Abstract
A 5-y-old male Poodle mix was presented with intermittent vomiting, anorexia, and weight loss. Physical examination revealed emaciation, lethargy, dehydration, hypothermia, respiratory distress, and splenomegaly. Based on clinicopathologic, serologic, and parasitologic findings, diagnoses of severe leishmaniosis and dirofilariasis were made. Extracellular, intraneutrophilic, and intramonocytic Leishmania amastigotes were observed on blood smear and buffy coat smear examination. In blood smears, 0.2% of neutrophils were observed to be infected; in buffy coat smears, 0.5% of neutrophils and 0.1% of monocytes were found to be infected. Leishmania amastigotes were also found engulfed by eosinophils and neutrophil precursors in bone marrow aspiration cytology. The detection of Leishmania amastigotes in blood smears is rare, and the clinical significance is uncertain. In circulating blood, Leishmania amastigotes are primarily found phagocytized by neutrophils. Although debatable, there is growing evidence that neutrophils are used as carriers enabling the “silent entry” of the protozoa into macrophages (“Trojan horse” theory). To date, cytologic screening of blood smears for the diagnosis of canine leishmaniosis is not a routine practice. Clinical pathologists and practitioners should be aware that Leishmania amastigotes may be present in neutrophils and less frequently monocytes during blood smear evaluation; neutrophil precursors and eosinophils may also be parasitized in bone marrow specimens.
A 5-y-old, intact male, Poodle mix dog was presented to the Companion Animal Clinic (School of Veterinary Medicine, Aristotle University of Thessaloniki, Greece) with a 3-wk history of intermittent vomiting, anorexia, and weight loss. The dog had no history of travel outside Greece. Physical examination revealed emaciation, lethargy, moderate dehydration, mild hypothermia, and respiratory distress. An ulcer was found on the tongue; abdominal palpation suggested splenomegaly. Arterial blood pressure was measured serially (Doppler method) and found to be normal.
Blood was collected via jugular venipuncture into K3EDTA-containing tubes and tubes with clot accelerator and granule serum separator (Deltalab, Barcelona, Spain). Both tubes were filled to the defined level and gently inverted several times to ensure that blood would be adequately mixed with the anticoagulant or the clot accelerator. The sample in the tube containing clot accelerator was allowed to clot for 30 min at room temperature and was then centrifuged at 1,800 × g for 10 min. Complete blood count (Advia 120; Siemens Healthcare Diagnostics, Deerfield, IL) abnormalities included mild normocytic, normochromic anemia (HCT: 0.35 L/L, reference interval [RI]: 0.37–0.55 L/L) and moderate leukocytosis (35.7 × 109/L, RI: 6.0–17.0 × 109/L). A differential white blood cell (WBC) count on a Diff-Quik–stained (Merck, Darmstadt, Germany) blood smear revealed moderate neutrophilia (29.7 × 109/L, RI: 3.9–8.0 × 109/L) with mild-to-moderate left shift (band neutrophils: 1.8 × 109/L) and mild monocytosis (1.6 × 109/L, RI: 0.2–1.1 × 109/L); rare polychromatophils were observed, suggesting inadequate regeneration. The presence of a few microfilariae (Fig. 1) was also noted. Several round-to-oval organisms, 2–5 μm in length, with a discrete cell membrane, eccentrically placed nucleus, and a distinct kinetoplast, consistent with Leishmania spp. amastigotes, were observed extracellularly or phagocytized by neutrophils (Fig. 2). After thorough evaluation of several blood smears and a review of 1,000 neutrophils, 21 (0.2%) neutrophils were found to be parasitized by Leishmania amastigotes. Buffy coat smears were prepared by centrifugation of 0.3 mL of EDTA whole blood in a Wintrobe tube (Bacacos, Athens, Greece) at 600 × g for 20 min. The percentage of infected neutrophils was higher in buffy coat blood smears (0.5%; 48 infected of 1,000 reviewed neutrophils); occasional parasitized monocytes were also found (0.1%; 10 infected of 1,000 reviewed monocytes; Fig. 2). The additional blood and buffy coat smears were stained with Giemsa stain (Merck). Air-dried smears were fixed in methanol for 5 min, air-dried again, and then stained with Giemsa (dilution 1:20 with deionized water) for 20 min. Finally, the smears were rinsed briefly in deionized water and air-dried.
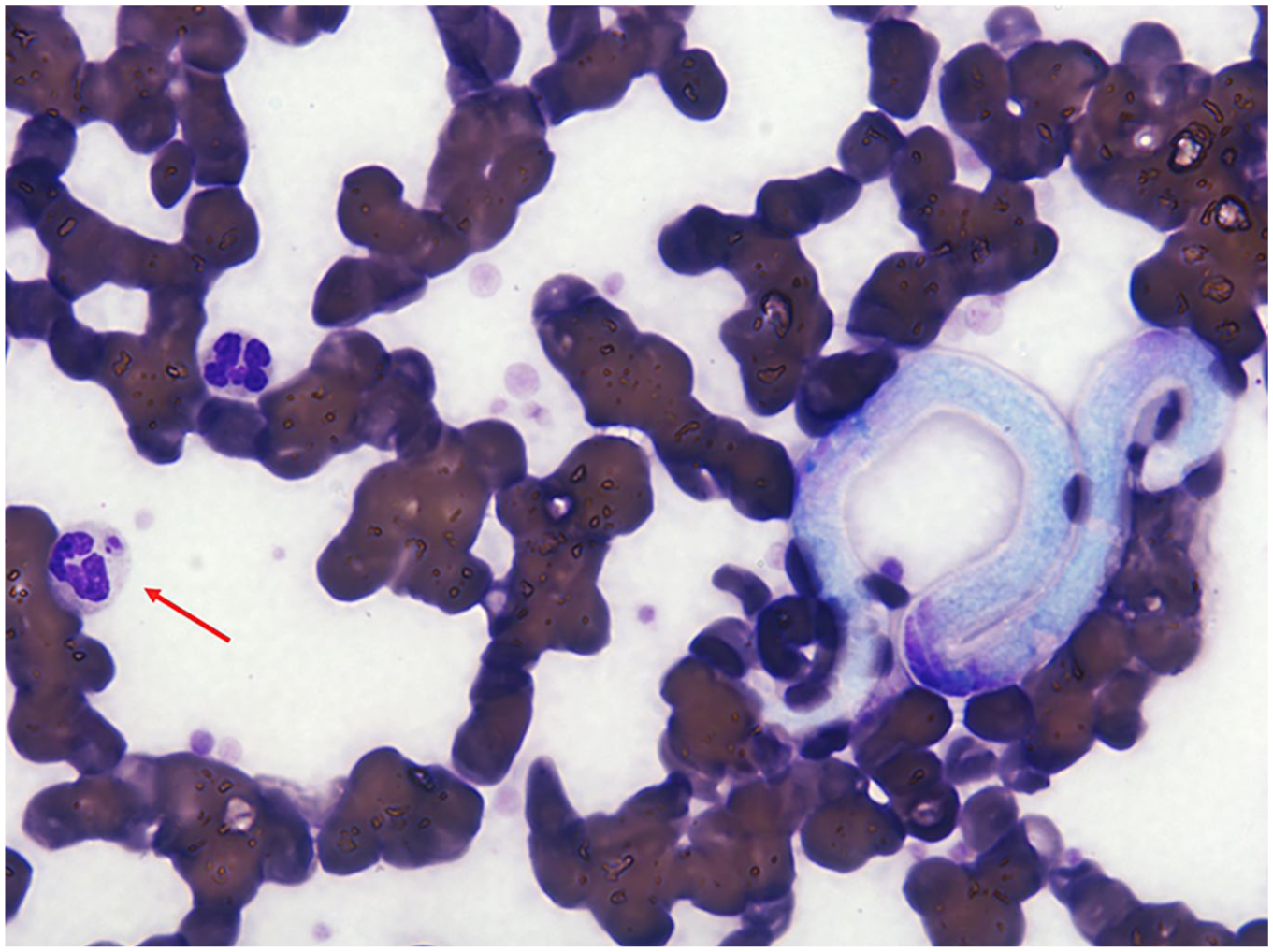
Blood smear from a dog diagnosed with dirofilariasis and leishmaniosis. One microfilaria and a Leishmania amastigote phagocytized by a neutrophil (arrow) are present. Diff-Quik stain. 600×.
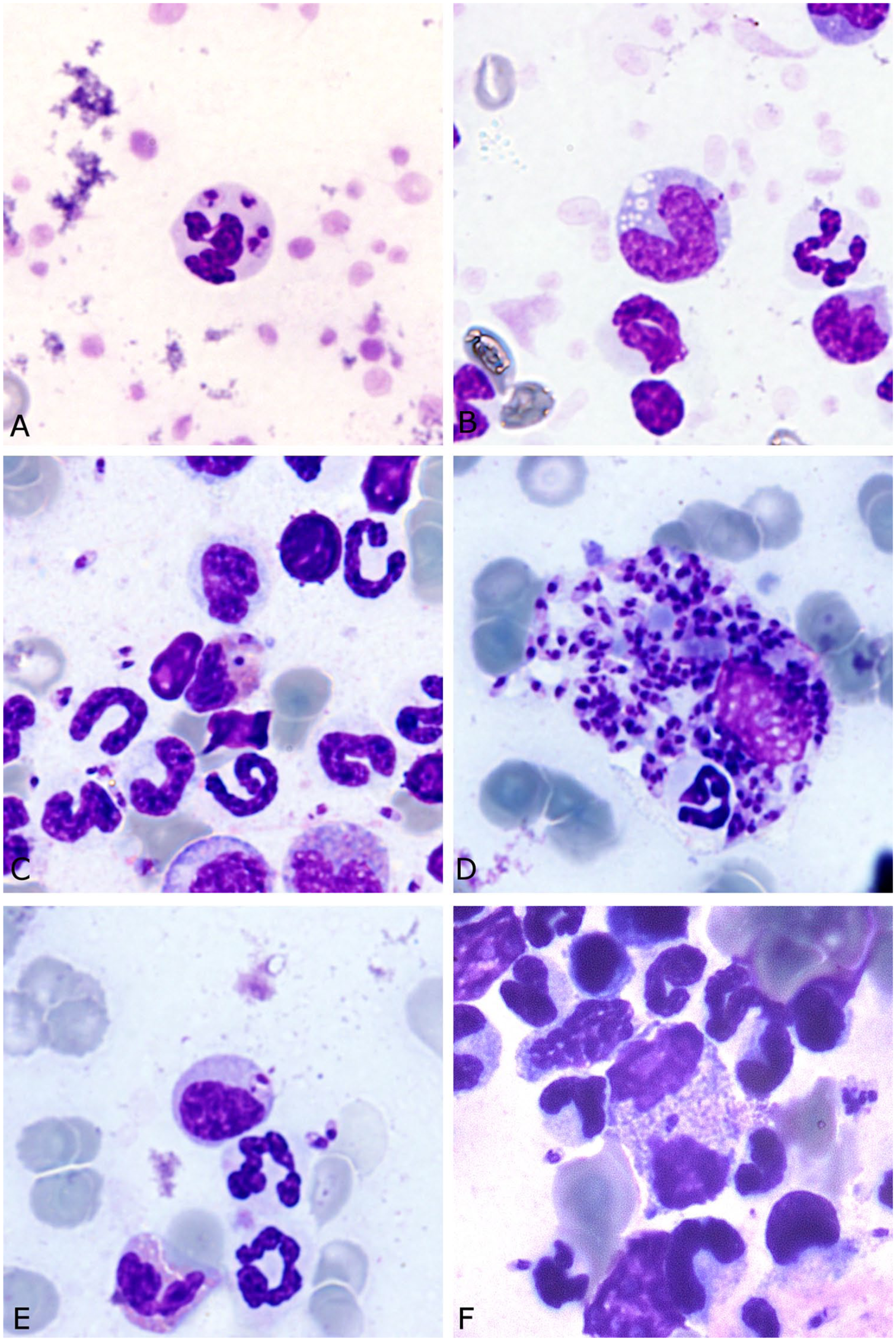
Blood, bone marrow, and buffy coat smears from a dog with leishmaniosis. Giemsa stain. 600×.
Routine serum biochemistry examination (Flexor E; Vital Scientific, Dieren, The Netherlands) revealed hypoalbuminemia, increased urea concentration, increased alkaline phosphatase activity, hyperbilirubinemia, hypocalcemia, hyperphosphatemia, hypercholesterolemia, and hypertriglyceridemia (Table 1). Thoracic radiographs indicated diffuse interstitial lung infiltrates; abdominal ultrasonography revealed peritoneal effusion and a diffusely hypoechogenic splenic parenchyma. Point-of-care ELISA (Snap 4Dx Plus; IDEXX Laboratories, Westbrook, MA) was negative for Dirofilaria immitis antigen and for antibodies to Ehrlichia spp., Borrelia burgdorferi, and Anaplasma spp. A few microfilariae, ~300 μm in length with a conical front end and a straight rear end, were found on a Knott test; these were morphologically consistent with D. immitis. 11 An indirect immunofluorescent antibody test for anti-leishmanial IgG antibodies was positive (titer: 1,600; positive titer cutoff was ≥100). For this assay, the anti-dog IgG, produced in rabbits, is conjugated with fluorescein isothiocyanate (Sigma-Aldrich, St. Louis, MO) and slides coated with Leishmania promastigotes (Agrolabo, Scarmagno, Italy). Positive and negative controls (Agrolabo) were run on the same slide used for examination of patient serum. Canine pancreas-specific lipase (Spec cPL test; IDEXX Laboratories) was markedly elevated (1,471 μg/L, RI: 0–200 μg/L, confirmatory for pancreatitis is >400 μg/L). Urine was collected via cystocentesis. Urine specific gravity measured on a refractometer (Atago, Tokyo, Japan) was 1.020; microscopic examination of the sediment was unremarkable; urine protein-to-creatinine ratio measured using a spectrophotometric analyzer (Flexor E) was markedly elevated (11.5, RI: <0.5).
Serum biochemistry results of a dog with leishmaniosis.
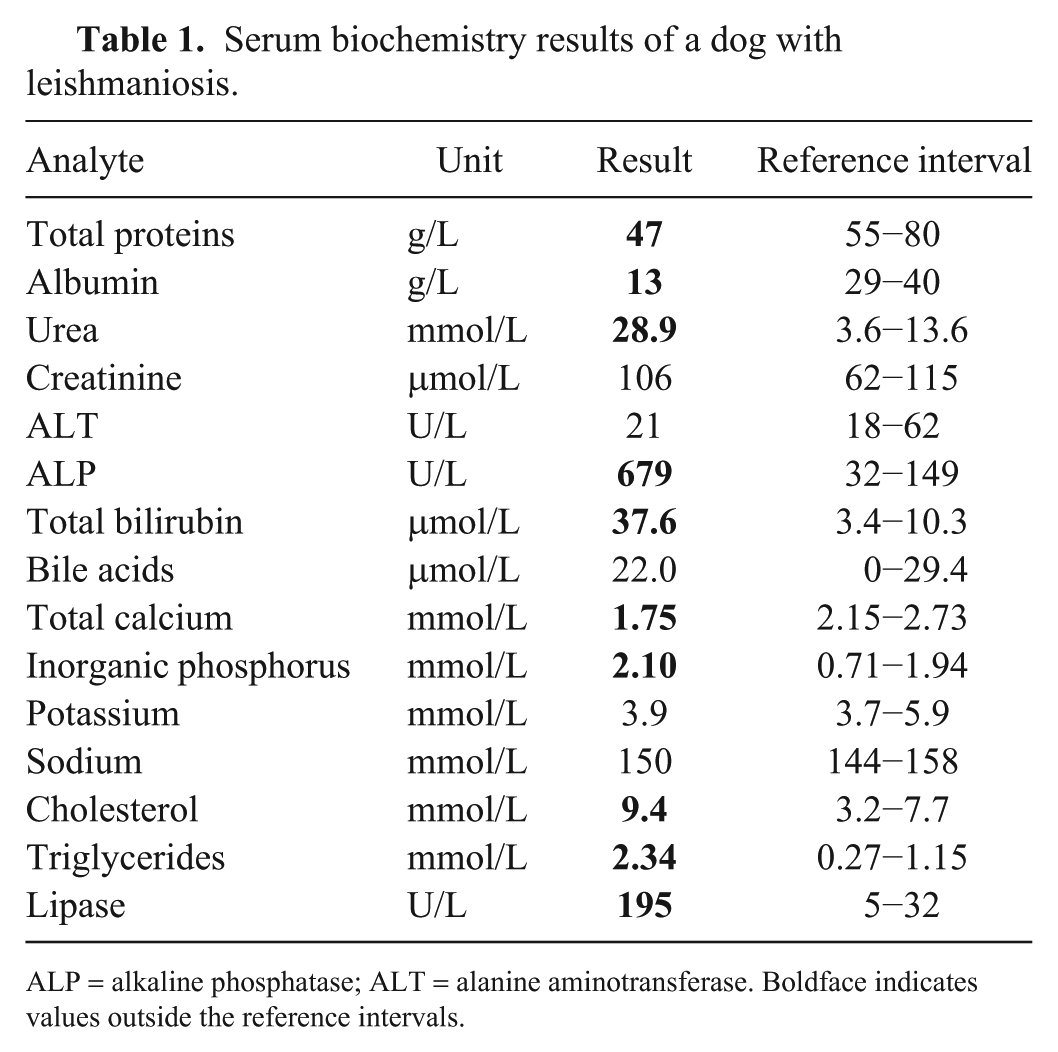
ALP = alkaline phosphatase; ALT = alanine aminotransferase. Boldface indicates values outside the reference intervals.
A small amount of peritoneal fluid was aspirated and collected into a K3EDTA-containing tube, a tube with clot accelerator and granule serum separator, and a sterile tube (Deltalab). The peritoneal fluid was immediately analyzed and was considered a pure transudate, based on total nucleated cell count (0.1 × 109/L, Advia 120), total protein concentration (0 g/L, refractometer), and cytologic evaluation of Giemsa-stained direct and cytospin smears (presence of a scant population of non-degenerate neutrophils, macrophages, and small lymphocytes). Peritoneal fluid lipase measured by a colorimetric method (Flexor E) was 1,579 U/L. Both aerobic and anaerobic bacterial cultures of the urine and the peritoneal fluid were negative.
Bone marrow was aspirated from the iliac crest using a 15-gauge Illinois-type aspiration needle and a 10-mL syringe rinsed with 0.15 mL of 2% EDTA solution. Prior to sampling, local anesthesia was applied by infiltrating the subcutaneous and subperiosteal tissues with 2% lidocaine. The sample was placed into a K3EDTA-containing tube (Deltalab). The compression (squash) technique was used for preparation of the smears, which were then stained with Giemsa as previously described for the blood smears, but for an extended time (40 min). Cytologic examination of bone marrow smears revealed increased cellularity, adequate iron stores, and moderately increased myeloid-to-erythroid ratio (4.8). All of the marrow lineages showed orderly and complete maturation. The numbers of plasma cells were mildly increased (6% of total nucleated cells). Although the proportion of macrophages appeared slightly increased focally, overall, their numbers were within normal limits. Importantly, numerous Leishmania spp. amastigotes were seen extracellularly and primarily inside macrophages (Figs. 2, 3), but also occasionally phagocytized by segmented neutrophils and rarely engulfed by neutrophil precursors and eosinophils (Fig. 2). The bone marrow cytologic diagnosis was leishmaniosis with hypercellular marrow and moderate myeloid and mild plasmacytic hyperplasia.
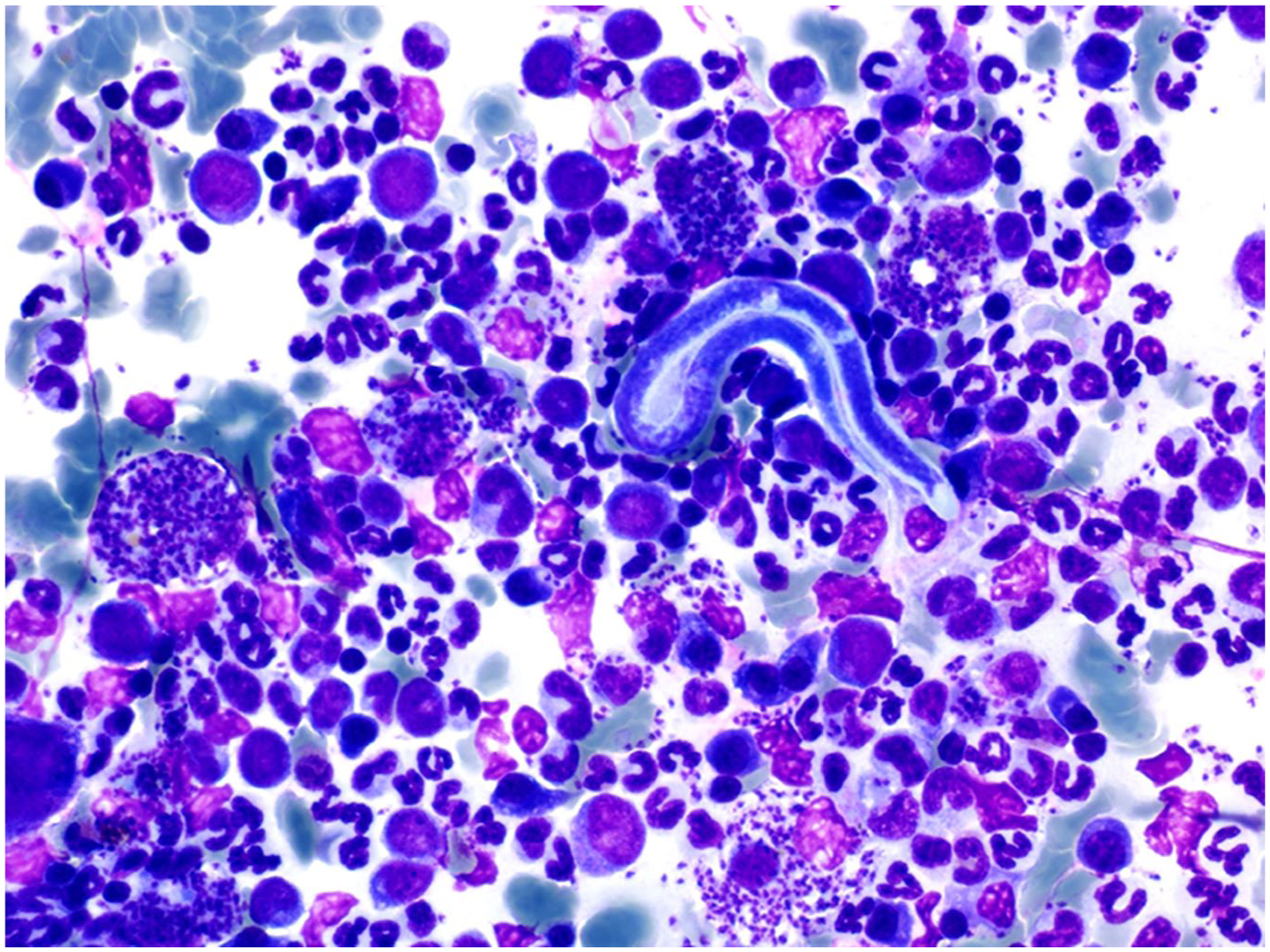
Bone marrow smear from a dog with leishmaniosis. Numerous Leishmania amastigotes are present extracellularly and inside macrophages. A microfilaria is present at the center of the picture. Giemsa stain. 400×.
A tentative diagnosis of nephrotic syndrome, chronic kidney disease (International Renal Interest Society [IRIS] stage I, severe proteinuria, no hypertension), and pancreatitis (markedly increased canine pancreas-specific lipase and higher lipase activity in peritoneal fluid than serum) was made, in the context of a definitive diagnosis of leishmaniosis and dirofilariasis. The dog was hospitalized for 4 d and treated with crystalloids, antibiotics, allopurinol, tramadol, and acetylcysteine. Given clinical deterioration and poor long-term prognosis, the dog was euthanized, and an autopsy was performed the same day with the owner’s consent. On gross examination, lungs were solid, gritty, and pale. Aorta, pulmonary vein, endocardium (mainly left), and carotids were extensively mineralized. Four adult heartworms were found in the right ventricle of the heart, which confirmed the diagnosis of dirofilariasis. The stomach was hyperemic with focal ulceration, and intestines contained bloody content. Kidneys were firm and pale, and the pancreas was atrophic.
Histologic examination of the kidneys showed diffuse glomerular hypercellularity and hypertrophy of the parietal and visceral epithelium. The interstitium had multifocal infiltration by moderate numbers of lymphocytes and plasma cells. Multifocally, tubules had homogeneous brightly eosinophilic intraluminal protein casts, and tubular epithelium was swollen with vacuolated cytoplasm. The pancreas had a few focally extensive areas of necrosis, replaced by cellular debris admixed with degenerate neutrophils and surrounded by macrophages, lymphocytes, plasma cells, and a peripheral rim of reactive fibroblasts. The inflammatory infiltrate focally extended to the adjacent adipose tissue. The morphologic diagnosis was chronic glomerulonephritis with tubular degeneration and interstitial nephritis, accompanied by chronic granulomatous pancreatitis.
Although the molecular detection of Leishmania spp. in peripheral blood samples is commonly successful in infected animals, cytologic documentation of amastigotes in blood smears is considered a rare finding in both symptomatic and asymptomatic infected dogs. In one study involving 1,438 infected dogs, the diagnostic sensitivity of blood smear cytology in detecting Leishmania amastigotes was extremely low (0.28%). 4 Interestingly, the diagnostic sensitivity of buffy coat smears for the cytologic detection of Leishmania amastigotes is reportedly 50% 13 and, although essentially higher than that of blood smears, it is still considered low. 4 It should be noted that the diagnostic sensitivity of lymph node and bone marrow aspiration cytology in detecting Leishmania amastigotes in symptomatic dogs has been found to be 92.6% and 88.2%, respectively; however, our patient’s nodes were hypoplastic, and aspiration was not attempted. 17
The presence of Leishmania amastigotes in the blood is consistent with hematogenous spread of the infection. However, a correlation has not been found between the presence of Leishmania amastigotes in the peripheral blood and the clinical and clinicopathologic findings of affected dogs, although in humans with subclinical leishmaniosis, amastigotes are rarely encountered in peripheral blood smears as opposed to those with clinical disease. 13 In our case, Leishmania amastigotes were identified in blood and buffy coat smears both extracellularly and phagocytized primarily by neutrophils and to a lesser extent by monocytes (0.5% neutrophils vs. 0.1% monocytes infected in buffy coat blood smears). Leishmania amastigotes are typically phagocytized by macrophages given that macrophages constitute the major protozoan cellular host. Nonetheless, in peripheral blood, Leishmania amastigotes are seen engulfed primarily by neutrophils.3,4,10,13,16,19
Although the role of neutrophils in the pathogenesis of leishmaniosis has not been completely elucidated, there is growing evidence supporting the so-called “Trojan horse” model. 9 According to this theory, infection with Leishmania promastigotes results in rapid recruitment of neutrophils via chemokines produced by the host and the protozoa (Leishmania chemotactic factor [LCF]). 21 LCF has a chemotactic effect exclusively on neutrophils and is associated with enhanced silent uptake and intraphagocytic survival of Leishmania major promastigotes. 21 Recruited neutrophils eventually phagocytize the protozoa and induce the release of interleukin-8, amplifying the neutrophil migration to the site of the infection. 9 Phagocytosis of L. major promastigotes by neutrophils is done via both opsonin-dependent and -independent mechanisms. 9 However, protozoa that are phagocytized by the opsonin-independent mechanism can survive intracellularly, presumably by blocking respiratory burst induction and/or escaping the lytic compartments of the neutrophil cytoplasm. 7 Spontaneous apoptosis of parasitized neutrophils is then delayed for up to 1 or 2 d, a process involving the inhibition of caspase-3 activation. 1 Infected neutrophils then become apoptotic, and macrophages are recruited to the primary site of infection, attracted by chemokines (e.g., monocyte-attractant chemokine-1β). Macrophages eventually phagocytize the apoptotic neutrophils infected with viable protozoa (i.e., the “Trojan horses”). 9 Apoptotic cells are not considered a danger by the host’s immune system, and their uptake does not result in the activation of antimicrobial functions of macrophages but instead silences them, 20 enabling the survival and multiplication of protozoa inside their final host cell and thus disease development.
In our case, Leishmania amastigotes were also observed inside neutrophil precursors and eosinophils on cytologic examination of bone marrow. To our knowledge, the phagocytosis of Leishmania amastigotes by these cells has not been described previously in dogs; increased eosinophil numbers have been documented very uncommonly in the blood or lymph nodes of dogs with leishmaniosis.12,14 In contrast to helminthic parasitic infections, eosinophils usually are not implicated in the immune response against protozoa. However, there is still evidence implying the involvement of eosinophils in the immune responses elicited during Leishmania infection. 15 Specifically, it has been observed experimentally that eosinophils are recruited to the site of the primary lesion, and they may participate in the immune response by enhancing the anti-leishmanial activity of macrophages and by directly phagocytizing protozoa. 15 Moreover, eosinophils had the ability to phagocytize Leishmania mexicana mexicana amastigotes in an experimental murine model. 6 Phagocytosis of Leishmania donovani amastigotes has also been demonstrated by neutrophil and eosinophil metamyelocytes in human bone marrow, although without a clear explanation. 6 Although reported extremely rarely, it has been proven that even some myeloblasts may have the ability to phagocytize. 5
Canine leishmaniosis is a well-documented cause of chronic kidney disease and nephrotic syndrome, with the major pathogenetic drive being immune-complex deposition in the glomeruli, renal interstitium, and tubular basement membranes.14,18 Importantly, chronic kidney disease is the leading cause of death or euthanasia in canine leishmaniosis, similar to the outcome of our case. 18 The dog also exhibited clinical and clinicopathologic findings consistent with pancreatitis, which was confirmed by histologic examination of the pancreas. Although an association between pancreatitis and leishmaniosis is not substantiated by the current literature, in our case, chronic granulomatous pancreatitis could potentially be attributed to leishmaniosis, 8 although amastigotes were not identified in the granulomatous pancreatic inflammation.
D. immitis infection was probably an incidental finding in this dog, given that historical, clinical, radiographic, or postmortem evidence of advanced disease was not documented. Interestingly, serologic examination for D. immitis antigen was negative; false-negative results have been reported and may occur when there is a very low number of worms, the female worms are immature, or only male worms are present (American Heartworm Society. Current canine guidelines for the prevention, diagnosis, and management of heartworm (Dirofilaria immitis) infection in dogs. 2018. Available at: https://www.heartwormsociety.org/images/pdf/2018-AHS-Canine-Guidelines.pdf). Immunosuppression has been reported in the context of canine clinical leishmaniosis, 2 and potential implication in D. immitis coinfection is suspected, although this supposition cannot be substantiated.
Our case report has 2 limitations. The presence of Leishmania amastigotes inside neutrophils, neutrophil precursors, and eosinophils was not confirmed using immunocytochemistry or immunohistochemistry; however, the observed microorganisms exhibited the typical appearance of Leishmania amastigotes and were morphologically identical to the numerous microorganisms observed extracellularly or inside macrophages in bone marrow aspirates. The second limitation is the lack of species identification of Leishmania using a molecular technique, such as PCR in blood or bone marrow samples. However, the dog did not have a history of travel outside Greece, and only Leishmania infantum is endemic in the Mediterranean region.
Although the cytologic detection of Leishmania amastigotes in blood smears is rare, the clinical relevance of this finding is elusive. Routine cytologic screening of blood smears in dogs suspected of leishmaniosis is not currently advised for establishing a diagnosis, because of substantially lower diagnostic sensitivity compared to bone marrow or lymph node cytology. Evaluation of blood buffy coat smears may be of higher diagnostic sensitivity. Although rare, eosinophil and neutrophil precursors may also be seen engulfing Leishmania amastigotes; thus, clinical pathologists and practitioners should be aware of the nonspecific cytotropism of Leishmania amastigotes, when cytologically evaluating blood or bone marrow specimens.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
