Abstract

We read with interest the report of Mullins et al 1 on the importance of causality assessment tools in the adjudication of drug-induced liver injury (DILI) in micafungin-treated patients. 1 In this report, utility of the updated RUCAM (Roussel Uclaf Causality Assessment Method) Assessment Scale on a DILI-drug pair earlier adjudicated by Naranjo resulted in significant change to the final point prevalence estimate (10.6%) of micafungin-associated DILI. 1 Sequential assessment with an adverse drug reaction (ADR) tool (Naranjo) followed by a DILI-specific algorithm strengthens the process of causality determination. Additionally, it is our opinion that this process may be further strengthened by proceeding to determine if any of the Micafungin-DILI pairs were “avoidable.” The concept of avoidability of ADR has been strengthened recently following the validation of a novel risk preventability tool (called the Liverpool adverse drug reactions avoidability tool [LAAT]). This tool adjudicates whether ADRs could potentially be avoidable.2,3 Utility of this tool has potential implication for reexamination of prescribing practices as well as therapeutic commissioning around specific therapeutic agents such as micafungin. Indeed, we are the first to explore the potential utility of this tool in a cohort of patients (65 years and older) to ascertain if, indeed, Naranjo and updated RUCAM Scale adjudicated DILI-drug pairs were avoidable or not. In an examination of 38 DILI-drug pairs (using the LAAT) from an elderly cohort of patients, we found that about 32.3% (50/152) of them were rated as “avoidable” (“probably” or “definitely”). 4 We found that the overall median Krippendorf’s κ with the tool was 0.61 (SE = 0.12; CI = 0.36, 0.85), with an interrater correlation coefficient of 0.50 [0.32, 0.65]. The demonstration of >32% of DILI cases as “avoidable” raises the prospect for further examination of these cases (including potentially micafungin cases) with the view to identifying areas for target therapeutic intervention. We will suggest incorporation of this tool (LAAT) into the adjudication algorithm by Mullins et al 1 ; we have provided a schema as shown in Figure 1. 5 This is likely to provide the physician at the bedside as well as therapeutic commissioners with robust information regarding the possible areas of intervention.
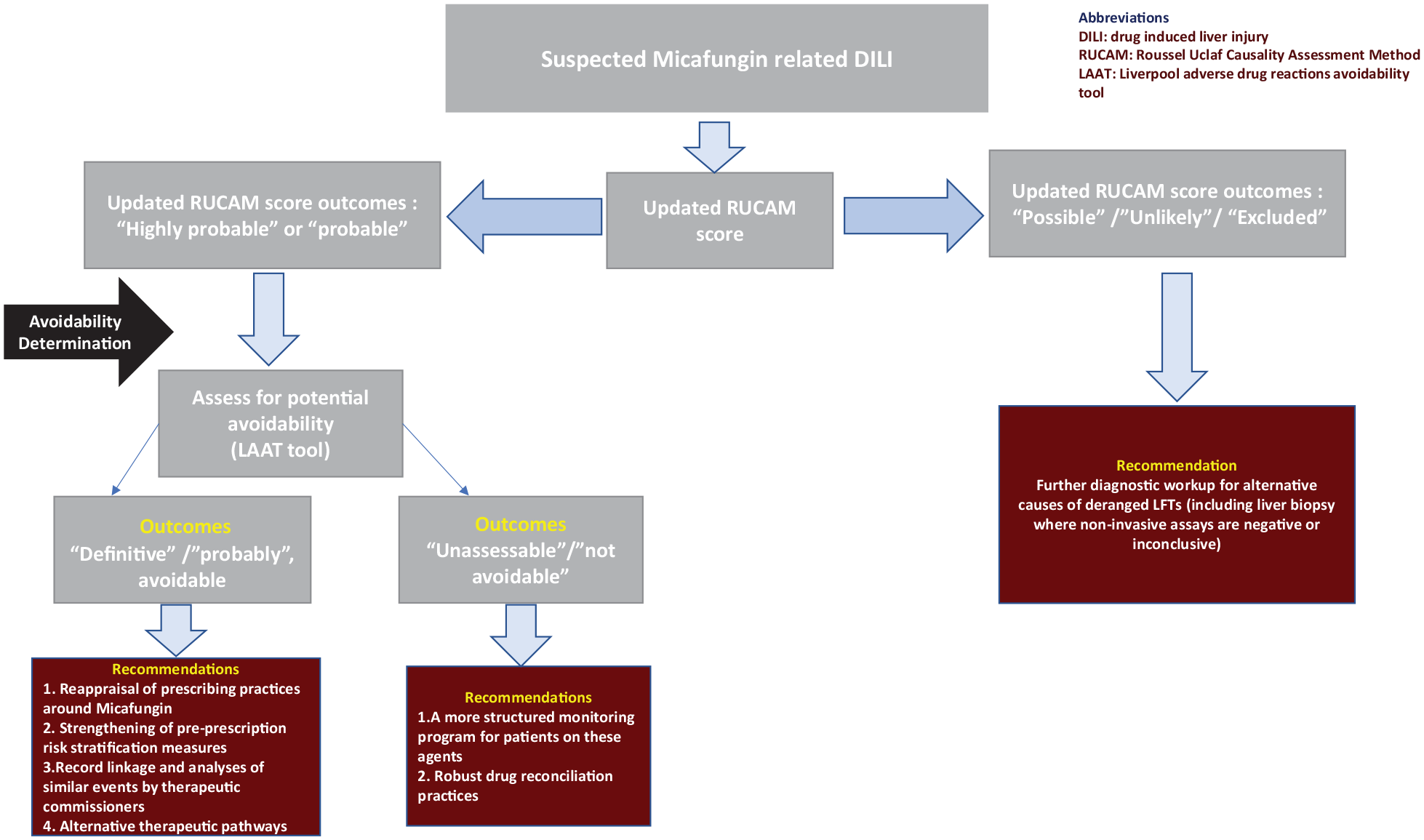
Suggested algorithm for the integrated adjudication of DILI caused by micafungin with the updated RUCAM Scale and the determination of its potential preventability utilizing the LAAT (adapted from Danjuma et al 5 ).
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
