Abstract
Background:
Rivaroxaban has predictable pharmacokinetics and pharmacodynamics. However, monitoring rivaroxaban concentrations should be provided for special patients with hepatic insufficiency, high bleeding risk, and high thrombotic risk.
Objective:
This study aimed to correlate chromogenic anti-Xa assay, prothrombin time (PT), activated partial thromboplastin time (APTT), thromboelastogram reaction time (TEG R-time), and rivaroxaban concentration measured by high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) (MS-Riva).
Methods:
Peripheral venous blood was collected from recruited patients 30 minutes before and 2 to 4 hours after drug administration. High-performance liquid chromatography-tandem mass spectrometry and chromogenic anti-Xa assay measured rivaroxaban concentration. Different assays were compared by Pearson correlation coefficient and Bland-Altman analysis.
Results:
A total of 104 patients with 191 plasma were included in the study. Overall analysis shows that chromogenic anti-Xa assay, PT, APTT, and TEG R-time strongly correlated with MS-Riva (r = 0.986; r = 0.884; r = 0.741; r = 0.739; P < 0.001). Rivaroxaban peak concentration detected by HPLC-MS/MS (MS-peak) showed a very strong correlation with the chromogenic anti-Xa assay (r = 0.977, P < 0.001) and moderate correlation with PT, APTT, and TEG R-time (r = 0.670; r = 0.571; r = 0.481, P < 0.001). Rivaroxaban trough concentration detected by HPLC-MS/MS (MS-trough) correlated strongly with the chromogenic anti-Xa assay (r = 0.884, P < 0.001), weakly with APTT (r = 0.313; P = 0.043), and not significantly with PT and TEG R-time (P = 0.140; P = 0.341).
Conclusion and Relevance:
High-performance liquid chromatography-tandem mass spectrometry/MS is the preferred choice for monitoring peak and tough concentrations, followed by anti-Xa, while PT is only suitable for peak concentrations. This study can help the clinicians to better adjust the medication regimen and reduce the risk of recurrence of thrombosis as well as the risk of bleeding.
Keywords
Introduction
Rivaroxaban is a selective factor Xa inhibitor that inhibits free and bound coagulation factor Xa and prothrombin activity, thus exerting an anticoagulant effect. 1 Compared with the traditional oral anticoagulant warfarin, direct oral anticoagulants (DOACs) have the advantages of high bioavailability, rapid onset of action, wide therapeutic window, no food interactions, few drug interactions, predictable pharmacokinetic and pharmacodynamic profiles, and are clinically approved for use at fixed doses without the need for routine laboratory monitoring. 2 However, some studies have found different drug concentrations in individuals depending on compliance, age, gender, concomitant use of certain drugs, and renal function. 3 The inability to accurately predict differences in drug efficacy between individuals with rivaroxaban may exacerbate the risk of thrombosis or bleeding. 4 Therefore, rivaroxaban concentration monitoring should be performed in patients with specific conditions, such as advanced age, abnormal liver and kidney function, perioperative period, underweight or overweight, high risk of bleeding or thrombosis, gastrointestinal malnutrition, drug overdose or omission, recurrence of thrombosis, concomitant administration of drugs affecting metabolism and transport of DOACs, and irregular drug usage.5,6
High-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) is the gold standard for detecting rivaroxaban because of its high specificity, sensitivity, and selectivity. This method is used for the direct detection of concentrations of rivaroxaban. 7 Although HPLC-MS/MS is the most accurate method, long turn around times, expense and lack of availability of this test limits is applicability. 8 Chromogenic anti-Xa assays are most commonly used for clinical assessment of heparin-antithrombin or low-molecular heparin-antithrombin chromogenic anti-Xa activity and can be used for quantitative assessment.9,10 However, the activity of rivaroxaban is significantly higher than regular and low molecular heparin, so specific calibrators and controls are required. 4 One study showed a good correlation between chromogenic anti-Xa assays and HPLC-MS/MS measurements of rivaroxaban concentrations in 50 to 200 ug/L. 8 Routine clinical monitoring of anticoagulation efficacy indicators includes prothrombin time (PT), activated partial thromboplastin time (APTT), and so on. Thromboelastography (TEG) is anathor anticoagulation monitoring modality to monitor and quantify the coagulation status of whole blood samples, providing information about the kinetics of clot formation, the physical strength of the clot, and the rate of fibrinolysis.11,12 Increasing evidence suggests that altered TEG parameters can be used to assess the anticoagulant effect of oral anticoagulants.13-17 Thromboelastography reaction time (TEG R-time, the time from the placement of the blood sample into the analyzer to the start of clot formation), using kaolin as a clotting activator, is the most appropriate TEG parameter for assessing the antithrombotic effect of novel anticoagulants, and the most sensitive to the effect of rivaroxaban.18,19 However, the conclusions of different studies on the correlation between rivaroxaban blood concentrations and these coagulation indices are different.4,18,20,21
Therefore, this study aimed to correlate chromogenic anti-Xa assay, PT, APTT, TEG R-time, and rivaroxaban concentrations measured by HPLC-MS/MS (MS-Riva).
Methods
Study Design and Participants
This prospective study compared chromogenic anti-Xa assays, PT, APTT, and TEG R-time with MS-Riva. Patients were recruited between September 2018 and September 2019 at Fujian Medical University Union Hospital. Inclusion criteria were (1) age ≥ 18 years, (2) patients with atrial fibrillation (AF) or venous thromboembolism (VTE) who had been taking rivaroxaban continuously for more than 2 days, and (3) able to provide informed consent. Exclusion criteria were (1) pregnancy and (2) patients with chronic liver disease or hereditary coagulation disorders. Data collection included age, patient gender, the dose of oral rivaroxaban, indication and duration of rivaroxaban treatment, comorbidities, and concomitant medications. The study complied with the Declaration of Helsinki and was authorized by The Ethics Committee of Fujian Medical University Union Hospital (Number: 2018YF005-02, date: 2018-3-14). All patients provided informed consent.
Sample Collection
To ensure the accuracy of the results, we followed a detailed protocol for rivaroxaban intake and blood collection. Patients taking 10 mg tablets have no restrictions on whether rivaroxaban is taken with food. For tablets of 15 or 20 mg, rivaroxaban is taken with food. After patients had taken rivaroxaban (10/15/20 mg once a day) for at least 2 days, peripheral venous blood was collected in the morning within 30 minutes (min) before (trough) and 2 to 4 hours after (peak) dosing. Blood samples were collected using the aseptic technique in blue-capped polypropylene tubes (containing 0.5 mL of 3.2% sodium citrate, pH 7.4). 22 Fresh whole blood was used for the TEG R-time assay.
Sample Determination
All laboratories have implemented strict quality management guidelines, including regular internal and external quality assessments, and have thoroughly evaluated assay results. Plasma concentrations were directly detected by HPLC-MS/MS. 23 The blood samples were processed by the protein precipitation method. The method used rivaroxaban-d4 as an internal standard with a linear range of 2 to 500 ng/mL. The concentration of rivaroxaban was estimated indirectly by chromogenic anti-Xa assay. Rivaroxaban plasma samples were tested with STA-Liquid Anti-Xa Reagent (Diagnostica Stago, France), and plasma samples were calibrated with STA-Rivaroxaban Calibration Kit (Diagnostica Stago, France). According to the manufacturer’s recommendations, the procedure was performed using the STA-R EVERSION fully automated coagulation analyzer. Commercial rivaroxaban chromogenic anti-Xa assays have demonstrated good accuracy and acceptable precision. The calibration range is 25 to 500 ng/mL, with a lower limit of 25 ng/mL. Before sample analysis, each new set of reagents is used with an STA-Rivaroxaban control to ensure the validity and comparability of the assay. The PT was determined using STA-Neoplastine (Diagnostica Stago). The APTT assay was performed with STA APTT-Automate (Diagnostica Stago, Asnieres, France). Thromboelastography Hemostasis System (Haemonetics Corporation, Braintree, MA, USA) measured thrombosis parameters. After sampling, whole blood specimens were activated with kaolin clay, and calcium citrate was added to form a thrombin-induced clot. The reaction time was determined and compared with the manufacturer’s control values because a standardized kit for establishing reference ranges was used.
Statistical Methods
Statistical analyses were performed using SPSS Statistics version 22 (IBM Corporation, Armonk, NY, USA). P < 0.05 was considered statistically significant. Descriptive statistics for continuous variables are expressed as mean ± standard deviation (SD) and for discrete variables are expressed as counts or percentages. Pearson correlation analysis determined the correlation of MS-Riva with the chromogenic anti-Xa assay, PT, APTT, and TEG R-time. The Pearson coefficient (r) absolute value between 0 and 0.09 is a negligible correlation, 0.1 to 0.39 is a weak correlation, 0.4 to 0.69 is a moderate correlation, 0.7 to 0.89 is a strong correlation, and 0.9 to 1.0 is a very strong correlation. 24 Bland-Altman analysis assessed the agreement of the chromogenic anti-Xa assay, PT, APTT, TEG R-time, and MS-Riva. Bland-Altman plots were plotted with the difference between the two assays as the vertical coordinate and the mean as the horizontal coordinate. The 95% confidence interval (95%CI) is the mean ± 1.96 SD. Data that do not have comparable units are transformed by normalized values (i.e., z-transformation). 25
Result
Baseline Characteristics
A total of 104 patients (including 80 patients with AF and 24 patients with VTE) were included in this study, including 59 male patients and 45 female patients with a mean age of 65 (13) years. Of the 80 patients with AF, 51 were male, and 29 were female, with a mean age of 68 (13) years. Of the 24 patients with VTE, 8 were male, and 16 were female, with a mean age of 58 (11) years. The baseline characteristics of the patients are shown in Table 1.
Baseline Patient Characteristics.
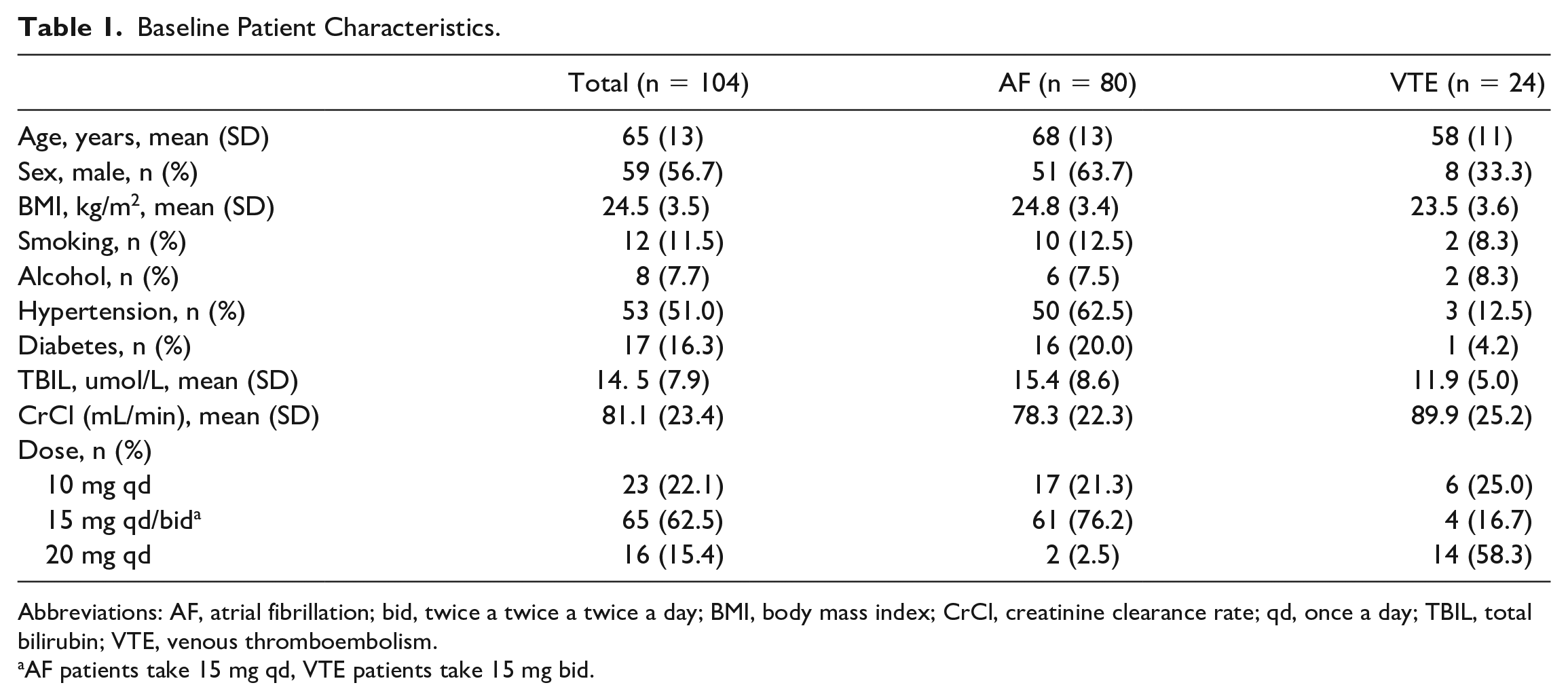
Abbreviations: AF, atrial fibrillation; bid, twice a twice a twice a day; BMI, body mass index; CrCl, creatinine clearance rate; qd, once a day; TBIL, total bilirubin; VTE, venous thromboembolism.
AF patients take 15 mg qd, VTE patients take 15 mg bid.
Total Patients
Table 2 shows the Pearson correlation of Anti-Xa assay, PT, APTT, and TEG R-time compared to MS-Riva. A total of 191 plasma samples were detected for concentration by HPLC-MS/MS and chromogenic anti-Xa assays, 104 plasma samples were used for PT, 80 plasma samples were used for APTT, and 138 whole blood samples were used for TEG R-time measurements. Figure 1 was a Bland-Altman plot of the chromogenic anti-Xa assay versus the MS-Riva. The mean difference between the two tests was 1.6 ng/mL (95% CI: −33.9, 37.1). The MS-Riva showed a significant and very strong correlation (r = 0.986, P < 0.001) with chromogenic anti-Factor Xa assay (Figure 2). The Bland-Altman plot (Figure 3) reflected the limits of agreement for PT (mean difference, 0.0; 95% CI: −0.9, 0.9), APTT (mean difference, 0.0; 95% CI: −1.1, 1.1), TEG R-time (mean difference, 0.0; 95% CI: −1.3, 1.3) and MS-Riva. There was a significant and strong correlation between PT, APTT, TEG R-time, and MS-Riva (r = 0.884, P < 0.001; r = 0.741, P < 0.001; r = 0.739, P < 0.001).
Pearson Correlation of Anti-Xa Assay, PT, APTT, TEG R-Time Compare to MS-Riva.
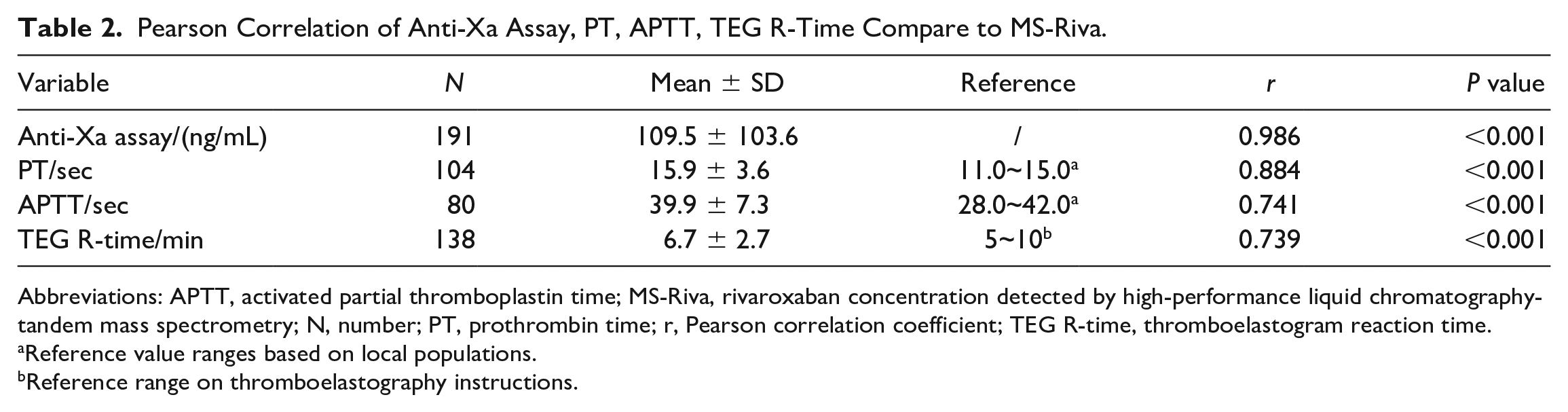
Abbreviations: APTT, activated partial thromboplastin time; MS-Riva, rivaroxaban concentration detected by high-performance liquid chromatography-tandem mass spectrometry; N, number; PT, prothrombin time; r, Pearson correlation coefficient; TEG R-time, thromboelastogram reaction time.
Reference value ranges based on local populations.
Reference range on thromboelastography instructions.
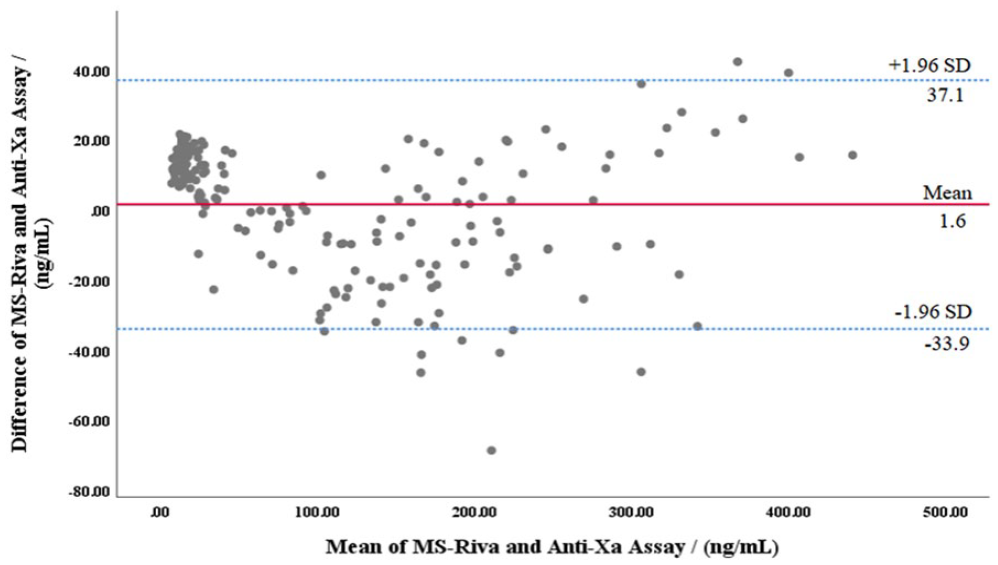
Bland-Altman analysis for agreement between MS-Riva and Anti-Xa assay.
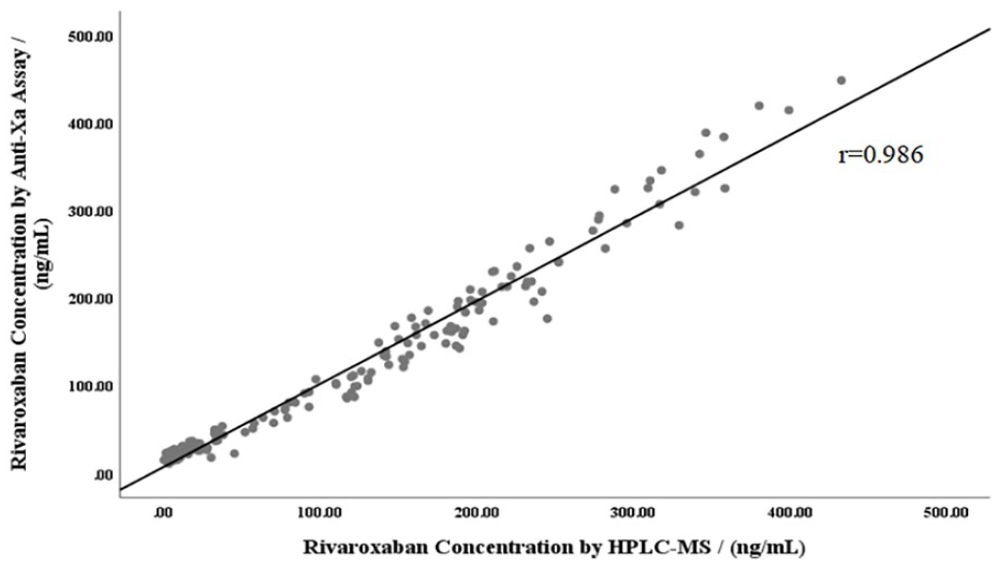
Scatter plot of rivaroxaban concentration measured by HPLC-MS/MS and anti-Xa assay.
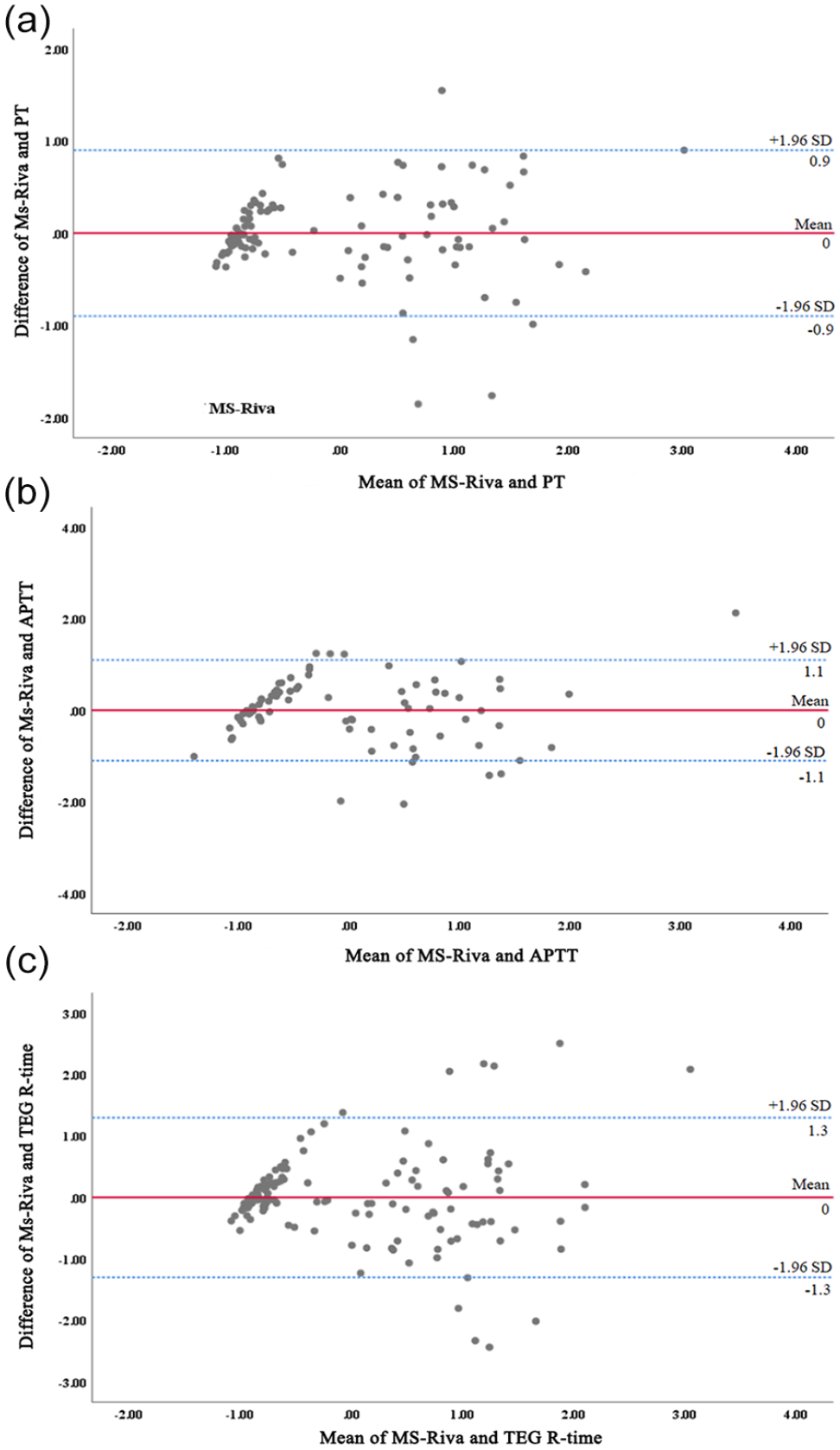
Bland-Altman analysis for agreement between (a) MS-Riva and PT. (b) MS-Riva and APTT. (c) MS-Riva and TEG R-time.
Table 3 shows the Pearson correlation of Anti-Xa assay, PT, APTT, and TEG R-time compared to MS-Riva in different ranges. The chromogenic anti-Xa assay showed no significant correlation with MS-Riva below 25.0 ng/mL (r = 0.241, P = 0.07) and a significant and very strong correlation with MS-Riva in the range of 25.0 to 448.5 ng/mL (r = 0.980, P < 0.001). Prothrombin time was significantly and moderately correlated with MS-Riva in the normal population reference range (11.0-15.0 s) and range 15.1 to 29.0 seconds (r = 0.510, P < 0.001; r = 0.654, P < 0.001). Activated partial thromboplastin time in the normal population reference range (28.0–42.0 s) and range 42.0 to 56.0 seconds had a significant moderate correlation with MS-Riva (r = 0.464, P < 0.001; r = 0.431, P = 0.025). There was a significant moderate correlation between TEG R-time and MS-Riva (r = 0.596, P < 0.001) in the normal population reference range (5-10 min), and no significant correlation with MS-Riva below 5 min and in the range of 10.1 to 17.9 min (r = 0.248, P = 0.079; r = 0.026, P = 0.923).
Pearson Correlation of Anti-Xa Assay, PT, APTT, TEG R-Time Compared to MS-Riva in Different Ranges.
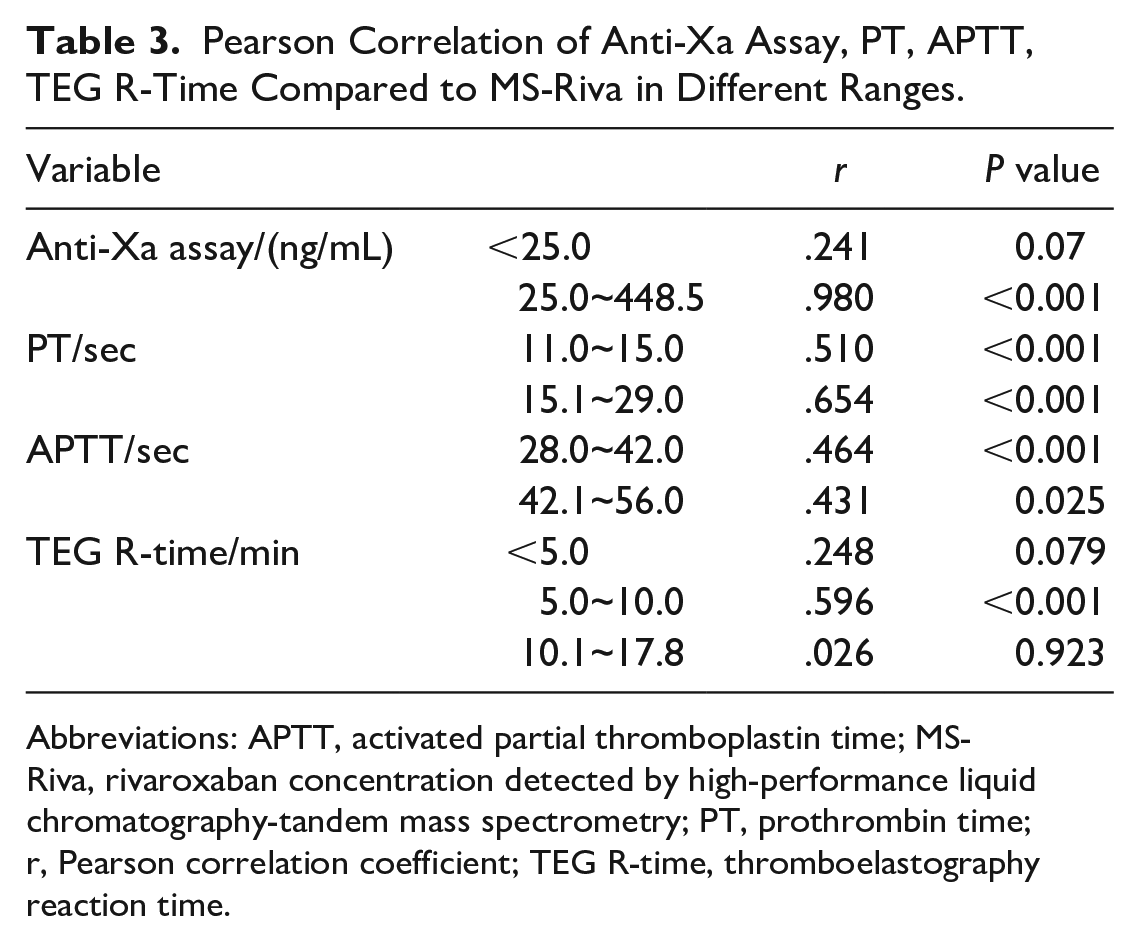
Abbreviations: APTT, activated partial thromboplastin time; MS-Riva, rivaroxaban concentration detected by high-performance liquid chromatography-tandem mass spectrometry; PT, prothrombin time; r, Pearson correlation coefficient; TEG R-time, thromboelastography reaction time.
Peak Concentration and Trough Concentration
Table 4 shows the Pearson correlation of Anti-Xa assay, PT, APTT, and TEG R-time compared to rivaroxaban peak and trough concentrations detected by HPLC-MS/MS (MS-peak; MS-trough). Mass spectrometry peak showed a significant and very strong correlation with the chromogenic anti-Xa assay (r = 0.977, P < 0.001) and a significant moderate correlation with PT, APTT, and TEG R-time (.670, P < 0.001; r = 0.571, P < 0.001; r = 0.481, P = 0.001). Mass spectrometry trough had a significantly strong correlation with the chromogenic anti-Xa assay (r = 0.884, P < 0.001), a significant weak correlation with APTT (r = 0.313; P = 0.043), and a weak and nonsignificant correlation with PT and TEG R-time (r = 0.217, P = 0.140; r = 0.115, P = 0.341).
Pearson Correlation of Anti-Xa Assay, PT, APTT, TEG R-Time Compare to MS-Peak and MS-Trough.
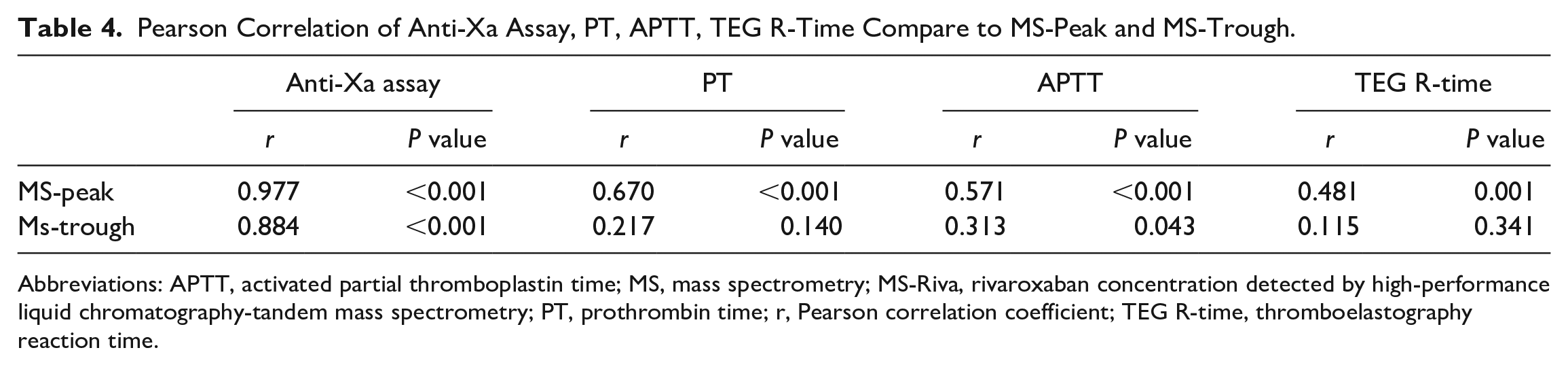
Abbreviations: APTT, activated partial thromboplastin time; MS, mass spectrometry; MS-Riva, rivaroxaban concentration detected by high-performance liquid chromatography-tandem mass spectrometry; PT, prothrombin time; r, Pearson correlation coefficient; TEG R-time, thromboelastography reaction time.
Patient With AF
Table S1 is a Pearson correlation of Anti-Xa assay, PT, APTT, and TEG R-time compared to MS-Riva in patients with AF. A total of 147 plasma samples were detected for concentration by HPLC-MS/MS and chromogenic anti-Xa assays, 66 plasma samples were used for PT, 54 plasma samples were used for APTT, and 98 whole blood samples were used for TEG R-time measurements. Bland-Altman plots (Figure S1) showed the limits of agreement between the chromogenic anti-Xa assay (mean difference, 2.3; 95% CI: −33.7, 38.3), PT (mean difference, 0.0; 95% CI: −0.7, 0.7), APTT (mean difference, 0.0; 95% CI: −0.9, 0.9), TEG-R time (mean difference, 0.0; 95% CI: −1.2, 1.2), and MS-Riva in AF patients. MS-Riva had a significant and very strong correlation with the chromogenic anti-Xa assay (r = 0.986, P < 0.001) and a significant and strong correlation with PT, APTT, and TEG R-time (r = 0.895, P < 0.001; r = 0.794, P < 0.001; r = 0.746, P < 0.001).
Patient With VTE
Table S2 assesses the Pearson correlation of Anti-Xa assay, PT, APTT, and TEG R-time, compared to MS-Riva in patients with VTE. Forty-four plasma samples were detected for concentration by HPLC-MS/MS and chromogenic anti-Xa assays, 38 plasma samples were used for PT, 28 plasma samples were used for APTT, and 42 whole blood samples were used for TEG R-time measurements. Bland-Altman plots (Figure S2) showed the limits of agreement between the chromogenic anti-Xa assay (mean difference, −0.6; 95% CI: −34.2, 33.0), PT (mean difference, 0.0; 95% CI: −0.6, 0.6), APTT (mean difference, 0.0; 95% CI: −1.0, 1.0), TEG R-time (mean difference, 0.0; 95% CI: −0.8, 0.8) and MS-Riva in VTE patients. MS-Riva had a significant and very strong correlation with the chromogenic anti-Xa assay (r = 0.985, P < 0.001), a significant and strong correlation with PT and TEG R-time (r = 0.871, P < 0.001; r = 0.730, P < 0.001), and a significant moderate correlation with APTT (r = 0.583; P = 0.001).
Discussion
Our study had the following findings: (1) The chromogenic anti-Xa assay, PT, APTT, and TEG R-time significantly and strongly correlated with MS-Riva. (2) The chromogenic anti-Xa assay strongly correlated with MS-Riva when concentrations were in the range of 25.0~448.5 ng/mL. PT was moderately correlated with MS-Riva when the times were in the range of 11.0 to 29.0 seconds. Activated partial thromboplastin time was moderately correlated with MS-Riva when the times were in the range of 28.0 to 56.0 seconds. Thromboelastogram reaction time was moderately correlated with MS-Riva when the times were in the range of 5 to 10 minutes. (3) Mass spectrometry peak showed an extremely strong correlation with the chromogenic anti-Xa assay and a significant moderate correlation with PT, APTT, and TEG R-time. Mass spectrometry trough was strongly correlated with the chromogenic anti-Xa assay, a significantly weak correlation with APTT, and an insignificant correlation with PT and TEG R-time. (4) In patients with AF, the chromogenic anti-Xa assay correlated highest with MS-Riva, followed by PT, APTT, and TEG R-time. The chromogenic anti-Xa assay correlated highest with MS-Riva in patients with VTE, followed by PT, TEG R-time, and APTT. Bland-Altman analysis plots of the chromogenic anti-Xa assay showed acceptable agreement. Bland-Altman analysis plots of PT, APTT, and TEG R-time showed acceptable agreement when data from different units were converted to normalized values for comparison.
This study showed a significant and very strong correlation between the chromogenic anti-Xa assay and MS-Riva when concentrations were in the range of 25.0 to 448.5 ng/mL (r = 0.980) and no significant correlation when concentrations were below 25.0 ng/mL (r = 0.241). That suggests chromogenic anti-Xa assay are unsuitable for assessing rivaroxaban concentrations below 25.0 ng/mL. Studt et al 8 found that the chromogenic anti-Xa assay correlated well with HPLC-MS/MS (r = 0.99), similar to our study’s results. However, Studt et al found a strong correlation between 50 and 200 µg/L, while our study found the correlation in a wider range (25.0~448.5 ng/mL). The difference in concentration ranges may be explained by the fact that Studt et al included healthy volunteers, whereas this study included patients with AF or VTE. In addition, this study found that HPLC-MS/MS had a significantly strong correlation with the chromogenic anti-Xa assay at peak and trough concentrations.
Secondly, we found that PT and APTT were both significantly and strongly correlated with MS-Riva, and the correlation between PT and MS-Riva was higher than that of APTT (r = 0.884, r = 0.741). In addition, we found a significant moderate correlation between PT and APTT when the times were in the normal population reference range of 11.0 to 15.0 seconds and 28.0 to 42.0 seconds (r = 0.510; r = 0.464). A moderate correlation was also found when the times were in the range of 15.1 to 29.0 seconds and 42.1 to 56.0 seconds (r = 0.654; r = 0.431). Those results suggest that PT and APTT can assess MS-Riva within the normal population reference range and above the normal population reference range. The results of Rathbun et al 21 differed from ours. Rathbun et al found a strong correlation between PT and MS-Riva, but APTT correlated poorly with MS-Riva and was insignificant. The reason for the difference may be that Rathbun et al included 1/3 of healthy volunteers and 2/3 of AF and VTE patients and analyzed blood samples from healthy volunteers together with AF and VTE patients. Meanwhile, blood samples were collected at hours 2, 4, and 6 after dosing (peak concentrations are 2 to 4 hours after dosing, and concentrations of rivaroxaban begin to decline 4 hours after dosing). In contrast, in this study, blood was collected 30 minutes before and 2 to 4 hours after dosing to measure peak and trough concentration. At the same time, this study found that PT and APTT had a significant moderate correlation with MS-peak, PT had no significant correlation with MS-trough, and APTT had a weak correlation with MS-trough. Therefore, the different time of blood collection leads to different rivaroxaban concentration, thus affecting the correlation between PT, APTT, and MS-Riva. However, Douxfils et al 26 found a poor linear correlation between PT and MS-Riva, whether the results were in seconds, in ratios, or ng/mL, and calibrating each reagent with rivaroxaban did not improve the correlation. The difference may be that the study was retrospective and used stored blood samples, which may have undergone deterioration during storage. It is also not clear when the samples were collected. Rivaroxaban may have already undergone a period of metabolism at the time of blood collection, resulting in lower plasma concentrations, which affects the correlation between PT and MS-Riva.
Our study found a strong correlation in TEG R-time with MS-Riva (r = 0.739). There was a significant moderate correlation (r = 0.696) between TEG when the times were in the normal population reference range (5.0-10.0 min) and MS-Riva. However, there was no significant correlation with MS-Riva when the times outside the normal population reference range (<5 min and 10.1-17.8 min) (r = 0.248; r = 0.026). This means that MS-Riva can be assessed when TEG R-time is within the normal population reference range but are not suitable for assessing MS-Riva outside the normal population reference range. The results of Myers et al 4 are inconsistent with ours. Myers et al found no significant correlation between TEG R-time and MS-Riva (r = 0.34). The difference may be because, in this study, rivaroxaban concentrations were measured 30 minutes before and 2 to 4 hours after dosing, that is, at trough and peak concentrations. While Myers included patients who took rivaroxaban within 48 hours and measured TEG R-time 24 hours after admission, at which time rivaroxaban concentrations had decreased.
Meanwhile, this study found a significant moderate correlation between TEG R-time and MS-peak, while there was no significant correlation with MS-trough. Therefore, different blood collection times can affect rivaroxaban concentration and thus lead to different correlations between TEG R-time and MS-Riva. Rathbun et al 21 also found conclusions inconsistent with this study: MS-Riva was essentially uncorrelated with TEG R-time (r = 0.099). The possible reason for this difference is that the TEG parameters in Rathbun’s study were assessed randomly during the patient’s hospitalization and did not record the patient’s most recent dose of rivaroxaban. The anticoagulant effect may have dissipated, causing the TEG R-time to converge to the value when the drug was not taken. 4
Although the routine use of rivaroxaban does not require monitoring, assessment of anticoagulation effects may be helpful in many clinical situations, including patients with impaired renal function, elderly patients, patients with acute bleeding episodes, patients who are excluded from overdosing, and monitoring treatment compliance in the event of an unexpected thromboembolic event. 27 In this study, chromogenic anti-Xa assay correlated highest with MS-Riva in patients with AF, followed by PT, APTT, and TEG R-time, while APTT correlated lowest with MS-Riva in patients with VTE. Rivaroxaban-calibrated chromogenic anti-Xa assays can accurately identify high and low rivaroxaban concentrations during treatment and have superior utility in all clinical situations where anticoagulation status assessment may be beneficial. 28
According to the results of this study, PT correlates better with MS-Riva than APTT, which is not suitable as a clinical assay for rivaroxaban plasma concentration compared to PT, especially in VTE patients. In contrast, TEG R-time correlated most poorly with MS-Riva. And since TEG assay is time consuming and relatively expensive, it is not recommended to monitor rivaroxaban concentration by TEG when the above assay can be performed. In general, HPLC-MS/MS is preferred for clinical detection of rivaroxaban concentration, but its detection time is longer. According to our study, in time-sensitive emergency situations (e.g., need for urgent surgery, deciding whether to give t-PA for a stroke victim, etc.), chromogenic anti-Xa assays may be preferred over PT, and if chromogenic anti-Xa assays cannot be performed due to limitations, PT may be used to assess the anticoagulant effect of rivaroxaban.
Meanwhile, when assessing rivaroxaban concentration using chromogenic anti-Xa assay, blood samples at trough concentration (30 min before dosing) or peak concentration (2~4 h after dosing) can be collected. In contrast, when using PT to assess rivaroxaban concentration, blood samples should be collected at peak concentration, that is, 2 to 4 hours after drug administration, and blood samples at trough concentration (30 min before drug administration) are not suitable. Monitoring peak and trough concentrations are important for predicting thrombotic recurrence and bleeding risk. For example, the 2021 guidelines state that the expected range of peak plasma concentrations in AF patients taking 20 mg qd rivaroxaban is 184 to 343 ng/mL and the expected range of trough concentrations is 12 to 137 ng/mL. 29 The 2018 International Council for Standardization in Hematology (ICSH) states that the expected range of peak plasma concentrations in VTE patients taking 20 mg qd rivaroxaban is 189 to 419 ng/mL and the expected range of trough concentrations is 6 to 87 ng/mL. 10 When the peak and trough concentrations detected in patients taking 20 mg qd rivaroxaban are not in the expected range, it may mean that the treatment is not effective, too low may be a risk of thrombosis, too high may be a risk of bleeding. By measuring blood concentration, the risk of bleeding and thrombosis can be assessed, which can help clinicians better adjust the dosing regimen, reduce the risk of recurrence of thrombosis, and reduce the risk of bleeding.
This study is the first to correlate multiple coagulation indices with different reference value partition segments and comprehensively compare coagulation indices with MS-peak and MS-trough. This study also adds important insights to the existing literature on the correlation of the chromogenic anti-Xa assay, PT, APTT, and TEG R-time with the peak and trough concentrations of rivaroxaban and the correlation of the chromogenic anti-Xa assay, PT, APTT, and TEG R-time with rivaroxaban concentrations within and outside the normal range of detection. One of the strengths of this study is the prospective nature, with detailed records of the time of administration of rivaroxaban and the time points of blood collection for each test indicator. At the same time, this study has some limitations. First, for TEG R-time measurements, the conclusions are based on a comparison with the reference range provided by the manufacturer. Second, slightly fewer patients with VTE were included in this study, and further validation of the results may be needed with a larger sample. Finally, patients in emergency situations were not included in this study, and it is hoped that more studies on coagulation parameters in patients in emergency situations will be available in the future.
Conclusion and Relevance
This study shows that HPLC-MS/MS is the preferred choice for monitoring peak and tough concentrations, followed by anti-Xa, while PT is only suitable for peak concentrations. This study can help the clinicians to better adjust the medication regimen and reduce the risk of recurrence of thrombosis as well as the risk of bleeding.
Supplemental Material
sj-docx-1-aop-10.1177_10600280231158929 – Supplemental material for Comparison of the Correlation Between Coagulation Indices and Rivaroxaban Concentrations
Supplemental material, sj-docx-1-aop-10.1177_10600280231158929 for Comparison of the Correlation Between Coagulation Indices and Rivaroxaban Concentrations by Tingting Wu, Shuyi Wu, Meijuan Li and Jinhua Zhang in Annals of Pharmacotherapy
Footnotes
Author Contributions
JZ initiated the study. JZ, TW, and SW collected and entered the data. TW and SW performed data collation, data extraction, and analyses. TW drafted the first version of the manuscript. JZ, SW, and ML critically reviewed the manuscript and revised it. ML and JZ worked on data validation. TW, SW and ML performed the graphical revisions. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study complies with the Declaration of Helsinki and was authorized by The Ethics Committee of Fujian Medical University Union Hospital (number: 2018YF005-02, date: 2018-3-14).
Informed Consent
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
