Abstract
Background:
Neonatal fragment crystallizable receptor (FcRn) blockers selectively inhibit FcRn-mediated IgG recycling, resulting in degradation of IgG autoantibodies, including thyrotropin receptor autoantibodies (TSH-R-Ab).
Methods:
We describe a case involving a 55-year-old woman with a 17-month history of Graves’ disease (GD) who was hyperthyroid despite 15 months on methimazole (MMI) 15–25 mg/day. She received the investigational FcRn blocker batoclimab in a clinical study and was subsequently followed in clinic.
Results:
Within 1 week of starting weekly subcutaneous batoclimab, serum free triiodothyronine (FT3) and thyroxine (FT4) were normalized. Thyrotropin (TSH) was normal within 4 weeks. MMI was discontinued by study week 6. Twenty-three months after stopping batoclimab, the patient remained off MMI, with normal FT3, FT4, and TSH. Total IgG returned to approximate baseline levels 18 months post discontinuation of batoclimab, whereas TSH-R-Ab, which was elevated at baseline, remained below the upper limit of normal 23 months post batoclimab discontinuation.
Conclusions:
FcRn blockade may be an effective and potentially disease-modifying treatment for GD.
Introduction
Graves’ disease (GD) and its extrathyroidal manifestations are driven by IgG autoantibodies (Ab) targeting the thyrotropin receptor (TSH-R).1–3 Current therapies are limited to antithyroid drugs (ATD) and definitive treatments, including radioactive iodine (RAI) and thyroidectomy.1,4 After decades of limited progress, new non-thyroid-ablating treatments targeting the underlying immune dysregulation of GD are in development. 5 Neonatal fragment crystallizable receptor (FcRn) blockers are a novel class of therapeutic agents that selectively inhibit FcRn-mediated IgG recycling, thereby accelerating the degradation of IgG, including pathogenic IgG Ab. As such, FcRn blockers represent a target-specific immunotherapy with potential broad application across multiple IgG Ab-mediated autoimmune diseases.6,7
Batoclimab is an FcRn blocker that has been investigated as a treatment for a variety of autoimmune disorders.8–10 Findings from a phase 2a proof-of-concept study and a phase 2b randomized placebo-controlled trial enrolling adult patients with moderate-to-severe Graves’ orbitopathy (GO, also known as thyroid eye disease) showed batoclimab administration resulted in decreases in TSH-R-Ab and total IgG, as well as improvements in eye signs, including proptosis and diplopia.8,11 In the phase 2b trial, statistically significant reductions from baseline in free serum triiodothyronine (FT3) and free serum thyroxine (FT4) were observed within 1–2 weeks of initiating batoclimab treatment, 8 suggesting a possible effect of batoclimab on GD itself.
Based on these observations, an open-label, single-center, proof-of-concept study (NCT05907668) was designed to investigate whether batoclimab treatment could reduce elevations in FT3 and FT4, without the need to increase ATD dose, in hyperthyroid, TSH-R-Ab–positive adult participants with GD. Study participants were to receive 12 weeks of treatment with batoclimab 680 mg, administered subcutaneously (SC) once a week (QW), followed by 12 weeks of treatment with batoclimab 340 mg SC QW. At the end of the treatment period, those with FT3 and FT4 levels ≤ upper limit of normal (ULN) entered a 24-week batoclimab-free follow-up period. Preliminary results showed that by week 2, 60% of the study population achieved FT3 and FT4 ≤ULN, without an increase in ATD dose. 12 By week 12, 80% of participants met this endpoint, including 60% who were off ATD. Euthyroidism and reductions in ATD dose observed during batoclimab treatment were sustained in many participants for at least six months after treatment was stopped. 12 Completed results from the full study are forthcoming.
Here, we present long-term follow-up outside the study for a participant who became euthyroid and ATD-free after an abbreviated course of batoclimab treatment. This participant prematurely discontinued batoclimab and withdrew from the study due to a nontreatment-related adverse event (elevated liver enzymes related to choledocholithiasis). However, subsequent standard-of-care monitoring post-study discontinuation demonstrated persistent ATD-free euthyroidism for nearly two years after receiving the last dose of batoclimab. Case details represent the first-ever clinical evidence of long-term disease remission after FcRn blockade in a patient with Graves’ hyperthyroidism.
Ethics
The clinical study in which this patient took part was conducted in accordance with the Declaration of Helsinki, the Council for International Organizations of Medical Sciences International Ethics Guidelines, and all applicable laws and regulations, including Title 21 of the Code of Federal Regulation and EU Clinical Trials Regulation No. 536/2014. The protocol was approved by the Ethics Committee of the Rhineland-Palatinate Medical Association. The patient described herein consented to the publication of this report.
Patient
The patient is a woman who was 55 years old with a 17-month history of GD at the time of study entry. She was a non-smoker and had a baseline body mass index of 25.9 kg/m2. She had previously received 15 months of therapy with methimazole (MMI) at doses ranging from 15 to 25 mg/day; nevertheless, she was overtly hyperthyroid. Multiple attempts to titrate off ATD in the previous 15 months, as well as attempts to control with block-and-replace therapy, were unsuccessful, and she continued to deteriorate clinically. Extrathyroidal symptoms and signs included anxiety, retrobulbar pressure, headache, tremor, and bulging eyes. Beyond the clinical symptoms, the patient reported a substantial negative impact of GD on her quality of life (QoL), including social isolation due to self-consciousness about her appearance and an inability to work for the previous three months. Despite the unrelenting, long-term nature of her disease, definitive therapy was not introduced due to the potential for worsening GO with RAI, 13 as well as patient concerns about the potential impact of thyroid surgery on her ability to speak, which would have significantly affected her job performance. The treating clinician (and principal study investigator, G.J.K.) therefore recommended enrollment in the proof-of-concept batoclimab study, with a view toward reducing TSH-R-Ab levels and stimulation of the thyroid gland.
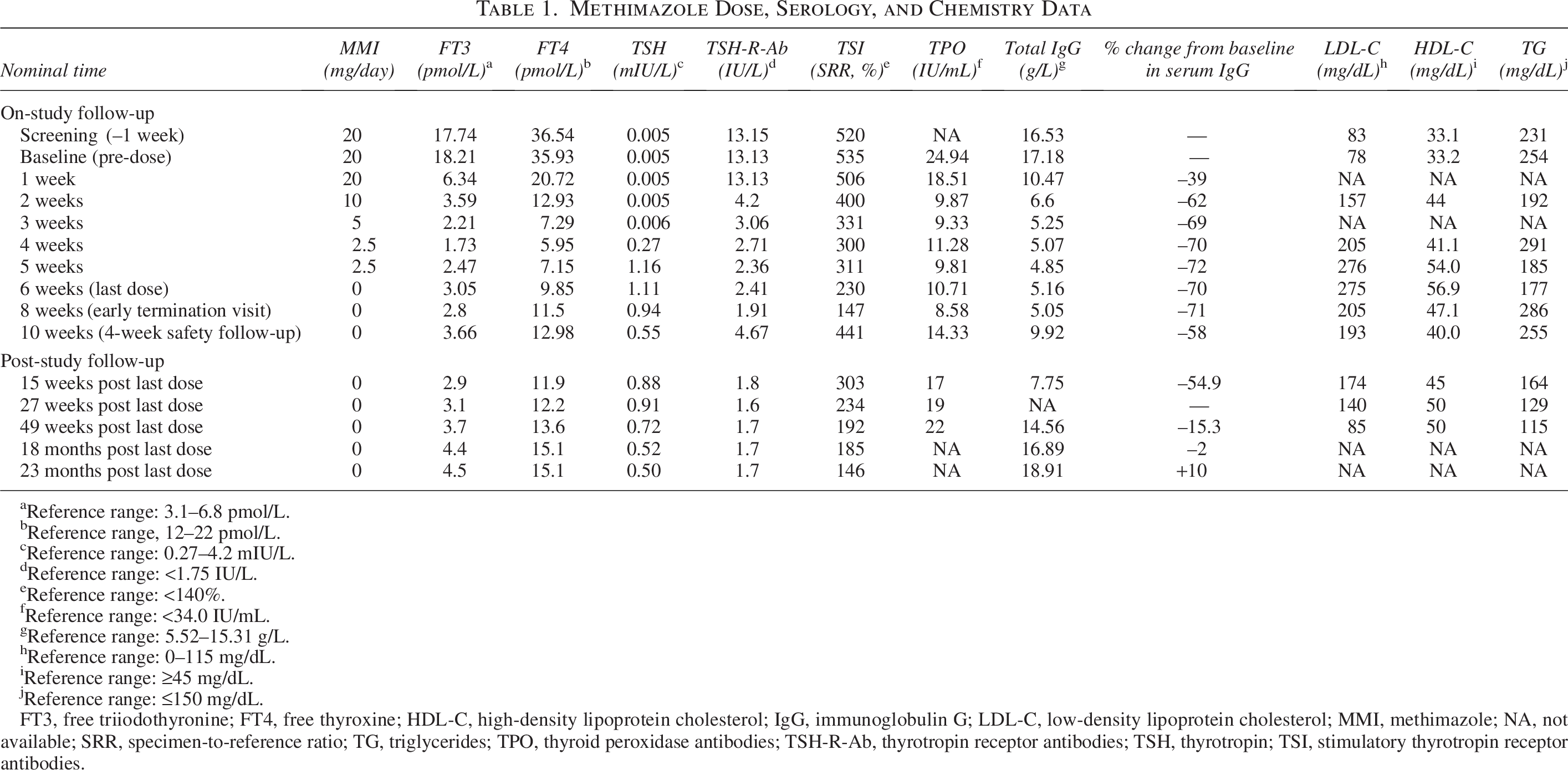
At the screening visit (week –1), serum FT3, FT4, total TSH-R-Ab (COBAS® eCLIA, 14 Roche, Switzerland), and stimulatory TSH-R-Ab (TSI) (Thyretain®,14–16 QuidelOrtho, CA, USA) were elevated, and thyrotropin (TSH) was suppressed (Table 1). Low-density lipoprotein cholesterol (LDL-C) was within the reference range, while high-density lipoprotein cholesterol (HDL-C) was low. The participant was on an MMI dose of 20 mg/day. These observations remained stable through the baseline visit (week 0), which occurred one week later (Table 1).
Methimazole Dose, Serology, and Chemistry Data
Reference range: 3.1–6.8 pmol/L.
Reference range, 12–22 pmol/L.
Reference range: 0.27–4.2 mIU/L.
Reference range: <1.75 IU/L.
Reference range: <140%.
Reference range: <34.0 IU/mL.
Reference range: 5.52–15.31 g/L.
Reference range: 0–115 mg/dL.
Reference range: ≥45 mg/dL.
Reference range: ≤150 mg/dL.
FT3, free triiodothyronine; FT4, free thyroxine; HDL-C, high-density lipoprotein cholesterol; IgG, immunoglobulin G; LDL-C, low-density lipoprotein cholesterol; MMI, methimazole; NA, not available; SRR, specimen-to-reference ratio; TG, triglycerides; TPO, thyroid peroxidase antibodies; TSH-R-Ab, thyrotropin receptor antibodies; TSH, thyrotropin; TSI, stimulatory thyrotropin receptor antibodies.
The patient subsequently received seven doses of batoclimab 680 mg SC QW. Within two weeks of the first dose, marked reductions in total IgG, TSH-R-Ab, and TSI serum levels were observed, with binding TSH-R-Ab decreasing to minimally elevated serum levels by week 8 (Table 1). Assessments of thyroid peroxidase antibodies were negative at baseline and remained so throughout treatment (Table 1), and thyroglobulin antibodies were consistently below the lower limit of detection. Serum FT3 and FT4 reached normal levels by week 1, and TSH was normal by week 4. By week 6, the patient discontinued MMI treatment (Table 1). Consistent with previous reports,8,17 batoclimab treatment was associated with an increase in LDL-C that reversed once treatment was stopped. Increases in HDL-C were also observed with treatment (Table 1).
Serological changes were accompanied by clinically relevant improvements, including reductions in thyroid volume (10 mHz Hitachi ultrasound, Japan), proptosis and lid aperture (Table 2). In addition, QoL improvements were recorded using the 39-item Thyroid-Related Patient-Reported Outcome scale. At baseline, the patient reported that her GD affected her appearance “very much,” with “some” impact on her overall QoL; after completing her final dose, she indicated that her GD affected her appearance “at little,” and overall QoL “not at all.” Notably, she was able to return to full-time professional work within a few weeks of initiating batoclimab, after having been on sick leave for the previous three months.
Clinical Outcomes Over Time
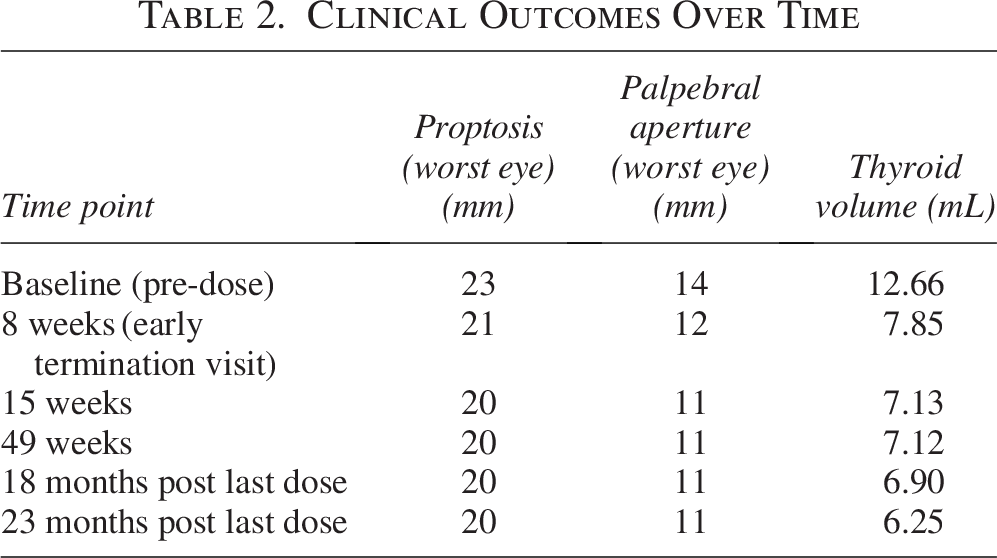
Batoclimab treatment was permanently discontinued after the seventh dose due to a gallstone-induced increase of liver enzymes. This adverse event was considered unrelated to batoclimab, as the patient had a prior history of cholelithiasis that was ongoing at the time of study entry; in addition, liver enzymes rapidly normalized after an uncomplicated endoscopic cholecystectomy. The patient completed the safety follow-up visit 4 weeks after the last batoclimab dose and was prematurely discontinued from the study; thereafter, the principal investigator continued to monitor her closely as part of routine clinical practice. During that time, the patient received no additional treatment for GD, GO, or any other systemic illness, and there were no significant life events or comorbid conditions that could have impacted the trajectory of her GD. Twenty-three months after her last dose of batoclimab, she remained euthyroid (i.e., normal FT3, FT4, and TSH) and ATD-free. Serum TSH-R-Ab levels continued to trend down and were minimally elevated at the last follow-up (compared with > 7.5×ULN at screening), and TSI levels were reduced by 72%; conversely, total IgG levels returned to pre-treatment baseline values (Table 1). Clinically relevant improvements in thyroid volume, proptosis, and palpebral aperture were also maintained off batoclimab (Table 2). Her clinical observation is ongoing, with the current trajectory showing excellent physical and self-reported psychological well-being, as well as continued ability to work full-time.
Discussion
This case represents the first-ever clinical evidence that FcRn blockade is associated with long-term remission in a patient with GD who was previously hyperthyroid despite 15 months on moderate-to-high-dose ATD, including an attempted block-and-replace regimen. Improvements in FT3 and FT4 were observed after a single dose of batoclimab, and normal thyroid hormone levels were maintained after MMI was discontinued at week 6. The patient discontinued batoclimab after seven doses due to a nontreatment-related episode of worsening choledocholithiasis. Nevertheless, she has remained euthyroid for at least 23 months posttreatment, consistent with clinical remission of disease.
The underlying immunological mechanisms driving long-term disease remission are currently unclear, although several areas of biology may be involved. Thyroid hormones are known to promote a pro-inflammatory state, leading to the activation of macrophages, T cells, B cells, and dendritic cells. 18 Consequently, a positive feedback loop may develop whereby the secretion of TSH-R-Ab increases the production of T3 and T4, which in turn further induces the production of TSH-R-Ab. 19 There is also evidence suggesting that TSH may directly stimulate increased production of CD19+ B cells and natural killer T cells. 20 By rapidly reducing the levels of circulating TSH-R-Ab, FcRn blockade may help break this cycle, allowing the restoration of self-tolerance and the reduction of thyroid hormone-driven inflammation and production of TSH-R-Ab.
The authors acknowledge the scientific limitations of interpreting results based on observations from a single patient. However, this patient’s robust response to batoclimab treatment within a few weeks, after 15 months of severe, intransient disease, is compelling. In addition, while long-term follow-up for this patient was not captured as part of the clinical study, subsequent standard-of-care monitoring provides strong evidence of disease remission lasting nearly two years after stopping batoclimab treatment. Definitive evidence linking these improvements in Graves’ hyperthyroidism to FcRn blockade may come from currently ongoing randomized placebo-controlled clinical trials (NCT07018323, NCT06727604).
Conclusions
Collectively, these results support FcRn blockade as a potentially effective mechanism for the treatment of GD and suggest the possibility of disease modification. While these results are from a single case, they are promising, and if confirmed in ongoing randomized placebo-controlled clinical trials, would represent important progress in a treatment landscape that has remained unchanged for decades.
Authors’ Contributions
G.J.K.: Conceptualization, methodology, investigation, formal analysis, writing—original draft, review, and editing. J.W.: Investigation, writing—review and editing. M.L.: Investigation, writing—review and editing. K.A.: Formal analysis, writing—review and editing. W.M.: Conceptualization, methodology, formal analysis, writing—original draft.
Footnotes
Acknowledgments
The authors thank Liz Rockstein, PhD, of Immunovant, Inc., for editorial support in the development of this publication. Interim results from the full study were previously presented at the 2025 Annual Meeting of the American Thyroid Association: Kahaly GJ, Wolf J, Ganz AL, et al. Batoclimab induces very rapid and sustained post-treatment euthyroidism in Graves’ disease Thyroid. 2025;35(Suppl 1), e1-e63.
Author Disclosure Statement
G.J.K.: The JGU Medical Center receives research-associated funding from Immunovant, Inc., and G.J.K. is a consultant and clinical investigator for Immunovant, Inc. J.W.: Nothing to disclose. M.L.: Nothing to disclose. K.A.: Employee of Immunovant, Inc. W.M.: Employee of Immunovant, Inc.
Funding Information
This study was funded by Immunovant, Inc. G.J.K.: Nothing additional to disclose. J.W.: Nothing additional to disclose. M.L.: Nothing additional to disclose. K.A.: Nothing additional to disclose. W.M.: Nothing additional to disclose.
