Abstract
Background
While there is strong evidence for botulinum toxin-A (BoNT-A) in post-stroke spasticity, there is a paucity of data in multiple sclerosis and other conditions in real-world settings.
Objective
Document the use of BoNT-A in the management of spasticity, with focus on the treatment of spasticity due to stroke and multiple sclerosis.
Methods
This noninterventional, retrospective study included all adults treated for upper and/or lower limb spasticity (any etiology) at three centers in Italy who received ≥3 BoNT-A injection cycles between 2008 and 2018.
Results
Injection data from 149 patients were analyzed (n = 67 post-stroke, n = 47 MS, n = 35 other etiologies). The median treatment duration was 54.9 months in the post-stroke population and 41.9 months in the MS population. Total doses for the commercially available BoNT-A formulations were typically lower than approved for use in spasticity; we also observed clinically relevant differences in the muscle patterns treated between the MS and post-stroke indications. Regardless of etiology, most patients were satisfied with treatment.
Conclusions
This retrospective study provides a snapshot of spasticity management for patients referred for BoNT-A treatment. Most patients were satisfied with their treatment over several cycles and the data support the effectiveness of BoNT-A for focal spasticity regardless of etiology.
Lay Summary
While there is a lot of high-quality evidence supporting the efficacy (how well a medication works) and safety of botulinum toxin-A in treating spasticity, most of the research has been done in stroke survivors and there is a need to understand how botulinum toxin-A is used in people living with spasticity due to other causes.
In this paper, a group of rehabilitation specialists from Italy describe a study where they reviewed the medical records of all patients who had received repeated botulinum toxin-A treatment for spasticity over a period of 10 years. They found that the two most common causes of the spasticity they treated were stroke and multiple sclerosis (MS). The authors made a number of important observations. Firstly, the doses of botulinum toxin used were typically on the lower end of the recommended dosing range in spasticity. Secondly, the way that stroke survivors and patients with MS are treated with botulinum toxin (e.g., the muscles injected) are different. Nevertheless, most patients said they were overall satisfied with repeated treatment and the findings support the use of botulinum toxin in managing spasticity in patients with a variety of conditions.
Keywords
Introduction
Spasticity is a common symptom of the Upper Motor Neuron Syndrome (UMNS) and has been defined as “disordered sensory-motor control, resulting from an upper motor neuron lesion, presenting as an intermittent or sustained involuntary muscle activation” (Pandyan et al., 2005). Acknowledging the complexity of the UMNS, this definition has been broadened to encompass, not just spasticity, but also related phenomena such as spastic dystonia and co-contraction, stretch-sensitive paresis, and soft tissue contracture (Gracies, 2005). When present, spasticity can cause substantial patient and caregiver burden (Jacinto et al., 2020; Patel et al., 2020). In an online survey, patients living with spasticity reported a range of signs and symptoms (in addition to ‘stiffness’) including muscle spasms and pain, as well as unwanted limb movements – causing a variety of functional impacts including difficulties in walking and using their affected arm(s)/hand(s) in routine daily activities such as eating and dressing (Jacinto et al., 2020).
Spasticity is a common feature of several conditions involving an upper motor neuron lesion, including stroke, cerebral palsy, anoxia, traumatic brain injury (TBI), spinal cord injury (SCI), multiple sclerosis (MS), and other neurodegenerative diseases. For example, it is estimated that 25% of stroke survivors will eventually develop symptomatic spasticity (Zeng et al., 2020), with larger lesion sizes in patients with hemorrhagic versus ischemic stroke (Andersen et al., 2009). By contrast, it is estimated that 60–80% of patients with MS will develop spasticity (Hugos & Cameron, 2019), which is thought to be caused by lesions in either the brain or spinal cord. The clinical presentation may vary, depending on the damage localization, and following typical patterns (Esquenazi et al., 2017). However, even when they have spasticity of the same etiology, no two patients present in the same way, and this heterogeneity makes the rehabilitation process complex and challenging, requiring tailored interventions (Turner-Stokes et al., 2018).
Regardless of etiology, current spasticity management is focused on enabling patients to achieve their current goals and to prevent secondary consequences such as soft-tissue shortening and contractures which compound the problem. The mainstay of therapy is physical rehabilitation often facilitated by pharmacological interventions. Evidence-based guidelines recommend botulinum toxin type A (BoNT-A) injections as an effective first-line treatment for focal spasticity of the upper and lower limbs (Haselkorn et al., 2005; Royal College of Physicians (UK), 2018; Simpson et al., 2016). While the evidence for BoNT-A is strong in stroke survivors (Simpson et al., 2016), it is only considered moderate in MS – primarily because of the paucity of controlled clinical studies (Comi et al., 2020; Dressler et al., 2017). As such, practioners treating MS patients may have to rely on data collected in other etiologies, even though the potential differences between populations have not been properly investigated.
Observational studies, conducted in real-world settings, are useful to inform about the current management of spasticity, including the various etiologies, and allowing for different clinical practices. Such studies also provide important information on individualized administration strategies (e.g., dosing per muscle) and effectiveness that are not possible to capture in controlled clinical trials. We present here BoNT-A treatment data from a retrospective, ‘real-world’, multicenter study conducted in Italy to document the routine use of BoNT-A in the long-term management of upper- and/or lower-limb spasticity. An additional aim was to compare injection parameters in post-stroke and MS spasticity patients treated in three experienced rehabilitation centers in Italy.
Methods
Study Conduct
This was a multicenter, non-interventional, retrospective study conducted at three expert centers in Italy. The study was conducted in compliance with the International Society for Pharmacoepidemiology (ISPE) Guidelines for Good Pharmacoepidemiology Practices. Independent Ethics Committee/ Institutional Review Board approval was obtained prior to each center initiation and data collection. The study is registered at Clinicaltrials.gov (NCT04340336).
Population
This retrospective study included all consecutive adult patients (≥18 years old), treated at the three aforementioned centers, who received at least three consecutive BoNT-A injection cycles with any of the currently available marketed BoNT-A for upper and/or lower limb spasticity between January 2008, and December 2018, and who had follow-up data available in their medical record. Database lock was performed on February 7, 2022. The only exclusion criteria were spasticity due to cerebral palsy and treatments and assessments being performed at more than one center.
As this was a retrospective study, all treatments were per routine practice.
Assessments
All assessments, including demography and baseline clinical characteristics, were extracted from medical records. BoNT-A treatment records including injection schedules and reasons for injection as well as reasons for changes in treatment schedule, permanent interruption of BoNT-A treatment, and/or discontinuation (defined as any temporary break in BoNT-A treatment being received other than for persisting effectiveness of treatment) were routinely collected. In addition, the three centers routinely documented Global Treatment Satisfaction (GTS) data collected at the first and final post-injection visit of the treatment cycle; possible responses were rated on a Likert scale ranging from “not satisfied” to “very satisfied.” Adverse events as recorded in the medical records were also reported.
Data Analysis
Relevant data collected as part of routine medical care were captured on an electronic case report form (eCRF). Pseudonymized data were reviewed for completeness and analyzed with each patient identified by a subject number. Data are presented for the overall cohort, post-stroke population, and the MS population using descriptive summary statistics; spasticity due to other etiologies (e.g., brain or spinal cord injury, tumor, or inflammatory/infective conditions) were not analyzed separately due to the limited number of cases. The duration of treatment was estimated for new (previously naïve to BoNT-A treatment) patients. Treatment intervals were calculated for patients who remained on the same BoNT-A brand. Mean and standard deviation (mean ± SD) or median [Q1, Q3] measures are used to summarize continuous variables, and absolute and relative frequencies expressed as percentage (%) are presented for categorical information.
Results
Patient Characteristics
A total of 152 patients from across the three sites were included. Of these, three patients were excluded from the analyses because they did not receive at least 3 BoNT-A injection cycles. Overall, 67 patients had post-stroke spasticity, 47 had a diagnosis of MS, and 35 had spasticity due to other etiologies. Baseline demographic and clinical characteristics are presented in Table 1. The proportion of male vs. female patients differed by etiology; whereas two-thirds (66%) of the post-stroke population were male, the converse was true for the MS population. The mean ± SD age at first BoNT-A injection was 58.2 ± 13.0 years for the post-stroke population, 47.1 ± 11.4 years for the MS population, and 53.1 ± 14.7 years for the total cohort. Whereas patients with MS were considerably more likely to have lower limb spasticity than upper limb spasticity (92% vs 21%, respectively), most post-stroke patients (73%) experienced both.
Patient Characteristics.

Patients with spasticity due to other etiologies were not analyzed separately due to the limited number of cases.
In general, patients of all etiologies had experienced spasticity for a prolonged period (median time since spasticity onset, 5.7 years) before receiving their first BoNT-A injection, with the MS population waiting the longest period (median of 13.6 years vs 2.2 years in the post-stroke population). Key BoNT-A treatment parameters are summarized in Table 2. Most patients (95%) were new to BoNT-A treatment. For these patients, the median overall duration of treatment during the data collection period was 41.1 months, with 72% still undergoing treatment at the end of the study. The median duration of treatment was 54.9 months in the post-stroke population, with 87% still ongoing, and 41.9 months in the MS population, with 52% still ongoing. In accordance with the protocol, all patients had ≥3 treatment cycles (median [range] 9 [3–35] cycles). Overall 39 (26%) patients had a permanent interruption to treatment, with the main reason being loss to follow-up. Most patients (n = 91, 61%) switched between BoNT-A formulations, and the proportion of ‘switch’ patients was similar regardless of etiology (89% of switches were due to lack of product availability during the index period). Of 2192 recorded visits, most were performed according to predefined hospital schedules (n = 1947 visits, 89%) with 11% (n = 244 visits) performed according to medical need. The median [Q1, Q3] injection interval was 20.0 [16.7, 21.5] weeks in the post-stroke population and 20.5 [16.7, 23.1] weeks in the MS population.
Treatment of Spasticity With BoNT-A.

Patients who switched BoNT-A type during the observation period were counted in each BoNT-A type used.
Only patients who did not switch BoNT-A type during the observation period were included.
Only patients who were previously naive to BoNT-A treatment were included.
The denominator is the number of patients who permanently interrupted treatment.
On average, patients with post-stroke spasticity had a median of 4 (mean, 5) muscles injected with almost three-quarters (74.6%) being injected in both the upper and lower limbs. In line with the predominance of lower limb spasticity, most patients with MS were injected in the lower limb with a median of 3 (mean, 4) muscles injected. Regarding the total doses administered, the mean total doses for all three formulations were lower than approved for use in spasticity. Of note, higher mean [95% CI] doses of aboBoNT-A were used in the treatment of MS patients compared with the post-stroke group (1049 [957, 1140] U vs 734 [638, 829] U), while they were more similar between the two subpopulations for incoBoNT-A and onaBoNT-A.
Figures 1 and 2 provide an overview of the muscles injected in post-stroke and MS patients (see Table e1 for overall population), and Tables e2 and e3 describe the doses used in each of the lower and upper limb muscles, respectively. For post-stroke patients, the most commonly injected upper limb muscles were the flexor digitorum superficialis (injected in 83.6% of patients), biceps brachii (80.6%), and pectoralis major (58.2%), and the most commonly injected lower limb muscles were the tibialis posterior (56.7%), gastrocnemius medial head (53.7%), and soleus (50.7%). For patients affected by MS, the most commonly injected lower limb muscles were the gastrocnemius (medial and lateral head, both injected in 83.0% of patients), soleus (76.6%), and hamstrings (40.4%).
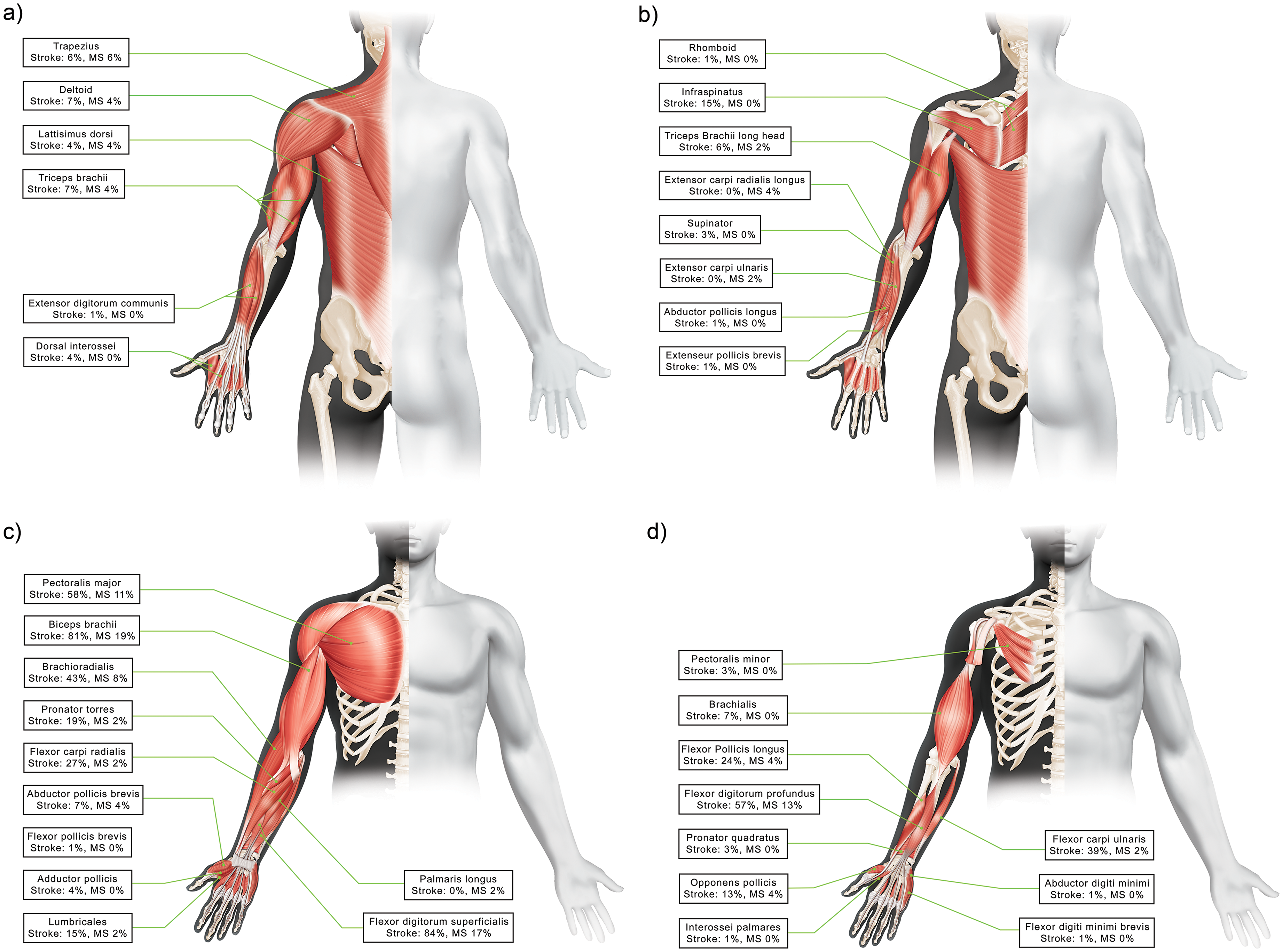
Upper Limb Muscles Injected (a) Posterior View Superficial Muscles, (b) Posterior View Deep Muscles, (c) Anterior View Superficial Muscles, (d) Anterior View Deep Muscles.
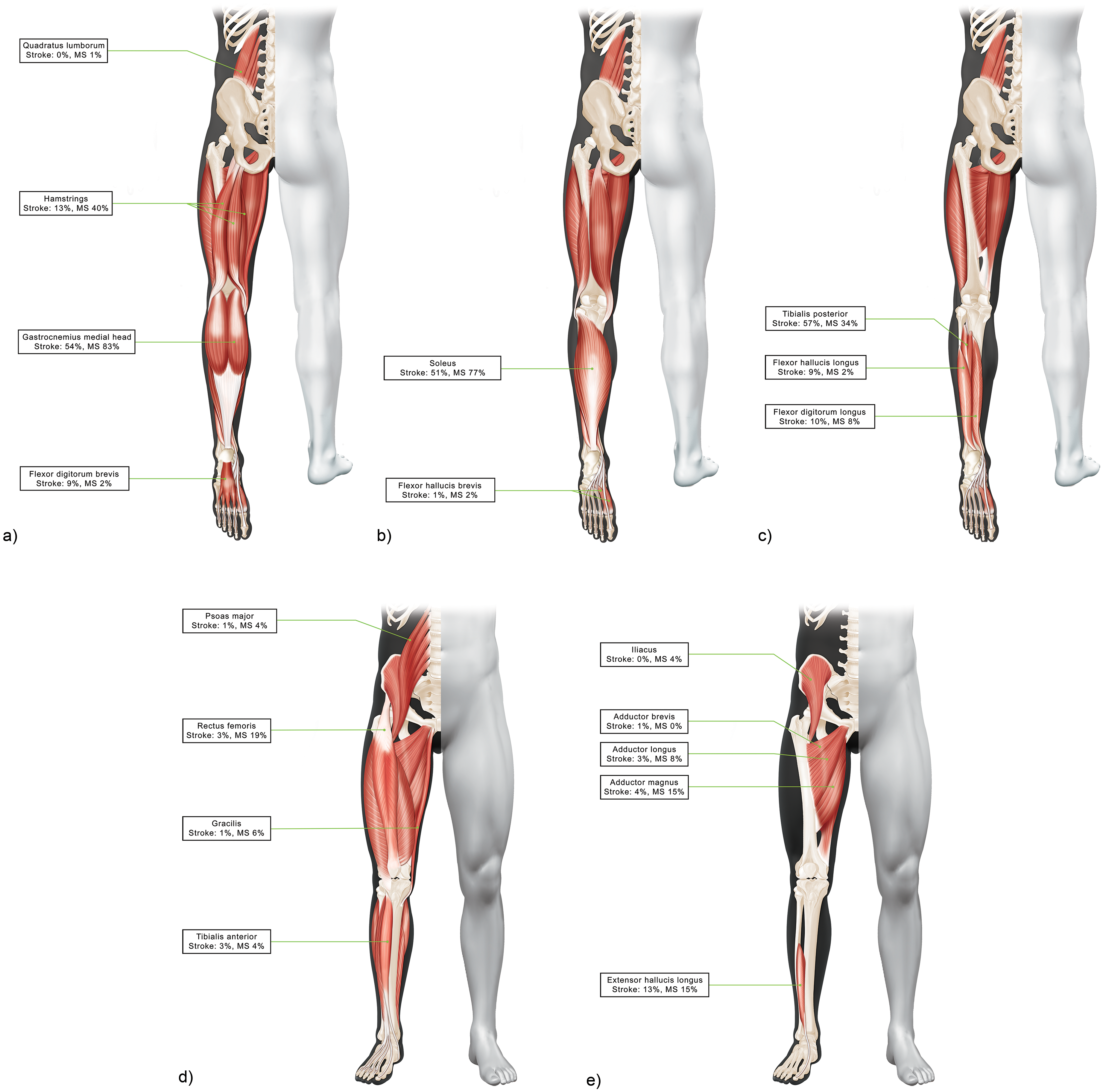
Lower Limb Muscles Injected (a) Posterior View Superficial Muscles, (b) Posterior View Intermediary Muscles (c) Posterior View Deep Muscles, (d) Anterior View Superficial Muscles, (e) Anterior View Deep Muscles.
Overall, the vast majority of patients with available GTS ratings were satisfied with their treatment at both visits, with a slight tendency to higher ratings of satisfaction in the post-stroke vs MS populations (Figure 3).
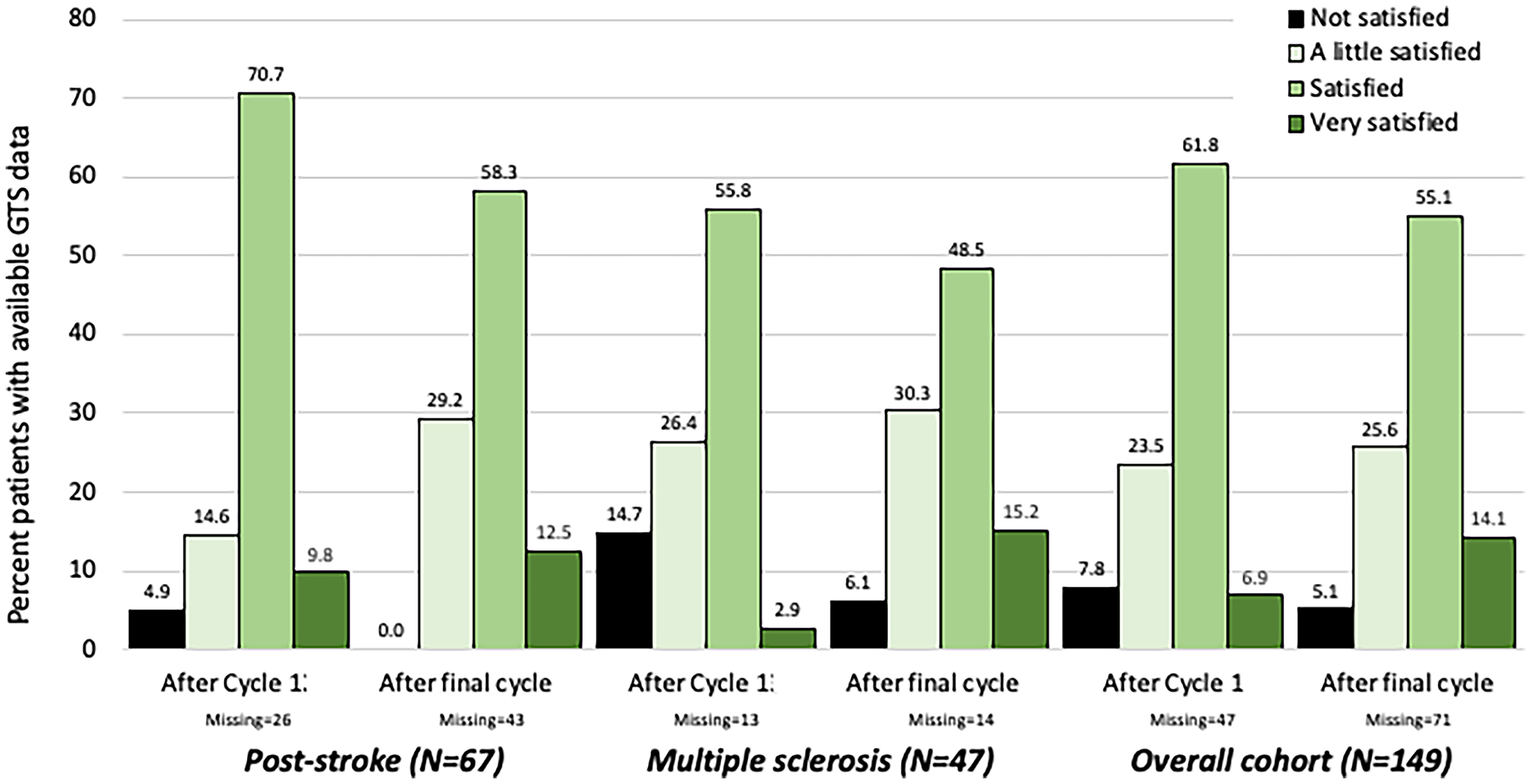
Global Treatment Satisfaction Ratings After Cycle 1 and After the Final Cycle.
Twenty-one patients (14%) temporarily discontinued treatment (n = 8 [9%] treated for upper limbs and n = 16 [12.4%] treated for lower limbs) at least once during the study, predominantly due to clinical reasons (e.g., no spasticity, change in treatment priority, adverse events). Three out of 92 aboBoNT-A treated patients included in the Safety Population had adverse events considered treatment-related by the treating clinician; all 3 patients had MS (asthenia n = 2, diplopia n = 1, and muscular weakness, n = 1). Two AEs of progressive MS and muscular weakness in one patient led to treatment interruption; neither of these events was considered as treatment-related. There were no deaths, serious AEs, or other significant AEs with aboBoNT-A during the index period.
Discussion
This retrospective study provides a snapshot of spasticity management for patients referred to Italian expert neurorehabilitation clinics for treatment with BoNT-A. While the efficacy and safety of BoNT-A is often collectively described under the umbrella term of ‘adult spasticity’, our data highlight some important differences in spasticity management between the MS and post-stroke populations. Nevertheless, the majority of patients were overall satisfied with repeated treatment over several cycles and the data support the effectiveness of BoNT-A injections in focal spasticity management regardless of etiology.
Whereas the use of BoNT-A is well embedded as a first-line option for managing post-stroke spasticity, this is not the case in MS – with less than 10% of MS patients receiving treatment with BoNT-A (Bensmail et al., 2023; Dressler et al., 2017). Potential reasons for this relatively low use include uncertainty about its effectiveness and tolerability in MS (Hui & Argáez, 2021; Moccia et al., 2020). Instead, MS-related spasticity management typically combines physiotherapy with anti-spasticity agents, including baclofen (oral or intrathecal administration), tizanidine, dantrolene, benzodiazepines, and cannabinoids (Carod-Artal et al., 2022; Comi et al., 2020). In line with current practice, patients with MS in our study only received their first BoNT-A injections after an average of 13 years living with spasticity. This extended delay may, at least in part, explain the higher BoNT-A doses required in MS compared with spasticity of other etiologies. Importantly however, as shown in this study, once patients with spasticity due to MS started their BoNT-A treatment, they were generally satisfied, most patients with MS safely remained on treatment over many cycles, and no new safety or tolerability issues were identified in this population.
In our study, the median duration of treatment in MS was 41.9 months (3.5 years), which is somewhat shorter than observed in patients with post-stroke spasticity (54.9 months /4.6 years), but longer than the 1.2 years (MS population) and 5 months (several etiologies, including MS) reported by other Italian studies (Latino et al., 2017). Whereas the main reason for a permanent interruption of therapy in our study was loss to follow-up (both indications), the earlier studies found that patients with MS mainly stopped treatment due to loss of efficacy in the context of a progressive disease, logistic problems, and advancement to surgical treatments, with factors such as absence of caregiver, lack of regular rehabilitation, spasticity severity, and cognitive dysfunction being significant predictors for BoNT-A discontinuation (Cinone et al., 2022; Latino et al., 2017). While satisfaction with treatment was high in both the post-stroke and MS populations, our observations of a slightly lower level of satisfaction in the MS population indicate the need for more work to better tailor treatments to their specific needs. Recently, the Italian network study showed that routine use of BoNT-A in MS depends on symptom burden (Moccia et al., 2020), which often worsens with greater gait disability, as well as with disease progression and relapses, and some disease modifying treatments (Hugos & Cameron, 2019). As such, it has been suggested that BoNT-A injection schedules in MS should be flexible to allow for fluctuating symptom burden (Bensmail et al., 2023), and future work is needed to understand how this impacts scheduling.
Our injection data highlight the diversity of muscle patterns treated in real-life practice compared to randomized controlled trials (RCTs). To date, most adult BoNT-A RCTs have been performed in patients with post-stroke spasticity and as such, injectors working with MS patients must use their personal experience to extrapolate data from post-stroke studies to devise their own treatment paradigms for MS. However, as demonstrated in this observational study, the muscle patterns treated differ substantially between the two etiologies. For example, whereas equinovarus deformities predominate post stroke (Li, 2020), patients with MS are much more likely than stroke survivors to require injections targeted to the hamstrings, rectus femoris, and other proximal muscles. For muscles not included in the labelling, recent expert-opinion based dosing tables have been proposed for incoBoNT-A and onaBoNT-A (Dressler et al., 2021), and a systematic review has recommended a simple algorithm where the aboBoNT-A dose for spasticity is 11.5 times the muscle volume (e.g., 100 to 150 U for a muscle volume of 100 cm3) (Schnitzler et al., 2022).
It is also noteworthy that, despite the fact that three-quarters of stroke survivors were injected in both the upper and lower limbs, the total dose for all three formulations in post-stroke spasticity was overall lower than approved in the management of spasticity (Botox Summary of Product Characteristics (Italy); Dysport Summary of Product Characteristics (Italy); Xeomin Summary of Product Characteristics (Italy)). Recent international studies have also shown a tendency to inject both upper and lower limbs at the lower end of the dosing range (Esquenazi et al., 2023; Turner-Stokes et al., 2021). On the other hand, most patients with MS were only treated for lower limb spasticity and previous retrospective studies have found that patients with MS typically require higher BoNT-A doses than other indications (Phadke et al., 2014). In our study, while the total doses of incoBoNT-A and onaBoNT-A were similar between the MS and post-stroke populations, patients with MS appeared to be injected with higher total aboBoNT-A doses than stroke survivors (and closer to maximal dosing for the lower limb). Reasons for this discrepancy are unclear, and a limitation of this study is that we were unable to correlate treatment data with the severity of spasticity.
Strengths of this observational study include its ‘real-world’ design which improves generalizability to clinical practice. Limitations of this study include those inherent to retrospective, observational studies such as the lack of a control group and the limiting of outcomes to those routinely recorded at the participating sites. For example, we did not systematically collect data on goal setting and goal attainment. The study was originally designed to document BoNT-A use in adult spasticity of any etiology and the comparison of post-stroke vs MS subpopulations was performed as an exploratory objective. Unfortunately, the sample size for adult patients with spasticity due to other causes was too small for meaningful analysis. Due to changes in availability (often at the central pharmacy), 61% of patients switched between BoNT-A formulations in this study, making it difficult to compare effectiveness in any given population. However, switching formulation did not appear to affect patient satisfaction with treatment.
In summary, this study provides important information for clinicians regarding the muscles and doses used to treat adult patients with spasticity due to MS and stroke. By highlighting the differences in injection practice, the study can also be considered a call to action for more MS specific work to guide treatments in this population.
Supplemental Material
sj-pdf-1-nre-10.1177_10538135251329322 - Supplemental material for Spasticity Management with Botulinum Toxin in Post-stroke and Multiple Sclerosis Patients: A Retrospective, ‘Real-world’, Multicenter Study
Supplemental material, sj-pdf-1-nre-10.1177_10538135251329322 for Spasticity Management with Botulinum Toxin in Post-stroke and Multiple Sclerosis Patients: A Retrospective, ‘Real-world’, Multicenter Study by Calogera Butera, Marcello Esposito, Trinchillo Assunta, Morena Giovannelli, Anna Rivaroli, Mario Pata, Mathieu Beneteau, Massimo Filippi, Patrizia M Caglioni and Ubaldo Del Carro in NeuroRehabilitation
Footnotes
Acknowledgements
The authors thank all patients involved in the study, as well as their caregivers, care team, investigators and research staff in participating institutions. The authors thank Anita Chadha-Patel, PhD, of ACP Clinical Communications Ltd (Hertfordshire, UK) for providing medical writing support, and Virginie Denis Greiss of Illustration Medicale (Paris, France) for producing the figures, which were funded by Ipsen (Paris, France, and Milan, Italy) in accordance with Good Publication Practice guidelines.
Ethical Considerations
The study was conducted in compliance with the International Society for Pharmacoepidemiology (ISPE) Guidelines for Good Pharmacoepidemiology Practices. Independent Ethics Committee/ Institutional Review Board approval was obtained prior to each center initiation and data collection.
Informed Consent
Informed consent was not required for this retrospective analysis of medical records, however patients who were still undergoing treatment at the centers were asked to provide written consent prior to data extraction.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by Ipsen.
Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Calogera Butera, Marcello Esposito, Trinchillo Assunta, Morena Giovannelli, Massimo Filippi, and Ubaldo Del Carro, were investigators in the RESULT study, and they or their institutions received payment from Ipsen. In addition, Massimo Filippi is Editor-in-Chief of the Journal of Neurology, Associate Editor of Human Brain Mapping, Neurological Sciences, and Radiology; and received compensation for consulting services from Alexion, Almirall, Biogen, Merck, Novartis, Roche, Sanofi; speaking activities from Bayer, Biogen, Celgene, Chiesi Italia SpA, Eli Lilly, Genzyme, Janssen, Merck-Serono, Neopharmed Gentili, Novartis, Novo Nordisk, Roche, Sanofi, Takeda, and TEVA; participation in Advisory Boards for Alexion, Biogen, Bristol-Myers Squibb, Merck, Novartis, Roche, Sanofi, Sanofi-Aventis, Sanofi-Genzyme, Takeda; scientific direction of educational events for Biogen, Merck, Roche, Celgene, Bristol-Myers Squibb, Lilly, Novartis, Sanofi-Genzyme; he receives research support from Biogen Idec, Merck-Serono, Novartis, Roche, the Italian Ministry of Health, the Italian Ministry of University and Research, and Fondazione Italiana Sclerosi Multipla. Anna Rivaroli, Mario Pata, Mathieu Beneteau, and Patrizia M. Caglioni are employed by Ipsen.
Data Availability
Qualified researchers may request access to patient-level study data that underlie the results reported in this publication. Additional relevant study documents, including the clinical study report, study protocol with any amendments, annotated case report form, statistical analysis plan and dataset specifications may also be made available. Patient level data will be anonymized, and study documents will be redacted to protect the privacy of study participants.
Where applicable, data from eligible studies are available 6 months after the primary manuscript describing the results has been accepted for publication. Further details on Ipsen's sharing criteria, eligible studies and process for sharing are available here (https://vivli.org/members/ourmembers/). Any requests should be submitted to ![]() for assessment by an independent scientific review board.
for assessment by an independent scientific review board.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
