Abstract
Background:
Sacubitril/valsartan (SV) is recommended for patients with heart failure (HF). In addition, a combination of 4 HF medications, including SV, is recommended in patients with HF with reduced ejection fraction (HFrEF). However, evidence on the characteristics of patients who could continue SV and its initiation methods is limited.
Objective:
To investigate the factors associated with SV continuation and methods of combining HF medications.
Methods:
This retrospective cohort study included HF patients who initiated with SV at our institution. The endpoint was SV continuation for 6 months after its initiation. Multivariate analysis was used to extract factors associated with SV continuation. The relationship between the methods of combining HF medications (renin-angiotensin system inhibitors, beta-blockers, mineralocorticoid receptor antagonists, or sodium-glucose cotransporter 2 inhibitors), including the number of HF medications, their combination patterns, and the timing of their initiation, and SV continuation was examined in patients with HFrEF.
Results:
Of 186 eligible patients, 68.8% had HFrEF, and 79.0% continued SV for 6 months. Significant factors associated with SV continuation were albumin ≥ 3.5 g/dL (odds ratio, 4.81; 95% confidence interval, 2.19-10.59), body mass index (BMI) ≥ 18.5 kg/m2 (4.17; 1.10-15.85), and systolic blood pressure (SBP) ≥ 110 mmHg (2.66; 1.12-6.28). In patients with HFrEF, the proportion of HF medications not initiated simultaneously with SV was significantly higher in the continuation group than in the discontinuation group (67.3% vs 33.3%, P = 0.002). The number of HF medications and their combination patterns were not significantly associated with SV continuation.
Conclusion and Relevance:
Albumin, BMI, and SBP are useful indicators for selecting patients who are likely to continue SV. In addition, initiating only SV without simultaneously initiating other HF medications in patients with HFrEF may lead to SV continuation.
Keywords
Introduction
The number of patients with heart failure (HF) is increasing annually worldwide. 1 In Japan, the 1-year rehospitalization rate due to worsening HF is 25%, and the 1-year all-cause mortality rate is 23%, with poor prognosis. 2 HF is classified into 3 categories based on the left ventricular ejection fraction (LVEF): HF with reduced ejection fraction (HFrEF), HF with mildly reduced ejection fraction (HFmrEF), and HF with preserved ejection fraction (HFpEF). HF treatment guidelines indicate the treatment for each category.3-5 For patients with HFrEF, a combination of 4 HF medications is recommended: angiotensin-converting enzyme inhibitors (ACEi)/angiotensin receptor blockers (ARB)/angiotensin receptor neprilysin inhibitor (ARNI), beta-blockers, mineralocorticoid receptor antagonists (MRA), and sodium-glucose cotransporter 2 inhibitors (SGLT2i).3-5 For patients with HFmrEF and HFpEF, treatment of comorbidities, such as hypertension, atrial fibrillation, and diabetes, and administration of diuretics for congestion is the standard treatment.3-5 This includes using medications, such as ACEi/ARB/ARNI, beta-blockers, MRA, SGLT2i, anticoagulants, antiarrhythmics, and hypoglycemic agents.3,4 Furthermore, patients whose LVEF is improved to > 40%, referred to as patients HF with improved ejection fraction, are recommended to continue HF treatment even after their LVEF has increased. 4
Sacubitril/valsartan (SV), an ARNI, reduce rehospitalization and cardiovascular death rates compared with enalapril, an ACEi, in patients with HFrEF. 6 Whereas, in patients with LVEF ≥ 45%, SV has not been shown to be more effective than ARB. 7 In another study of patients with symptomatic HF, SV reportedly reduced HF hospitalization and cardiovascular death in patients with HFmrEF compared with renin-angiotensin system inhibitors (RASi). 8 Therefore, SV is recommended in patients with HFrEF, HFmrEF, and those with LVEF closer to the lower normal limits.3,4
In a previous study, the SV prescription rate 1 year after discharge was 13% in patients first admitted for HF. 9 In addition, 12.7% of patients using ACEi or ARB switched to SV within approximately 1.5 years. 10 Furthermore, approximately 25% of patients started on SV discontinued within 1 year.10,11 Thus, despite the recommendations for SV in the guidelines, the initiation and continuation rates of SV are extremely low. This may be influenced by various factors, such as patient characteristics and adverse effects.
In outpatients with HFrEF, factors associated with SV discontinuation were unemployment, low scores on the Kansas City Cardiomyopathy Questionnaire, chronic kidney disease (CKD), and arrhythmias. 10 In patients admitted for HF, factors associated with SV continuation were LVEF ≤ 40% and a history of HF hospitalization. 12 SV continuation may be affected by physical characteristics, such as body mass index (BMI), blood pressure, and laboratory data. However, evidence on the physical characteristics of patients who could continue SV and initiation methods is limited.
In patients with HFrEF, the combination of 4 HF medications is recommended,3-5 and early initiation and titration of these medications have been shown to improve prognosis. 13 Furthermore, according to a meta-analysis, the combination of 4 HF medications (SV, beta-blockers, MRA, and SGLT2i) prolongs survival compared with the combination of ACEi/ARB and beta-blockers. 14 These findings suggest that patients using the 4 HF medications at SV initiation may have more stable HF symptoms due to their additive effects compared with those who are not. However, the relationship between the number of HF medications, their combination patterns, and the timing of their initiation and SV continuation are unclear. Therefore, clarifying the optimal method of combining the 4 HF medications may lead to SV continuation.
To select patients who could continue SV, we aimed to investigate the factors associated with SV continuation in patients with HF. We also investigated the relationship between methods of combining HF medications and SV continuation in patients with HFrEF.
Methods
Study Design and Patients
We performed a retrospective cohort study of HF patients who initiated with SV at the Department of Cardiology, Showa University Fujigaoka Hospital, from August 1, 2020, to December 31, 2022. The inclusion criteria were as follows: (1) HF diagnosis or confirmation based on the criteria in the guidelines, 3 (2) HF in all LVEF categories, (3) SV initiation twice daily or switching from SV once daily for hypertension treatment 15 to SV twice daily for HF treatment, and (4) SV initiation for HF treatment either in inpatient or outpatient settings. The exclusion criteria were as follows: death during hospitalization, participation in a clinical trial at SV initiation, unknown primary endpoints because treatment could not be continued at our institution, and age < 18 years. After the exclusion of 36 patients, 186 patients were included in this study (Supplementary Figure 1).
This study was approved by the Ethics Committee of Showa University, Japan (Approval No. 22-255-B), which waived the requirement for informed consent due to the retrospective nature of the study. Patients were informed that they could opt out of having their data used for research purposes on the Showa University Fujigaoka Hospital’s website.
Treatment of HF
The cardiologist prescribed HF medications following treatment guidelines. 3 SV was initiated twice daily for HF patients, either inpatient or outpatient settings. When switching from ACEi to SV, SV was administered at least 36 hours after the last dose of ACEi. Subsequently, SV was titrated up to 400 mg/day, as tolerated by the patient. The physician made the decision to discontinue SV based on the patient’s condition.
Data Collection
Data on patient characteristics, physical findings, laboratory data, and medications were collected from the medical records at SV initiation. Patient characteristics included age, sex, BMI, past admission for HF, LVEF, past smoking and drinking, primary causes of HF, and comorbidities. Physical findings included systolic and diastolic blood pressure. Laboratory data included hemoglobin, blood urea nitrogen, serum creatinine, estimated glomerular filtration rate (eGFR), albumin, sodium, potassium, and brain natriuretic peptide (BNP) levels. Data at the closest point to SV initiation were collected if physical findings and laboratory data at SV initiation were unavailable.
The following medications used before and at SV initiation were investigated: RASi (ACEi/ARB/ARNI), beta-blockers, MRA, SGLT2i, loop diuretics, tolvaptan, thiazide diuretics, antiarrhythmic agents, antiplatelet agents, anticoagulants, statins, hypoglycemic agents, calcium channel blockers, and potassium adsorbents. Medications before SV initiation were defined as those prescribed in ambulatory care just before SV initiation.
We also collected information on the setting of SV initiation (inpatient or outpatient), duration of SV use in the discontinuation group, and reasons for SV discontinuation from the medical records. The dose at SV initiation, discontinuation, and 6 months after SV initiation were also collected.
In patients with HFrEF, information on the number of HF medications (RASi, beta-blockers, MRA, and SGLT2i) 6 months after SV initiation was collected for the continuation and discontinuation groups.
Endpoint
The endpoint was SV continuation for 6 months after its initiation. Patients who discontinued SV even once within 6 months after SV initiation were assigned to the discontinuation group.
Statistical Analysis
Sample size
We estimated that a sample size of 131 patients would be required to detect an odds ratio of 2.0, with 80% statistical power at a significance level of 0.05 in an equivalence test of the hypothesis, assuming the SV continuation rate to be 85%.12,16-19 The sample size calculated using the actual SV continuation rate of 79% was 102 patients. Therefore, the sample size in this study was sufficient to arrive at a valid conclusion. 20
Stratification
For convenience in clinical use, the continuous variables were dichotomized and treated as categorical variables. Patients were divided into 2 groups based on the cutoff values according to the World Health Organization’s definition for age (65 years), BMI (18.5 kg/m2), and hemoglobin (13 and 12 g/dL in males and females, respectively). There is no report of an appropriate cutoff value for systolic blood pressure (SBP); therefore, the cutoff value was explored and set at 110 mmHg. Patients were divided into 2 groups based on eGFR (cutoff: 60 mL/min/1.73 m2) according to the CKD guidelines, 21 albumin levels (cutoff: 3.5 g/dL) according to the criteria for hypoalbuminemia, 22 and BNP levels (cutoff: 200 pg/mL) as an indicator of the need for HF treatment. 23 Other laboratory data were divided into 2 groups according to the upper or lower limits of the reference range values for clinical laboratory tests established by the Japanese Association of Clinical Laboratory Standards. 24
Factors affecting SV continuation
Patient characteristics, physical findings, laboratory data, medications, the dose of SV initiation, and the setting of SV initiation were compared between the SV continuation and discontinuation groups. Univariate analysis was performed using the χ2 test or Fisher’s exact test. Significant variables extracted by univariate analysis were entered into the multivariate analysis. Ischemic heart disease and antiplatelet agents (r: 0.698), the setting of SV initiation and albumin levels (r: -0.545), and albumin and BNP levels (r: –0.491) were correlated; thus, ischemic heart disease and albumin were entered into the multivariate analysis. Ischemic heart disease was selected because it is a common cause of HF. Albumin was selected because its odds ratio for SV continuation was higher than that in the setting of SV initiation and BNP. SBP was not correlated with hypertension (r: 0.121); however, both factors reflect blood pressure; thus, SBP, which was used to determine SV initiation, was selected. Finally, age < 65 years, BMI ≥ 18.5 kg/m2, SBP ≥ 110 mmHg, albumin ≥ 3.5 g/dL, and ischemic heart disease were entered into the multivariate analysis. Multivariate analysis was performed using logistic regression analysis. Independent and significant factors associated with SV continuation were extracted using a stepwise method. P < 0.05 was considered statistically significant. Statistical analyses were performed using SPSS version 28 (IBM, Tokyo, Japan).
Relationship between methods of combining HF medications and SV continuation in patients with HFrEF
In patients with HFrEF (LVEF < 40%), the relationship between the methods of combining HF medications (RASi, beta-blockers, MRA, and SGLT2i) and SV continuation was investigated in the following 4 ways.
The relationship between the number of HF medications before and at SV initiation and SV continuation was investigated using the χ2 test or Fisher’s exact test. “Heart failure medications before SV initiation” were defined as those prescribed in ambulatory care just before SV initiation. When SV was initiated in an outpatient setting, the HF medications before SV initiation were defined as those prescribed in the last ambulatory care visit. When SV was initiated in an inpatient setting, the HF medications before SV initiation were defined as those prescribed in the ambulatory care visit immediately preceding the admission.
The combination patterns of HF medications in the SV continuation and discontinuation groups were compared using the χ2 test or Fisher’s exact test.
The relationship between initiating any of HF medications other than SV (beta-blockers, MRA, and SGLT2i) at the same time as SV initiation and SV continuation was investigated using the χ2 test. “At the same time as SV initiation” was defined as the same day as SV initiation when SV was initiated in an outpatient setting or the period from admission to SV initiation when SV was initiated in an inpatient setting.
In the inpatient group, the period from admission to SV initiation between the SV continuation and discontinuation groups was compared using the Mann-Whitney U test.
Results
Patient Characteristics
Patient characteristics are shown in Tables 1 and 2. The patients’ median age was 74 (interquartile range; 61-81) years, 137 (73.7%) were male, and 128 (68.8%) had HFrEF. Ten patients (5.4%) had previously used SV for hypertension, 15 and 81 patients (43.5%) initiated with SV during hospitalization. The initiation dose of SV was 100 mg/day in 164 patients (88.2%, Supplementary Table 1). No factor was missing in more than 10% of patients.
Patient Characteristics and Medications Before SV Initiation (n = 186).
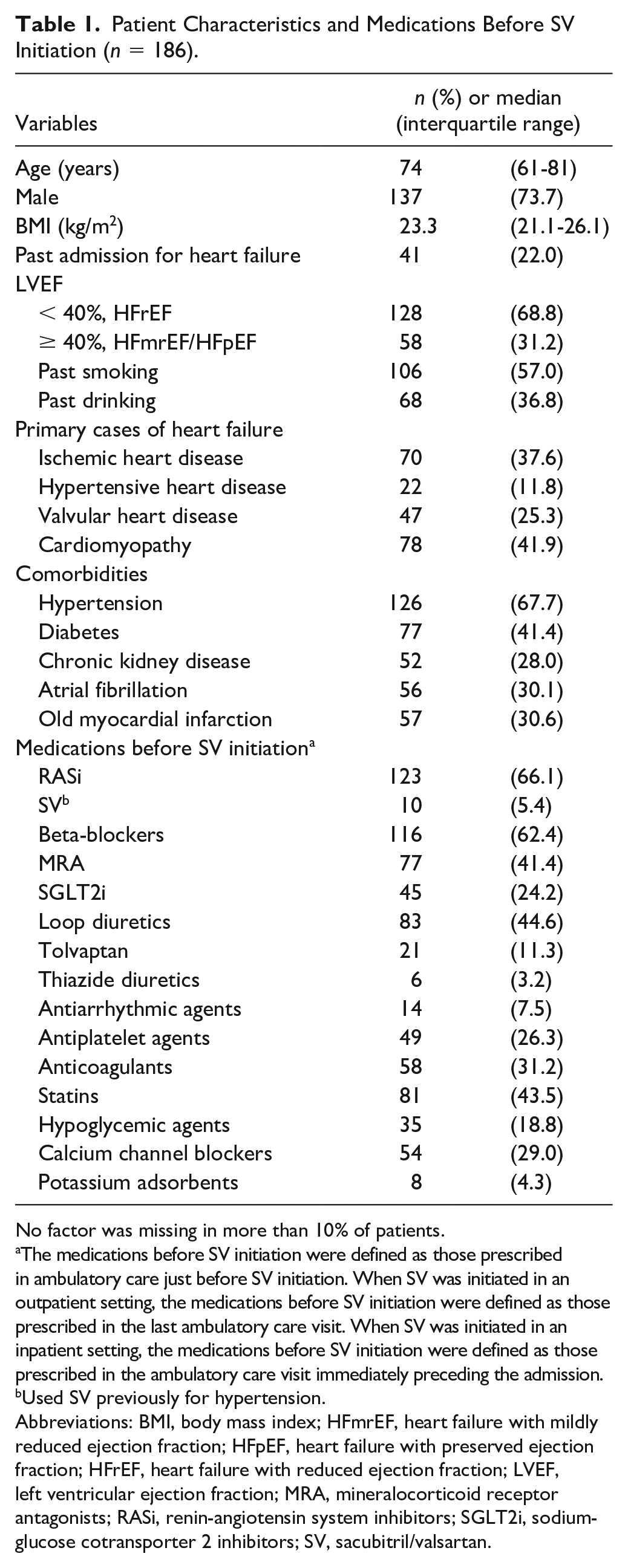
No factor was missing in more than 10% of patients.
The medications before SV initiation were defined as those prescribed in ambulatory care just before SV initiation. When SV was initiated in an outpatient setting, the medications before SV initiation were defined as those prescribed in the last ambulatory care visit. When SV was initiated in an inpatient setting, the medications before SV initiation were defined as those prescribed in the ambulatory care visit immediately preceding the admission.
Used SV previously for hypertension.
Abbreviations: BMI, body mass index; HFmrEF, heart failure with mildly reduced ejection fraction; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; LVEF, left ventricular ejection fraction; MRA, mineralocorticoid receptor antagonists; RASi, renin-angiotensin system inhibitors; SGLT2i, sodium-glucose cotransporter 2 inhibitors; SV, sacubitril/valsartan.
Patient Findings and Medications at SV Initiation (n = 186).
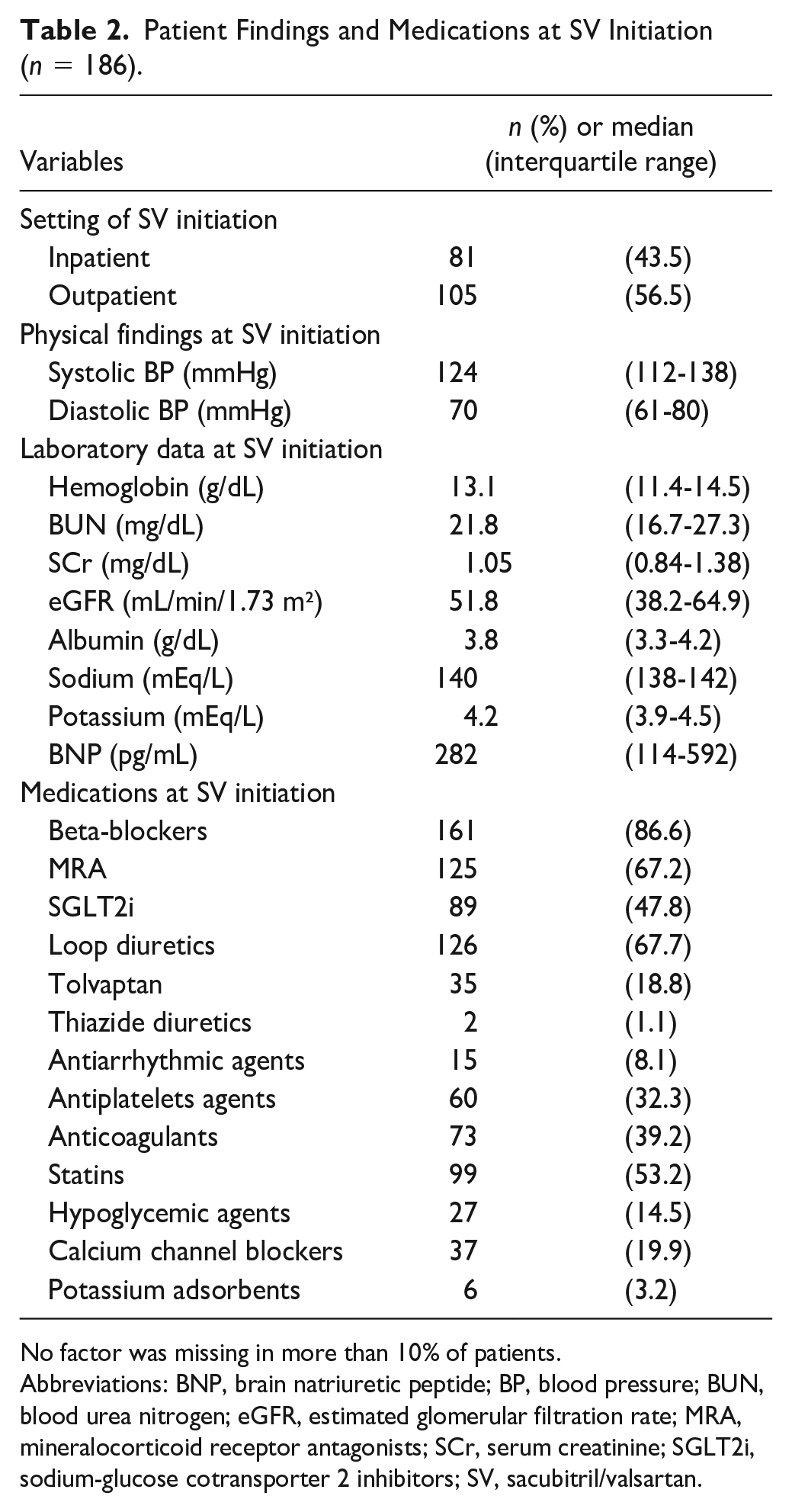
No factor was missing in more than 10% of patients.
Abbreviations: BNP, brain natriuretic peptide; BP, blood pressure; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; MRA, mineralocorticoid receptor antagonists; SCr, serum creatinine; SGLT2i, sodium-glucose cotransporter 2 inhibitors; SV, sacubitril/valsartan.
Endpoint
SV was continued for ≥ 6 months in 147 patients (79.0%). In contrast, 39 patients (21.0%) discontinued SV within 6 months. The most common reason for SV discontinuation was hypotension (74.4%, Supplementary Table 2). In the discontinuation group, the median duration of SV use was 26 days (Supplementary Table 2). Furthermore, in the continuation group, only 33.3% of the patients achieved the SV target dose at 6 months after SV initiation (Supplementary Table 1). The rate of patients receiving SV 100 mg/day at discontinuation was 79.5% (Supplementary Table 1).
In patients with HFrEF, the rate of using the 4 HF medications 6 months after SV initiation was 44.2 and 40.0% in the continuation and discontinuation groups, respectively (Supplementary Figure 2).
Factors Affecting SV Continuation
In the univariate analysis, the proportions of patients with age < 65 years, BMI ≥ 18.5 kg/m2, hypertension, SV initiation in the outpatient setting, SBP ≥ 110 mmHg, albumin ≥ 3.5 g/dL, and BNP < 200 pg/mL were significantly higher in the continuation group than in the discontinuation group (Table 3). In contrast, the proportions of patients with ischemic heart disease and taking antiplatelet agents were significantly lower in the continuation group than in the discontinuation group (Table 3).
Relationship Between Patient Characteristics and SV Continuation (n = 186).
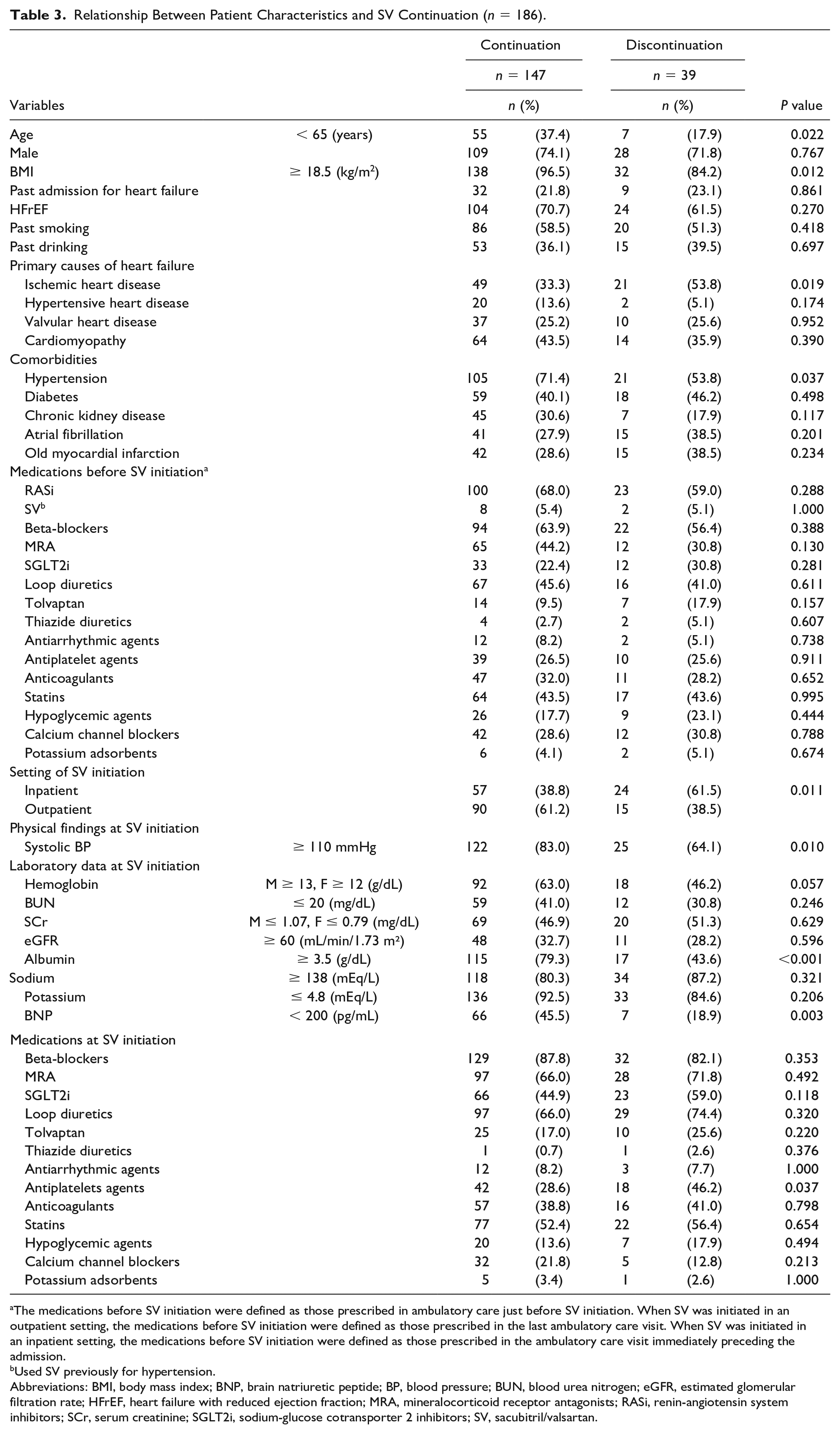
The medications before SV initiation were defined as those prescribed in ambulatory care just before SV initiation. When SV was initiated in an outpatient setting, the medications before SV initiation were defined as those prescribed in the last ambulatory care visit. When SV was initiated in an inpatient setting, the medications before SV initiation were defined as those prescribed in the ambulatory care visit immediately preceding the admission.
Used SV previously for hypertension.
Abbreviations: BMI, body mass index; BNP, brain natriuretic peptide; BP, blood pressure; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; HFrEF, heart failure with reduced ejection fraction; MRA, mineralocorticoid receptor antagonists; RASi, renin-angiotensin system inhibitors; SCr, serum creatinine; SGLT2i, sodium-glucose cotransporter 2 inhibitors; SV, sacubitril/valsartan.
In the multivariate analysis, albumin ≥ 3.5 g/dL, BMI ≥ 18.5 kg/m2, and SBP ≥ 110 mmHg were independent significant factors associated with SV continuation (Table 4).
Factors Associated With SV Continuation in Patients With Heart Failure (n = 186).
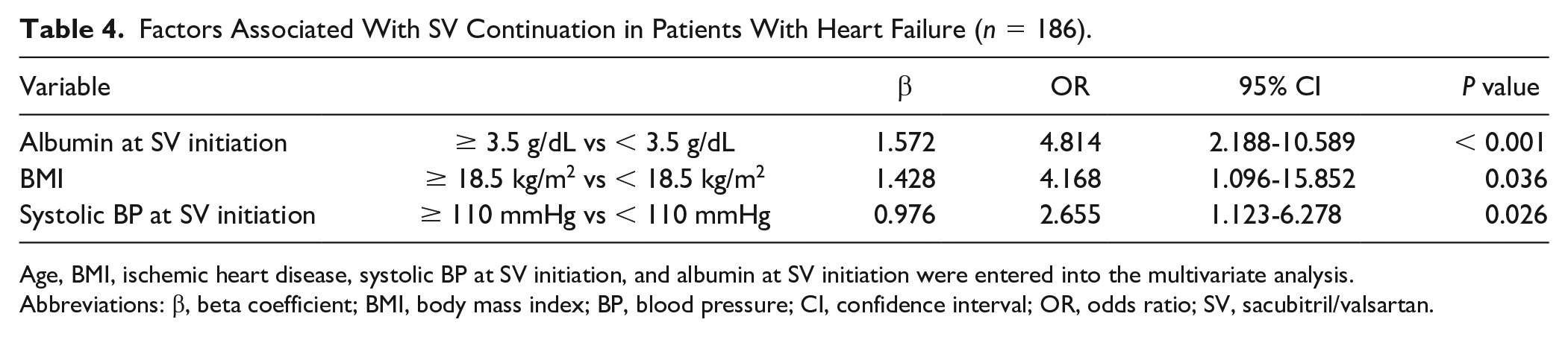
Age, BMI, ischemic heart disease, systolic BP at SV initiation, and albumin at SV initiation were entered into the multivariate analysis.
Abbreviations: β, beta coefficient; BMI, body mass index; BP, blood pressure; CI, confidence interval; OR, odds ratio; SV, sacubitril/valsartan.
Relationship Between Methods of Combining HF Medications and SV Continuation in Patients With HFrEF
The number of the 4 HF medications before or at SV initiation was not significantly different between the SV continuation and discontinuation groups (Figure 1).
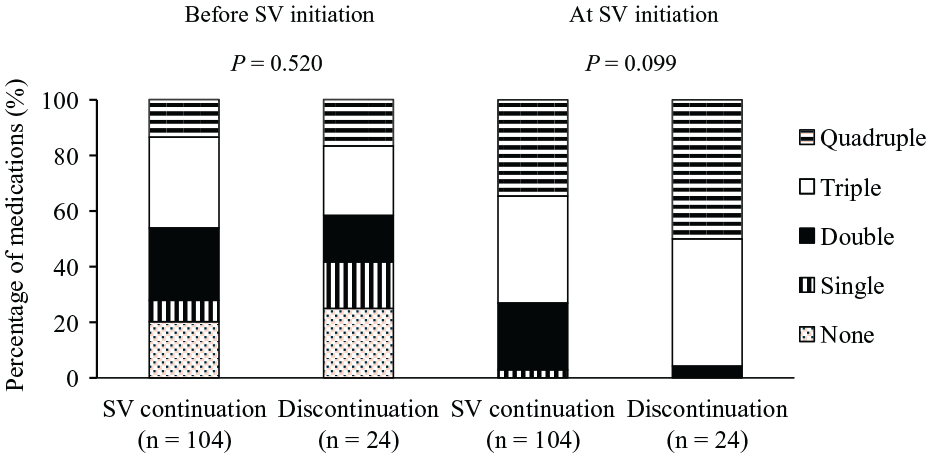
Relationship between the number of heart failure medications initiated before and at SV initiation and SV continuation in patients with HFrEF. The heart failure medications included RASi, beta-blockers, MRA, and SGLT2i. “Heart failure medications before SV initiation” were defined as those prescribed in ambulatory care just before SV initiation. When SV was initiated in an outpatient setting, the heart failure medications before SV initiation were defined as those prescribed in the last ambulatory care visit. When SV was initiated in an inpatient setting, the heart failure medications before SV initiation were defined as those prescribed in the ambulatory care visit immediately preceding the admission.
The combination patterns of the 4 HF medications at SV initiation were not significantly different between the SV continuation and discontinuation groups (Supplementary Table 3).
The proportion of patients who had not initiated any of HF medications other than SV (beta-blockers, MRA, and SGLT2i) at the same time as SV initiation was significantly higher in the SV continuation group than in the discontinuation group (67.3% vs 33.3%, P = 0.002, Figure 2).
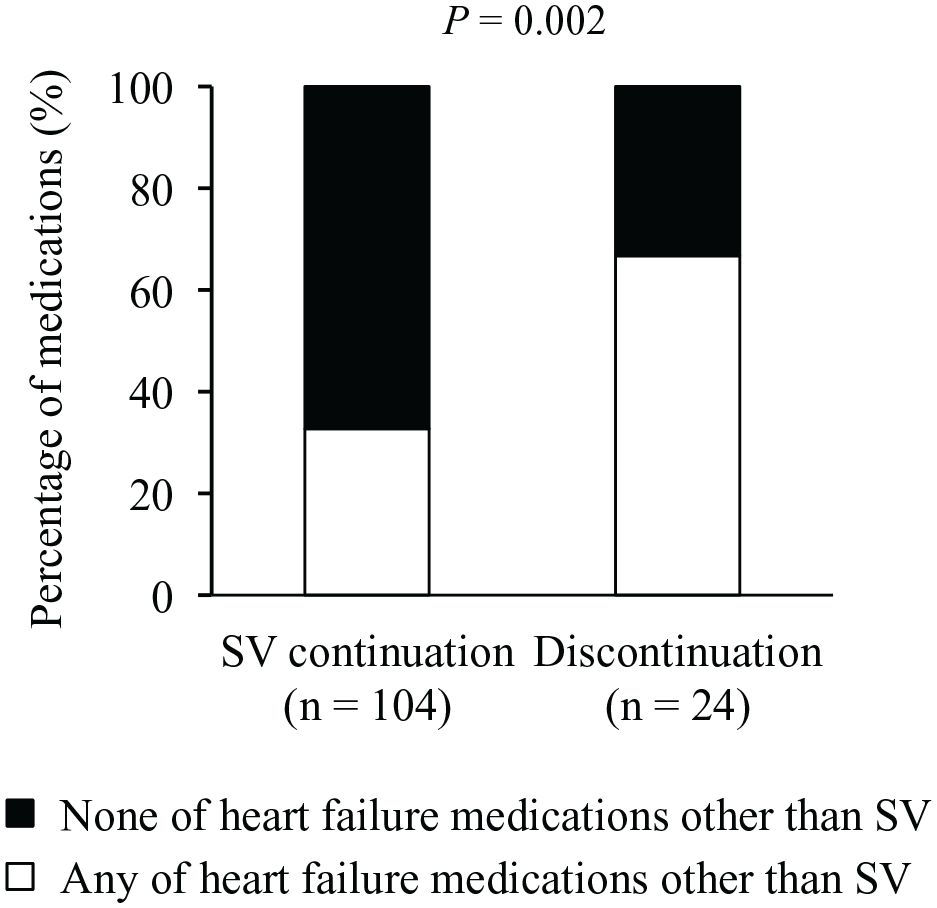
Relationship between initiation of any of heart failure medications other than SV at the same time as SV initiation and SV continuation in patients with HFrEF. The heart failure medications other than SV included beta-blockers, MRA, and SGLT2i. “At the same time as SV initiation” was defined as the same day as SV initiation when SV was initiated in an outpatient setting or the period from admission to SV initiation when SV was initiated in an inpatient setting.
In patients who initiated SV during hospitalization (n = 60), the period from admission to SV initiation was not significantly different between the SV continuation and discontinuation groups (median [interquartile range], continuation group vs discontinuation group, 5 [3-6] vs 6 [3-10], P = 0.184).
Discussion
Albumin ≥ 3.5 g/dL, BMI ≥ 18.5 kg/m2, and SBP ≥ 110 mmHg were factors associated with SV continuation in patients with HF. In addition, in patients with HFrEF, SV initiation as a single agent without initiating HF medications other than SV (beta-blockers, MRA, and SGLT2i) at the same time was associated with SV continuation. To the best of our knowledge, this is the first study to identify the physical factors associated with SV continuation in patients with HF and an effective combination of HF medications. These factors could be useful indicators for selecting patients who are likely to continue SV. Furthermore, it may lead to suggestions for effective methods of combining HF medications at SV initiation.
We found that lower albumin levels were associated with SV discontinuation. Patients with lower albumin levels might have discontinued SV because of their poor general condition. 25 Alternatively, SV may have been discontinued due to progressive fluid retention and inability to maintain circulation. 25 Therefore, we considered that patients with preserved albumin levels could continue SV because of their good general condition and stable circulation.
Patients who discontinued SV reportedly have a lower BMI than those who continued SV.16,17 There are 2 possible reasons that BMI was associated with SV continuation. The first is the effect of SV blood concentration. A systematic review of patients with cachexia, including those with BMI < 20 kg/m2, reported that patients with a lower BMI had a smaller volume of drug distribution than those with a higher BMI. 26 This suggests that, at the same dose, the blood concentration of SV in slimmer patients would likely be higher than that in normal- or overweight patients. Therefore, patients with low BMI may show stronger pharmacological effects of SV due to increased blood concentration, resulting in a higher rate of adverse effects. The second possible reason is the influence of SV tolerability. Patients with high BMI may have a better tolerance for the adverse effects of SV, such as hypotension, renal dysfunction, and hyperkalemia. A higher BMI has been reported as a determinant of achieving the SV target dose. 16 Therefore, patients with high BMI were possibly more tolerant of the adverse effects and could continue SV.
The relationship between low blood pressure at SV initiation and SV discontinuation has been reported,17,18,27 and similar results were found in this study. A previous study has shown that, in patients with HF, SBP at 2 to 4 weeks after SV initiation was 6 to 10 mmHg lower than that at SV initiation. 18 Therefore, patients with blood pressure ≥110 mmHg at SV initiation may have been able to continue SV because of the low risk of symptomatic hypotension. In this study, we found a cutoff value of 110 mmHg for SBP at SV initiation, which may be a critical indicator for determining SV initiation.
In patients with HFrEF, initiating HF medications (beta-blockers, MRA, or SGLT2i) simultaneously with SV initiation may have increased the risk of adverse effects leading to SV discontinuation. There are 2 possible reasons for this increased risk. First, there is the additive effect of HF medications. Adverse effects of beta-blockers include hypotension and bradycardia, those of MRA include hyperkalemia and renal dysfunction, and those of SGLT2i include hypotension and renal dysfunction. 5 Therefore, initiating HF medications simultaneously with SV initiation may have led to a stronger effect on hypotension, renal dysfunction, and hyperkalemia due to the additive effects of the medications. Second, inpatients may have had unstable blood pressure and HF. During periods of unstable blood pressure and HF, initiating SV and HF medications other than SV at the same time may have increased the risk of adverse effects. Therefore, to reduce the risk of discontinuation due to adverse effects, it may be better to initiate SV as a single agent. Particularly for inpatients, it is preferable to initiate SV as a single agent after blood pressure and HF have stabilized. In addition, a few patients discontinued any HF medications at SV initiation (6 patients in the continuation group and 3 in the discontinuation group); thus, discontinuing HF medications at SV initiation is not necessary.
In the STRONG-HF study, patients in the high-intensity care group were prescribed several HF medications at discharge, and a few patients were not receiving HF medications after 6 weeks (1.0%-5.0%). 13 This suggests that several HF medications could be initiated at discharge. However, the inclusion criteria for our study differed from those for the STRONG-HF study, so that, this study included patients with unstable blood pressure and HF. This may have contributed to the finding that SV initiation as a single agent leads to SV continuation.
In patients with HFrEF, the use of 4 HF medications is recommended.3-5 It has been reported that early initiation and up-titration of HF medications improved the prognosis of patients with HF. 13 Therefore, we hypothesized that the number of 4 HF medications at SV initiation would lead to SV continuation, but no relationship was observed. This could be because this was a retrospective study with uncontrollable confounding factors.
Limitations
This study had 4 limitations. First, the external validity of the factors could not be verified as this was a single-center study. Second, data on the severity of HF in outpatients based on the New York Heart Association were unavailable; thus, the relationship between HF severity and SV continuation could not be investigated. To complement this limitation, the setting of SV initiation was included in the analysis. Third, owing to the retrospective nature of the study, standardizing the period between before and at SV initiation and the frequency of clinic visits after SV initiation was impossible. Fourth, no information was collected on the duration of use and doses of HF medications other than SV, SV adherence during the 6 months after SV initiation, and past preferences.
Conclusion and Relevance
Albumin ≥ 3.5 g/dL, BMI ≥ 18.5 kg/m2, and SBP ≥ 110 mmHg were factors associated with SV continuation in patients with HF. These factors can be useful indicators for selecting patients who are more likely to continue SV. In addition, in patients with HFrEF, SV initiation as a single agent without initiating beta-blockers, MRA, or SGLT2i at the same time as SV initiation may lead to SV continuation. This result can serve as suggestions for methods of combining HF medications at SV initiation. Furthermore, this information will be useful for healthcare professionals when initiating SV.
Supplemental Material
sj-docx-1-aop-10.1177_10600280241277354 – Supplemental material for Factors Associated With Sacubitril/Valsartan Continuation and the Methods of Combining Heart Failure Medications in Patients With Heart Failure
Supplemental material, sj-docx-1-aop-10.1177_10600280241277354 for Factors Associated With Sacubitril/Valsartan Continuation and the Methods of Combining Heart Failure Medications in Patients With Heart Failure by Erika Iwasaki, Noriko Kohyama, Mayumi Inamoto, Michiru Nagao, Tomiko Sunaga, Hiroshi Suzuki, Mio Ebato and Mari Kogo in Annals of Pharmacotherapy
Footnotes
Acknowledgements
The authors are grateful to Yusuke Kato for his efforts in collecting the data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
