Abstract
Malakoplakia is an uncommon granulomatous disease reported to affect mainly the urinary bladder and, less frequently, the urogenital tract, gastrointestinal tract, and lymph nodes. We describe cases of malakoplakia within the renal medulla of 2 dogs. Both dogs had a history of recurrent urinary tract infections with urine culture growth of Escherichia coli. Autopsy findings in dog 1 included ~60 tan-to-yellow, irregular nodules within the renal medulla, and tan-to-red, semi-firm nodules within the urinary bladder mucosa. Dog 2 had no gross changes within the kidneys but rather had ~20 red, semi-firm nodules along the surface of the apex of the urinary bladder. Histologically, the renal medulla of both dogs had sheets of foamy IBA1-positive macrophages with cytoplasmic granules and inclusions of intensely periodic acid–Schiff (PAS)-positive and occasional intracytoplasmic von Kossa– and Prussian blue–positive Michaelis–Gutmann bodies, consistent with malakoplakia. Fluorescence in situ hybridization and/or immunohistochemistry confirmed E. coli within macrophages. An underlying endocrinopathy could be a predisposing factor in the development of malakoplakia in older dogs.
Malakoplakia is a poorly understood and uncommon granulomatous disease typically affecting the urinary bladder. Lesions have been described mainly in the genitourinary and gastrointestinal tract in people; in the urinary bladder, vagina, large intestine, lymph node, and systemically in multiple domestic species; and in the kidney of a cynomolgus macaque.1 –4,6,8,9,13,14,16 –18,20 Clinical signs of urinary malakoplakia can vary from subclinical to urinary incontinence, dysuria, hematuria, pollakiuria, and stranguria. Malakoplakia can range from no gross lesions to characteristic soft, yellow-to-brown plaques or nodules with or without umbilication on the mucosal surface.16,20
The pathogenesis of malakoplakia is not fully understood, but has been postulated to be a defect in the phagolysosomal function of macrophages to digest microorganisms, such as Escherichia coli.12,16,20 One proposed theory is that phagolysosomal dysfunction results from decreased cyclic guanosine monophosphate, which plays a role in microtubular assembly and degranulation of lysosomes for phagocytic killing of bacteria.16,20 Incompletely digested bacteria accumulate within phagolysosomes and over time cause minerals, such as calcium, phosphorus, and iron, to deposit—identified histologically as Michaelis–Gutmann (
Two dogs from separate veterinary institutions were evaluated for systemic disease and ultimately euthanized and submitted for autopsy. Dog 1 was a 12-y-old spayed female Vizsla presented to Texas A&M University (College Station, TX, USA) with a chronic history of pneumonia, an ~1-y history of recurrent bacterial urocystitis, and mixed bowel diarrhea. On presentation, the patient had decreased body and muscle condition, with crackles noted caudoventrally on thoracic auscultation. Thoracic radiographs revealed a nonspecific diffuse interstitial pattern. Abdominal ultrasound revealed bilateral chronic degenerative nephropathy and an irregularly marginated urinary bladder lumen with numerous nodular proliferations. An additional finding was mild bilateral adrenomegaly. Analysis of urine collected by cystocentesis revealed isosthenuria [specific gravity (SG) 1.010] with a few WBCs; a quantified urine culture grew 600 E. coli cfu/mL.
Dog 2 was a 5-y-old intact female Great Dane that was presented to The Ohio State University (Columbus, OH, USA) because of an acute onset of paraplegia. Additionally, the patient had historic diabetes mellitus, hypothyroidism, and an ~2-y history of chronic relapsing bacterial urocystitis with multidrug-resistant organisms. On presentation, the patient was a non-ambulatory paraplegic with pain elicited on thoracolumbar palpation. Urine collected by cystocentesis was minimally concentrated (SG 1.013) with many WBCs and bacteria; a quantified urine culture grew >100,000 E. coli and Enterococcus faecium cfu/mL.
On gross examination of dog 1, the mucosa of the urinary bladder contained approximately sixty 1 × 1 × 1-mm to 3 × 3 × 5-mm, tan-to-red, semi-firm nodules. Both kidneys were small and firm, with irregularly pitted cortical surfaces and pale-tan to white, firm, 1–3-mm, acquired cysts. On cut section, the renal medulla contained 2–5-mm yellow, nodular areas (
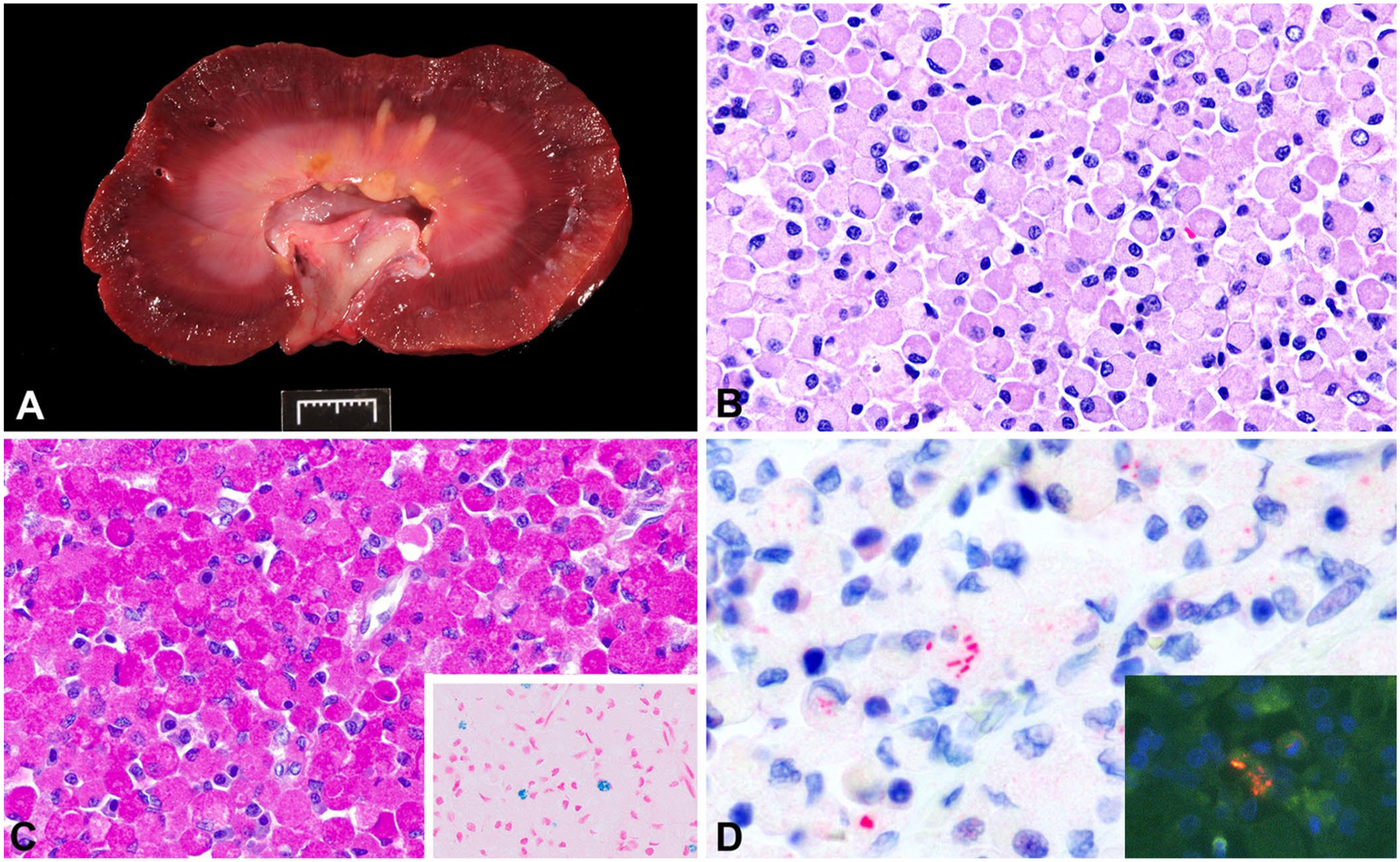
Escherichia coli–associated renal malakoplakia in a dog.
Histologically, dense sheets of eosinophilic foamy macrophages (
Sheets of foamy macrophages forming well-demarcated nodules were found within the urinary bladder of dog 1, lifting the overlying urothelium, expanding the lamina propria, and dissecting the smooth muscle. The mucosal surface overlying these nodules was multifocally eroded. The adjacent lamina propria contained moderate numbers of lymphocytes, plasma cells, fewer neutrophils, foamy macrophages, and prominent lymphoid follicles. The lamina propria of dog 2 showed a similar focal population of foamy macrophages and moderate numbers of lymphocytes and hemosiderophages, plus edema.
Additional histologic findings in dog 1 included marked numbers of lymphocytes and plasma cells with fibrosis and crypt dilation in the small intestine, consistent with chronic inflammatory enteropathy. The adrenal glands had moderate bilateral cortical hyperplasia, the lungs had diffuse alveolar septal mineralization, and the pituitary gland was unremarkable. In dog 2, small numbers of lymphocytes and plasma cells expanded and replaced cells in the thyroid glands, a finding consistent with the history of hypothyroidism.
Based on the histopathologic findings within the renal medulla and urinary bladder of both dogs, we performed periodic acid–Schiff (PAS), von Kossa, and Prussian-blue staining, and immunohistochemistry (IHC) for ionized calcium-binding adapter molecule 1 (IBA1), a marker for histiocytes. Macrophages had diffuse cytoplasmic labeling for IBA1 and cytoplasmic inclusions stained intensely PAS positive (
In people, malakoplakia most often affects the genitourinary tract, with 40% of cases involving the urinary bladder; the gastrointestinal tract is the second most common location.16,19,20 Women tend to be more commonly affected than men, with individuals typically >50-y-old.16,20 In approximately two-thirds of human patients, malakoplakia is linked with persistent infection by E. coli, as seen in our cases and in 50% of the previously reported cases in veterinary species.16,19 Other proposed causes for the development of malakoplakia in people include immunosuppression secondary to chronic disease, neoplasia, or immunosuppressive therapy, most commonly related to renal transplantation.16,19,20 In veterinary species, the development of malakoplakia is less commonly diagnosed than in human medicine. The few species reported to develop malakoplakia include 7 dogs, 3 cats, 3 pigs, and a cynomolgus monkey, in which the urogenital tract was most commonly affected.1 –4,6,8,9,13,14,16 –18,20 Of the 7 reported cases in dogs, 6 were brachycephalic breeds, including 2 French Bulldogs, 1 Pug, 2 English Bulldogs, and 1 Bulldog. Similar to human cases and our own study, these veterinary patients were more likely to be females. However, most veterinary species tend to be younger, with an age range of 6-wk-old to ~4-y-old.
In malakoplakia, large numbers of foamy macrophages (i.e., von Hansemann macrophages) expand the tissue parenchyma due to an ineffective inflammatory response by macrophages to bacteria. These cells contain basophilic-to-eosinophilic cytoplasmic MG bodies considered pathognomonic for malakoplakia. MG bodies can be difficult to identify with H&E and are better appreciated with PAS. These inclusions are believed to form when bacteria are incompletely digested by macrophages; the subsequent accumulation of bacteria in the cytoplasm then becomes a nidus for gradual deposition of calcium and other minerals, as identified with von Kossa and Prussian-blue stains. In the acute stage of the disease, MG bodies may not always be apparent; however, their absence should not exclude the diagnosis of malakoplakia.15,16 Experimental injection of E. coli into the kidneys and testes of rats initially showed an absence of MG bodies, which formed later in the disease process. 5 Interestingly, E. coli–associated granulomatous colitis was referred to as “malakoplakia” in a French Bulldog due to the presence of MG bodies. Clinicians widely recognize the condition in predisposed French Bulldogs and Boxer dogs as E. coli–associated granulomatous colitis; however, “malakoplakia” may not be the most appropriate term for describing the gastrointestinal lesions. 10
Besides the positive urine culture of E. coli in both dogs, IHC confirmed clusters of E. coli antigen in their kidneys. However, FISH was negative in dog 2 for both E. coli and the eubacterial probe EUB338. FISH is a molecular technique used to detect nucleic acids and is considered a gold-standard method for visualizing intracellular and intramucosal bacteria. 11 FISH has been used in the diagnosis of granulomatous colitis as well as E. coli–associated malakoplakia. Possible explanations for negative DNA detection by FISH but positive immunolabeling for E. coli on IHC include degeneration of the bacterial DNA or low permeabilization of the intracellular bacteria, preventing probe hybridization; or less likely, cross-reactivity of the antibody with other bacteria, resulting in a false-positive IHC. Both IHC and FISH are considered sensitive in situ techniques for detecting invasive E. coli in granulomatous colitis of dogs. 11
Electron microscopy has also been used conjunctively in the diagnosis of malakoplakia. Findings can include macrophages with abundant lysosomes and phagolysosomes and containing bacteria and/or MG bodies, described as being concentrically laminated with an irregular outer surface and less electron-dense core containing calcium deposits.12,16 In dog 2, many macrophages with cytoplasmic vacuoles were observed in the kidney, but no identifiable bacteria were found. Given the chronicity of the lesions, it is likely that the phagocytized E. coli were partially digested and no longer recognizable as bacteria.
Malakoplakia is considered a sporadically induced disease in veterinary species. However, given the age and comorbidities of these 2 dogs, additional causes for this disease should be considered. In human patients with malakoplakia, 21% were immunosuppressed, suggesting that multiple mechanisms were involved in the development of this disease beyond the simple presence of bacteria. 7 Both dogs in our report had a history of recurrent urinary tract infections, from which E. coli was consistently grown. Dog 2 had been diagnosed with diabetes mellitus and hypothyroidism, which have been correlated with recurrent urinary tract infections and altered immune status. Similarly, dog 1 had ultrasonographic, gross, and histologic evidence suspicious for hyperadrenocorticism, which is known to compromise immune function; although confirmatory testing was not performed. Thus, an underlying immune-compromising endocrinopathy, resulting in altered urogenital microbiota, altered immune status, and upregulation of microbial growth, may have contributed to the development of renal malakoplakia in our 2 cases.
Footnotes
Acknowledgements
We thank the personnel of the Pathology Diagnostic Laboratory, Diagnostic Histotechnology Laboratory, and Gastrointestinal Laboratory at Texas A&M University, and The Ohio State University histotechnology laboratory for their technical support. We also acknowledge the contributions of Dr. Lori Lind, who died in 2023.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
