Abstract
Duchenne muscular dystrophy (DMD) is characterized by progressive systemic muscle wasting, leading to respiratory paralysis and early death. This X-linked disease is caused by DMD mutations, encoding dystrophin. 1 There is little information regarding gastrointestinal abnormalities in patients with DMD. However, since the esophageal wall includes smooth and skeletal muscle it is also vulnerable to suffering the effects of muscle wasting in patients with DMD. After finding dyskeratosis and parakeratosis restricted to the proximal and middle esophagus with distal sparing in an 18-year-old patient with DMD, we performed an archive search of a large academic hospital and identified four additional patients with DMD who had also undergone esophageal biopsy. The patients consisted of five boys, ranging from 7 to 19 years of age. Esophageal injury was present in two patients, consisting of mild esophagitis in one, and spongiosis with dyskeratosis and parakeratosis in another. These patients were both older and had been diagnosed with DMD for greater than 15 years, while the three patients with histologically normal biopsies were younger and been diagnosed with DMD for 7, 9, and 13 years, respectively. Although the data is limited and the changes are subtle, they can be explained by the underlying muscular dystrophy pathophysiology.
Keywords
Introduction
DMD is the most common muscular dystrophy, occurring in approximately one in 5,000 males. 1 It is an X-linked disorder with mutations in the DMD gene, resulting in a non-functional dystrophin protein.1,2 It typically presents in early childhood with ascending and progressive proximal weakness, gait abnormalities, gross motor delay, or difficulty rising from the ground. 1 On clinical examination, the afflicted patients often have either a waddling gate or the classic Gower's sign, the need to use their arms to “walk up” their thighs to stand. The diagnosis can be suspected when elevated creatine kinase (CK) levels are found in combination with the above-mentioned clinical picture. Molecular studies identifying DMD mutations are the confirmatory gold standard. 2
The weakness is progressive, and most patients are wheelchair bound by 12 years of age.1,2 Previously, DMD patients died in their late teens or early twenties from cardiopulmonary insufficiency. With advancements in medical care and support, the life expectancy is now between 20 and 40 years of age. 2 While there have been previous studies demonstrating gastrointestinal motility dysfunction in patients with DMD and other muscular dystrophies, to the best of our knowledge there have been no studies that investigate the histopathologic changes that occur in the esophagus in these patients.3-7
Index Case
An 18-year-old male with DMD, obstructive sleep apnea, mild gastroparesis, osteoporosis, anxiety, and depression presented to the local emergency department with decreased oral intake, decreased urine output, dysuria and dysphagia. The patient was on daily proton pump inhibitor (PPI) therapy, without resolution of his dysphagia symptoms. He was admitted to the hospital with an acute kidney injury and concerns for sepsis, with a blood pressure of 94/53 mm Hg, a heart rate of 114 beats/minute, and a respiratory rate of 32/min. He had a prior percutaneous endoscopic gastrostomy (PEG) tube placed due to malnutrition, decreased oral intake, dysphagia, and anxiety with eating. An esophagogastroduodenoscopy (EGD) was performed. Endoscopically, the upper and lower thirds of the esophagus were normal, while the middle third appeared congested and friable. The biopsies from the proximal, middle, and distal esophagus were taken. Histologically the biopsies from the proximal and middle esophagus showed markedly hyperplastic squamous epithelium with prominent parakeratosis, spongiosis, and mild dyskeratosis; the distal esophagus was histologically unremarkable (Figure 1). Shortly afterward, the patient had a barium swallow study that showed no features of obstruction, and a gastric emptying study showed only mild delayed emptying. Manometry was not performed.
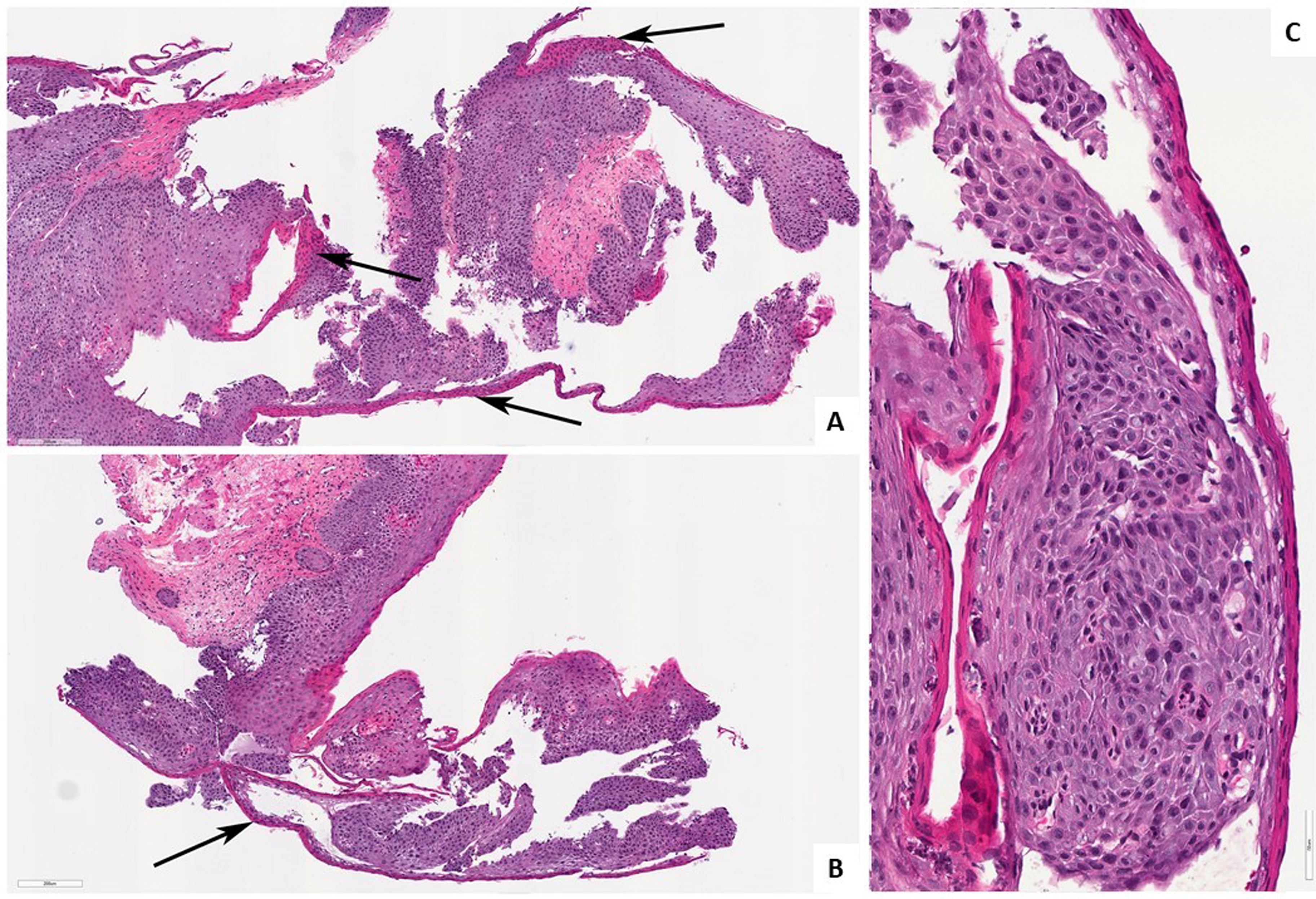
Biopsies from the proximal (A) and middle (B) esophagus displaying prominent parakeratosis (arrows), mild dyskeratosis, and spongiosis. Hematoxylin and eosin. 10× magnification. Proximal esophagus, hematoxylin and eosin, 40× magnification (C).
We then reviewed the University of Pittsburgh Medical Center system for patients with DMD who had also previously undergone EGD with biopsy. We found four additional cases and reviewed the biopsies histologically. Three were unremarkable microscopically, while the fourth contained mild esophagitis with spongiosis, increased intraepithelial lymphocytes, and basal cell hyperplasia at an unspecified location in the esophagus. The patient’s age, gender, presenting symptoms, time since diagnosis, biopsy site, and histologic interpretations are summarized in Table 1.
Clinical Presentation and Pathologic Esophageal Changes.

Discussion
While there are a handful of studies that describe gastrointestinal changes that occur in the context of DMD, our index case is the first to describe histologic abnormalities of the esophagus in these patients.3-7 In some cases, smooth muscle dysfunction can be the only clinical manifestation of a muscular dystrophy. 5 As the esophagus contains skeletal muscle in the upper and middle thirds, it is logical that this progressive systemic disease will eventually involve the esophagus. Table 1 offers some evidence that esophageal changes in DMD may not occur early in the disease. Indeed, both of the patients with histological changes were over 15 years old and had been diagnosed for at least 15 years, while two of the patients with negative diagnoses were either younger or had shorter times from diagnosis.
Table 1 also offers insight into the various upper gastrointestinal complaints that patients afflicted with DMD can have. These included decreased oral intake, dysphagia, chest pain, early satiety, nausea, vomiting, and distention. Jaffe et al also report that nasal quality of voice, dysphagia choking, and vomiting occur in a higher frequency of patients with DMD compared to age-matched controls. 5 While these are all relatively non-specific, they likely result from impaired contraction and peristalsis secondary to disrupted skeletal muscle fibers. 3 The biopsies provided in our case were superficial, containing only mucosa and submucosa, and thus we were not able to evaluate the muscularis propria.
The biopsies from the case presented contained hyperplastic epithelium with parakeratosis, spongiosis, and mild dyskeratosis in the upper two-thirds of the esophagus. We found a second case with pathologic changes that included mild esophagitis associated with spongiosis and basal cell hyperplasia. While these histologic findings are non-specific, we propose that they are secondary to impaired peristalsis leading to food impaction and abrasion of the esophageal mucosa. The friable nature of the mucosa observed endoscopically from the presented case, and the fact that both cases with pathologic changes demonstrate spongiosis support this idea. However, these changes may also be partially attributed to reflux, as Jaffe et al found that heartburn occurs more commonly in patients with DMD compared to healthy controls, 5 although in our biopsies an important negative finding is the absence of intraepithelial eosinophils, which militates against reflux.8,9 The fact that the observed changes occurred in the proximal and middle esophagus while the distal esophagus was histologically normal, and while on daily PPI therapy also argue against reflux as the mechanism driving these changes. The current patient's steroid therapy had been discontinued for multiple years due to previous fractures.
The second patient found on record review also suffered from dysphagia refractory to steroid and PPI therapy. Unfortunately, the endoscopist note was unavailable, but an esophagram and barium study performed shortly after the biopsy was interpreted as normal. This patient was not malnourished, and was instead in the 98th percentile for weight at the time of biopsy.
In conclusion, we present two cases describing histopathologic changes that occur in the esophageal mucosa of pediatric patients with longstanding Duchene muscular dystrophy. These patients are at high risk for anesthetic complications given their likelihood to have cardiopulmonary insufficiency. Thus, these findings are important to be aware of so the esophageal dysmotility workup can begin with studies that do not require anesthesia such as manometry or impedance. Given our limited patient cohort, additional research into this topic is needed to further understand the changes that can occur in this context, so we can provide proper care to ensure that patients with this ultimately fatal disease have the highest quality of life possible.
Footnotes
Acknowledgements
Informed consent was obtained in accordance with our institutional IRB policies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
