Abstract
Diffuse alveolar damage (DAD)—one histologic manifestation of severe acute interstitial lung injury—includes a subset of cases with the clinical diagnosis of acute respiratory distress syndrome (ARDS). ARDS and DAD both involve acute damage to endothelial and alveolar epithelial cells, resulting in pulmonary edema. DAD has well-defined histologic stages, including cell exudation and hyaline membranes, followed by type II pneumocyte hyperplasia. More severe lesions progress to chronic interstitial fibrosis. ARDS and DAD have diverse causes in humans and animals, yet historically were viewed as universal pathways of tissue dysfunction irrespective of cause. Molecular data have suggested that ARDS has heterogeneous signatures in epithelial, endothelial, and inflammatory cells that can characterize the specific pathogenesis of individual cases and therefore support targeted treatment. The signatures are grouped into endotypes classified according to the mechanism of primary damage. The proposed ARDS endotypes are epithelial injury, endothelial injury, angiopathy, systemic inflammatory response, and local inflammatory response. We present the pathogeneses that form the foundation of the ARDS endotypes, including evidence from dogs and cats. Specific canine and feline causes of ARDS can be assigned to an ARDS endotype. For some etiologies, multiple endotypes are applicable, highlighting the need for increased resolution of the underpinning evidence to best support the accurate application of ARDS endotypes to clinical cases.
The lung is composed of airways, alveolar airspaces, the interstitium, and the pleura. The interstitium incorporates pneumocytes and other cells of the alveolar walls, blood vessels, and supporting stroma, including interlobular septa where present in relevant species.
27
Inflammation targeting the lung interstitium is classified as interstitial pneumonia.
27
Diffuse alveolar damage (
Definitions relating to acute lung injury.
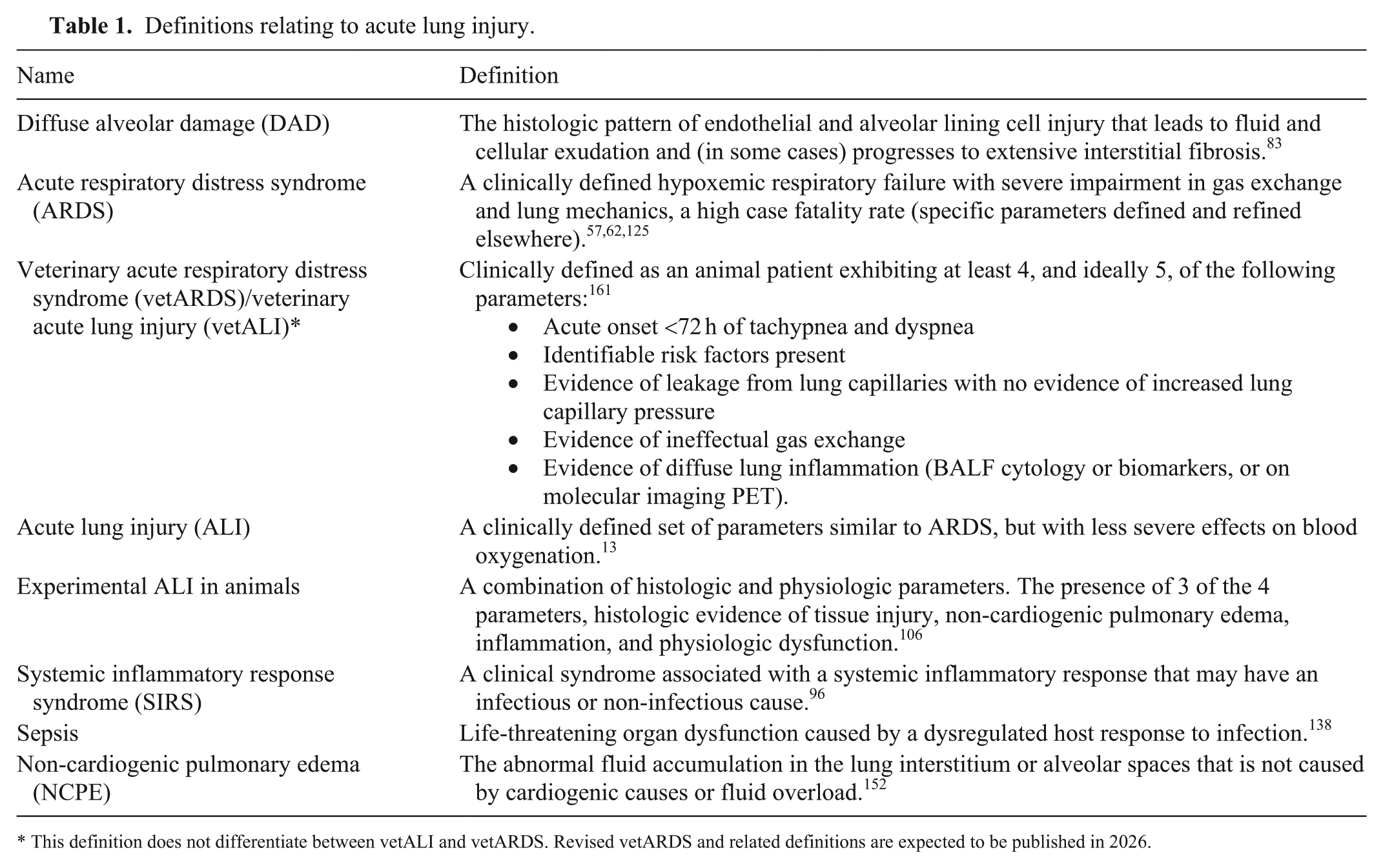
This definition does not differentiate between vetALI and vetARDS. Revised vetARDS and related definitions are expected to be published in 2026.
ARDS may develop from progression of other clinical syndromes, such as sepsis, acute lung injury (
Historically, ARDS and DAD were thought to be universal pathways of tissue dysfunction independent of the initiating cause. Experimental models have identified therapeutic targets that reduce the severity of ARDS; however, those potential therapies have often failed in clinical trials. In human cases of ARDS, reduced ventilator tidal volume, plateau pressure, and driving pressure have improved outcomes1,3; however, supportive treatment has been the mainstay.
23
The lack of translational relevance of pre-clinical ARDS models to the clinics has re-focused research on clinical cases of ARDS and DAD. In humans, post hoc analysis of 6 cohorts of randomly controlled clinical trials separated ARDS cases into 2
For veterinary species, ARDS subphenotypes have been re-created in a pre-clinical ovine experimental model with broadly similar features of hyperinflammatory and hypoinflammatory phenotypes.112,160 Subphenotypes of ARDS in dogs and cats have not been defined.
Variation in ARDS is now thought to occur on etiologic, physiologic, and biological levels.18,127 Although the classifications have an arbitrary basis, they link to clinical test and therapeutic outcomes, highlighting potentially treatable traits that may be modeled experimentally.
18
Mining clinical information for pre-clinical biomarkers has been successful in sepsis research; specific biomarkers related to sepsis mortality in children were predictive of mortality in mice challenged with experimental sepsis.
166
Understanding subphenotypes of heterogeneous conditions may improve the analysis of pre-clinical models and, ultimately, the development of new therapeutic strategies. With that aim, the European Respiratory Society chose a research target to further divide ARDS subphenotypes into
The pursuit of ARDS endotypes is facilitated by advances in molecular techniques, such as single-cell multi-omics and spatial transcriptomics. These tools have revealed that, even with similar clinical manifestations, different causes of ARDS induce heterogeneous cellular and molecular signatures in epithelial, endothelial, and inflammatory cells. 103 Endotypes classified by mechanism of primary damage have been proposed, grouped into epithelial injury, endothelial injury, angiopathy, systemic host response, and local host response. 18
For veterinary patients, the aim of subclassification of ARDS cases into endotypes is to lay the groundwork for developing targeted treatment trials and point-of-care testing. Veterinary diagnosticians working in pathology or interfacing fields can contribute to the creation and analysis of these data. Developing our understanding of the lesions initiated by ARDS endotypes will contribute to that goal. After reading this review, readers should be able to:
Describe the histologic features of the classical phases of DAD.
Differentiate the subphenotypes and endotypes of ARDS.
Describe the classification of ARDS endotypes using examples from canine and feline pathology.
The histologic features of the phases of DAD
The phases of DAD occur within a defined sequence (
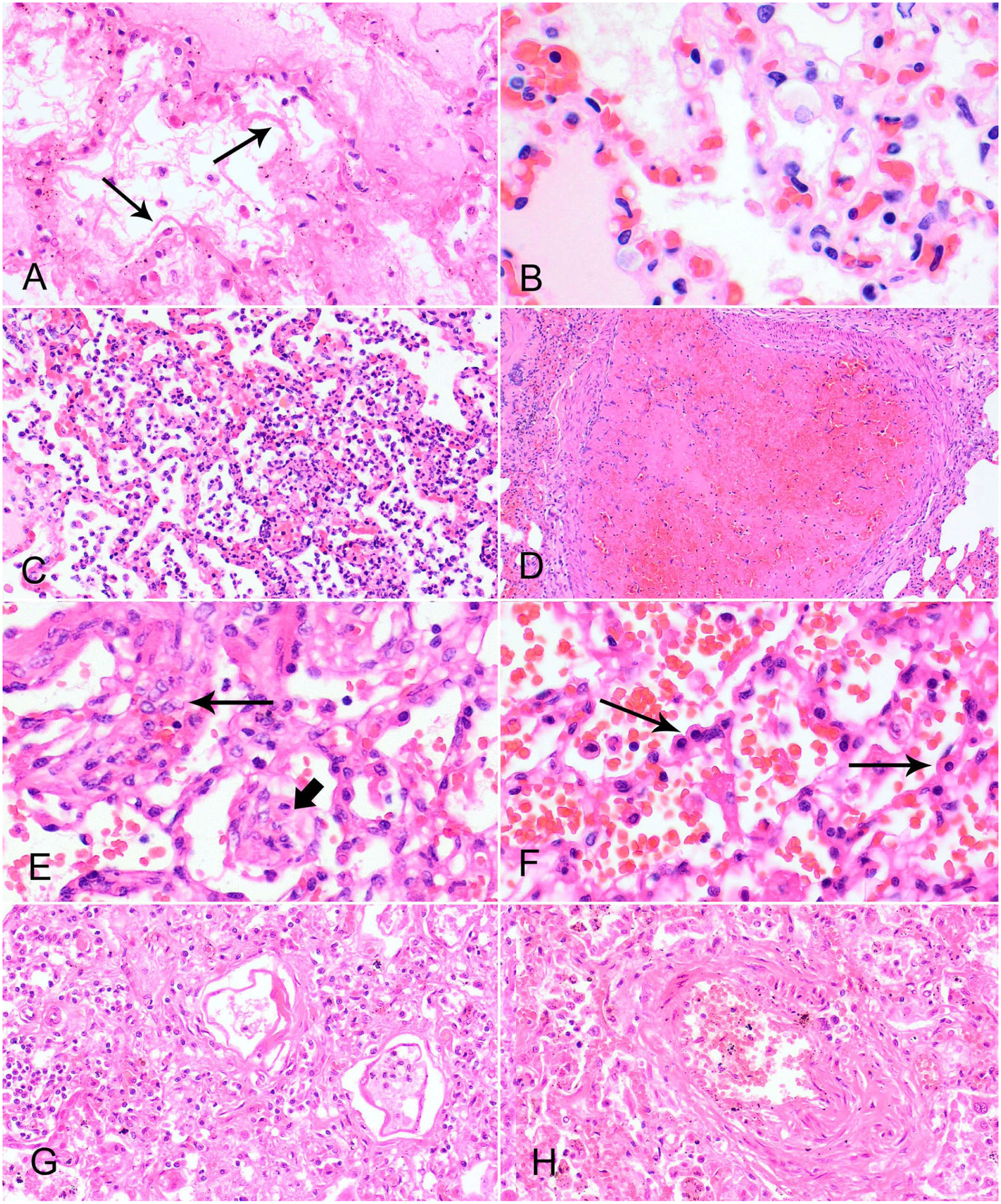
Classical histologic features of diffuse alveolar damage in dogs and cats. H&E.
Stages and key histologic features of diffuse alveolar damage.

Exudative (acute) stage
Hyaline membranes are an essential feature of DAD.26,27 However, if the damage is <24 h old, histologic changes may be minimal. Furthermore, regardless of time post injury, hyaline membranes in experimental animal models of acute lung damage are variable; therefore, diagnosing experimental acute lung injury in the absence of hyaline membranes is possible but requires supporting physiologic data (Table 1). 106 In veterinary diagnostic cases, histology suggesting acute lung damage—particularly in the absence of hyaline membranes—requires integration with the clinical context to achieve a diagnosis. This multidisciplinary approach is essential because of the nonspecific nature of the component histologic features.
Proliferative or organizing (subacute) stage
Type II pneumocyte hyperplasia is an increase in cell number with concurrent hypertrophy. The hyperplastic type II pneumocytes may also demonstrate cellular atypia, 27 including cytomegaly, karyomegaly, pleomorphism, and multinucleation. 165 Type II pneumocyte hyperplasia occurs initially at 2–3 d post insult, becoming extensive at 7 d post insult. 83 Individualized hypertrophied type II pneumocytes mimic reactive macrophages and may only be distinguishable by cytokeratin immunohistochemistry (IHC). 29 TGFβ produced by injured type II pneumocytes and macrophages initiates myofibroblast and fibroblast activation and collagen deposition. 58
Fibrosing (chronic) stage
Fibroblasts and myofibroblasts proliferate within alveolar septa and infiltrate intra-alveolar fibrin, thereby generating extensive granulation tissue and contributing to fibrotic lung disease. 27 The fibrosed alveolar exudates eventually get epithelialized and remodeled into the interstitium. Paracrine signaling from interstitial fibroblasts and alveolar macrophages can prevent type II pneumocytes from differentiating into type I pneumocytes. 156 The production of TGFβ from alveolar macrophages and tissue factor from injured endothelial cells induces the recruitment of fibroblasts to the alveolar space, followed by deposition of collagen.58,153 Depending on the extent of damage and whether injury is ongoing, collagen deposition may contribute to repair of the alveolar barrier or may lead to extensive fibrosis, resulting in a permanent decrease in lung function.
Pathophysiology of ARDS endotypes
The aim for both human and veterinary medicine is to use the ARDS endotypes to improve diagnostic and therapeutic precision in the treatment of ARDS. The premise of the ARDS endotypes is that original injury confers a retained molecular signature to the ARDS process that can be differentiated from other types of primary injury. Based on the foundation of the ARDS subphenotypes in people, ARDS etiologies were classified into 5 prospective endotypes (
Epithelial—alveolar epithelial cells are injured, impairing fluid clearance.
Endothelial—vascular barrier disruption.
Local inflammatory response—focal airway or interstitial lung damage propagate to diffuse alveolar injury.
Angiopathy—multi-organ or systemic pro-thrombotic endothelial phenotype leads to microvascular injury.
Systemic inflammatory response—ARDS develops secondary to systemic vascular dysregulation induced by systemic insults, such as sepsis or pancreatitis.
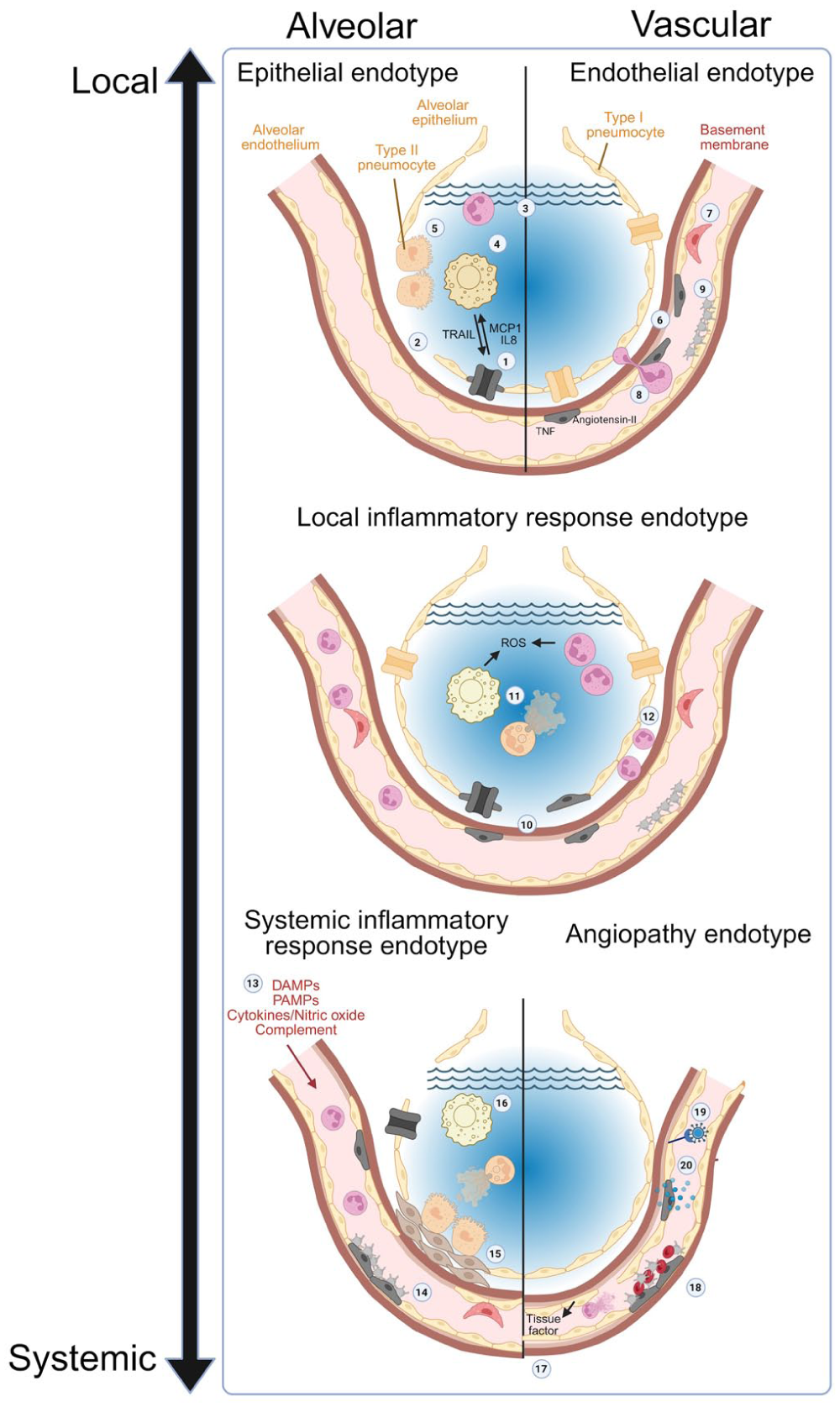
Hallmark aspects of distinct acute respiratory distress syndrome endotypes.
Determining the unique features of different endotypes is challenging because of the close spatial and functional relationships between the lung epithelial and endothelial cells, which are closely opposed on either side of a fused basement membrane to optimize gas exchange.
79
Alveolar edema can be initiated through endothelial or epithelial changes. Endothelial activation and damage increases vascular permeability.
114
Alveolar epithelial damage decreases alveolar fluid clearance through a loss of ion transporters and disruption to impermeable tight junctions.105,107 To further complicate the distinction between endotypes, primary damage in one compartment can induce inflammation in another compartment by releasing danger-associated molecular patterns (
Acute respiratory distress syndrome (ARDS) endotypes with clinical examples from cats and dogs.
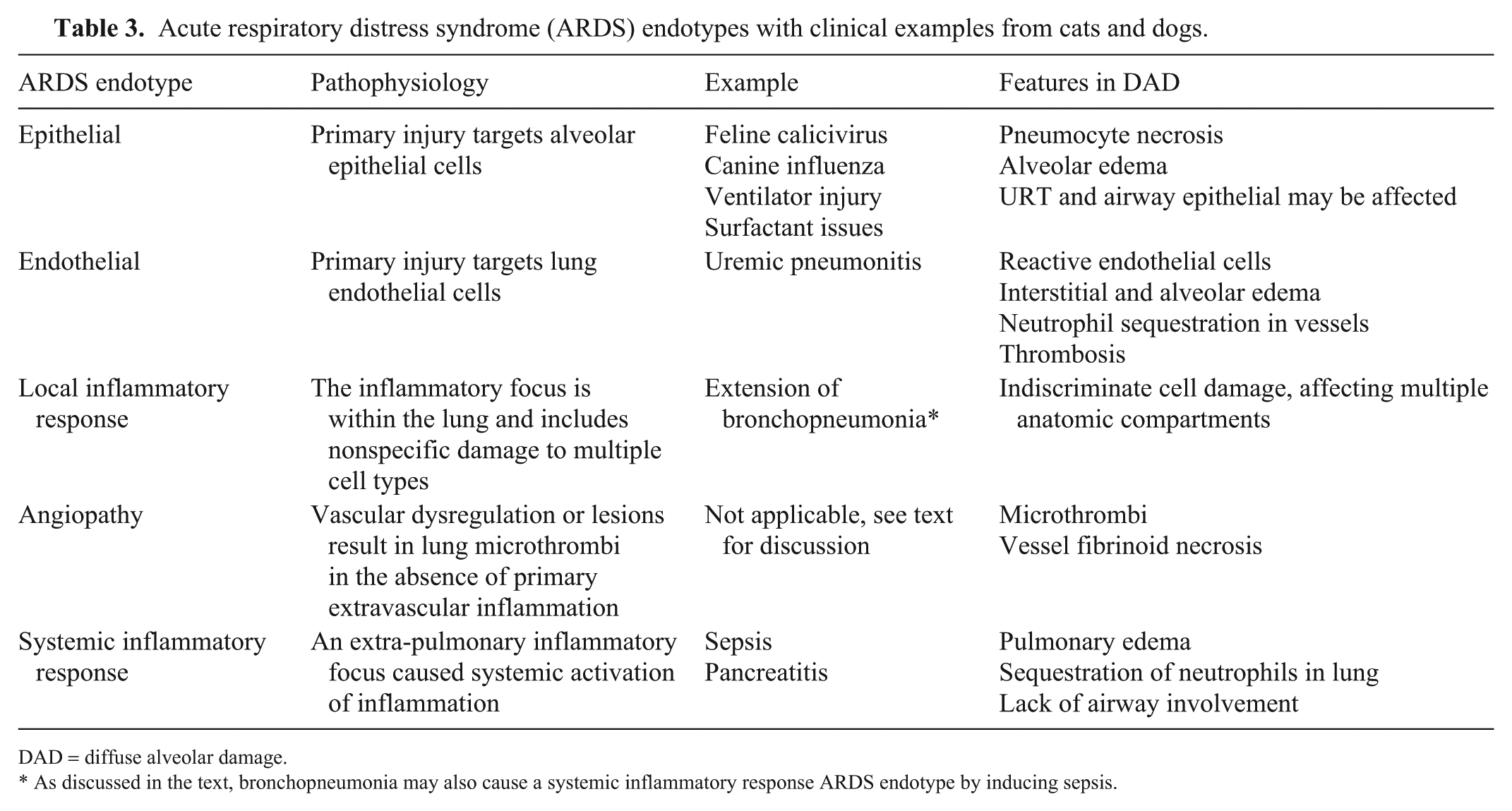
DAD = diffuse alveolar damage.
As discussed in the text, bronchopneumonia may also cause a systemic inflammatory response ARDS endotype by inducing sepsis.
Epithelial endotype
In the epithelial endotype, the primary insult damages alveolar epithelial cells. Both type I and type II pneumocytes play roles in the removal of alveolar fluid, via expression of ion transporters and water channels. 128 Paracrine signaling—for example, by catecholamines—can increase alveolar fluid clearance. Cell injury results in down-regulation or loss of transporters and decreased expression of tight and adherens junctional proteins, resulting in alveolar edema.65,107 Type I pneumocytes are very sensitive to injury, including biomechanical factors such as alveolar stretch. 108 Activation of alveolar macrophages induces release of TNF-related apoptosis-inducing ligand (TRAIL), which results in the paracrine-initiated apoptosis of alveolar epithelial cells, further potentiating the primary damage. 73 Damaged alveolar epithelial cells release cellular proteins that, alongside surfactant and plasma proteins, contribute to hyaline membrane formation. 147 Activated alveolar epithelial cells also recruit leukocytes independently of alveolar macrophage signaling through the release of the chemokine monocyte chemoattractant protein 1 (MCP1) and IL8. 150
Endothelial endotype
In the endothelial endotype, processes cause primary damage to lung vascular endothelial cells, resulting in dysregulation of lung function that ultimately progresses to ARDS. Endothelial cells have multiple key functions in lung homeostasis that may be dysregulated in ARDS and DAD. These functions include water transport out of vessels and production of inflammatory and vasoactive compounds.101,111 Endothelial cells respond to injury and inflammation by transforming from an anti-adhesive, low-permeability barrier to an adhesive, high-permeability layer. This transformation is achieved through expression of adhesion molecules and pro-thrombotic activators, and response to (and paracrine production of) vascular and inflammatory mediators, such as TNF, IL8, and angiotensin II. Heterogeneity in the responses from spatially distinct areas in the lung is influenced by the local microenvironment, 101 potentially contributing to the multifocal distribution of tissue injury in DAD.
Endothelial damage exposes the basement membrane and initiates coagulation, facilitating the development of ARDS and DAD, because activated platelets are effective activators of neutrophils. 21 Inhibition of platelet-derived neutrophil activation in an experimental mouse model of acute lung injury reduced neutrophil recruitment, lung permeability, and improved survival. 170 Mechanical factors also increase the sequestration of activated neutrophils in alveolar capillaries. Activated neutrophils undergo cytoskeletal re-arrangements that make them more resistant to deformation when traveling through alveolar capillaries that are narrower than a single neutrophil.22,47 The loss of elasticity—combined with changes in expression of endothelial adhesion molecules—facilitates cross-talk between endothelial cells and neutrophils, priming neutrophils to emigrate into the lung tissue. 22
Local inflammatory response endotype
Lung inflammation that extends locally may damage the pulmonary interstitium; and if severe, it may induce ARDS and/or DAD. Induction of ARDS and DAD in this way is predominantly secondary to airway inflammation. Inflammation in airspaces may directly or indirectly damage epithelial cells—for example, when neutrophilic exudates release reactive oxygen species (ROSs) or activate alveolar macrophages. Alveolar macrophages are central modulators of alveolar inflammation by controlling immune responses and epithelial barrier permeability, and by influencing apoptosis or necrosis of alveolar epithelial cells. 119
Angiopathy ARDS endotype
In the angiopathy endotype, endothelial dysfunction results in vasoconstriction and a procoagulant state, potentially inducing microthrombi and causing organ ischemia. The ARDS angiopathy subgroup primarily reflects the angiopathy diagnosed by imaging in human patients with COVID-19.30,118 In these patients, SARS-CoV-2–infected endothelial cells result in lung angiopathy and microthrombosis.
154
Interactions between complement and tissue factor–enriched neutrophil extracellular traps (
Outside of SARS-CoV-2 models, angiopathies as a primary cause of vetARDS are less well-recognized, partly because most primary veterinary vasculopathies could be classified as an endothelial endotype. Potentially relevant angiopathies for this endotype could include disseminated intravascular coagulation, progressing to ARDS not associated with sepsis or pneumonia.42,45 Severe cases of babesiosis occasionally result in ARDS,37,45,95 and some causes of NCPE that progress to ARDS may be relevant to this endotype. 165 Cases of canine cutaneous and renal glomerular vasculopathy have not been associated with substantial lung involvement. 74 Considering the low incidence of angiopathies not otherwise attributed to primary endothelial injury in veterinary medicine, we suggest it may be more appropriate to consider the angiopathy vetARDS endotype as a subset of the endothelial endotype, rather than a distinct group.
Clotting dysfunction is a known component of ARDS. Consistent differences in coagulation parameters are present between the P1 hypoinflammatory and P2 hyperinflammatory ARDS subphenotypes in humans, with P2 patients having elevated procoagulant and antifibrinolytic markers. 98 However, in ARDS, complement activation exacerbates inflammation induced by other primary triggers—rather than complement dysfunction being the primary disease. 7 In veterinary medicine, this is corroborated by the finding that primary coagulopathies are not considered a risk factor for canine vetARDS. 42
Systemic inflammatory response endotype
In ARDS developing from sepsis or SIRS, a pre-existing systemic inflammatory response and endothelial activation are present. The pathogenesis of the endothelial endotype also applies here and because of widespread endothelial activation, septic-ARDS cases are at high risk of systemic circulatory dysfunction. Sepsis and SIRS also amplify inflammation through the neuroendocrine-immune network, supporting further widespread homeostatic dysfunction and inflammation. Septic-ARDS and SIRS-ARDS include endothelial injury, pulmonary edema, cell death, oxidative stress, pulmonary microcirculation dysfunction, cytokine release, inflammatory signaling pathways, complement activation, and coagulation.109,169 Some of these factors are explored in more detail in Part 2, where we describe the subsequent cellular processes that follow the primary damage. 165
Endothelial injury and pulmonary edema
The key feature in the progression of sepsis to septic ARDS is the sustained increase in pulmonary vascular permeability.
169
Single-cell RNA sequencing of peripheral blood macrophages from human sepsis patients with and without ARDS had different expression patterns. ARDS patients had significantly upregulated genes related to endothelial barrier disruption, interferon responses, and fibrosis and genes that block neutrophil clearance.
80
Sheep exposed to bacterial pneumonia–induced sepsis progressed to ARDS with pulmonary vascular hyperpermeability caused by vascular endothelial growth factor (
Cell death and oxidative stress
Cell-death pathways are activated during sepsis and SIRS. In a mouse model of sepsis-induced lung injury, caspase-dependent apoptosis and pyroptosis of pulmonary microvascular endothelial cells contributed to tissue injury.31,67 Conversely, sepsis extends the lifespan of neutrophils through suppression of neutrophil cell death and debris clearance pathways. Human cases of septic shock and sepsis-ARDS have significantly lower rates of neutrophil apoptosis compared with uncomplicated sepsis, non-septic ventilated patients (including ARDS patients), and healthy controls. 59 The finding has been recapitulated in ARDS patients, where genes that block neutrophil clearance were upregulated. 80
During sepsis and SIRS, redox homeostasis is disrupted, resulting in oxidative stress. ROSs produced by NADPH oxidase complexes in neutrophils are a major contributor to the disruption.33,121 Oxidative stress induces a prothrombotic state in lung endothelial cells, while simultaneously impairing vasodilation, increasing capillary permeability, and increasing leukocyte and platelet endothelial adhesion. 81 These factors increase the likelihood of lung capillary endothelial injury.
Microvascular dysfunction
Lung microvascular abnormalities occur during sepsis and are caused by a range of processes, including endothelial dysfunction, glycocalyx alterations, WBC and platelet adhesion, and changes to RBC deformity. 39 The mechanical influences on activated neutrophils already described in the endothelial endotype section also apply to the angiopathy endotype. Together, these changes support the aggregation of neutrophils 116 and RBCs 120 in the pulmonary circulation, contributing to vascular inflammation, thrombosis, and dead-space generation. Dead space further potentiates microvascular dysfunction because hypoxic environments cause neutrophil degranulation and increase neutrophil-mediated injury in the lung. 71
Inflammation and cytokine release
Activation of classical proinflammatory innate immune signaling pathways, such as NF-κB, JAK2/STAT3, MAPK, mTOR, and Notch, in pulmonary-resident or infiltrating cell populations support the transition of sepsis into sepsis-associated ARDS. 97 Many cytokines have roles in septic-ARDS, including IL1β, IL18, IL6, IL12, IL17, and TNF. 169 Levels of IL1β, IL6, IL8, IL12, IFNγ, GMCSF, and TNF were higher in human non-survivors of sepsis compared with survivors, with IL8 levels predictive of a fatal outcome in multivariate analysis. 110 The association between IL18 elevation and clinical severity is evidenced by higher IL18 levels in human sepsis cases compared with SIRS; levels also are significantly higher in septic-ARDS than in sepsis alone. 48 In a randomized controlled clinical trial in human medicine, elevated IL18 levels were associated with mortality in sepsis-induced ARDS. 126 Circulating proinflammatory cytokines impact lung permeability by directly influencing transcription of genes that help control endothelial permeability (e.g., IL1β can suppress transcription of VE-cadherin in lung endothelial cells, contributing to sepsis-induced lung injury). 168
Pulmonary intravascular macrophages, complement and coagulation
Pulmonary intravascular macrophages (
Innate immune responses in sepsis also activate complement that results in thromboinflammation. 102 A cohort of human sepsis cases had increased levels of circulating cleaved complement protein C3a and higher C3a/C3 ratios, compared with both healthy controls and cases of SIRS without an identified inflammatory focus. 145 ARDS does not appear to induce levels of complement activation beyond that induced by sepsis, given that C3a levels and the C3a/C3 ratio did not differ between sepsis cases who developed ARDS and those who did not. 145 Vascular-related biomarkers (generated from canine patient data) for canine sepsis and SIRS have been proposed; higher levels of angiopoietin-2 at hospital admission was correlated with a negative outcome in both SIRS and sepsis, and VEGF was elevated in dogs with sepsis compared with healthy dogs. 89
Immune dysregulation
SIRS and sepsis are processes of immune dysregulation, given that hyperinflammation occurs contemporaneously with a compensatory anti-inflammatory response syndrome (
Specific examples of causes of vetARDS in dogs and cats classified by ARDS endotype
Cytolytic respiratory viruses—epithelial ARDS endotype
Influenza A virus preferentially infects respiratory epithelia in dogs28,35 and contributes to the canine infectious disease complex. 38 Infection in the cat is often subclinical. 135 For both dogs and cats, influenza can progress to clinical signs consistent with ARDS,35,143 but this requires specific strains and relatively high exposure doses. In dogs, highly pathogenic influenza A(H5N1) virus induces clinical signs consistent with ARDS and acute neutrophilic interstitial pneumonia with fibrin exudation35,143; in cats, it causes confirmed DAD with clinical signs consistent with ARDS. 86 In these cases, virus was detected using IHC within alveolar epithelial cells and luminal leukocytes, suggesting that viral-induced epithelial damage was a factor in the lung damage.
Feline calicivirus—an RNA virus that is part of the feline respiratory disease complex—causes rhinitis, pneumonia, and oral ulcers, with high morbidity and low mortality in immunocompetent cats. Feline calicivirus can induce fatal acute pneumonia with pneumocyte necrosis, marked fibrin exudation, alveolar infiltrates of macrophages and neutrophils. 142 These cases are speculated to have developed ARDS based on the fatal severity and acute nature of the histologic changes. Hyaline membranes were not described; hence, DAD was not confirmed. Viral antigen, detected by IHC, was present within pneumocytes and macrophages within alveolar spaces, supporting an ARDS epithelial endotype.
Ventilator-induced lung injury and surfactant disorders—epithelial ARDS endotype
Ventilator injury and surfactant disorders are both classified as inducing epithelial endotype ARDS, ultimately through mechanical damage to pneumocytes. Ventilator injury can cause ARDS 158 by overstretching of alveoli, 136 or through sheer forces induced by repetitive collapse and re-expansion, 50 both of which result in pneumocyte damage. Mechanical ventilation also induces surfactant dysfunction, similar to that caused by congenital surfactant disorders, which increases surface tension on pneumocytes and further increases sheer forces generated through breathing cycles. 2 Presumed ARDS with confirmed DAD caused by a congenital surfactant disorder was identified in a family of Airedale Terriers. 46
Inhaled toxic gases and metabolized hematogenous agents—epithelial ARDS endotype
Inhalation of toxic gases and hematogenous agents, which are metabolized to forms that are toxic to pneumocytes, are reported causes of ARDS in veterinary species. 27 The primary target is lung epithelial cells; thus, this process is classified as an epithelial ARDS endotype. For dogs and cats, toxic gases include 100% oxygen, 6 nitrogen dioxide 70 and chlorine. 72 Paraquat exposure in dogs is an example of a hematogenous toxin metabolized by pneumocytes.36,162
For domestic animals, the source of many toxic gases is smoke inhalation,60,144 which is a documented cause of suspected ARDS in cats 51 and dogs. 52 Smoke inhalation may also cause thermal inhalation trauma to respiratory epithelia, although because of rapid cooling of air and smoke once in the upper respiratory tract, thermal burns are more often seen in cases of steam inhalation. 164
Aspiration pneumonia—epithelial and local inflammatory response ARDS endotype
Acid aspiration
Gastric acid aspiration results in damage to epithelium, followed by neutrophilic inflammation, loss of pulmonary microvascular integrity, and pulmonary edema. 122 The volume of the aspirate contributes to the severity of the lesion. 123 At very low volumes, when microinjected directly into rodent alveoli, acid causes pore formation in epithelial cells and hydrogen peroxide release, subsequently increasing ROSs in the perialveolar microvascular endothelium. 159 A second study demonstrated primary acid-induced epithelial injury—without endothelial injury—using live confocal microscopy in an experimental mouse model of acid alveolar microinjection; however, it similarly found that epithelium-derived hydrogen peroxide induced endothelial cell retraction and barrier failure, resulting in pulmonary edema. 75 In an experimental rat model, the pH and the volume of aspirated content influenced mortality. Low pH fluid caused mortality even at relatively low volumes, whereas higher pH content was tolerated in larger volumes before mortality was induced. 78 Acid inhalation studies in rats indicated a 2-stage injury process. Damage occurring within the first hour was attributed to a direct chemical effect on the epithelium; damage ~4 h post-inhalation that coincided with infiltration of the interstitium and alveolar spaces by neutrophils. 85 In an experimental rabbit model of acid inhalation and acid-induced lung injury, 61 IL8 was a key chemotactic factor that mediated neutrophil recruitment to the lungs; IL8 neutralizing antibodies protected against clinical signs, mortality, and airspace neutrophil accumulations. These findings support the interpretation that neutrophil influx and interstitial inflammation contribute most of the damage in some experimental models. 61
Primary acid injury may also facilitate colonization of lung tissue by opportunistic bacteria present in the lower respiratory tract, 113 complicating the primary sterile event with a secondary bacterial infection. We suggest that the initial acid-related damage is an epithelial ARDS endotype, more likely to induce ARDS when large volumes of very-low pH content are aspirated. In a canine acid-inhalation model, aspiration of a moderate volume of a low pH acid (1 mg/kg, pH 1.8) caused compromise of gas exchange (measured by fractional intrapulmonary physiologic shunt) within 10 min and remained significantly elevated compared with the control for 3 h post injury. 66 We suggest that damage caused by neutrophil influxes constitutes a local inflammatory ARDS endotype through neutrophil-induced immunopathology on the interstitium.
Aspiration of bacteria
Aspiration of colonized oropharyngeal or gastric content can result in acute and severe pneumonia. Acute aspiration pneumonia in racing sled dogs (
Feline aspiration pneumonia causes acute-onset tachypnea and an alveolar pattern on imaging, with bacterial involvement detected in some cases; however, a lack of routine testing limits the assessment of the true incidence. 41
Uremia—endothelial ARDS endotype
In humans, acute kidney injury (
In humans, lung injury caused by renal failure may progress to ARDS. 100 In veterinary medicine, uremia is considered a potential cause of ARDS in dogs and cats, 34 albeit uncommon, especially because cats appear to be relatively resistant to systemic manifestations of uremia. 4 Based on human and experimental data, uremia is considered an endothelial ARDS endotype, given the effect of circulating factors on the lung endothelium. Lung abnormalities comparable to human uremic pneumonitis were present in 104 dogs with renal azotemia of both acute and chronic causes. 94 Six of the 104 dogs had respiratory distress or dyspnea, including those with sudden onset of signs; however, clinical detail is not sufficient to retrospectively confirm ARDS. The histologic appearance of uremic lung damage in dogs includes alveolar edema and mineralization of alveolar septa with necrosis of both epithelial and endothelial cells. 94 In a survey of 78 cats with non-renal lesions of uremia, the most common finding was pulmonary edema (40 of 78 cats). Five of those 40 cats also had uremic pneumopathy with mineralization of alveolar septa, neutrophilic infiltrates, and fibrin deposition. The cats were not in respiratory distress; the most frequent clinical signs were anorexia and apathy. Hyaline membranes were not reported in either the canine or feline study, and therefore these cases did not have concurrent DAD.4,94
Bacterial bronchopneumonia—local inflammatory response ARDS endotype and systemic inflammation ARDS endotype
Bacterial pneumonia is a common cause of ARDS. Hypoxemia and clinical features supportive of vetARDS were common findings in 64 confirmed cases of canine bacterial pneumonia presented to a veterinary teaching hospital. 163 In dogs and cats, bacterial pneumonia is often precipitated by an event that compromises barrier function, such as an aspiration event, viral infections, inhaled foreign bodies, nosocomial or states of immune dysfunction, or immunosuppression. 40 From an ARDS endotype perspective, 2 endotypes are potentially applicable: the local inflammatory response endotype, in which local extension of inflammation results in ARDS, and the systemic inflammatory response endotype, in which the primary pneumonia results in sepsis that progresses to sepsis-related ARDS.11,12
An understanding of the mechanism by which bacterial bronchopneumonia transitions to ARDS from different primary etiologies is hampered by a lack of prospective controlled studies. 11
Local inflammation in bacterial bronchopneumonia can be intense and contribute to clinical signs. In single-cell profiling of BALF samples from human patients with bacterial pneumonia, patients with severe bacterial pneumonia, including suspected ARDS cases, experienced a cytokine storm that was not present in patients with mild bacterial pneumonia or healthy controls. 167 In a mouse model of acute gram-positive pneumonia, lungs had acute severe interstitial pneumonia with extensive neutrophilic infiltrates induced higher levels of IL6, IFNγ, and TNF than those with mild subclinical bacterial bronchopneumonia. 166 In humans, comparison of cytokine levels between BALF and serum offer evidence of cytokine compartmentalization, indicating that local inflammatory responses in pneumonia could cause ARDS in the absence of sepsis. 12 For individual veterinary clinical cases, adopting a multidisciplinary approach could help provide evidence to support a specific endotype.
Pancreatitis—systemic inflammatory response ARDS endotype
Pancreatitis causes respiratory signs that may progress to vetARDS, 33 of 109 client-owned dogs with acute pancreatitis had respiratory signs, including 3 cases of vetARDS 92 ; 9 of 26 client-owned dogs with pancreatitis had vetALI. 68 The American College of Veterinary Internal Medicine consensus statement on feline pancreatitis states that breathing difficulties are common in pancreatitis cases and can be associated with ALI or ARDS. 63 Clinical cases of canine acute pancreatitis had reduced antithrombin, and elevated TNF, IL6, and C-reactive protein in blood samples, compared with normal controls. 91 Human cases of pancreatitis also have respiratory complications. In a retrospective study of 359 cases of acute pancreatitis, 31 of 359 (9%) patients developed respiratory failure. 49 ARDS usually developed 2–7 d after the onset of human pancreatitis and was a major contributor to deaths from acute pancreatitis occurring before hospital admission. 90
Conclusion
Causes of ARDS can be conceptually classified by the relative involvement of alveolar, vascular, pulmonary, and systemic inflammation in the primary injury. 19 The 5 endotypes—epithelial, endothelial, local inflammatory response, angiopathy, and systemic inflammatory response—offer clinically relevant combinations of these attributes. The core premise of ARDS endotypes is that the original injury confers a persistent molecular signature to the ARDS process that differs from those associated with other primary injuries, thereby facilitating precision testing and therapeutics.
In exploring clinical examples of ARDS endotypes applicable to dogs and cats, we highlight that assigning an endotype can be complicated by the multiple potential endotypes and knowledge gaps. We recognize that our designations will be modified in response to future published work, and advances in ARDS understanding and treatment will be iterative. We also believe that work toward defining ARDS endotypes will improve our understanding of the histology of ALI outside of classical DAD, further refining diagnostic accuracy.
We encourage readers to access part 2 165 of this series, which explores beyond ARDS and DAD to look at other types and causes of NCPE, including the diagnostic challenges presented by pulmonary edema. We also describe downstream immune and cell-death processes involved in ARDS that amplify the primary insult across cellular systems and anatomical locations. In addition, we review the current understanding of comparative immunology of humans, mice, cats, and dogs. 165
Footnotes
Acknowledgements
Figure 2 was created in Biorender.com. Boyle, J. (2025), ![]() . The histology images are from diagnostic cases from the Diagnostic Anatomic Pathology Service, Department of Veterinary Medicine, University of Cambridge, UK, where written consent was obtained for use of case material in teaching and research. We thank Andrea Starling, Emma Ward, Yvonne Pratt, Hugh Balmer, Mathew Rhodes, and Logan Marsh for technical assistance and expertise in the histology laboratory and the postmortem room.
. The histology images are from diagnostic cases from the Diagnostic Anatomic Pathology Service, Department of Veterinary Medicine, University of Cambridge, UK, where written consent was obtained for use of case material in teaching and research. We thank Andrea Starling, Emma Ward, Yvonne Pratt, Hugh Balmer, Mathew Rhodes, and Logan Marsh for technical assistance and expertise in the histology laboratory and the postmortem room.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
