Abstract
The pathogenesis of meningoencephalomyelitis of unknown origin (MUO) and steroid-responsive meningitis-arteritis (SRMA) in dogs remains enigmatic. Numerous studies have attempted and failed to identify (viral) pathogens in samples from MUO- or SRMA-diagnosed dogs. Orthoflavivirus-associated meningoencephalitis or meningoencephalomyelitis has been diagnosed in dogs in several European countries. We investigated serologic evidence for orthoflavivirus infection in dogs with clinical diagnoses of MUO or SRMA in the Netherlands. Twelve dogs with a clinical diagnosis of MUO based on signalment, neurologic examination, MRI studies, CSF analysis, and response to treatment were included in the study (age range: 1–11 y; 4 females, 8 males; weight range: 8–44 kg). Serum samples from all 12 dogs tested negative in a commercial competitive ELISA and virus neutralization tests for West Nile virus, Usutu virus, and tick-borne encephalitis virus. We did not find serologic evidence of orthoflavivirus infection in dogs with MUO or SRMA in the Netherlands.
The terms meningoencephalomyelitis of unknown origin (MUO) and steroid-responsive meningitis-arteritis (SRMA) refer to inflammatory disorders of the CNS. MUO variants are characterized and classified histologically as necrotizing meningoencephalitis, necrotizing leukoencephalitis, and granulomatous meningoencephalitis. 8 MUO can account for ~50% of CNS inflammatory disorders in dogs. 11 To date, it is deemed most likely that MUO and SRMA are autoimmune disorders.2,3,8 Indeed, autoantibodies against neuronal and glial antigens have been identified in some cases.2,3 Nevertheless, it is possible that, in some patients, MUO has an infectious cause that is not identified by clinical or histologic tests. Orthoflaviviruses such as tick-borne encephalitis virus (TBEV; Flaviviridae, Orthoflavivirus encephalitidis) and West Nile virus (WNV; Flaviviridae, Orthoflavivirus nilense) have caused meningoencephalitis in human patients in the Netherlands since ~2015.19,23 Recognizing the emerging relevance of orthoflaviviruses in clinical veterinary medicine,9,10,12,13,15,16,18,20,21 we investigated serologic evidence for orthoflavivirus infection in dogs with MUO in the Netherlands.
Sampling of the animals in our study was performed under legislation (license AVD40100202114384) of the Dutch Central Authority for Scientific Procedures on Animals (CCD), and the experimental plan was approved by the Animal Welfare Body of Wageningen University and Research prior to the start of the sampling. For our prospective study, we included dogs with a clinical diagnosis of MUO or SRMA based on signalment, neurologic examination, and either or both MRI studies, and CSF analysis between May 2021 and May 2022. This subset of dogs was part of a larger serosurvey set up to investigate the seroprevalence of orthoflaviviruses in horses and dogs in the Netherlands. 22 Serum samples were screened for antibodies against orthoflaviviruses by a commercial multi-species WNV competitive ELISA (ID Screen; IDvet). Additionally, virus neutralization tests (VNTs) were performed for WNV, Usutu virus (USUV; Flaviviridae, Orthoflavivirus usutuense), and TBEV as described previously. 22
We included 12 dogs (age range: 1–11 y; 4 females, 8 males; weight range: 8–44 kg) in our study. All dogs spent 0–6 h outdoors per day. Of these 12 dogs, 8 were diagnosed with MUO and the remaining 4 with SRMA. Four dogs recovered fully, 3 recovered partially, and 5 dogs were euthanized. All serum samples tested negative for the presence of orthoflavivirus antibodies (Table 1). Seven dogs were tested for canine distemper virus (CDV) among other pathogens, for which all results were negative.
Signalment and case information of dogs with a clinical diagnosis of meningoencephalitis of unknown origin, from which serum samples were obtained and included in the study.
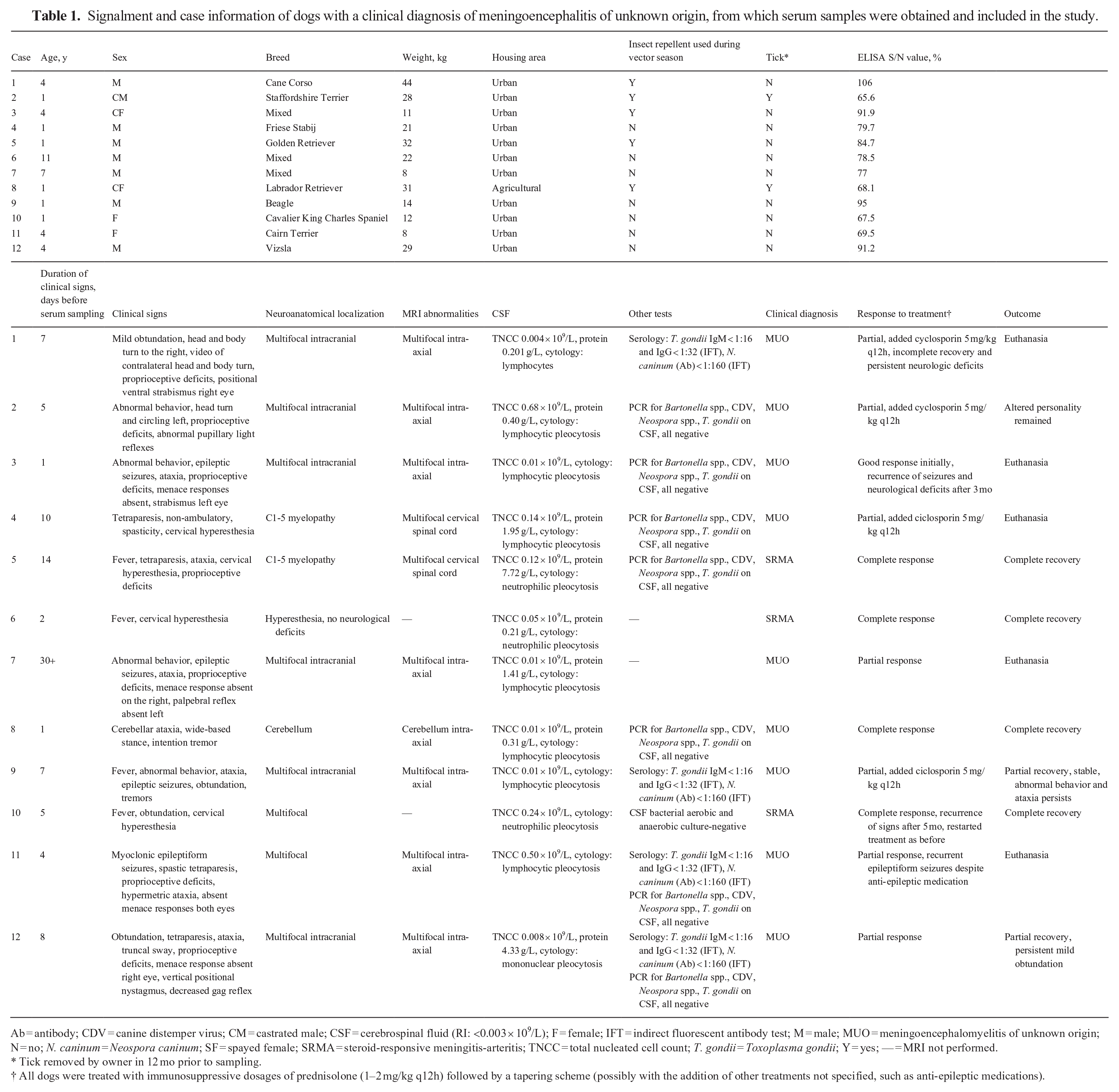
Ab = antibody; CDV = canine distemper virus; CM = castrated male; CSF = cerebrospinal fluid (RI: <0.003 × 109/L); F = female; IFT = indirect fluorescent antibody test; M = male; MUO = meningoencephalomyelitis of unknown origin; N = no; N. caninum = Neospora caninum; SF = spayed female; SRMA = steroid-responsive meningitis-arteritis; TNCC = total nucleated cell count; T. gondii = Toxoplasma gondii; Y = yes; — = MRI not performed.
Tick removed by owner in 12 mo prior to sampling.
All dogs were treated with immunosuppressive dosages of prednisolone (1–2 mg/kg q12h) followed by a tapering scheme (possibly with the addition of other treatments not specified, such as anti-epileptic medications).
Numerous studies have attempted but largely failed to identify evidence for the presence of, or role for, various pathogens, such as astroviruses, bornaviruses, CDV, Toxoplasma gondii, and Borrelia burgdorferi, in samples of dogs with a clinical diagnosis of MUO or SRMA.2,3,7 When an infectious cause is identified, the term MUO or SRMA is discarded. Orthoflaviviruses may cause meningoencephalomyelitis in dogs as well as other mammals, including humans.1,6,12 The Orthoflavivirus genus (positive-strand RNA viruses—arthropod vector-borne) includes WNV, USUV, TBEV, dengue virus, and yellow fever virus, among others. 14 Dog ownership has been identified as a risk factor for TBE in humans. 17 Ticks, such as Ixodes ricinus, are involved in the infection of dogs with TBEV.4,21 Diagnosis of TBE has been reached in other studies of dogs in Europe by identification of antibodies in serum and CSF and/or testing for viral RNA.1,4,13 Even though experimental WNV infections in dogs did not result in clinical signs, 5 clinical WNV infections have been reported in which animals had signs of encephalitis and myocarditis, among other signs. 6 We found no records of clinical cases of USUV in dogs in the literature.
The seroprevalence of orthoflaviviruses in dogs in Europe varies per country but may be up to 16.4% for TBEV and 55.5% for WNV.10,16 TBEV, USUV, and to a lesser extent WNV, are known to circulate in the Netherlands, as infections have been proven in both vectors and dead-end hosts.9,18,20 As we did not detect seropositive dogs, our study does not support a role for orthoflavivirus infection in dogs clinically diagnosed with MUO or SRMA in the Netherlands. Our results can, however, not exclude the possibility of orthoflaviviruses (or other unidentified pathogens) causing meningoencephalitis in dogs in the Netherlands, as we were not able to perform viral testing on relevant tissues and, for some of the dogs, the time to develop proper antibody responses may have been too short. Unfortunately, we were unable to perform additional sampling of the surviving animals and postmortem examination of dead animals.
As the search for pathogens involved in meningoencephalomyelitis in dogs continues, future studies should consider and possibly address several limitations of our study, including lack of concurrent antibody testing of CSF, small cohort size (12 dogs), imperfect sensitivity and specificity of the used serologic test, and timing of sampling (repeated sampling from the same animals was not performed to detect seroconversion). We encourage histologic confirmation of clinical diagnoses of MUO in future cases.
Footnotes
Acknowledgements
We thank colleagues in the clinic and from the neurology department of the involved animal hospitals for the care of the patients included in our study and for the contribution of patient samples.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Testing of the samples was part of the project “Preparing for Vector-Borne Virus Outbreaks in a Changing World: a One Health Approach” (NWA.1160.18.210), which is partly financed by the Dutch Research Council (NWO).
