steps per care event. The number of steps per care sequence varied by unit with the burn unit having the highest median (13) and the SNICU having the lowest (6) (P < .001; Figure 1).
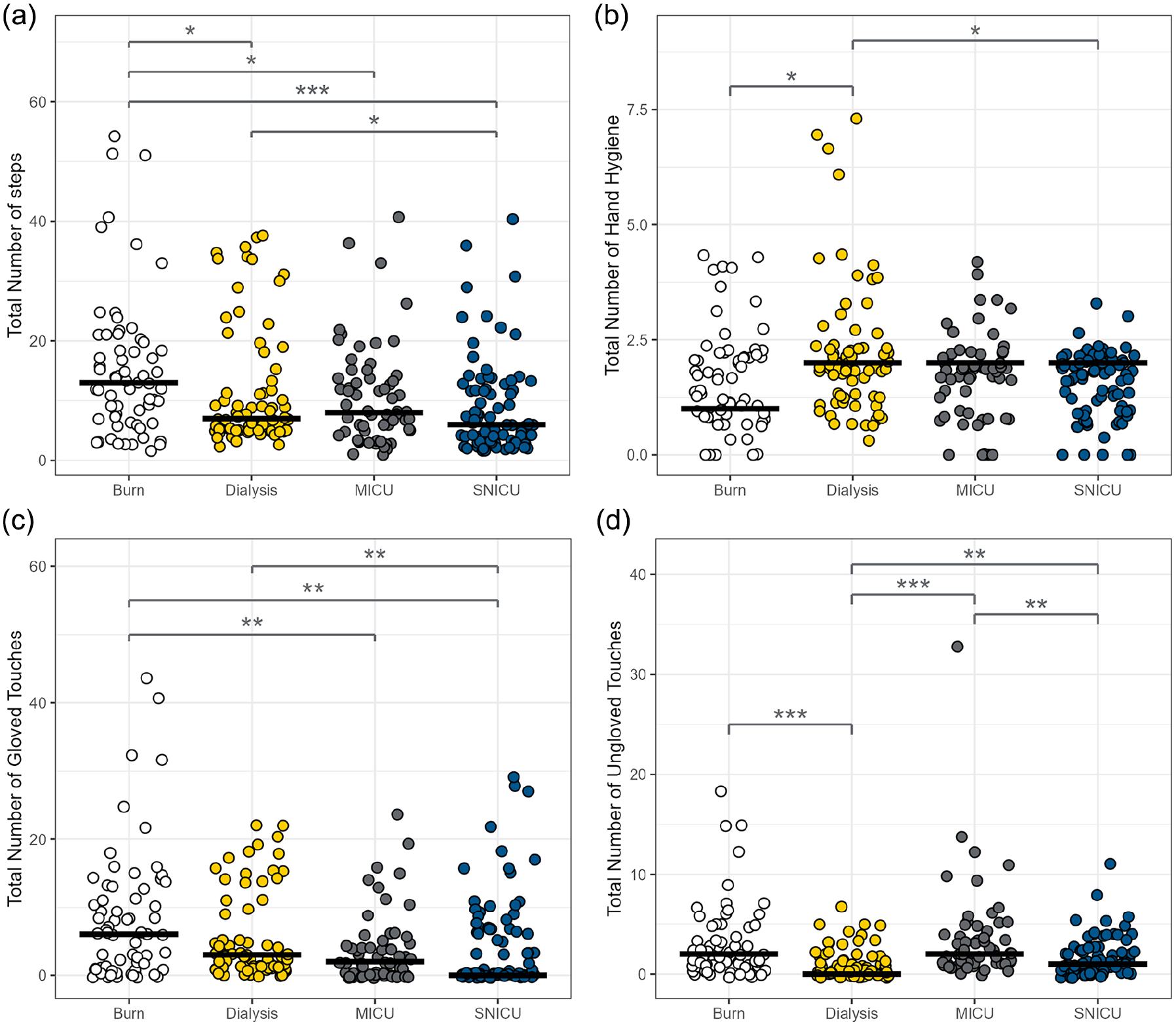
Jitter plots for the total number of steps (a), hand hygiene episodes (b), gloved touches (c), and ungloved touches (d) during care events per unit.
HCPs wore gloves for 1,531 (70.7%) of the 2,166 touches; gloved touches were significantly more frequent than ungloved touches (29.3%; P < .001). Gloved-touch categories B (clean patient sites; n = 615) and A (clean equipment and supplies; n = 386) followed by ungloved-touch category B (n = 256) were the most frequent touch types (Figure 2).
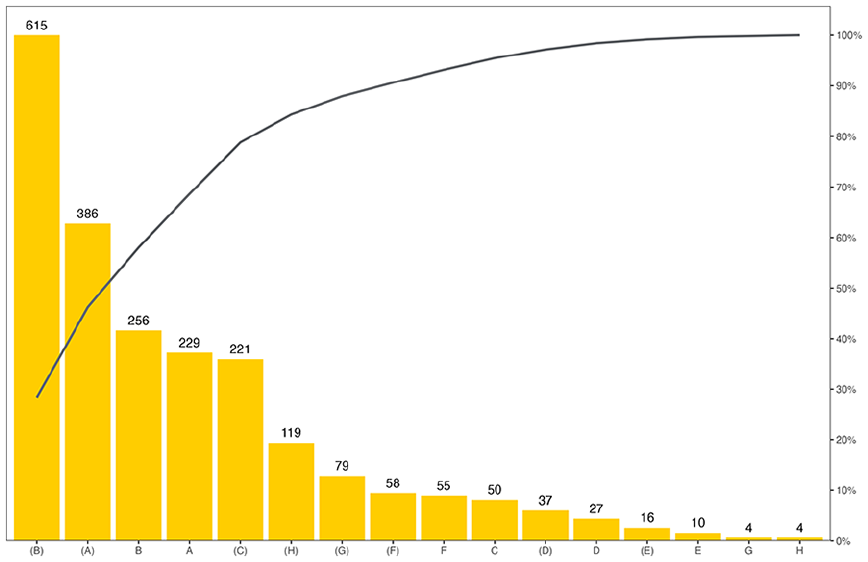
Pareto chart for the total frequencies of gloved and ungloved touches during care events per category.
Category B gloved touches were the most frequent touch points in the burn unit (n = 208), dialysis unit (n = 186), and SNICU (n = 142) whereas category A ungloved touches were the most common touch points in the MICU (n = 90; Figure 3). Categories G (contaminated sites) and H (surgical or percutaneous sites)—deemed critical risk (i.e., HCPs must do HH before and after these touches)—both gloved and ungloved, were most frequent in the burn unit (n = 60), followed by the dialysis unit (n = 54), SNICU (n = 52), and MICU (n = 40; Figure 3). On average, HCP completed 1.8 HH episodes per care event. The burn unit had a lower median for HH (1) per care sequence compared with the dialysis unit (2), MICU (2) and SNICU (2) medians (P < .05; Figure 1). Overall, HH episodes occurred more often toward the initial and/or the final steps of care sequences.
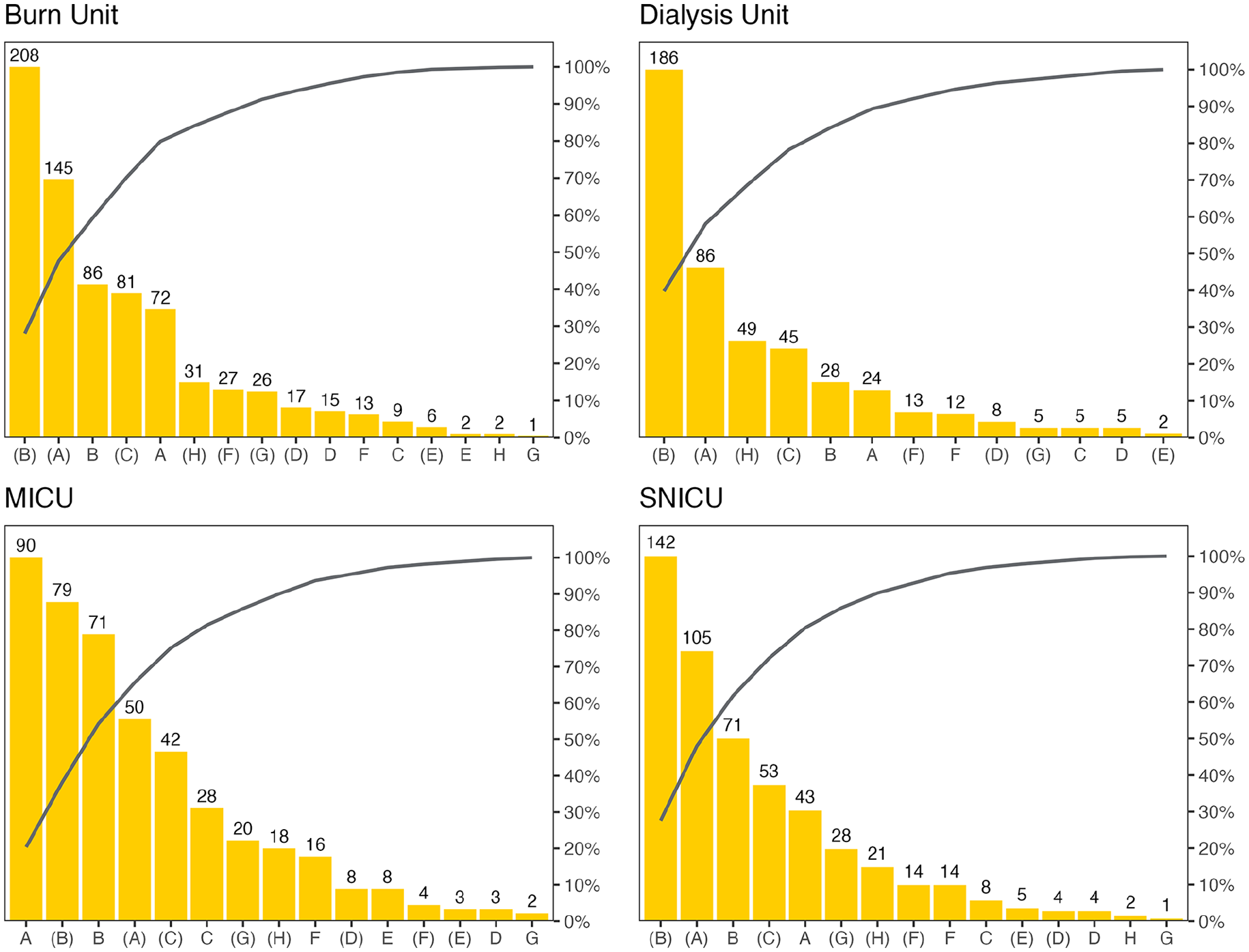
Pareto charts for the frequencies of gloved and ungloved touches during care events per category, by unit.
Discussion
Published templates designed for collecting data on patient care sequences often have significant limitations. Given these challenges, our objective was to create an optimized and innovative system for gathering detailed data crucial for enhancing IPC practices. The ACESO system streamlines the collection, entry, and initial analysis of granular data related to patient care sequences and HCPs use of IPC practices within those sequences. Preliminary results from our study revealed that HH compliance was suboptimal and occurred infrequently during a series of contacts. Notably, HCPs touched a diverse range of surfaces with potentially contaminated gloves, increasing the risk of cross-contamination. These contaminated sites and surfaces may act as reservoirs for pathogenic organisms, contributing to the potential transmission throughout the healthcare facility and the broader community. Furthermore, HCPs frequently touched patients’ body sites with potentially contaminated gloves, inadvertently introducing contaminants to patients’ clean body sites, wounds, or percutaneous devices. We designed an adaptable tool and system—ACESO—that can be utilized by various investigators, including researchers and staff, to observe HCPs’ care sequences in diverse settings, thereby, expanding our ability to study and improve HCPs’ integration of IPC practices into these sequences.
Conclusion
Given that existing templates for collecting data on patient care sequences have significant limitations, we developed the ACESO system to address these challenges. The ACESO system, designed for use by researchers and staff across diverse healthcare settings, is an adaptable tool for collecting data on HCPs’ care sequences and using these data to improve the integration of IPC practices into HCPs’ care processes. ACESO enables observers to optimize the collection, entry, and preliminary analysis of detailed data on patient care sequences and HCPs adherence to IPC practices. Our preliminary findings indicate that HH compliance is subpar during patient care and that HCPs often touch various surfaces with potentially contaminated gloves, thus increasing the risk of cross-contamination.
Footnotes
Acknowledgements
The authors thank Maria Emilia Schulter Silva, Carolina Gonzalez Bravo, and Melissa Ward for contributing to this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Centers for Disease Control and Prevention (CDC) (1 NU50CK000623-01-00). In addition, Loreen Herwaldt receives funding from CDC, and the Agency for Healthcare Research and Quality, unrelated to the work reported here. PDI Healthcare and 3M have provided products for two of Herwaldt’s studies, which are unrelated to the work reported here.
