Abstract
Tapinarof is a novel topical formulation approved recently, in May 2022, by the United States Food and Drug Administration to treat plaque psoriasis. Existing topical therapies for psoriasis are limited by systemic and local adverse effects, medication cost and repeated administration, thus significantly hampering the compliance of patients to therapy. These limitations can be resolved by tapinarof owing to its better efficacy and favourable safety profile in psoriasis management. Tapinarof was developed with a unique mechanism targeting the aryl hydrocarbon receptor (AhR) involved in inflammation and modulation of skin barrier integrity in inflammatory dermatological disorders such as psoriasis and atopic dermatitis. The efficacy and safety outcomes of tapinarof in psoriasis were justified through the two pivotal clinical trials, namely, PSOARING 1 and PSOARING 2. The common adverse effects observed with tapinarof are folliculitis, contact dermatitis and headache. The literature search was conducted for efficacy and safety of tapinarof in the electronic databases of PubMed and Cochrane using a combination of keywords such as tapinarof, psoriasis and AhR. This review will delineate the molecular mechanisms underlying the action of tapinarof and also summarise the trial data supporting the claim that tapinarof is replacing the existing standard of care in psoriasis management.
Introduction
Psoriasis is often considered a dermatologic autoimmune disorder but is also associated with cardiovascular impairment, inflammation of the joints and psychiatric disturbances. It affects nearly 7.5 million (3%) adults in the United States, with 70.9% and 27.3% having mild to moderate and moderate to severe psoriasis, respectively. 1 Currently, biological agents are often employed for moderate to severe psoriasis. The Food and Drug Administration approved existing topical drugs for the treatment of plaque psoriasis include corticosteroids like clobetasol propionate, halobetasol propionate, retinoids (tazarotene) and vitamin D analogues such as calcitriol. The clinical applications of these agents are restricted by various factors, such as the insufficient resolution of moderate to severe lesions, inadequate improvement of quality of life, serious application site reactions and specific reactions such as vitamin D-induced hypercalcemia and steroid-induced hyperglycaemia. Low compliance rates due to expensive therapy and an increased frequency of administration are the challenges faced in advocating topical therapies. Topical drugs with better efficacy, limited side effects and sustained duration of action are often warranted by psoriasis patients.
Tapinarof cream (proprietary name—VTAMA) was approved by the United States Food and Drug Administration (US FDA) on May 23, 2022, for the treatment of plaque psoriasis in adults. 2 This drug was developed by Dermavant Sciences, a company based in the United States. This review will discuss the molecular mechanisms, clinical trials, pharmacokinetics and adverse effects of tapinarof.
Search Strategy and Data Collection
Articles from PubMed/MEDLINE and Cochrane clinical databases were collected using search terms like tapinarof, psoriasis and aryl hydrocarbon receptor (AhR). Articles elucidating the role of tapinarof in psoriasis and other skin disorders were considered for the review. The US FDA webpage was used to obtain the prescribing information and data on clinical pharmacology from tapinarof drug label. The United States National Library of Medicine Registry, and ClinicalTrials.gov, were also used for obtaining the results of clinical trials.
Molecular Mechanism
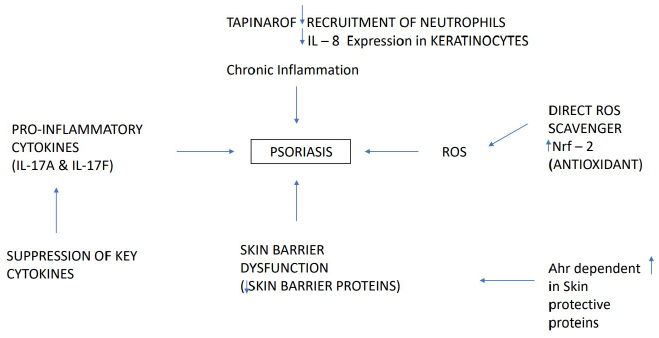
Tapinarof is chemically termed as 5-[(E)-2-phenylethenyl]-2-[propan-2-yl]benzene-1,3-diol), a synthetic stilbene molecule with a hydroxyl group. 3 It is a relatively novel prototypic topical agent that acts as an AhR modulator and antioxidant and is employed in the treatment of psoriasis and atopic dermatitis. The AhR plays a role in the development of normal epidermal barriers and regulates inflammation through IL-10, IL-21 and IL-22 expressions.4, 5
The AhR is a ligand-dependent transcriptional factor that is expressed widely in keratinocytes, regulating differentiation of dermal cells and maintenance of skin barrier homeostasis. Upon ligand binding, this cytoplasmic receptor translocates into the nucleus and undergoes dimerisation with the AhR-Nuclear Translocator (ARNT). This cascade activates the transcription of various cytochrome P450 genes and genes for antioxidant enzymes. 6 The AhR pathway requires the aid of skin barrier proteins such as filaggrin and involucrin to maintain skin barrier integrity. Psoriasis is mainly characterised by abnormal differentiation and hyperproliferation of keratinocytes and impairment of the skin barrier by downregulation of skin barrier protective proteins. Coal tar is one of the age-old regimens in psoriasis, found to target AhR in a non-specific manner but limited by its unpleasant odour and tissue staining property. It also exacerbates pustular psoriasis. Hence, new molecules modulating AhR can be used in the management of autoimmune dermatologic disorders like psoriasis and atopic dermatitis.7–10
Tapinarof: The First Therapeutic Aryl Hydrocarbon Receptor Modulating Agent (TAMA) in Psoriasis
Tapinarof activates AhR owing to a reduction in the levels of pro-inflammatory cytokines such as IL-17. It also regulates the expression of skin barrier proteins like filaggrin, which promotes the normalisation of the skin barrier. By activating the nuclear factor erythroid 2-related factor 2 (Nrf-2) pathway, it also has antioxidant activity. Tapinarof can be termed as a novel agent in the myriad of therapeutic options due to the restrictions imposed by existing topical medications for psoriasis in terms of difficult to apply areas, duration of use and increased adverse effects that can interfere with patient adherence further impairing the quality of life in a patient with psoriasis.11–16 AhR activation induces epidermal differentiation and hyperproliferation of keratinocytes, leading to adverse effects like chloracne with compounds like coal tar and dioxins. Similarly, tapinarof-induced folliculitis could also be attributed to the stimulation of epidermal differentiation via AhR. 15 The molecular mechanism of tapinarof is depicted in Figure 1.
Preclinical Data
Tapinarof was shown to bind and activate AhR in various cell types apart from keratinocytes and also reduced the expression of pro-inflammatory cytokines and inhibited gene expression for skin barrier proteins. The binding of tapinarof to the target receptor was confirmed by its affinity towards ARNT–Per-Arnt-Sim domains in human and mouse models. The antioxidant property of tapinarof was demonstrated with a decrease in reactive oxygen species generation in oxidative stress-induced keratinocytes. The anti-inflammatory property of tapinarof was predominantly expressed through the decline in the level of pro-inflammatory cytokines and markers of skin inflammation such as redness and epidermal thickening in imiquimod-induced psoriasis and lipopolysaccharide-induced ear oedema mice models. A skin sensitisation study with 8% tapinarof cream in guinea pigs did not induce any adverse skin irritation after 6 hours of application. Topical photosensitivity testing of tapinarof cream performed using ultraviolet radiation in naked mice models declared the absence of signs of phototoxicity with this drug. 17
Clinical Trials
Phase 1: Clinical Trial
Bissonnette et al. 18 conducted an open-label sequential phase 1 study (Study 201851) with tapinarof cream (1% and 2%) twice a day dosing among patients with moderate-to-severe atopic dermatitis to determine the systemic exposure, safety and efficacy. Participants were admitted to the trial site on the 1st and 21st days for pharmacokinetic evaluation. Participants also had eight trial visits for laboratory evaluation and clinical examination on 1, 2, 3, 4, 7, 14, 21 and 22 days. A total of 11 subjects with an initial Investigator’s Global Assessment (IGA) score ≥ 3 were recruited; five of them were allocated to receive 2% cream and six were administered 1% cream. Study medications were applied for 21 days [20 days of twice daily (BD) dosing and 21st day of once daily (OD) dosing] all over the affected body areas except the scalp and periorbital region. Plasma samples were collected at different timepoints such as 0, 1, 2, 4, 8, 10 and 12 hours for determining pharmacokinetic parameters. Steady-state samples were collected on days 2, 3, 4, 7, 14 and 21 before drug application. The mean Cmax for 2% and 1% cohorts were 2.5 ng/ml and 1.2 ng/ml on day 1 and 0.3 ng/ml and 0.15 ng/ml on day 21, respectively. The decrease in plasma concentration on repeated application represents the absence of drug accumulation and restoration of the epidermal barrier halting the drug absorption over a period. The median Tmax of 2% and 1% cream were 2 hours and 3 hours on day 1, respectively. The median Tmax on day 21 was 4 hours and 0.66 hours in 2% and 1% cohorts, respectively. Mean AUC0–24 with 2% and 1% cream on day 1 were 23.4 ng.h/ml and 10.5 ng.h/ml, respectively. Tapinarof concentration was found to be higher in the 2% cohort. The common adverse events observed were headache, folliculitis, diarrhoea, nausea and vomiting. Adverse events (AE) resolved within 24 hours of the cessation of treatment. Side effects are more frequently encountered with 2% cream than with 1% formulation. Progressive improvement in efficacy outcomes such as a 50% reduction in eczema area and severity index (EASI), IGA, pruritus and body surface area (BSA) were documented with both concentrations of tapinarof over 21 days. The maximum reduction of pruritus score from baseline 6.3 to 1.3 was on day 7 and percentage of BSA was affected by 15% on day 21 in the 1% cream cohort. These efficacy parameters were observed concurrently and strongly confirmed the systemic absorption and pharmacokinetic effects. From this study, it was declared that 1% cream achieved an efficacy threshold similar to that of 2% cream with a better safety profile and patient tolerability. The limited sample size and open-label study design were the limitations of this study. 18
Phase 2: Clinical Trial
A phase 2 randomised double-blinded clinical trial of tapinarof cream (GSK2894512 cream) with vehicle control was conducted to evaluate its safety and efficacy among adult patients with plaque psoriasis at 40 sites in Canada, Japan and the United States by Robbins et al. Tapinarof was administered as either 0.5% or 1% tapinarof cream for 12 weeks. Patients were allocated into 6 groups, such as twice daily 1% tapinarof, once daily 1% cream, twice daily 0.5% cream, once daily 0.5% cream, vehicle twice daily and vehicle once daily. Patients with BSA involvement of 1–15% and a Physician Global Assessment (PGA) score > 2 were considered for study inclusion. The primary efficacy endpoints are the percentage of patients with PGA 0 or 1 and an improvement in PGA scores of at least 2-grade points at the end of the treatment phase. The percentage of patients reporting improvement in the Psoriasis Area and Severity Index (PASI) was represented as a secondary endpoint. The study period was divided into three phases, comprising a screening phase (4 weeks), a treatment phase (12 weeks) and a follow-up phase (4 weeks). Patients visited the trial during screening and treatment periods on weeks 1, 2, 4, 8 and 12 and made two visits during the 2nd and 4th weeks of the follow-up phase. In this study, out of 290 screened patients, 227 participants were randomised and 196 from the modified intent to treat study population were included for analysis. At 12 weeks, patients on tapinarof showed significantly higher PGA response rates such as 65%, 56%, 46% and 36% with 1% BD, 1% OD, 0.5% BD and 0.5% OD treatments, respectively, compared to vehicle groups (11% in BD and 5% in OD groups). Tapinarof 1% cream produced the highest improvement in PGA score. Tapinarof patients also exhibited a significantly higher 75% improvement in PASI (65% and 56% after BD and OD applications) than the vehicle group. The onset of effect started at 2 weeks and persisted for 4 weeks after the cessation of treatment, denoting the sustained action of tapinarof. The decrease in the percentage of BSA affected in tapinarof and vehicle groups were 3.6–4.9% and 1–1.6%, respectively. The most common treatment-related adverse effects in tapinarof groups were folliculitis (10%), contact dermatitis (3%), local irritation, allergic dermatitis and headache (1%). Serious adverse effects noted were heart failure, coronary vascular disease and haemolytic uremic syndrome but unrelated to tapinarof. Treatment-related discontinuation was observed in 10% of tapinarof patients versus 1% in the vehicle group. Contact dermatitis was the major cause of permanent discontinuation. 19
Phase 3: Clinical Trial
PSOARING 1 (NCT03956355) & PSOARING 2 (NCT03983980) Trials
The outline of PSOARING 1 and PSOARING 2 trials is presented in Figure 2. Two randomised vehicle-controlled identical phase 3 clinical trials were conducted to assess the efficacy and safety of tapinarof 1% cream at 97 sites in North American countries with an allocation ratio of 2:1 among plaque psoriasis patients between 18 and 75 years of age. Adult patients with stable disease involving 3–20% BSA and PGA scores of 2, 3 and 4 were recruited for the study and stratified based on the PGA score. The trial drug was applied once daily to the affected areas as well as the sites of new lesions and lesions that disappeared during the trial. Drugs like methotrexate and corticosteroids were withheld for 4 weeks. The primary efficacy end-points were a PGA score of 0 or 1 and a reduction of 2 or more points in the PGA score during the 12th week. Secondary outcomes assessed were the proportion of patients with a 75% reduction in Psoriasis and Area Severity Index (PASI 75) score, the average decrease in total BSA percentage, and the proportion of patients with a 90% reduction in PASI (PASI 90) at 12 weeks since baseline. Peak Pruritus Numeric Rating Scale score, Dermatology Life Quality Index total score and Psoriasis Symptom Diary score were also directly collected from patients. Safety data were obtained from patient interviews, physical findings and laboratory values. These trials were conducted between March 2019 and May 2020. In trial 1, 674 patients were screened, 510 were randomised and 340 received tapinarof, and in trial 2, out of 692 screened patients, 515 and 343 patients were randomised and allocated to tapinarof. In trial 1, the mean percentage of patients who achieved PGA scores in tapinarof and vehicle groups were 35.4 and 6, respectively, and 40.2% and 6.3% were the proportion of patients with target PGA in tapinarof and vehicle groups, respectively, in trial 2. All the secondary endpoints showed a significant difference between the tapinarof and vehicle groups. In trials 1 and 2, the proportion of adverse events in tapinarof groups was 50.3% and 54.5% versus 22.4% and 26.2% in the vehicle group respectively. The adverse drug reactions observed during PSOARING 1 and PSOARING 2 clinical trials among 1,025 adult plaque psoriasis patients were folliculitis (20%), nasopharyngitis (11%), contact dermatitis (7%), headache (4%), pruritus (3%), influenza (2%), urticaria (1%) and drug eruption (0.9%). Folliculitis and contact dermatitis lead to treatment discontinuation in more than 1% of participants. Tapinarof 1% cream was found to be better than the vehicle group in both trials. 20
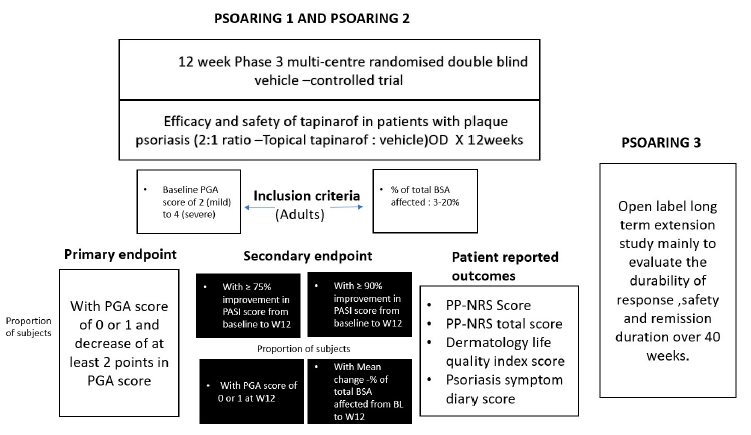
PSOARING 3
In this open-label study, patients with mild-to-moderate psoriasis were administered tapinarof 1% cream for 40 weeks followed by 4 weeks off-treatment to determine the efficacy, safety, status of remission and tolerability of tapinarof. This study was conducted in continuation with PSOARING 1 and 2 with tapinarof 1% cream for 2 weeks. Nearly 763 patients were recruited from PSOARING 1 and 2 trials, with the majority having PGA scores of 1 (32.4%) and 2 (32.6%), average BSA of 4.7% and PASI of 4.8. The efficacy data declared complete disease control in 40.9% (312/763) of participants. The mean (SD) duration of sustainable therapeutic effect after treatment discontinuation (remittive effect) in patients with PGA 0 was determined as 130.1 (89.4) days. Tapinarof therapy for 52 weeks did not demonstrate any signs of tachyphylaxis, unlike corticosteroids and biologics therapy. Similar to PSOARING 1 and 2, mild folliculitis (22.7%) and contact dermatitis (5.5%) were the most common AE leading to treatment discontinuation in 1.4% and 1.2% of patients, respectively, in this trial. Thus, this study concluded that tapinarof 1% cream once daily application was found to be safe and effective for 52 weeks with significant remittive effect for up to 4 weeks in mild-to-severe psoriasis. The limitations of this study were open-label design with a lack of a control group, leading to diminished external validity. 21
Formulation-specific Clinical Studies
Skin residency studies were conducted to determine the residency time, safety and tolerability of tapinarof cream (1%) in healthy volunteers after 7 days of once-daily application. Skin biopsies were used for pharmacokinetic analysis of concentration. Tapinarof was found in samples collected 48 hours after dosing, unlike in preclinical studies that showed extended residency time. 17
The maximal use study conducted among adult plaque psoriasis patients with tapinarof 1% cream for 29 days revealed that the plasma concentration was lower than 50 pg/ml (below the lower limit of quantification) throughout the treatment period and did not cause a significant change in QT interval. A skin irritation study with tapinarof 1% cream in healthy Japanese subjects also reported a systemic concentration of less than 40 pg/ml. 17
Dosage and Administration
Tapinarof cream (proprietary name—VTAMA) is available as a 1% cream and administered once daily over the skin lesions. It should not be used for oral, vaginal and ophthalmic purposes. The desired storage temperature ranges between 20°C and 25°C (permissible fluctuations of 15–30°C). 22
Pharmacokinetics
Tapinarof does not accumulate at the application site with continuous use. Plasma concentration was found to be lower than the quantifiable limit. In-vitro studies confirmed 99% plasma protein binding and metabolism in the liver, mainly by oxidation, sulfation and glucuronide conjugation reactions. Tapinarof does not influence the activity of cytochrome P450 enzymes and transporters. 22
Adverse Effects
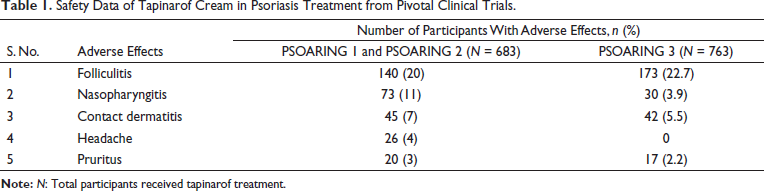
The most common adverse effects of tapinarof cream are summarised in Table 1 based on results retrieved from phase 3 clinical trials such as PSOARING 1, 2 and 3.20, 21
Safety Data of Tapinarof Cream in Psoriasis Treatment from Pivotal Clinical Trials.
Considering Tapinarof in Special Population 22
Pregnancy
Tapinarof safety in pregnancy has not been fully explored to rule out the risk of congenital anomalies, abortion or other harmful effects on the foetus and mother. During preclinical reproductive toxicity studies in pregnant rabbits and rats, no significant teratogenic effects were noted at doses higher than the maximum recommended dose for humans.
Lactation
No clinical data on the excretion of tapinarof through breast milk and its negative impact on lactation in humans have been documented. Nevertheless, preclinical studies declared the presence of tapinarof in animal milk following subcutaneous administration to pregnant rats.
Paediatric Population
Tapinarof was not investigated for its efficacy and safety outcomes in paediatric psoriasis patients aged less than 18 years. Preclinical testing in juvenile rats at a dose 11 times higher than the maximum recommended human dose did not produce any adverse effects.
Elderly Patients
Based on the results of PSOARING 1 or PSOARING 2 trials, efficacy, safety or tolerability aspects of tapinarof did not differ between elderly and young adult patients.
Tapinarof in Atopic Dermatitis
The phase 2b randomised vehicle-controlled trial evaluated the efficacy of tapinarof (1% and 0.5% cream) in patients between 12 and 65 years of age with atopic dermatitis. Improvement in IGA score, EASI and numeric rating scale were considered the measures of treatment success, and tapinarof was found to be effective and safe in the study population. 23 After the successful results from the PSOARING trials, phase 3 clinical trials of tapinarof such as ADORING 1 (NCT05014568) and ADORING 2 (NCT050328659) were formulated to evaluate the efficacy, safety, degree of remission and tolerability in moderate-to-severe atopic dermatitis patients of age greater than or equal to 2 years. The Validated Investigator Global Assessment Scale for Atopic Dermatitis and EASI were the efficacy endpoints evaluated in these trials. In continuation with these trials, an open-label extension study (ADORING 3) with the same participants was initiated to establish long-term safety and remission and safety outcomes of tapinarof. These studies are ongoing. 24
The Benvitimod Controversy
In the review by Assaf et al., the two medications, namely, tapinarof and benvitimod, were attributed to being the same due to the identical active component (3,5-dihydroxy-4-isopropyl-trans-stilbene). 14 This statement was contradicted by Gold et al. 25 , who claimed that tapinarof 1% cream is exclusively licensed by Dermavant Sciences, while benvitimod 1% cream is neither associated with Dermavant Sciences nor GlaxoSmithKline. While tapinarof comprised a novel vehicle with specific excipients, benvitimod differed in its non-specific excipients and twice-daily dosing schedule. The pivotal trials for these two compounds varied with the aspect of study design and disease remission that were only observed in the phase 3 trial (PSOARING 3) with tapinarof. Though the benvitimod trial reconfirmed the biological property of the active principle, this would not substantiate the interchangeable use of these two molecules. 25
Conclusion
Tapinarof is the first-in-class TAMA approved for the treatment of plaque psoriasis. The clinical trials of tapinarof in atopic dermatitis and paediatric patients with psoriasis are currently in progress. Furthermore, data is required in terms of adverse event profile and efficacy compared to existing topical treatments in psoriasis. The question of whether tapinarof is just another cosmetically elegant product in psoriasis management or whether it will replace the standard of care in the treatment of psoriasis can be solved only with the real-world data of this fortuitous discovery. Thus, accumulating evidence suggests that tapinarof has the potential to become a key topical drug in the treatment of autoimmune skin disorders.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not applicable
