Abstract
Objective:
This article reviews the efficacy and safety of 1% tapinarof cream for plaque psoriasis.
Data sources:
A literature search was conducted from August 2022 to February 2023. The terms tapinarof, VTAMA, benvitimod, GSK2894512, DMVT-505, and WBI-1001 were queried in PubMed. ClinicalTrials.gov was searched to identify ongoing or unpublished studies.
Study selection and data extraction:
All clinical trials written in English and relevant to pharmacology, efficacy, and safety were included.
Data synthesis:
In two 12-week phase III clinical trials, disease severity assessed by a Physician’s Global Assessment (PGA) score of clear or almost clear and a 2-point PGA improvement was 35.4% and 40.2% at week 12 in the 2 trials, respectively. In the 40-week, open-label extension trial, the efficacy and safety results were similar: 40.9% of patients achieved a PGA of 0 at least once during the trial, and 58.2% of patients with PGA ≥ 2 achieved PGA 0/1 at least once.
Relevance to Patient Care and Clinical Practice in Comparison to Existing Drugs:
Tapinarof is a topical aryl hydrocarbon receptor agonist and a first-in-class, potentially promising treatment for plaque psoriasis recently approved by the U.S. Food and Drug Administration.
Conclusion:
Compared with placebo, tapinarof may be an effective and safe topical treatment for mild to severe plaque psoriasis. Head-to-head trials to compare the efficacy and adverse effect profile of tapinarof to other topical treatments are still needed, as are investigation in patients with recent or current use of phototherapy or biologic or nonbiologic systemics. Cost and adherence to treatment may be barriers for treatment efficacy.
Introduction
Psoriasis is a chronic, immune-mediated disease with a prevalence of 3.0% among adults in the United States. 1 Plaque psoriasis is the most common subtype, typically presenting with salmon-colored plaques with silvery scales beginning in middle adulthood. Although cutaneous involvement may be the most well-recognized manifestation, psoriasis is a multisystem inflammatory disorder with an extensive impact on quality of life and a high annual burden of cost in the United States that was estimated at 112 billion dollars in 2013. 2
The pathogenesis of plaque psoriasis involves infiltration and activation of CD4+ T cells including T helper (Th)17 and Th22 cells, which results in aberrant differentiation and hyperproliferation of keratinocytes. Psoriasis is also associated with an abnormal skin barrier and impaired barrier function. Oxidative stress has also been implicated in the disease and may result in additional inflammation and further impaired skin barrier function.3,4 Dysfunction of the interleukin (IL)-23/IL-17A pathway is considered the central pathogenic axis and is the target of many targeted systemic biologics.
A range of treatment modalities are used for psoriasis including topical corticosteroids, topical calcineurin inhibitors, topical vitamin D analogs, topical and systemic retinoids, light therapy, and systemic immunosuppressants and immune modulators. Because mild and localized psoriasis is the most common form of the disease, topical therapies are the mainstay of treatment for up to 80% of patients.5,6 However, many topical corticosteroids have restrictions on application site or duration of use, while topical calcineurin inhibitors and tazarotene are associated with skin irritation. There is, therefore, a potential unmet need for a well-tolerated, nonsteroidal topical therapy.
The aryl hydrocarbon receptor (AhR) is a cytoplasmic, ligand-activated transcription factor that is involved in terminal differentiation of CD4+ Th17 and Th22 cells, production of IL-17, development and maintenance of the skin barrier, and the response to external environmental signals. 7 Tapinarof 1% cream is a nonsteroidal AhR agonist that was approved by the U.S. Food and Drug Administration (FDA) for the treatment of adults with plaque psoriasis in May 2022. 8 Tapinarof cream is a first-in-class topical treatment in development for atopic dermatitis and psoriasis although systemic AhR agonists have been available for other inflammatory conditions. 8 This review will evaluate published clinical studies to summarize the pathophysiology of tapinarof, as well as the efficacy, safety, and implications for the treatment of plaque psoriasis.
Methods
A review of the literature was performed from August 2022 to February 2023. The terms tapinarof, VTAMA, benvitimod, GSK2894512, DMVT-505, and WBI-1001 were queried in the MEDLINE (PubMed) and EMBASE databases and yielded 16 results. Available studies were considered for inclusion if written in English and relevant to pharmacology, clinical trials, efficacy, adverse reactions, and safety. References of the reviewed articles were also searched for additional studies for inclusion. A total of 8 articles were included in this review. The website ClinicalTrials.gov was searched to identify ongoing or unpublished studies.
A range of clinical metrics will be discussed in this review, of which several are psoriasis severity measures. The extent of body surface area (BSA) involved is calculated using the patient’s closed hand from the wrist to the fingers and thumb to represent approximately 1% of the patient’s BSA. This metric does not incorporate body location, clinical characteristics of the plaques, or symptoms or quality of life. The Psoriasis Area and Severity Index (PASI) incorporates 3 plaque characteristics—erythema, induration, and scaling—with the BSA affected to give a score from 0 to 72. A score ≥10 is considered moderate to severe psoriasis; PASI is inaccurate for mild psoriasis. An expert panel convened by the National Psoriasis Foundation defined “treatment success” as a 75% improvement from baseline or total involved BSA of less than 3%. This 75% improvement from baseline is studied and reported using the PASI-75. The 50% and 90% improvements from baseline, given by PASI-50 and PASI-90, respectively, are also commonly used endpoints in clinical trials. 5 PASI is highly correlated with the Dermatology Life Quality Index (DLQI), a quality-of-life assessment tool.
The Physician Global Assessment (PGA) also uses erythema, induration, and scale, with scores of 0 (clear), 1 (almost clear), 2 (mild), 3 (moderate), and 4 (severe). BSA, PASI, and PGA do not incorporate symptoms or impact on quality of life. The DLQI is a 10-item questionnaire that is widely used to measure the impact of skin disease on an individual’s quality of life. Scores range from 0 to 30. The Peak Pruritus Numeric Rating Scale (PP-NRS) is a 1-item, patient-reported outcome (PRO) of itch severity ranging from 0 to 10. A change ≥2 to 4 points is the threshold for identifying a clinically relevant, within-person response. 9 The Psoriasis Symptom Diary is an electronic, 20-item daily assessment to measure patient-perceived psoriasis severity and disease-related impacts. 10
Results
Drug Pharmacology
Mechanism of action
Tapinarof is a naturally derived polyphenol distinct from plant-based polyphenols that is produced by the Photorhabdus luminescens bacterial symbionts of Heterorhabditis nematodes. 11 The nonsteroidal anti-inflammatory properties of tapinarof occur through activation of the AhR, a cytoplasmic, ligand-activated transcription factor that is widely expressed in the skin and activated by a variety of ligands. Activation causes the AhR-ligand complex to heterodimerize with the AhR nuclear translocator, which is a high-affinity DNA-binding transcription factor that can cause transcription of AhR-responsive genes. 7 AhR signaling in healthy skin impacts multiple biological processes. The AhR is a regulator of innate and adaptive immune responses including the terminal differentiation of Th17 and Th22 cells and the expression of cytokines including IL-10, IL-21, and IL-22. AhR signaling has a role in formation and maintenance of the skin barrier through keratinocyte differentiation and expression of the skin barrier proteins filaggrin, hornerin, and involucrin and response to environmental signals such as ultraviolet (UV) B exposure, environmental toxins, and microbial products.11,12
Tapinarof also exhibits antioxidant activity: The molecule contains 2 phenol groups that directly scavenge superoxide anions and hydroxyl radicals, and further antioxidant activity may additionally derive from activation of AhR and partial activation of the nuclear factor erythroid 2-related factor 2 (NRF2) pathway.7,11-13
Pharmacokinetics
Systemic absorption of tapinarof was low in patients with moderate to severe atopic dermatitis following application of both 1% and 2% creams. In a phase I study (NCT02466152), the day 1 maximum concentration (Cmax) for the 2% cream was 2.5 ng/mL, and the day 21 value was 0.3 ng/mL, while the 1% values were 1.2 ng/mL and 0.15 ng/mL for days 1 and 21, respectively. The median time to peak drug concentration (Tmax) for the 2% formulation was 2 hours on the first day and 4 hours on day 21, while the corresponding values for the 1% formulation were 3 hours on day one and 1 hour on day 21. Overall, the systemic concentrations decreased over time with continued administration. There is an indeterminate terminal elimination half-life (t1/2) due to the low systemic concentrations. 13
In the phase IIa maximal use study (NCT04042103) of patients with PGA ≥3 and BSA involvement ≥20% with once daily use of 1% tapinarof cream, systemic absorption of tapinarof was low, with systemic concentrations highest on day 1 and approximately tenfold lower on day 29 than on day 1. The mean Cmax on day 1 was 898 pg/mL and 116 pg/mL on day 29 following application of 1% creams. Levels were similarly low with application of the 2% cream. The reason for this decreased systemic exposure may be improved skin barrier function although there was no relationship between tapinarof exposure and %BSA. The Tmax was between 2 and 5 hours after receipt of dose. 14
Tapinarof is metabolized through oxidation, sulfation, and glucuronidation in the liver. 13 Cytochrome P450 enzymes (CYPs) are essential for drug metabolism and are heme-containing molecules that are bound to the mitochondria or endoplasmic reticulum. The enzymes CYP1A2 and CYP3A4 are the major enzymes responsible for oxidation of tapinarof in the hepatocytes although multiple other CYPs are involved. 9
Use in select populations
The available data on 1% tapinarof cream is insufficient to evaluate the safety in pregnant women. The safety and efficacy of 1% tapinarof cream has not been established in patients with psoriasis who are younger than 18 years. 15 In the phase III PSOARING 1 and 2 clinical trials, 15.5% of patients were ≥65 years old while 1.2% were ≥75 years old. There were no differences in efficacy or safety between elderly and younger subjects. 15 Cardiovascular effects were investigated by the mean change from baseline in QT interval corrected for heart rate using Fridericia’s formula (ΔQTcF), which was negative (ranging from −7.2 ms to −1.1 ms) at all time points on day 1 (the day with the higher Cmax). 14 In total, 3 patients had a QTcF ranging from 450 to 480, and none had a QTcF >480 ms. Therefore, there was no evidence of QT interval prolongation or other electrocardiogram parameters affected. 14
Summary of Clinical Trials
Phase II trial
A phase II, randomized, double-blind, vehicle-controlled trial (NCT02564042) was conducted to assess the efficacy and safety of tapinarof at 2 concentrations and 2 application frequencies. Inclusion criteria included age between 18 and 65 years, stable disease for ≥6 months prior to the study onset, body surface involvement ≥1% and ≤15% (excluding the scalp), and a baseline PGA score ≥2. Exclusion criteria included history of serious illness, signs of infection of psoriasis lesions, and use of the treatments including the following: UV light therapy within 4 weeks prior to the study visit; biologic agents or systemic immunosuppressive within 12 weeks or 5 half-lives; and topical treatments within the 2 weeks prior to treatment. A total of 227 adults were enrolled. The primary endpoint was a PGA score of 0 (clear) or 1 (almost clear) at week 12 with a 2-point improvement from baseline PGA.
A total of 175 patients completed the 12-week study. Treatment success based on PGA was higher in the 1% tapinarof groups (65% for twice daily and 56% for once daily) than that in the 0.5% groups (46% and 36%, respectively) and the vehicle groups (11% and 5%, respectively; Table 1). 16 From week 8 and later, the PASI-50 and PASI-75 response rates were significantly higher in all tapinarof groups (PASI-50: 71%-92%, PASI-75: 46%-65%) than those in all vehicle groups (10%-32% and 5%-16%, respectively), while the PASI-90 response rates were higher in the once- and twice-daily 1% and twice-daily 0.5% creams (18%-40% vs 0%). 17 The mean reductions in percent of affected BSA from baseline to week 12 were 3.6% to 4.9% in the tapinarof groups and 1% to 1.6% in the vehicle groups. 16
Efficacy Results from Phase II and III Clinical Trials of Tapinarof.
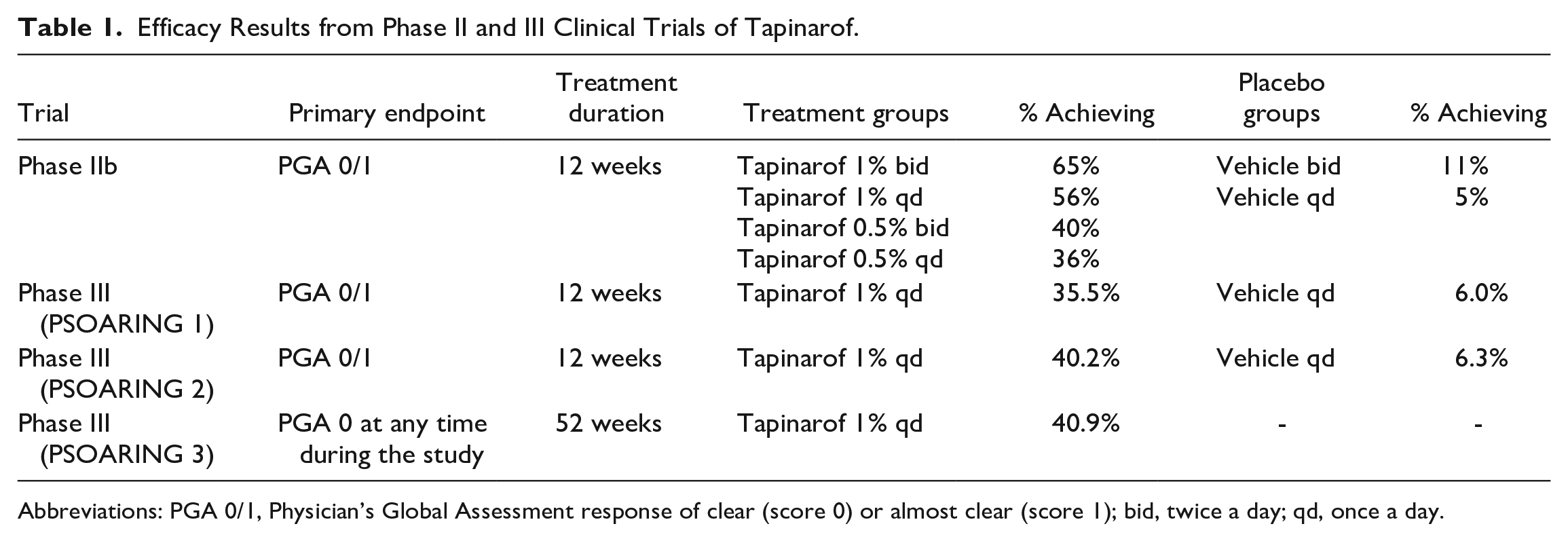
Abbreviations: PGA 0/1, Physician’s Global Assessment response of clear (score 0) or almost clear (score 1); bid, twice a day; qd, once a day.
Adverse events occurred in 53% of participants in the once-daily tapinarof 1% group compared with 29% in the once-daily vehicle group (Table 2). There were 8 instances of serious adverse events (alcoholic pancreatitis, dehydration, malignant melanoma [not at the application site], hemolytic uremic syndrome, coronary artery disease, enlarged uvula, acute cardiac failure, and atrial fibrillation). These occurred in 7 patients, all of whom were in the tapinarof groups; none were determined by the study investigators to be related to study treatments. There was a 13% discontinuation rate in the once-daily 1% tapinarof group and a 3% discontinuation rate in the once-daily vehicle group. Adverse events led to discontinuation in 7% of total participants: 10% in the tapinarof group and 1% in the vehicle group. Although the improvements in PGA, BSA, and PASI-75 were all higher with twice-daily administration of 1% tapinarof cream than those with once-daily administration, the authors concluded that once-daily administration would provide clinical improvement and encourage patient compliance. 16
Overview of the Safety Data of Phase II and III Trials for Tapinarof.

An AE is defined as any untoward medical occurrence in a patient under clinical investigation, temporarily associated with the use of a therapeutic product, whether or not considered related to the therapeutic product.
Abbreviations: AE, adverse event; bid, twice a day; no., number; qd, once a day; TEAE, treatment-emergent adverse event.
A TEAE is defined as an AE which occurred on or after study treatment start date and on or before the last visit.
Most frequent adverse events were defined by the investigators to occur in ≥2% of patients in any group.
Phase III trials
Two phase III, double-blind, multicenter, vehicle-controlled, randomized trials PSOARING 1 (NCT03956355) and PSOARING 2 (NCT03983980) evaluated the efficacy and safety of tapinarof for adult patients with moderate-to-severe plaque psoriasis. Participants were adults in the age range 18 to 75 years with a baseline PGA score between 2 (mild) and 4 (severe) and a percent BSA between 3% and 20% (not including scalp, palms, soles, fingernails, or toenails). Additional inclusion criteria included having stable disease for ≥6 months prior to the study onset. Exclusion criteria included psoriasis other than plaque variant; any sign of infection of psoriasis lesions; and concurrent conditions including other inflammatory skin conditions, chronic or acute infection, use of UV light therapy, systemic immunosuppressants, or topical psoriasis treatments within the 4 weeks prior to baseline visit and use of biologic agents within 5 half-lives of the agent. In trial 1, 510 patients were enrolled, and 515 were enrolled in trial 2; each group randomly assigned in a 2:1 ratio to receive tapinarof 1% cream or the vehicle control once a day for 12 weeks. The primary endpoint for efficacy was defined as both a PGA score of 0 (clear) or 1 (almost clear) and a decrease in the PGA of at least 2 points at week 12. After the initial 12 weeks, patients were able to enroll in a long-term extension trial PSOARING 3 (NCT04053387) or to opt at week 16 to complete a follow-up visit. A total of 763 participants were included in PSOARING 3. 18
A total of 399 patients completed the study in trial 1 (rates of discontinuation were 20.9% of the tapinarof group and 23.5% of the vehicle group), while 424 patients completed the study in trial 2 (17.8% and 17.4% discontinued in the tapinarof and vehicle groups, respectively). At baseline, 79.2% of patients in trial 1 and 83.9% of patients in trial 2 had a PGA of 3 (moderate). Baseline mean PASI for participants was 8.9 in trial 1 and 9.1 in trial 2, and the initial mean BSA affected by psoriasis of the 2 groups was 7.9% and 7.6%, respectively. In trial 1, 35.4% of patients on tapinarof and 6.0% of subjects on vehicle achieved the primary endpoint (a PGA score of 0 to 1 with ≥2-point improvement), compared with 40.2% and 6.3% in the treatment and vehicle groups, respectively, in trial 2 (P < 0.0001 for both). The PASI-75 responses at week 12 were 36.1% and 47.6% in trials 1 and 2, respectively, compared with 10.2% and 6.9% in the controls (P < 0.001 for both). A total of 37.8% of patients in the tapinarof group and 9.9% in the vehicle group in trial 1 had a PGA score of 0 or 1 at week 12. In trial 2, the corresponding values were 43.6% and 8.1% (P < 0.001 for both). There was a mean change in involved BSA from baseline of −3.5 percentage points and −4.2 percentage points in the tapinarof groups in trials 1 and 2, respectively, (compared with −0.2% and 0.1% in the vehicle groups; P < 0.001 for both). The time to initial clinical improvement was not reported. 18
Trials 1 and 2 also assessed PROs using the PP-NRS, DLQI, and the Psoriasis Symptom Diary scores. The PP-NRS score had a −3.6 mean change from baseline to week 12 in the tapinarof group compared with −2.7 in the vehicle group in trial 1. The corresponding values in trial 2 were −3.0 and −1.4, respectively. The DLQI mean change from baseline was −4.6 in the tapinarof group and −2.8 in the vehicle group for trial 1. The corresponding values for trial 2 were −4.4 and −1.1. The Psoriasis Symptom Diary scores at week 12 were −48.5 in the tapinarof group and −34.0 in the vehicle group, compared with −42.9 and −18.8 for trial 2, respectively. All PRO measures had a greater improvement in the treatment group than in the placebo group; however, these studies did not have a prespecified plan for adjustment of confidence intervals or imputation of missing data of these endpoints. These data were not adjusted for missing data, and therefore, no conclusions can be drawn. 18
Tapinarof was overall well tolerated with a favorable safety profile (Table 2). Common adverse reactions were folliculitis, nasopharyngitis, and contact dermatitis. There were no serious adverse events in either study considered to be related to tapinarof or to the vehicle. There was 1 severe adverse event of folliculitis in the tapinarof group in PSOARING 1, 1 serious adverse event of contact dermatitis in the tapinarof group of PSOARING 2, and 1 serious adverse event of headache in the tapinarof group of PSOARING 2 that occurred 30 days after the completion of tapinarof therapy.
Longer-term 52-week trial
A 52-week, multicenter, open-label, noncomparative, phase III trial PSOARING 3 (NCT04053387) evaluated the efficacy and safety of 1% tapinarof cream in patients with chronic plaque psoriasis. Criteria for inclusion included age between 18 and 75 years, BSA ≥3% and ≤20%, and completion of the 12-week PSOARING 1 or 2 pivotal studies. Exclusion criteria included the use of a prohibited product or procedure during the study (see exclusion criteria in the phase III trials), occurrence of a serious adverse event that led to treatment discontinuation during the pivotal study, and history of ongoing serious illness, medical, physical, or psychiatric conditions. Of 833 eligible patients from PSOARING 1 and 2, 763 patients elected to be enrolled in PSOARING 3 (91.6%). Patients had a mean percent BSA of 4.7% and a mean PASI of 4.8. Treatment was based on the PGA score. Patients with PGA greater than 1 (almost clear) received tapinarof until PGA = 0 (clear), while patients with PGA = 0 were discontinued from tapinarof and monitored. Treatment success was defined with the same endpoint as the PSOARING trials, with a PGA of 0 or 1 and a 2-point or more improvement from the patient’s baseline score. 19 Complete disease clearance (PGA = 0) occurred in 40.9% of participants at least once during the trial. Of patients who entered PSOARING 3 with PGA ≥ 1, 34.3% achieved PGA = 0 at least once, while 58.2% of patients who entered PSOARING 3 with PGA ≥ 2 achieved PGA = 0 one or more times. For patients with a PGA of 0 at any point during PSOARING 3, the average total duration of disease clearance once treatment was discontinued was around 130 days.
Tapinarof was well tolerated with a similar safety profile as in PSOARING 1 and PSOARING 2 (Table 1), and over 90% of patients had no irritation at any of their visits during the 40-week study. The most common adverse events were folliculitis (occurred in 22.7%), contact dermatitis (occurred in 5.5%), and upper respiratory tract infection (occurred in 4.7%), and neither the incidence nor the severity increased with long-term treatment with tapinarof (Table 2). There were no severe events reported. The rates of discontinuation due to adverse events were low, at 1.2% for folliculitis and 1.4% for contact dermatitis. 19
Of the 763 patients who enrolled in PSOARING 3, 599 completed a patient satisfaction questionnaire. Of these patients, 85.8% reported that they could easily manage their psoriasis with tapinarof, and 62.9% of patients indicated agreeing or strongly agreeing that tapinarof cleared their skin and prevented a recurrence of their psoriasis. 20
Clinical trials of benvitimod 1% cream
Tapinarof cream 1% and benvitimod cream 1% (WBI-1001) both have the same active pharmaceutical ingredient, 3,5-dihydroxy-4-isopropyl-transstilbene. 21 Benvitimod 1% cream was developed and approved for use in China in 2019 for twice-daily treatment of mild-to-moderate plaque psoriasis. A phase I trial investigated the maximum tolerated dose and pharmacokinetic characteristics in 56 healthy subjects. Participants received benvitimod cream (0.5%-2.0%) or placebo once or twice daily, and doses were increased from 5 to 30 mg daily. The study drug was well tolerated, and all adverse events were mild. There were no observable dose effects and no electrocardiographic or clinical laboratory changes, and the study medication was detected in fewer than 5% of the plasma samples. 22
Benvitimod is a twice-daily compound that was studied in 2 randomized, double-blind phase II trials in China and Canada. In the Chinese trial, 365 patients were randomized, and 52.5% of patients in the benvitimod group had a 75% reduction in PASI score at the end of treatment. 21 In the Canadian trial, 61 patients were randomized (2:1) to receive either WBI-1001 1% cream or placebo twice daily for 12 weeks. Of these, 50.0% of patients had a 75% reduction in PASI score, and 72.5% achieved PASI 50 (P = 0.0004 and P < 0.0001, respectively). 23 Both trials had similar safety profiles. The phase III trial was conducted in 686 patients who were randomized to receive 1% benvitimod cream, 0.005% calcipotriol ointment, or vehicle twice daily for 12 weeks. Of these patients, 59 were enrolled in a long-term extension study and followed for up to 40 weeks. The primary endpoint was the PASI-75, which was achieved in 50.4% of patients in the benvitimod group, compared with 38.5% in the calcipotriol group (P < 0.05). In the 40-week extension study, the median remission period of benvitimod cream was 36 weeks. 24
Summary of clinical trials: Future studies
A search on the clinicaltrials.gov website revealed 5 current studies evaluating the efficacy and safety of topical tapinarof in pediatric patients with plaque psoriasis, 1 in intertriginous plaque psoriasis, and 3 others for tapinarof in atopic dermatitis.
Relevance to Patient Care and Clinical Practice in Comparison to Existing Drugs
In the United States, approximately 3% of the population has psoriasis. 1 Psoriasis carries an increased risk of systemic inflammatory diseases and directly impacts patients’ quality of life, contributing to depression and suicidal ideation in 5% of patients. 25 Therefore, optimized management of psoriasis is important to reduce comorbidities and impact on quality of life, as well as to clear the visible skin lesions. Tapinarof 1% cream is a novel, AhR agonist, recently approved by the FDA for the topical treatment of plaque psoriasis in adults. Tapinarof downregulates IL-17, reducing the inflammatory infiltrate characteristic of plaque psoriasis, and offers antioxidant properties and increased expression of skin barrier proteins.
In the completed clinical trials, patients treated with tapinarof had improved disease severity compared with the control group. In the phase II studies, 56% of patients achieved the primary endpoint (a PGA score of clear or almost clear and a 2-point improvement from baseline PGA). In the phase III studies, disease severity assessed by the same 2-point improvement on the PGA score was 35.4% and 40.2% at week 12 in trial 1 and 2, respectively. At the same time point, a 75% improvement in PASI occurred in 36.1% of patients in trial 1 and 47.6% of patients in trial 2, and a PASI-90 occurred in 18.8% and 20.9% in the 2 trials. The results from the benvitimod studies were comparable, with approximately 50% of patients reaching PASI-75 compared to 45% to 65% with tapinarof. Compared with topical calcineurin inhibitors and topical vitamin D analogs, tapinarof 1% cream likely has similar or reduced efficacy. 5 However, the duration of effect of tapinarof after treatment discontinuation was approximately 4 months—an important factor that is not described with other topical psoriasis treatments. In addition, patient satisfaction from a survey conducted after PSOARING 3 was favorable with 83.6% of patients satisfied about how well tapinarof worked. 20
Adverse events were frequent in the tapinarof clinical trials: Over 50% of patients in the once-daily tapinarof 1% group had adverse events. Common adverse reactions were folliculitis, headache, nasopharyngitis, and contact dermatitis. Folliculitis was the most frequently occurring adverse effect, present in around 18% to 25% of patients. Headaches were rarer, occurring in <5%; however, because systemic absorption of tapinarof is low, it is concerning that there is no clear mechanism for this systemic adverse effect. The rate of discontinuation was 13% in the phase II study, 5.8% in PSOARING 1, and 5.6% in PSOARING 2, although the rates of adverse reactions causing discontinuation in the trial were <1%, except for 2.9% with contact dermatitis and 2.8% with folliculitis. Despite these low rates, it is possible that such adverse events experienced during trials may not lead to discontinuation as they might in real-life clinical practice. Differences in the care that patients in clinical trials receive, such as more frequent follow-up appointments, study incentives, and an awareness that they are being monitored, may lead to an increased adherence to the treatment regimen that would be unrealistic in clinical practice. 26 Therefore, in clinical practice the efficacy of tapinarof may actually be lower.
Determination of which patients may be most appropriate for use of tapinarof 1% cream necessitates consideration of a variety of factors including patient preference, extent of disease, other medical problems, and previous psoriasis treatments used. Tapinarof is a cream-based topical medication. Creams are water-based, typically nongreasy, thin, and easily spread topicals. Creams are typically less messy than other vehicles such as ointments and thus, are frequently preferred by patients. 27
Among the array of treatments for psoriasis are topical corticosteroids, which have restrictions on continual use, and topical calcineurin inhibitors, which have off-label recommendation for psoriasis involving the face and intertriginous regions. Topical calcineurin inhibitors and tazarotene are associated with irritation, and tazarotene is less well tolerated than topical calcineurin inhibitors. Vitamin D analogs are frequently used as combination treatment with topical corticosteroids. 5 Phototherapy can be time-intensive and impractical for patients to access. Systemic agents such as methotrexate are frequently used for moderate to severe psoriasis, particularly if there is psoriatic arthritis. Systemic immunosuppressants often require laboratory monitoring and consideration of adverse effects and toxicity such as hepatotoxicity with methotrexate. Frequently, patients are cotreated with a topical agent and either phototherapy or a systemic medication. 28
Tapinarof may be useful for patients who have conditions aggravated by topical corticosteroids such as acne, rosacea, perioral dermatitis, and tinea or for those hesitant to use topical corticosteroids. Patients with inverse psoriasis or disease located on areas of thin skin such as the eyelids may benefit from long-term use of a steroid-sparing topical such as topical calcineurin inhibitors or tapinarof cream. Unlike tazarotene, vitamin D analogs, and topical corticosteroids, topical tapinarof does not have restrictions on chronic use or sites of application. Topical medications are frequently used adjunctive therapies for patients on phototherapy and systemic or biologic therapies. Tapinarof may be a useful long-term treatment along topical corticosteroids, phototherapy, or systemic agents. Tapinarof might be appropriate for patients who prefer a nonsteroidal agent, have difficulty tolerating topical calcineurin inhibitors, or prefer a cream-based or once-daily treatment. However, the tapinarof clinical trials prohibited the use of biologic agents (for 5 half-lives before baseline visit) and other systemic treatments, including apremilast, methotrexate, and glucocorticoids for 4 weeks before baseline visit. This inclusion criterion renders these studies inadequate to identify which patients might be most likely to benefit from this medication.
Cost may be an inhibiting factor for patients using tapinarof: The average wholesale price is $1405 for a 60-gram tube. 29 However, there is a patient assistance plan for patients who qualify depending on the type of insurance. Eligible commercially insured patients may pay between $0 and $75. Prior authorization may be required for insurance coverage of tapinarof, and patients might be required to have previously failed multiple other treatments, such as the treatments reviewed in Table 3. Although prior authorizations would reduce primary nonadherence, which is defined as failure to fill the initial prescription, secondary nonadherence, wherein the patient has the medication but does not consistently use it, may still occur. It is, therefore, possible that patients may experience similar results with tapinarof as with other topical medications. 26
Classes of Topical Medications Prescribed for Plaque Psoriasis.
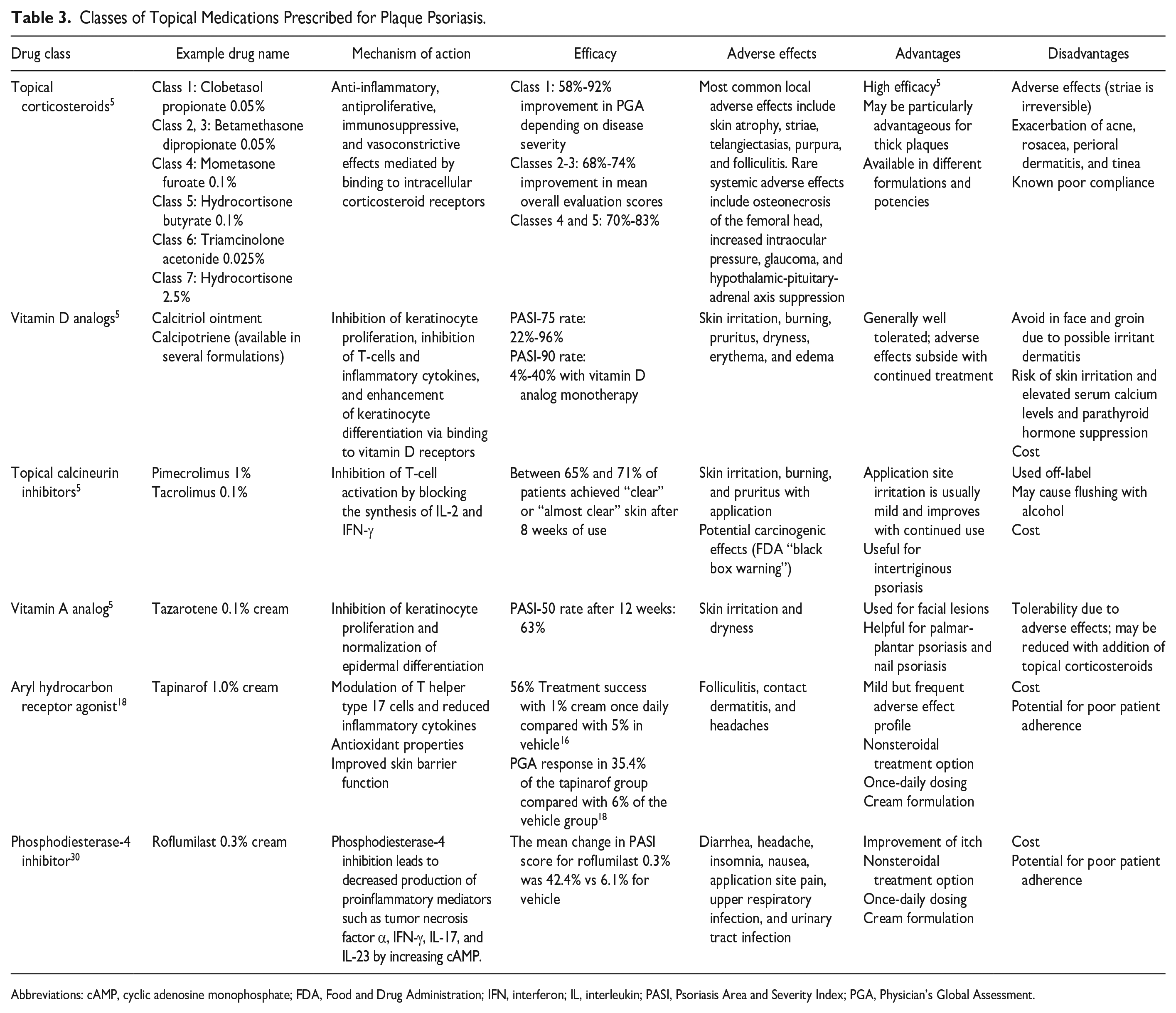
Abbreviations: cAMP, cyclic adenosine monophosphate; FDA, Food and Drug Administration; IFN, interferon; IL, interleukin; PASI, Psoriasis Area and Severity Index; PGA, Physician’s Global Assessment.
Although tapinarof 1% cream has not been compared with other topical psoriasis treatment in clinical trials, twice-daily benvitimod 1% cream, containing the same identical active pharmaceutical ingredient, was compared with calcipotriol in a phase II study in China, wherein a PASI-75 was achieved in 50.4% of patients in the benvitimod group, compared with 38.5% in the calcipotriol group. 24 This suggests that there may be higher efficacy from tapinarof than from topical vitamin D analogs.
Conclusion
Psoriasis is a common, chronic, inflammatory skin disease characterized by scaling plaques with variable morphology, distribution, and severity. Tapinarof, a first-in-class, FDA-approved topical treatment option for plaque psoriasis successfully reduced psoriasis severity after 12 weeks of treatment and had disease clearance for up approximately 4 months after treatment discontinuation. Limitations to treatment with tapinarof include high cost, as well as the potential for poor patient adherence that is inherent with all topical treatments. Although PSOARING 1 and 2 and PSOARING 3 used patient satisfaction questionnaires, trials 1 and 2 were not able to draw conclusions from the PRO tools. Limitations of this review include a lack of head-to-head trials with other psoriasis treatments. Future clinical trials are needed to elucidate the mechanism of headache in patients treated with 1% tapinarof cream; to evaluate treatment in patients previously or currently treated with other topicals, phototherapy, and systemic agents; and to compare tapinarof 1% cream with other psoriasis treatments. Patients with psoriasis involving regions with thin skin such as the eyelids or who have difficulty tolerating other psoriasis treatments may benefit from use of tapinarof 1% cream.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Feldman has received research, speaking, and/or consulting support from Eli Lilly and Company, GlaxoSmithKline/Stiefel, AbbVie, Janssen, Alovtech, vTv Therapeutics, Bristol-Myers Squibb, Samsung, Pfizer, Boehringer Ingelheim, Amgen, Dermavant, Arcutis, Novartis, Novan, UCB, Helsinn, Sun Pharma, Almirall, Galderma, Leo Pharma, Mylan, Celgene, Ortho Dermatology, Menlo, Merck & Co, Qurient, Forte, Arena, Biocon, Accordant, Argenx, Sanofi, Regeneron, the National Biological Corporation, Caremark, Teladoc, BMS, Ono, Micreos, Eurofins, Informa, UpToDate, and the National Psoriasis Foundation. He is a founder and part owner of Causa Research and holds stock in Sensal Health. Other authors declare no conflicts of interests.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
