Abstract
An unprecedented increase in the number and the size of yellow mealworm beetle (Tenebrio molitor) farms in North America and Europe has been driven, among other factors, by the demand for sustainable proteins. Insect farming offers ecologic advantages, including lower land and water use and reduced greenhouse gas emissions. As in conventional livestock farming, veterinarians are essential to maintain insect health because disease outbreaks can reduce productivity, compromise animal welfare, and cause monetary loss. Yet, the role of veterinary diagnosticians remains limited in this emerging sector because of the lack of standardized diagnostic tools and anatomic references. The autopsy of an invertebrate is a valuable tool that can, just as in any other species, serve to determine the cause of death, identify underlying disease processes, and guide herd health management. However, performing beetle autopsies is technically challenging given their small size, rigid exoskeleton, and fragile internal organs. Here, we offer an illustrated, step-by-step dissection protocol for adult T. molitor using standard laboratory equipment. Our method emphasizes an aqueous immersion approach that preserves tissue integrity, enhances visibility, and yields consistent results. We describe the abdominal anatomy of both sexes, including the digestive, reproductive, and nervous systems. Structures include 6 testicular follicles, bean-shaped accessory glands, and a spermathecal gland in females that exceeds the spermatheca in size. The abdominal nerve cord comprises 7 metameric ganglia, with caudal fusion.
The yellow mealworm beetle (Tenebrio molitor Linnaeus) is a widely introduced species in North America that is traditionally considered a pest in stored grain. T. molitor has been used as an alternative to vertebrate models in research, including preclinical trials, in part because of ethical concerns over the use of mammals, simpler maintenance without strict animal care regulations, and lower rearing costs.4,7 Furthermore, this insect has attracted increasing scientific interest for its role in addressing climate change and food insecurity. Over the past decade, research has highlighted its capacity to contribute to a circular economy by transforming organic waste, and even certain plastics, into frass that can be recycled as fertilizer within plant production systems. In addition, T. molitor efficiently converts waste into body mass, offering a cost-effective, high-quality alternative protein source.6,18,23,43 Insect farming is also more sustainable than conventional livestock production, requiring less land and water while generating lower ammonia and greenhouse gas emissions. 21
In recent years, T. molitor farms have increased in size and number across North America and Europe, reflecting the global growing attraction toward natural waste management and sustainable protein alternatives. 25 Supporting this expansion, the European Food Safety Authority recently approved frozen and dried yellow mealworm larvae as a novel food for human consumption. 11 Nutritionally, mealworms compare favorably to traditional protein sources such as beef, pork, poultry, and fish, and have higher digestibility than many plant-based proteins.9,21,32,37,39
Similar to conventional animal farming, veterinarians play a central role in safeguarding animal health in reared insects. 3 Indeed, viral and bacterial outbreaks have been reported in T. molitor herds, devastating insect farming operations.2,12 Microscopic examination revealed pathologic changes commonly observed across species, such as viral inclusions and necrosis in various organs, as well as more insect-specific alterations, including cuticular or gut discoloration or darkening and loss of body volume. Yet, because insect farming is still an emerging industry, diagnostic tools remain scarce, despite being essential for proper husbandry, as well as disease prevention and treatment. Therefore, a reliable baseline reference for the anatomy of this beetle is crucial for accurate diagnosis. Without such data, normal variations may be misinterpreted as lesions or, conversely, subtle but significant signs of disease may be overlooked. Standardized microdissection techniques are also essential for consistent sample harvesting and to maintain tissue integrity for subsequent examinations.
Autopsy, a cornerstone diagnostic tool in veterinary medicine, can also be applied to invertebrates. 10 However, its effectiveness relies on knowledge of normal anatomy and the availability of standardized dissection protocols. This is particularly important in insects, which have rigid exoskeletons and delicate internal organs suspended in hemolymph. To our knowledge, no comprehensive dissection manual for the adult yellow mealworm beetle exists. Searches in Google, PubMed, CAB Direct, Web of Science, and Scopus using the terms “dissection”, “anatomy”, and “yellow mealworm beetle” yielded no complete reference. Our objectives were therefore to 1) provide an illustrated and accessible dissection guide for the adult T. molitor and 2) map the male and female anatomy. Ultimately, we aim to standardize abdominal autopsy procedures and facilitate diagnostic efforts in insect farming.
Materials and methods
Adult T. molitor beetles were obtained (TriCycle Entotechnologies, Montréal, Canada) in 2024 and maintained in plastic bins at our laboratory in Saint-Hyacinthe, Québec, Canada. Colonies were kept under natural photoperiod conditions, with ambient temperature averaging 21°C (68.9°F) and relative humidity of 30–50%. Insects were given oat flakes and organic leafy greens ad libitum and were periodically supplemented with organic apples.
For dissection, we selected 2–4-mo-old adults with a fully dark-brown exoskeleton, indicative of sexual maturity. 22 The chosen individuals, ~50 males and 50 females, were free from apparent disease conditions or lesions at the time of euthanasia. Formal ethical approval is not required for experimental use of insects in Canada. Nevertheless, all procedures were designed to minimize suffering. Euthanasia was performed by 2-step immersion in isopropyl alcohol (5% then 99%; adapted from reference 17 ). Specimens were then air-dried for a few minutes before dissection.
For immobilization, each beetle was ventrally affixed with a drop of hot glue onto a piece of Styrofoam (DuPont). The foam was secured tightly at the bottom of a shallow plastic container that was subsequently filled with isotonic saline. Clothespins placed along the container’s rim prevented the foam from floating. Microdissections were carried out with the specimen fully submerged in isotonic saline, using a stereomicroscope (SMZ 161 Series; Leica), a microdissection instrument set (Imed Scientific), and a tungsten dissection needle (ultra fine tip; Roboz Surgical) mounted on a needle holder (RS-6063; Roboz Surgical).
Results
Abdominal dissection guide
The elytra and wings were completely removed using iris scissors. A horizontal incision was then made with a #11 scalpel blade or iris scissors along the pleural membrane of the abdomen, separating the sternites from the tergites while sparing the metathorax. To facilitate the cut, the pygidium was gently pinched with tweezers. At the level of the anus, the incision was directed slightly dorsally to avoid damaging the rectum. A fine dissection needle mounted on a holder could also be used to perform this step.
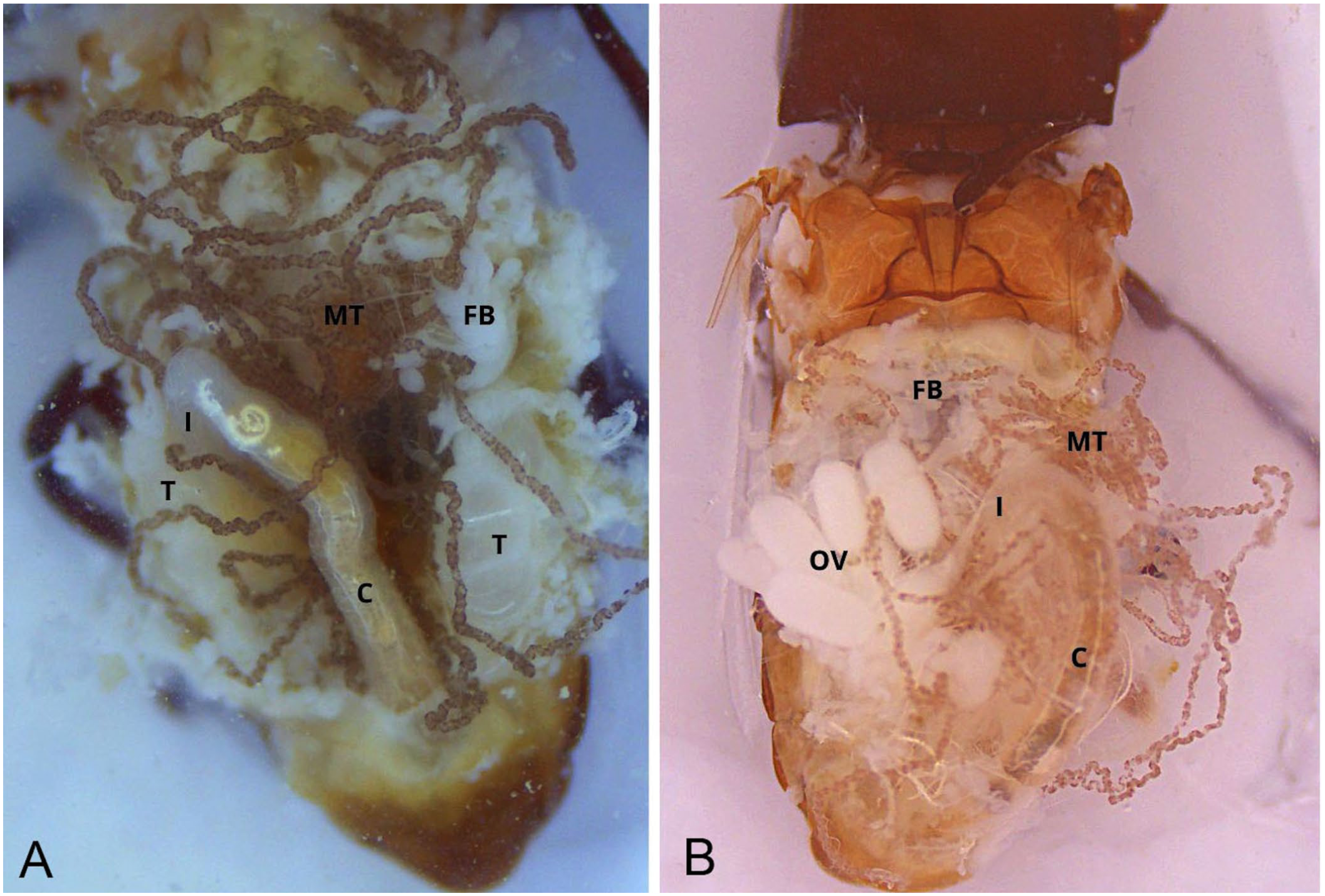
Once the incision was complete, the exoskeleton flap was lifted cranially with tweezers at the junction between the abdomen and the metathorax. As the flap was raised, tracheae and tracheoles, appearing as a delicate white web, were carefully detached using an ultra-thin needle to prevent damage. The exoskeleton was then severed at the base of the flap, exposing a clear dorsal view of the abdominal cavity ( Fig. 1 ). This view included the fat bodies, Malpighian tubules, gastrointestinal tract (midgut and hindgut), and the reproductive system (testes in males, ovaries in females; Fig. 2 )
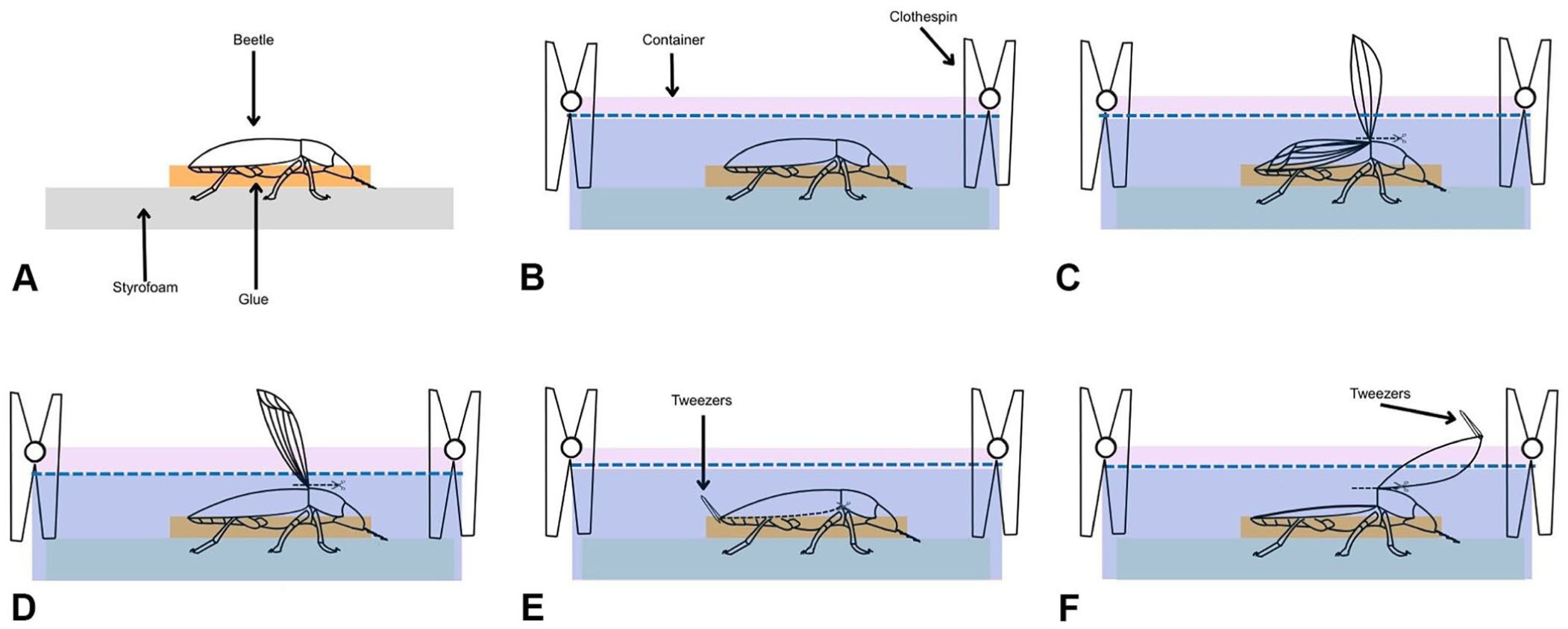
Stepwise dissection for exposure of the abdominal organs of Tenebrio molitor.
Removal of the abdominal tergites
For improved visualization, the rectum was detached with scissors from its ventral connection with the reproductive tract, and the gastrointestinal tract was extracted by pinching the rectum and pulling it outward in a controlled cranial motion. During this maneuver, the rectum was slightly lifted to allow better access to the connective tissue between the gut and the vagina. Most Malpighian tubules were typically removed with the gut at this stage ( Fig. 3 ). In females, the reproductive tract was then isolated by gently grasping the common oviduct with tweezers and applying light traction ( Fig. 4 ). Fat body tissue, which remained attached to the exoskeleton, was removed with the tweezers or a fine needle to reveal the mediodorsal nerve cord.
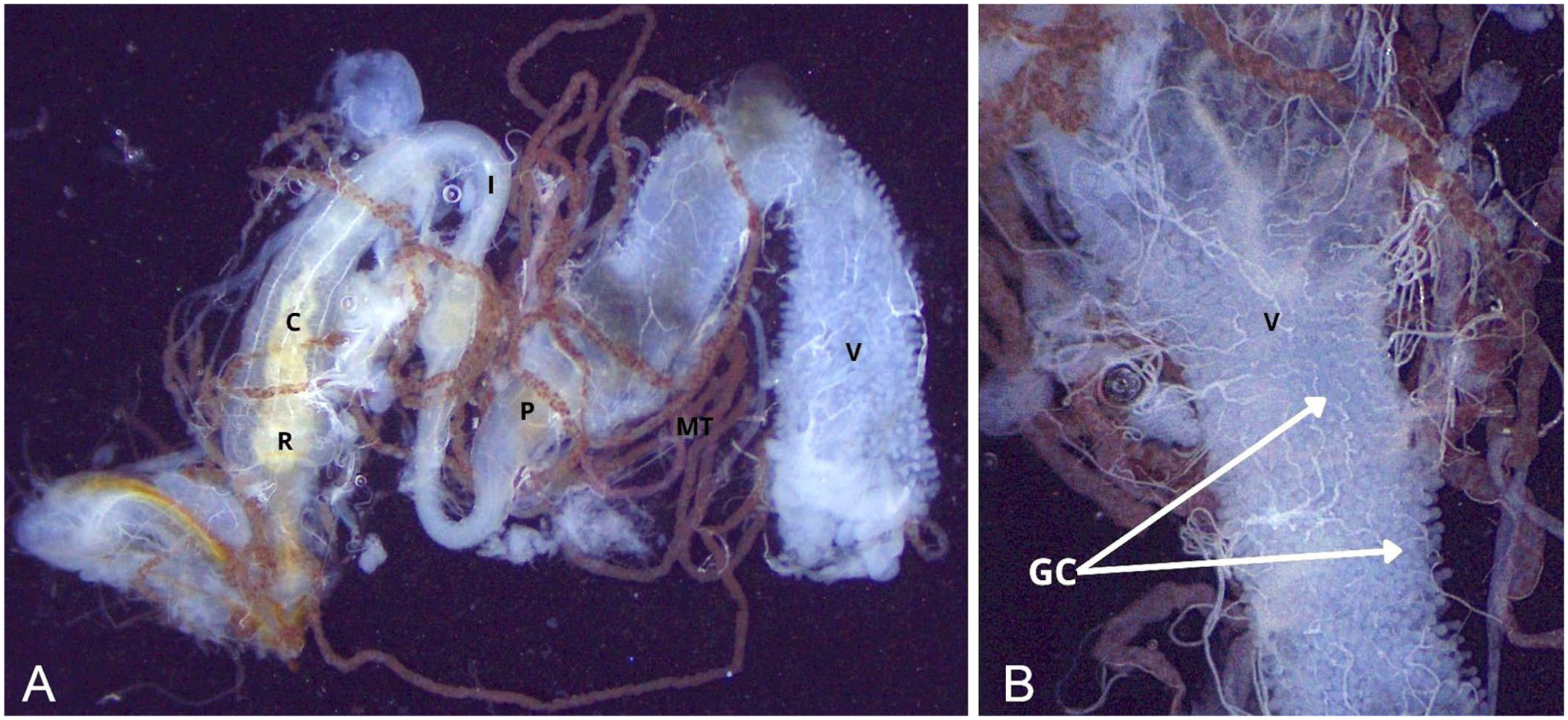
Isolation of the abdominal gastrointestinal tract.
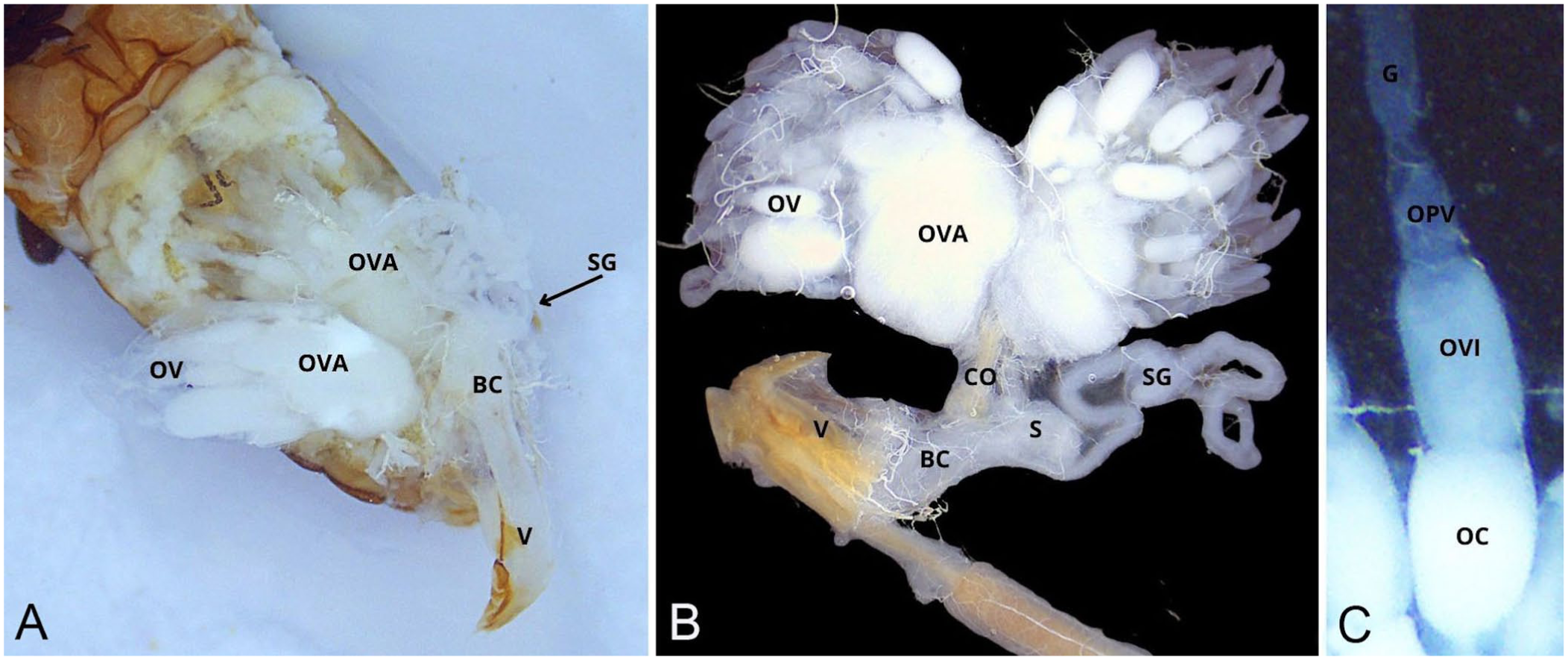
Isolation of the female reproductive tract.
In males, the digestive tract was removed following the same procedure. This allowed clear visualization of the testes, the bean-shaped accessory glands, and the tubular accessory glands ( Fig. 5 ). The respiratory system was severed between the ventral plane of the accessory glands and the nerve cord; the reproductive tract was displaced laterally. The aedeagus was detached from its abdominal anchorage using tweezers, and the fat bodies were carefully removed to expose the nerve cord ( Fig. 6 ).
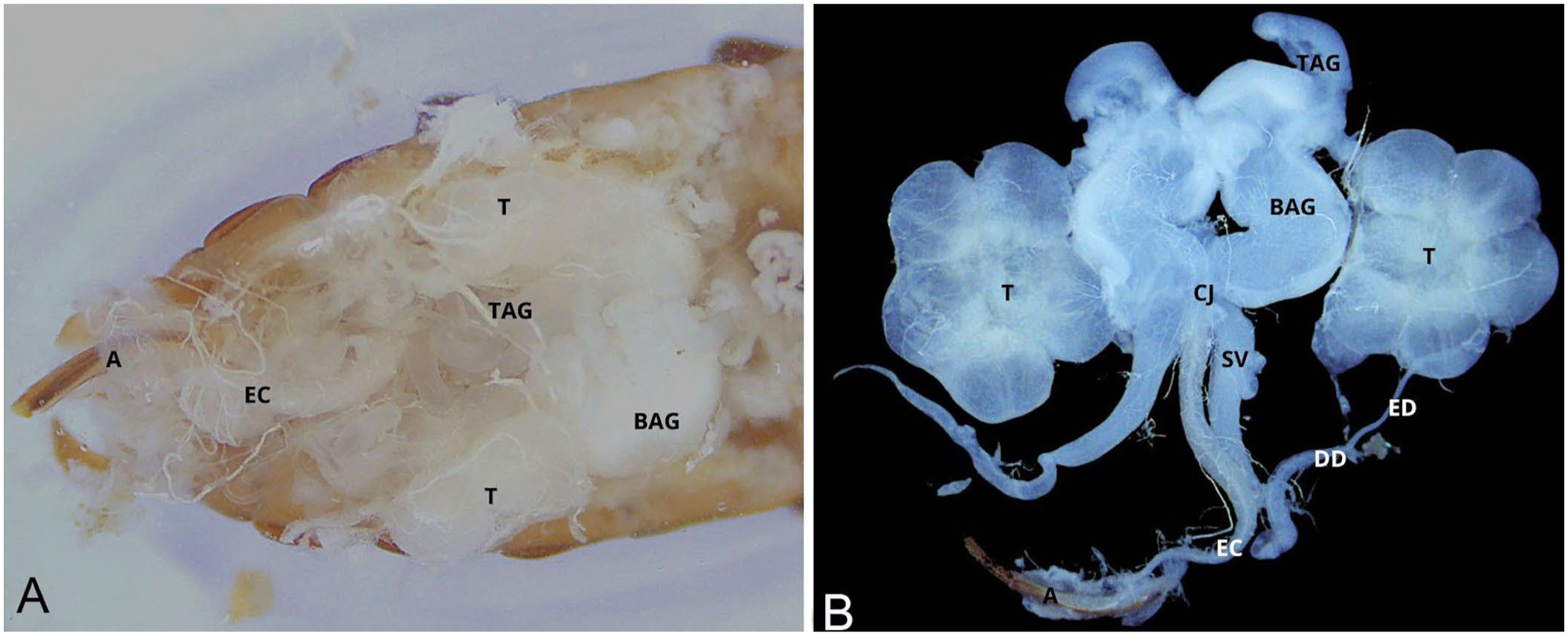
Isolation of the male reproductive tract.
Isolation of the nerve cord.
Capturing pictures
In situ images were captured with a camera and a ring light (Moticam S3, LED 60T-B; Bausch & Lomb) mounted on the stereomicroscope. To reduce light flares caused by the dissection medium, parchment paper was placed over the light source. Isolated organs were photographed directly on the microscope stage. For this purpose, 2 dissection needles were positioned parallel on the stage, flanking the specimen without contact. A 50 × 24 mm coverslip (Fisher) was then placed over both the organ and the needles. Drops of saline were added with a pipette along the coverslip’s edge, allowing the liquid to spread beneath by capillary action. Finally, a few drops of 70% isopropyl alcohol were added above the cover slip to enhance image clarity. All images were processed with software (Motic Images Plus 3.0; Bausch & Lomb).
Abdominal organ description
The fat body, resembling deposits of fat, can be seen in the abdominal cavity. From oral to aboral, the abdominal digestive tract includes the ventriculus, covered with small white gastric ceca; followed by the pylorus, marked by a sharp reduction in lumen diameter; then the ascending ileum, descending colon, and rectum. The digestive tract is mainly located dorsally, in close association with the tergites, on the median line. The rectum is slightly dilated and terminates at the anus, often still covered by the terminal tergite and sternite. At the junction between the ventriculus and the ileum are 6 Malpighian tubules. Functionally analogous to mammalian kidneys, the distal extremities of the Malpighian tubules form the cryptonephric complex, which is often only partially preserved given the fragile connection between the Malpighian tubules and the rectum.
The female reproductive tract is located ventral to the digestive tract. From craniolateral to caudomedial, the 2 ovaries are each connected to the common oviduct via a lateral oviduct. Each ovary contains multiple ovarioles, within which oocytes develop progressively through early germarial stages to fully mature forms. At its distal pole, the common oviduct merges with the bursa copulatrix, which opened into the vagina. The spermatheca lay caudoventrally to the bursa and are accompanied by a spermathecal gland at the base.
The male reproductive system is also located ventral to the digestive tract. Craniomedially, the tubular accessory glands are followed by the bean-shaped accessory glands slightly caudally. The testes are positioned caudolaterally to the accessory glands, and each resembles a flower composed of 6 follicles (lobes). A series of tubes connects the caudal pole of the testes to the common junction, situated medially. From the testes, the efferent ducts extend caudally to become the slightly wider deferent ducts. Widening but now extending cranially, the seminal vesicle ends at the common junction. Both types of accessory glands are also connected to the common junction at their caudal poles. The common junction continues caudally into the ejaculation canal, on the median line, which terminates at the aedeagus.
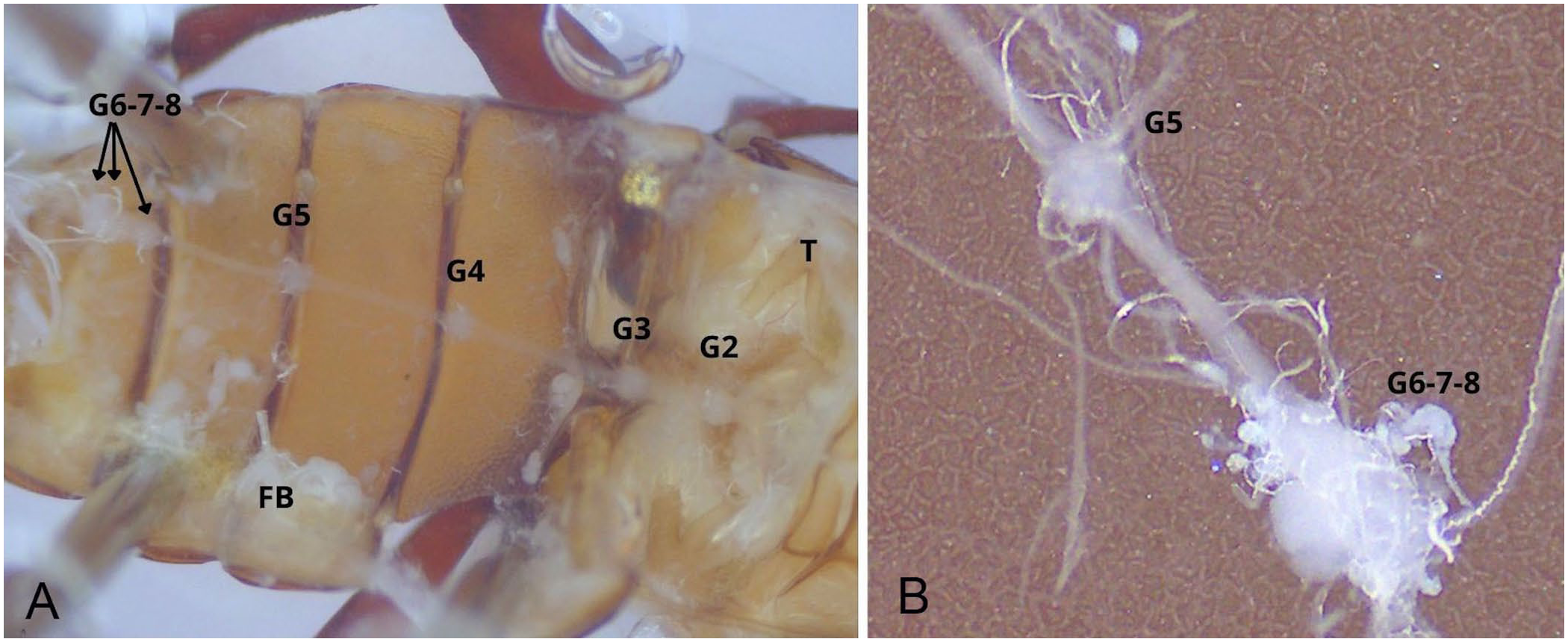
Lying ventral to the male or the female reproductive system, on the median line, the abdominal nerve cord consists of 2 parallel strands bearing 7 equally spaced bulges, or ganglia. The spacing between the first 2 ganglia is reduced. The terminal ganglion, which is the most caudal, is ~3× larger than the others, which are otherwise uniform in size ( Fig. 7 ).

Schematic representation of the dissection steps for male (A, B, and E) and female (C, D, and E) Tenebrio molitor specimens.
Discussion
A learning curve for insect autopsy is inevitable, particularly in developing the hand-eye coordination required to manipulate tools indirectly while observing through a microscope. We found that conducting dissections under aqueous immersion offered clear advantages: tissues remained hydrated, reducing the risk of tearing, and visibility was improved, facilitating the isolation of target organs. Furthermore, abdominal dissection yielded consistent and repeatable results, unlike dissections of the thorax and head (data not shown). The abdominal exoskeleton’s relative thinness and larger cavity likely explain this reproducibility. By contrast, the thorax proved difficult to dissect because of its dense musculature; attempts to dissect the head frequently resulted in organ rupture because of the limits of our instruments.
The anatomy of the adult abdominal digestive tract closely resembles that described in T. molitor pupae, likely reflecting dietary consistency throughout development.8,42 As in pupae, numerous small ceca surrounded the ventriculus. This contrasts with strictly herbivorous beetles, such as the African black beetle (Heteronychus arator) or the coffee berry borer (Hypothenemus hampei), in which more-developed ceca have been reported.1,38
Our review of major databases identified only one prior description of the T. molitor adult male reproductive tract. 19 That study provided limited, black-and-white illustrations and an incomplete organ description, restricting its diagnostic value. More recent work on other Coleoptera families, however, reveal considerable diversity that both supports and contrasts our observations. For instance, the number of accessory gland pairs varies widely across beetles: T. molitor has 2 pairs, as in Ceracis cornifer (Tenebrionidae), Hypothenemus hampei (Curculionidae), and Spasalus silvarum (Passalidae), whereas Meleo proscarabaeus (Meloidae), Silpha obscura (Silphidae), and Isotomus speciosus (Cerambycidae) have 3 pairs, and Lasioderma serricorne (Ptinidae) have 5.14,24,29,33 –35,41 T. molitor is unique in having bean-shaped accessory glands. Testicular morphology also varies among species: 1–6 follicles are present, with T. molitor having the maximum of 6, giving the testes a flower-like shape. A similar arrangement is described in Parastizopus armaticeps (Tenebrionidae). 5 Such morphologic diversity is presumably adaptive, enhancing reproductive success under different ecologic pressures. 20
The female reproductive tract of T. molitor resembles that of several other beetles, including Chrysolina herbacea (Chrysomelidae), Zygogramma exclamationis (Chrysomelidae), Chrysomela populi (Chrysomelidae), Gonioctena fornicata (Chrysomelidae), Omorgus suberosus (Trogidae), and Trypophloeus klimeschi (Curculionidae).13,15,16,27,28,30 A spermatheca is consistently observed, but the presence of a spermathecal gland, which nourishes and maintains the viability of sperm over time, varies among species.31,40 For example, no gland was reported in G. fornicata, C. herbacea, or T. klimeschi. In contrast, T. molitor has a spermathecal gland that not only is present but also larger than the spermatheca itself. This differs from the previously cited Coleoptera, in which the gland appears smaller. We hypothesize that a larger spermathecal gland may be a reproductive advantage, supporting long-term sperm viability and storage.
The insect nervous system broadly resembles a mammalian spinal cord, comprising a brain followed by a ventral chain of ganglia. In most insects, there is 1 subesophageal ganglion, 3 thoracic ganglia, and up to 8 abdominal ganglia. Variation arises through differences in spacing and fusion of these ganglia, with beetles having the greatest diversity. For example, Curculionidae often have a single fused thoraco-abdominal ganglion, whereas Lycidae has 8 distinct abdominal ganglia. 26
In our T. molitor specimens, the terminal ganglion was ~3× larger than the others, suggesting fusion of the 3 most caudal ganglia while retaining their neural mass. Only 7 abdominal ganglia were identifiable when counting the fused ganglia separately, consistent with observations in pupae. 42 Additionally, the spacing between the second and third abdominal ganglia was reduced by about half compared with other segments. This contraction may reflect evolutionary segmental rearrangements, such as shortened body regions innervated by these ganglia. Dissection also revealed that isolating the nerve cord was more difficult in males because of its proximity to the bean-shaped accessory glands, often leading to accidental removal with adjacent tissues.
One abdominal organ that we did not recover was the heart. Its dorsal placement likely caused it to be torn and lost when the abdominal flap was lifted. For future work, positioning specimens dorsally and performing dissections from a ventral approach may allow isolation of this structure.
We used commonly available laboratory tools in a practical guide for the dissection of adult T. molitor. By minimizing tissue damage and improving organ visibility, our method should contribute to a better understanding of beetle anatomy and facilitate both in situ examination and isolation of individual organ systems. Ultimately, such protocols may enhance the diagnostic capabilities of veterinarians and researchers working with farmed insects. Further studies are nevertheless required to examine diseased beetles and to better characterize the lesions and the information they can reveal
Footnotes
Acknowledgements
We thank TriCycle Entotechnologies (Montréal, Canada) for providing the insects used to start our colony.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study received financial support from Fonds du Centenaire from the Faculté de médecine vétérinaire, Université de Montréal and Programme Innov’Action Bioalimentaire (IA121693) from the Ministère de l’Agriculture, des Pêcheries et de l’Alimentation du Québec (MAPAQ).
