Abstract
Background:
A finely tuned balance between excitation and inhibition is essential for proper brain function. Disruptions in the GABAergic system, which alter this equilibrium, are a common feature in various types of neurological disorders. Understanding GABAergic neuron maturation processes is thus currently a major challenge in basic neuroscience. Thyroid hormones (THs) are required for the proper maturation of parvalbumin (PV)-expressing GABAergic interneurons in the mouse neocortex. However, the timeline of this TH action has yet to be elucidated. The aim of the present study was to define better the time window during which THs promote the postnatal maturation of PV neurons in the mouse neocortex.
Methods:
We used genetically engineered mouse models expressing dominant-negative mutations of the TH nuclear receptor α1 (TRα1). The consequences of blocking TH signaling at different times in development were assessed in PV neurons of the somatosensory cortex, in terms of histology and gene expression.
Results:
Histological observations in mice revealed that the action of THs during the first three postnatal weeks was necessary to initiate the expression of PV and the elaboration of a specialized extracellular matrix called the perineuronal net (PNN). By contrast, after the third postnatal week, TH action on PV neuron maturation appeared to be somewhat dispensable. Transcriptome analysis of neocortical GABAergic neurons two weeks after birth identified a small set of putative target genes for TRα1. Several of these genes are involved in the postnatal remodeling of the repertoire of ion channels within PV neurons and in the elaboration of PNNs.
Conclusions:
These data suggest that THs act as a timer to define the temporal boundaries of the critical period of heightened cortical plasticity, which plays a fundamental role in the development of neuronal circuits. Unveiling the molecular underpinnings of TH action in PV neurons may help understand better neurological disorders associated with alterations of TH signaling, such as hypothyroidism, resistance to THs, or Allan–Herndon–Dudley syndrome, but also more widely, neurological disorders associated with an imbalance in the excitation/inhibition ratio in the brain, including attention-deficit/hyperactivity disorder, autism, and epilepsy.
Introduction
Parvalbumin-expressing neurons (PV neurons) are GABAergic inhibitory interneurons that play a central role in the organization of neuronal circuits in the neocortex. 1 They are fast-spiking, synchronize network oscillations, and play an important role in information processing. In mice, PV neurons begin to express parvalbumin (PV) during the first postnatal weeks.2,3 During this time, the electrical activity of these neurons progressively increases, 4 and synaptogenesis is very active. This opens the so-called critical period of heightened synaptic plasticity 5 during which sensory input and experience shape neuronal networks. The critical period ends when circuit rewiring becomes actively dampened. 6 Notably, the elaboration of the perineuronal net (PNN) around PV neurons, a densely packed extracellular matrix enriched in proteoglycans, stabilizes synapses and restricts plasticity. 7 Defects in PV neuron maturation during the critical period cause a persistent imbalance between neuronal excitation and inhibition, which is at the origin of several pathological conditions, including attention-deficit/hyperactivity disorder, autism, and epilepsy. 8
Thyroid hormones (THs; including triiodothyronine [T3] or 3,3′,5-triiodo-
To gain more insight into the timeline of TH action on PV neuron maturation, we generated novel mouse models with restricted or inducible expression of mutant TRα1 receptors. We evidenced a lifelong persistence of defects in PV neuron maturation when TH signaling was blocked in GABAergic neurons from early stages of development. Transcriptome analysis identified a set of genes whose regulation is expected to be involved in TH-mediated maturation of PV neurons and in the closure of the critical period.
Materials and Methods
Mouse models and treatments
Experiments involving the use of live animals for the current project were approved by local ethics committees (C2EA015 and C2EA017) and subsequently authorized by the French Ministry of Research (Projects #33279-2021082516194165 and #25496-2020032811174506).
The ThraAMI allele allows the expression of the dominant-negative TRα1L400R after Cre/loxP-mediated deletion of a cassette with polyadenylation signals. 18 ThraSlox is identical, except for the presence of a C-terminus frameshift mutation, which leads to the expression of the dominant-negative TRα1E395fs401X. The ROSA-tdTomatolox reporter transgene (also known as Ai9, MGI Cat# 4436851, RRID: MGI: 4436851) drives the Cre-dependent expression of a red fluorescent protein, mainly localized in cytoplasm. 19 The ROSA-GSGFPL10lox transgene encodes the GS-EGFP-L10a protein, which is a green fluorescent protein fused to the N-terminus part of the large subunit ribosomal protein L10a, mainly localized in cell nuclei. 20 All transgenes were in the C57Bl6/J genetic background. Gad2-Cre and Gad2-CreERT2 are knock-in alleles in which the Cre recombinase reading frame is inserted into the Gad2 gene in order to restrict the expression of the recombinase in GABAergic neurons. 21 In Gad2-Cre mice, Cre induces DNA recombination from around E12.5, whereas in Gad2-CreERT2 mice, CreERT2 is translocated to the cell nucleus only after mice are given tamoxifen (T5648, Sigma, 10 mg/mL in sunflower oil), which allows for control of the time at which a mutated TRα1 starts to be expressed. PND7 mice received a single 50 µL subcutaneous tamoxifen injection, whereas 3-week-old mice received five daily intraperitoneal injections (daily dose: 7.5 µL/g).
Gestating mice were fed for 2 weeks with iodine-deficient food supplemented with 0.15% propyl-thiouracyl (PTU Envigo ref TD.95125) to cause deep hypothyroidism. TH levels were restored by a single intraperitoneal injection in half of the pups (10 µg T4 + 1 µg T3 dissolved in 50 µL of phosphate-buffered saline [PBS], all chemicals from Sigma Aldrich, France). The other PTU-treated animals were injected with 50 µL of PBS.
RNA sequencing analysis from sorted nuclei
Nuclei were isolated from whole individual cortex frozen in liquid nitrogen, and nuclear RNA was extracted as previously described. 20 Nuclear RNA was extracted from sorted nuclei (RNeasy Micro Kit, Qiagen ref 74004) and quantified using Tapestation4150 (Agilent). One nanogram of RNA was reverse-transcribed using the SMART-Seq v4 Low Input RNA kit (Takara). Complementary DNA (cDNA) was quantified and qualified using Qubit (Invitrogen) and Tapestation4150 (Agilent). Libraries were then prepared from 1 ng cDNA using the Nextera XT DNA Library Kit (Illumina). Libraries were sequenced (>2.107 reads/library) on a NextSeq500 DNA sequencer (Illumina). Reads were aligned on the mouse genome (mm10 GRCm38 release) with Bowtie2 (Galaxy Version 2.4.2 + galaxy 0). 22 Count table (Supplementary Table S1) was prepared using htseq-count (Galaxy Version 0.9.1 + galaxy 1, mode union, feature type: gene). 23 Differential gene expression analysis was performed with DEseq2 (Galaxy Version 2.1.8.3) 24 (Supplementary Table S1) using the following thresholds: false discovery rate <0.05; p-adjusted value <0.05; maximum adjusted basecount >30; and fold change >2.0. Hierarchical clustering was performed using Euclidean distance and complete distance with ClustVis. 25 Gene ontology analysis was performed with Gorilla (https://cbl-gorilla.cs.technion.ac.il/). Raw RNA sequencing (RNA-seq) data are available at NCBI Gene Expression Omnibus (GSE290072 and GSE290207).
RT-qPCR analysis
RNAs were extracted using TRIzol (Invitrogen, Carlsbad, CA). Total RNA was converted to cDNA using M-MLV reverse transcriptase (Promega, Madison, WI). RT-qPCR was performed using SYBRGreen mix (BioRad iQ supermix). The results were analyzed according to the ΔΔCT method. 26 The housekeeping gene Hprt was used as the reference.
Brain slice histology
Brain slice collection was performed as described. 17 Coronal sections (50 μm) were cut with a vibrating microtome (Integraslice 7550 SPDS; Campden Instruments, Loughborough, UK), in PBS at room temperature. Brain sections were stored at −20°C in cryoprotectant (30% ethylene glycol and 20% glycerol in 10 mM low-salt PBS). Immunohistochemistry was performed on free-floating brain sections. For PV labeling, a mouse anti-PV primary antibody (PARV19, Sigma P3088, 1:2000) and a secondary antibody made in donkey (anti-mouse DyLight 633, ThermoFisher Scientific, 1:1000) were used. For the concurrent PNN labeling, a biotinylated Wisteria floribunda lectin (WFL/WFA, Vector Laboratories B-1355, 20 µg/mL) and streptavidin coupled to DyLight 488 (Vector Laboratories, SA-5488, 1:1000) were used. Nonspecific binding sites were blocked by incubating sections for 1 hour in PBS with 1% bovine serum albumin (BSA) and 0.2% Triton X-100. Brain sections were incubated overnight at 4°C with PV antibody and biotinylated WFA, which were diluted in PBS with 1% BSA, 0.2% Triton X-100, and 1% dimethyl sulfoxide. Sections were washed in PBS and further incubated for 15 minutes at room temperature with 4’,6-diamidino-2-phenylindole (1:5000, Sigma). Incubation with the secondary antibody lasted for three hours at room temperature. Sections were mounted in Fluoroshield™ (Sigma), coverslipped, and imaged using an inverted confocal microscope (Zeiss LSM 780 and Zeiss LSM 800).
Image analysis
Image analysis was performed using ImageJ. Final images resulted from the z projection of three optical sections (maximum intensity tool) that were 2 µm apart (the resulting images thus reflected the fluorescence collected over a thickness of 4 µm). PV+ WFA+ Tomato+ cells were detected from final images using the Cellpose wrapper for Fiji. 27 Numbers of mono-, double-, or triple-labeled cells per slice were measured automatically from 3 to 4 slices per animal. The code of the original macro may be found at https://github.com/jbrocardplatim/MacroPV.
The densities of Tomato+, PV+, WFA+ cells, and their colocalizations were counted in the whole area of one image taken in the somatosensory cortex. Three to four images in each hemisphere were analyzed for each mouse. Quantitative data were analyzed using a two-tailed unpaired T-test. Data are expressed as mean ± standard error of the mean. Data analyses were performed with Excel 2016 and GraphPad Prism software (v.8). The level of significance was set at p < 0.05.
Results
TRα1E395fs401X expression in GABAergic neurons from an early developmental stage permanently inhibits PV neuron maturation
As the expression of TRα1L400R in the GABAergic lineage, starting at embryonic stage, is lethal after birth, 17 we used TRα1E395fs401X hypomorphic mutation, whose expression can be triggered by Cre/loxP recombination from the ThraSlox allele 28 to address the long-term consequences of preventing T3 response in GABAergic neurons. One-third of ThraSlox/+ ROSA-tdTomatolox/+ Gad2-Cre mice survived to adulthood, while all the Thra+/+ ROSA-tdTomatolox/+ Gad2-Cre littermates survived and were used as controls.
Red fluorescence allowed one to trace the entire GABAergic lineage and did not indicate any cell loss (Supplementary Fig. S1). On the contrary, the density of PV+ cells was drastically reduced in mutant mice (Fig. 1a, b). The density of WFA+ cells (Fig. 1a, c) and the ratio of PV+ cells labeled with WFA were reduced (Fig. 1a, d). Some recovery of PV+ cell density was observed at the adult stage (Fig. 1e, f), while the PNN defect persisted (Fig. 1e, g, h). Therefore, early TRα1E395fs401X expression permanently affects the maturation of PV neurons.
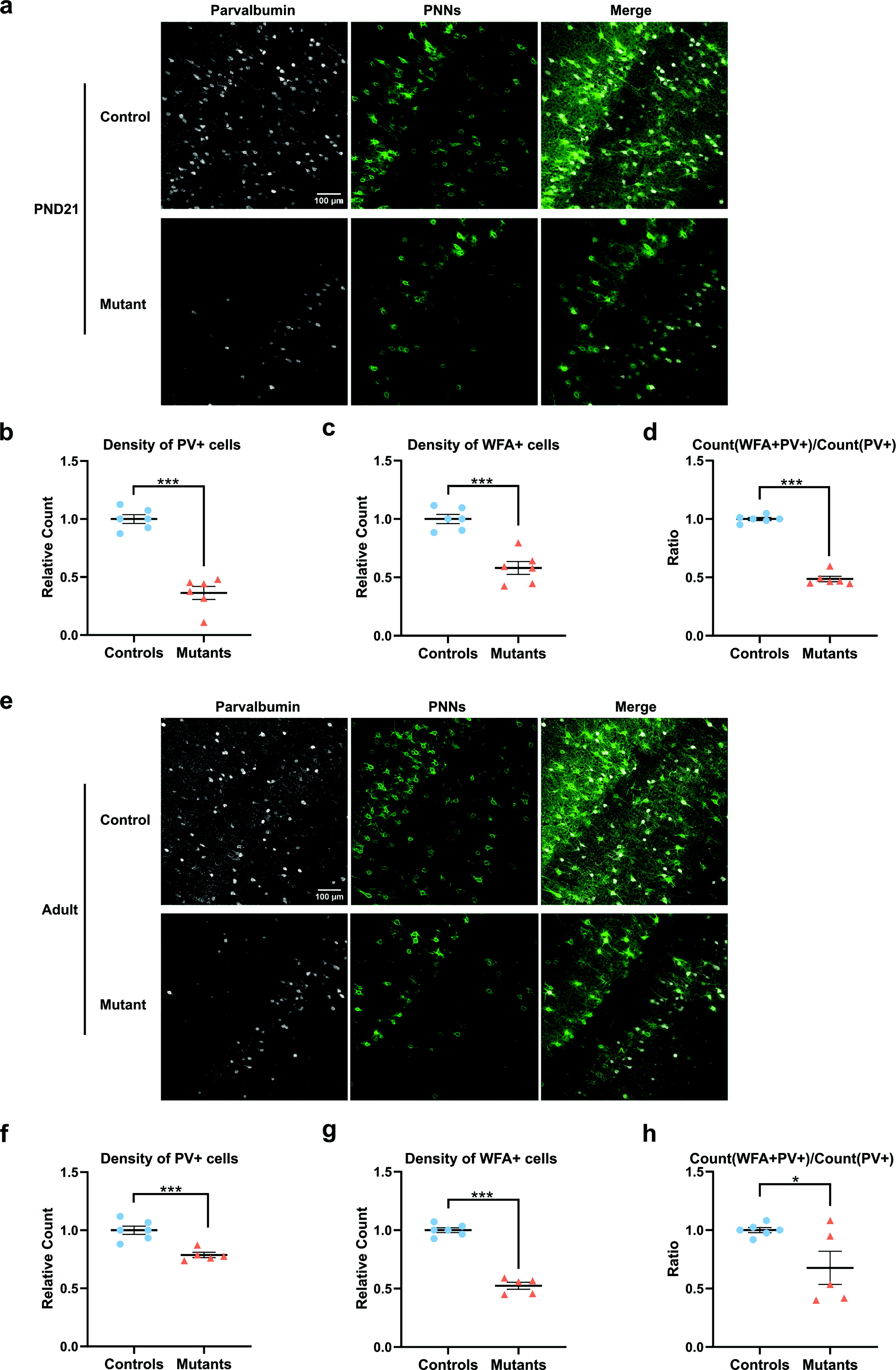
Persistent defect in PV neuron maturation in mice expressing TRα1E395fs401X. Thra+/+ ROSA-tdTomatolox/+ Gad2-Cre littermates (controls) and ThraSlox/+ ROSA-tdTomatolox/+ Gad2-Cre mice (mutants) express TRα1E395fs401X and a red fluorescent protein only in GABAergic neurons and in their progenitors from embryonic day 12.5. The neocortex of mutant mice did not display any major anatomical alteration. Histological analysis of the somatosensory neocortex at PND21 (
Postnatal expression of TRα1L400R in GABAergic neurons is sufficient to inhibit PV neuron maturation
We replaced the Gad2Cre driver with the Gad2-CreERT2 inducible version. This allows for the induction of TRα1L400R expression by tamoxifen treatment at a chosen time and addresses a possible temporal restriction on T3 action in GABAergic neurons. Treatment with tamoxifen at PND7 was sufficient to significantly reduce PV+ cell density at PND14 (Fig. 2a, b). The density of WFA+ cells (Fig. 2a, c) and the fraction of PV+ cells labeled with WFA (Fig. 2a, d) remained unchanged, perhaps because the elaboration of PNNs was not completed at this early stage. By contrast, a treatment with tamoxifen between PND21 and PND25 did not have visible histological consequences in the adult stage (Fig. 2e–h). Therefore, THs exert their influence during a postnatal time window, which includes the second postnatal week and precedes weaning (∼PND25).
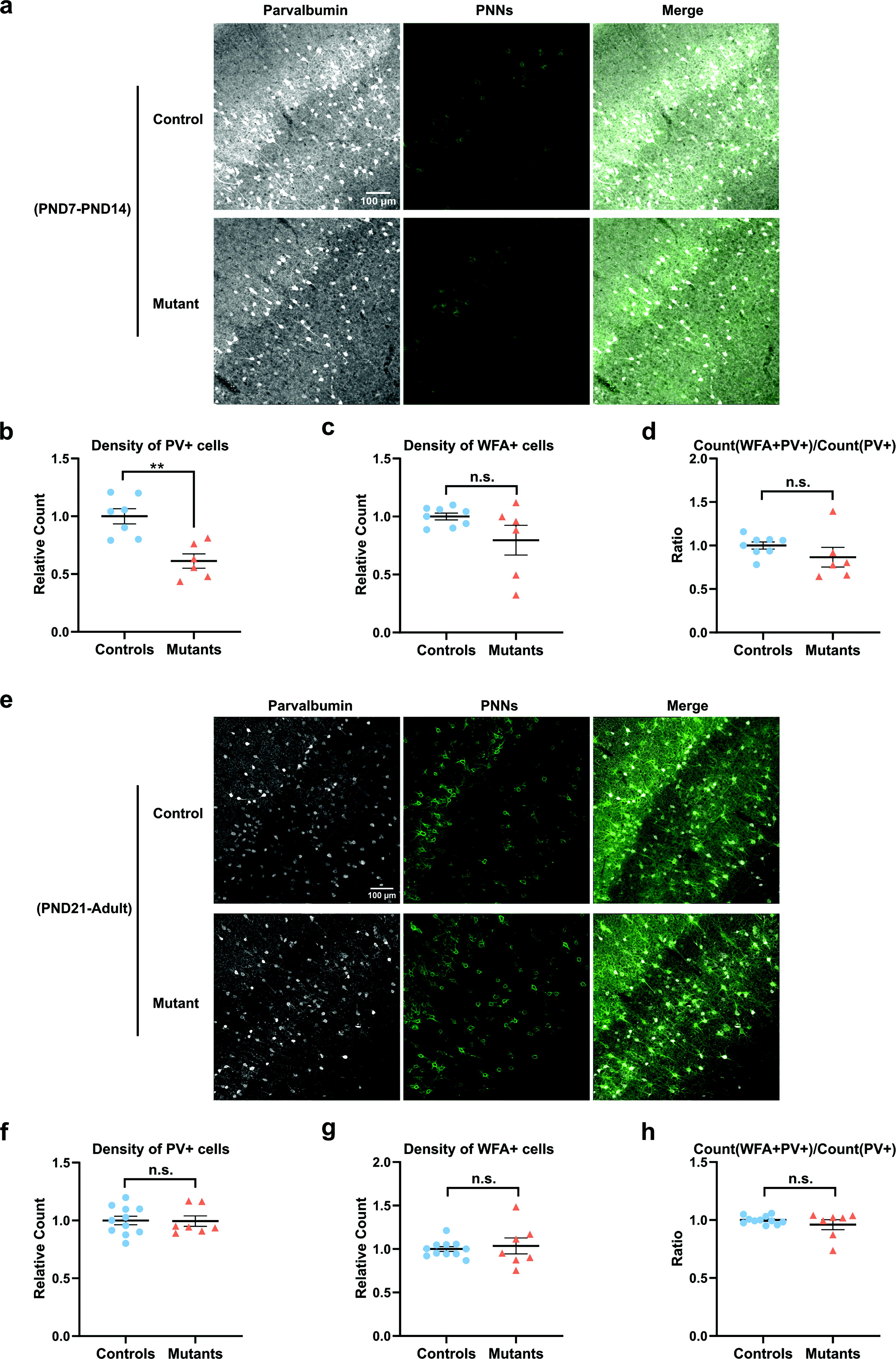
A time window for TH action on PV neuron maturation. Histological analysis of Thra+/+ ROSA-tdTomatolox/+ Gad2-CreERT2 or Gad2-CreERT2 negative littermates (controls) and ThraAMI/+ ROSA-tdTomatolox/+ Gad2-CreERT2 mice (mutants) that express TRα1L400R in the GABAergic lineage from PND7 to PND14 (
Consequences of TRα1L400R expression on gene expression in neocortical GABAergic neurons
We used the ROSA-GSGFPL10lox reporter transgene, which labels cell nuclei, to extract nuclear RNA from neocortical GABAergic neurons at PND15 and perform a bulk RNA-seq analysis. A total of 1306 genes were sensitive to either TRα1L400R expression, hypothyroidism, or TH stimulation (Deseq2 adjusted p-value >0.05; fold change >2.0; and basemean >30; Fig. 3a, b). Among them, a large set of genes was only deregulated in hypothyroid mice. In the end, only 61 genes behaved as bona fide TRα1 target genes, with a robust negative response to both TRα1L400R expression and hypothyroidism, and clear induction after short TH treatment of hypothyroid mice (Fig. 3a). Clustering analysis revealed a general trend toward recovery of hypothyroid mice after TH treatment (Fig. 3c, d). Five genes appeared to be negatively regulated by T3-bound TRα1 (Fig. 3b). qPCR analyses carried out on the cortex of mice with different circulating TH levels confirmed part of the results of the RNA-seq analysis (Supplementary Fig. S2). Notably, many of the genes identified as regulated by TH/TRα1 signaling are expressed in PV neurons (Supplementary Table S2).
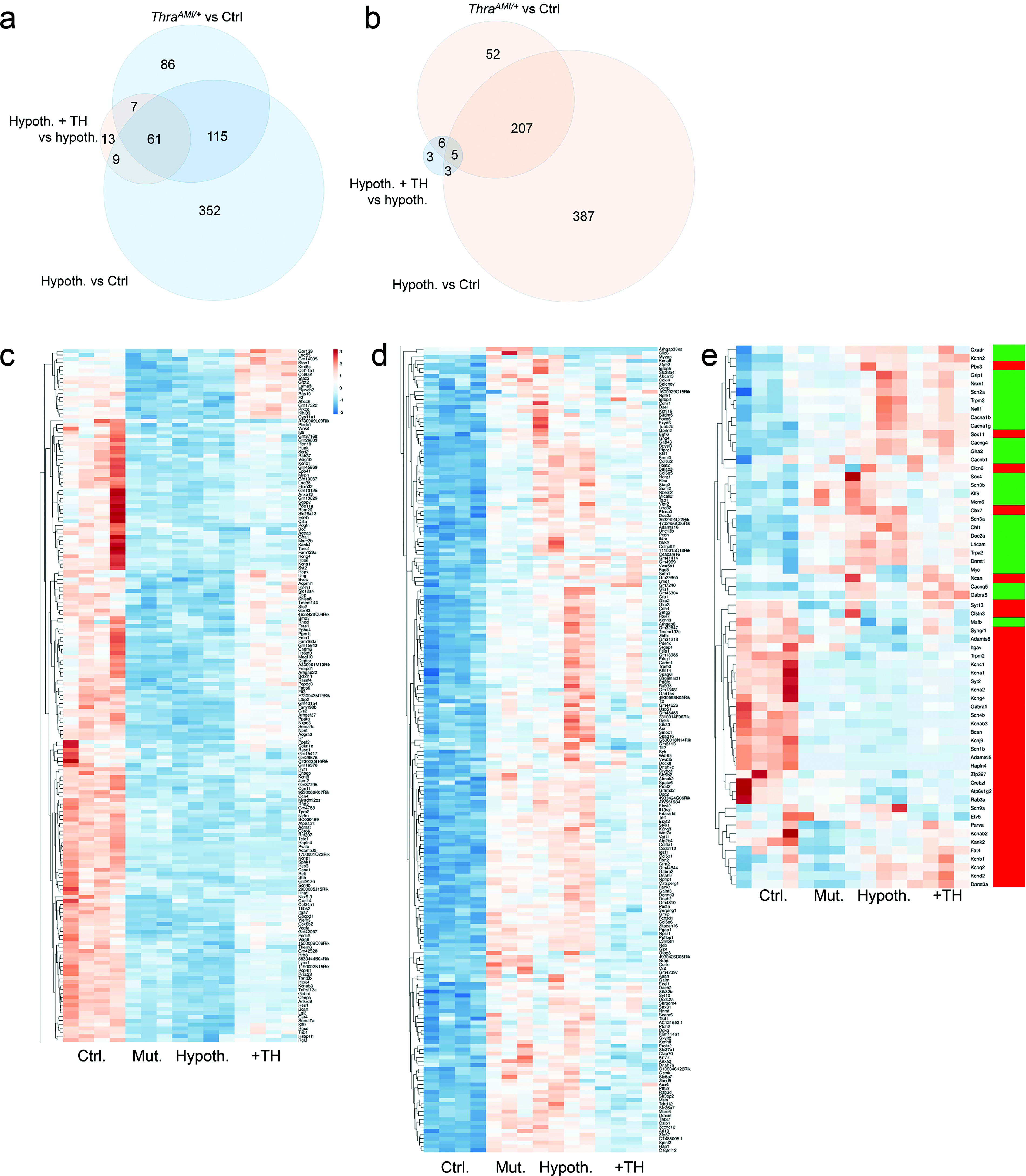
RNA sequencing analysis of gene expression in sorted GABAergic neuron nuclei. Taking advantage of the selective expression of a fluorescent protein present in the nuclei, RNA was extracted from nuclei of GABAergic neurons sorted from the neocortex of four groups of PND14 mice. Thra+/+ ROSA-GSGFPL10lox Gad2-Cre mice (Ctrl group) are control transgenic mice that express only fluorescent protein. ThraAMI/+ ROSA-GSGFPL10lox Gad2-Cre mice (Mut group) express TRα1L400R in GABAergic neurons. Thra+/+ ROSA-GSGFPL10lox Gad2-Cre mice were made hypothyroid by a PTU treatment started during gestation (Hypoth. group). Some of these hypothyroid Thra+/+ ROSA-GSGFPL10lox Gad2-Cre mice received a single TH injection at PND13 (hypoth. +TH group). The rapid response of genes after TH treatment is a hallmark of genes whose transcription is directly regulated by the liganded TRα1 nuclear receptor. (
Gene ontology analysis indicated that many of the genes deregulated in hypothyroid and mutant mice encode components of the extracellular matrix (GO:0031012; enrichment 4.4, p-value 3.10−13). This includes genes encoding collagens or proteins binding to the glycosaminoglycans of PNNs (GO:0005539; enrichment 4.2, p-value 5.10−6). The expression pattern of two of these genes (Sema7a, encoding semaphorin 7A and Hapnl4, encoding hyaluronan and proteoglycan link protein 4) identifies them as genuine TRα1 target genes. This observation is consistent with the hypothesis that TH/TRα1 signaling stimulates the elaboration of PNNs around PV neurons.
The large group of genes that are deregulated in hypothyroid and mutant mice but are not responsive to TH stimulation is also enriched in voltage-gated potassium channels (GO:0005249; enrichment 6.1, p-value 10−6). We extracted the genes that mainly encode ion channels and whose expression changes during postnatal maturation of GABAergic neurons. 4 This analysis (Fig. 3e) revealed that the immature status of PV neurons persists in hypothyroid and mutant mice at PND14.
Discussion
In the present study, we extend our previous observations showing that a cell-autonomous action of TH/TRα1 in GABAergic neurons is required for the maturation of neocortical PV neurons in mice. We identified a time window that spans from birth to the end of the third postnatal week, during which the action of THs is critical for the proper maturation of PV neurons. Analysis of gene expression also reveals that at this stage, TH/TRα1 normally activates the transcription of a small group of genes necessary for the elaboration of the PNN around PV neurons.
PV neurons show near-normal maturation when the blockade of TH/TRα1 signaling occurs at a late stage, or if it occurs only after the onset of the expression of Pvalb, the gene that encodes PV. 20 This confirms a previous whole-cortex RNA-seq analysis, which showed that gene expression changes rapidly in GABAergic neurons during the second postnatal week under the control of TH/TRα1 signaling. 17 Our novel transcriptome analysis indicates that blocking TH/TRα1 signaling compromises the expression of a number of genes involved in a well-documented transition, during which the repertoire of ion channels is extensively modified. 4 It is remarkable that the second postnatal week, during which THs seem to play a crucial role in PV neuron maturation, coincides with a peak in circulating levels of THs. 30 This strongly suggests that THs represent a temporal cue to synchronize the postnatal maturation of PV neurons.
We provide evidence that the postnatal maturation of PV neurons is promoted in a cell-autonomous manner. However, we did not notice any obvious change in the radial distribution of GABAergic neurons. Therefore, the reported defect in prenatal migration of GABAergic neuron progenitors might not be a cell-autonomous consequence of early TH deficiency. 31 A complete recovery of PV neuron density in adults has been reported for mice expressing TRα1R384C,14 which we did not observe for TRα1E395fs401. This discrepancy is probably explained by the residual capacity of TRα1R384C to bind T3 and activate transcription. 32 Furthermore, the late onset of TRβ1 expression in PV neurons 33 could be sufficient to counteract the negative influence of TRα1R384C.
The mode of action of THs in PV neurons during the second postnatal week is clarified by RNA-seq data. We found that PV neurons were much more sensitive to variations in TH signaling at PND14 than in adulthood, where the same method only showed minor changes in gene expression. 20 Another finding is that genes that are consistently deregulated by blocking TH/TRα1 signaling encode extracellular matrix proteins, specific components of or proteins interacting with PNN glycosaminoglycans in PV neurons. Rapid recovery of gene expression after TH treatment in hypothyroid mice provides a clear indication of direct transcriptional control by TRα1 for a small subset of genes. Deregulation of these genes is likely to represent the initial event preventing PV neuron maturation. Among these were genes encoding transcription factors or cofactors (Dbp, Hr, Klf9), also regulated by TH in other cell types 34 and again genes encoding PNN components. Thus, it appears that liganded TRα1 notably activates the transcription of genes encoding collagens (Col24a1 and Col9a2), ephrin A4 (Epha4), laminin alpha 3 subunit (Lama3), semaphorin 7A (Sema7a), hyaluronan, and proteoglycan link protein 4 (Hapln4), an essential component of PNNs.35–37
The novel data presented in this article provide strong support for the hypothesis that TH is a major temporal signal for the opening and closing of the critical period of heightened plasticity, as previously suggested. 38 The critical period occurs at slightly different times in different neocortical areas, as does the onset of TH signaling. 39 It is characterized by a series of cellular events that play a crucial role in the elaboration of functional neuronal circuits. 5 The beginning of PV synthesis marks the opening of the critical period, while the elaboration of PNNs around PV neurons stabilizes synapses and decides their closure. Thus, our findings suggest that both the opening and the closure of the critical period are directly stimulated by liganded TRα1. Therefore, THs appear to act in the postnatal neocortex as it does during amphibian metamorphosis, as an external cue that represents an important temporal signal. Of note, oligodendrocyte maturation, which is also a TH-dependent process,40,41 has also been shown to play a role in PV neuron maturation and in the closure of the critical period. 42
Patients with early TH deficiency caused by congenital hypothyroidism 43 or carrying TRα1 mutations 13 are currently treated with TH replacement therapy. Treatment can have a positive influence on cognitive development only if it is initiated soon after birth. This is an indication that the action of TH on the maturation of the human neocortex is restricted to an early temporal window, as we have found in mice. An alteration in the timing of the critical period is also suspected to cause autism spectrum disorders. 8 In that regard, it is noticeable that some of the rare patients in whom THRA mutations have been discovered were first diagnosed for autism spectrum disorders.44,45 Within the framework of our hypothesis, the irreversible cognitive and neurological defects that are seen both in patients with TH-related diseases and in patients with autism spectrum disorders might be similarly regarded as consequences of a disruption of the critical period of heightened cortical plasticity. Delving deeper into the detailed molecular events occurring in the cortex during the critical period will be key to finding therapeutic approaches to these debilitating pathologies.
Study limitation
Our study identifies TH-regulated genes in PV neurons but does not address chromatin binding of TRα1 to the neighboring regulatory sequences. This is not currently feasible with the small number of PV neurons sorted from the mouse neocortex. Also, the histological methods that we have used only indirectly inform on the timing of the critical period, which can be defined in a number of different ways.46,47
Conclusions
THs appear as major players of the critical period of heightened cortical plasticity, which is a decisive step in neurodevelopment. Unveiling the underpinnings of TH action in PV neurons should help understand neurological disorders deriving from alterations in TH signaling, as well as those associated with an imbalance in the excitation/inhibition ratio in the brain.
Authors’ Contributions
J.R.: Investigation, writing—original draft, and writing—review and editing. S.M., J.B., R.G., and D.A.: Investigation and writing—review and editing. J.W.: Supervision. F.F.: Conceptualization, funding acquisition, supervision, writing—original draft, and writing—review and editing. S.R.: Conceptualization, investigation, supervision, and writing—review and editing.
Footnotes
Acknowledgments
The authors acknowledge the contribution of SFR Biosciences (Université Claude Bernard Lyon 1, CNRS UAR3444, Inserm US8, ENS de Lyon): Nadine Aguilera and the Plateau de Biologie Expérimentale de la Souris (ANIRA-PBES) for mouse breeding; ANIRA-CYTOMETRIE and particularly Sébastien Dussurgey for nuclei sorting; and ANIRA-AGC for mouse genotyping. The authors thank Benjamin Gillet and Sandrine Hughes of the deep sequencing facility (PSI IGFL, Lyon). They also thank Elodie Martel for carrying out histology experiments as part of her internships and Isabelle Dusart (Institut de Biologie Paris Seine, France) for helpful discussions.
Author Disclosure Statement
The authors declare no conflicts of interest.
Funding Information
Funding for this research was provided by the China Scholarship Council (supporting Juan REN’s PhD project at ENS de Lyon), the European Union’s Horizon 2020 program (grant agreement No. 825753, ERGO), the Agence Nationale de la Recherche (Thyromut2 program, ANR-15-CE14-0011-01), and the Joint Research Institute for Science and Society (JORISS 2022, ![]() ).
).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
