Abstract
Psittacine beak and feather disease (PBFD) is caused by the psittacine beak and feather disease virus (PBFDV; Circoviridae, Circovirus parrot). This disease affects mainly young captive birds, inducing lesions mainly in the skin adnexa and lymphoid organs. Here we report PBFD in 2 free-living great green macaws (Ara ambiguus). The birds were depressed and had lost feathers and body condition. The diagnosis was confirmed by gross lesions and PCR testing in one bird, and by gross and microscopic lesions, PCR testing, viral sequencing, and in situ hybridization in the other bird. Gross lesions in both birds included mild beak discoloration and feather loss. Microscopic lesions in the bird whose tissues were examined histologically included bronchopneumonia and severe lymphoid depletion with intracytoplasmic and intranuclear botryoid inclusion bodies in the cloacal bursa. Sequences of the viral DNA obtained from paraffin-embedded cloacal bursa tissue had 100% nucleotide and 100% amino acid identity with several strains of PBFDV isolated from captive birds in multiple countries. To our knowledge, PBFD has not been reported previously in free-living great green macaws.
Psittacine beak and feather disease (PBFD) is an infectious disease that affects more than 60 psittacine species worldwide and is caused by psittacine beak and feather disease virus (PBFDV; Circoviridae, Circovirus parrot). PBFD is a progressive condition that affects individuals <5-y-old. The disease can be subclinical or clinical, mild or severe, but is usually fatal.29,43 A search of PubMed, Google Scholar, and CAB abstracts did not yield any previous report of PBFD in free-living great green macaws (Ara ambiguus). Here we describe 2 cases of PBFD in free-living great green macaws, with special emphasis on the diagnostic workup. In addition, we provide a literature review, focused on the main epidemiologic, clinical, and diagnostic aspects of PBFD.
Great green macaws are a large species of macaw, 85–90 cm in length. Sexual maturity is reached at 4–5 y of age, and the lifespan is up to 40 y. 37 This species has a distribution ranging from Honduras to Ecuador and is classified as critically endangered, with a wild population of 500–1,000 adult individuals in the wild and a recent steep reduction in its population, with a 99% decrease from 2009 to 2019. 1 The population in Sarapiqui, Costa Rica, is considered one of the last bastions of the species and is critical for conservation efforts. 1 One of the proposed steps for the conservation of the species is the implementation of captive breeding programs to reintroduce new genetics into the population. 1
In April 2021, 2 free-living great green macaws were rescued from Virgen de Sarapiquí county, Heredia province, Costa Rica. Case 1 was an ~7-wk-old female, aged by the typical growth curve of a green macaw with apparent stunting, 37 that weighed 450 g. The bird had fallen from its nest and had been taken to the Toucan Rescue Ranch (TRR; Heredia province, Costa Rica) for medical evaluation. On arrival at the TRR, the bird was inappetent, lethargic, had mild beak discoloration, and had lost a few feathers on the neck and abdomen. The bird died shortly after and, although an autopsy was not performed, feathers were collected.
Case 2 was an ~8-wk-old male, aged by the typical growth curve of a great green macaw with apparent stunting, 37 that was found on the ground near the site of discovery of the first bird and was also taken to the TRR for medical evaluation and treatment. On arrival at the TRR, the bird weighed 860 g, was lethargic, anorectic, and had mild beak discoloration and mild multifocal feather loss. He died shortly after, and the body was submitted for autopsy. The carcass was mildly autolyzed and in poor nutritional condition, with no fat reserves, serous atrophy of coronary fat, and severe skeletal muscle atrophy. Externally, the carcass was moderately infested with lice of the family Menoponidae, had small scabs on the face, and a moderate loss of feathers, mainly from the abdomen (Figs. 1, 2). The lice were identified based on their morphologic features, observed with a stereo microscope. The liver had mild diffuse atrophy; the lungs had multifocal, 0.1–0.3-cm foci of hemorrhage; and the cloacal bursa (bursa of Fabricius) was markedly atrophic (Fig. 3). Muscle, skin, spleen, heart, cloacal bursa, liver, lung, trachea, pancreas, adrenal gland, thyroid gland, ventriculus, proventriculus, small bowel, large bowel, kidney, and brain samples were collected and fixed in 10% buffered, pH 7.2, formalin before being processed routinely to produce 4-µm thick H&E-stained sections.

Gross lesions in a great green macaw with psittacine beak and feather disease.
Microscopically, the corneal layer of the epidermis was mildly hyperkeratotic and was infiltrated by a few heterophils. Within several feather follicles, organisms consistent with mites, with small, jointed appendages, a chitinized cuticle, and striated muscle, were observed. Surrounding these follicles were moderate numbers of lymphocytes and plasma cells, and hemorrhages. The cloacal bursa had severe atrophy of the lymphoid follicles, with abundant cell debris and severe fibrosis of the interstitium (Fig. 4). Multifocally, intracytoplasmic and intranuclear basophilic botryoid inclusion bodies were observed within macrophages (Fig. 5). In lung sections, the parabronchi, mesobronchi, and secondary bronchi contained many heterophils, and multifocally this inflammatory infiltrate was associated with fibrin and plant material. Blood vessels of both lungs and cloacal bursa were congested. No significant microscopic changes were observed in the other organs examined.
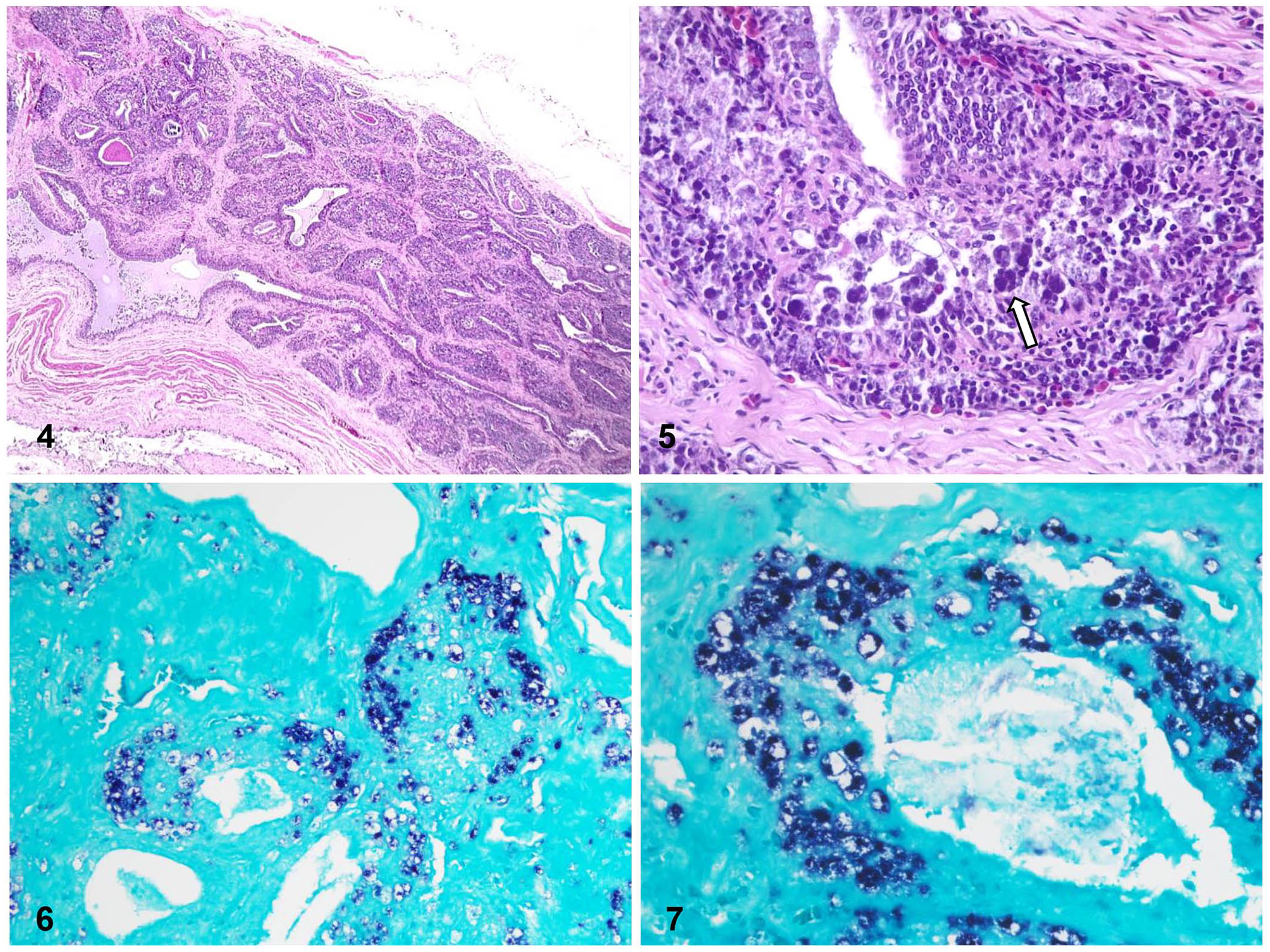
Microscopic lesions and in situ hybridization (ISH) for psittacine beak and feather disease virus detection in a great green macaw with psittacine beak and feather disease.
DNA was extracted from the feathers of case 1 and from frozen liver, kidney, and spleen samples of case 2 (Blood and tissue kit; Qiagen). As fresh cloacal bursa was collected during the autopsy, DNA was extracted from formalin-fixed paraffin-embedded (FFPE) cloacal bursa (NucleoSpin DNA FFPE XS kit; Macherey-Nagel), following the manufacturer’s instructions.
The detection of PBFDV was performed using conventional PCR. A region of 202 bp of the highly conserved gene orfC1 (encoding the capsid protein) was amplified following a protocol described previously, 28 using the following primers: circo-s (5′-CGGTGCCAGAAAATGGTATGTTAG-3′) and circo-as (5′-GAAGCTGAAGCCAATGCCGTA-3′). The amplification program consisted of an initial denaturation at 95°C for 5 min, followed by 35 cycles of denaturation (94°C for 30 s), alignment (60°C for 30 s), extension (72°C for 30 s), and a final extension at 72°C for 10 min. Positive control PBFDV DNA was kindly provided by the Clinic of Birds, Reptiles, Amphibians and Fish, Justus Liebig University, Giessen, Germany. Molecular biology–grade water (Fermentas) was used as the negative control. Amplicons were visualized in 1% agarose gels stained with GelRed nucleic acid gel stain (5 μg/mL; Biotium).
PCR for PFBDV was positive in feathers of case 1 and in paraffin-embedded cloacal bursa of case 2. Sequencing (Macrogen) of the viral DNA obtained from the FFPE cloacal bursa confirmed 100% nucleotide identity with viral strains isolated from several avian species located in multiple countries (Table 1) and was deposited in GenBank (PP781980). Analysis of the amino acid sequence showed also 100% identity with 12 strains (Suppl. Fig. 1), with modifications in 2 of 5 amino acids located on the hypervariable segment of the capsid protein C1. PCR on spleen, kidney, and liver of case 2 was negative. Regardless of possible false negatives inherent to any PCR technique, it is worth mentioning that PBFD lesions were only observed in the cloacal bursa and maybe the viral burden was minimal or absent in other organs, which could explain the negative results.
Characteristics of viral strains with 100% nucleotide identity to psittacine beak and feather disease virus (PP781980) isolated from formalin-fixed cloacal bursa of a great green macaw (Ara ambiguus) from Costa Rica.

FFPE cloacal bursa of case 2 was also processed for in situ hybridization (ISH) for PBFDV, based on modified protocols10,11 and using a manual capillary-action workstation (Fisher). Briefly, FFPE tissue blocks were sectioned at 3 μm. The tissue sections were mounted on 3-aminopropyltriethoxylane–coated slides (Probe-On Plus; Fisher) and placed in a holder for capillary gap formation (Fisher). The mounted tissues were deparaffinized by immersion in 100% d-limonene (Fisher) for 3 consecutive 15-min incubations. The deparaffinized tissues subsequently were rehydrated by 3 immersions in 100% ethanol and a final immersion in 70% ethanol. The slides were rinsed twice in Automation buffer, pH 2.0, (Biomedica) prior to digestion in 0.3% pepsin solution in Automation buffer (pH 2.0). Digestion occurred with incubation at 37°C for 10 min. The pepsin was heat-denatured by incubation at 100°C for 5 min and removed by 3 sequential washes in 1× Automation buffer (pH 7.0). A pre-hybridization step, using 200 μL of hybridization buffer (0.5% deionized formamide, 7.5% chondroitin sulfate, 5× saline sodium citrate [SSC], 50 mM phosphate buffer, and deionized water), was added to the slides to prevent nonspecific binding of the probes. After incubation at 100°C for 5 min, the pre-hybridization solution was replaced with 200 µL of hybridization buffer containing 2 µL of a 20-pmol solution of a PBFD 30-base oligo, digoxigenin end-labeled (Roche DIG oligonucleotide 3′-end labeling kit, 2nd generation; Sigma Aldrich). Washing and DIG detection followed a published method. 11 This technique successfully detected PBFDV DNA (Figs. 6, 7).
In case 1, a presumptive diagnosis of PBFD was established based on clinical signs and confirmed by PCR on feathers. In case 2, a presumptive diagnosis of the same disease was established based on clinical signs, and gross and microscopic lesions, and confirmed by PCR on FFPE cloacal bursa, sequencing analysis, and ISH.
The 2 birds in our study were young, which agrees with the literature, and indicates that PBFD can produce a fatal disease in young birds.13,29 Although some of the lesions of PBFD, such as dystrophic feathers, beak discoloration, and elongation with fractures, are suggestive of the disease, they are not pathognomonic, and it is necessary to complement autopsy and histology with other diagnostic techniques. This is even more important because lesions may vary depending on age, species of birds, and course of the disease. The lesions observed in the cloacal bursa of case 2 probably resulted in severe immunosuppression that can explain, at least partially, the secondary lesions of heterophilic bronchopneumonia, as well as the severe louse and mite infestations. The plant material within airways, together with an inflammatory reaction, is suggestive of aspiration pneumonia. No digestive abnormalities were seen in the autopsy that could explain this lesion. Neither encephalitis nor ganglioneuritis were seen in the examined sections, thus proventricular dilation disease (PDD) was discarded as a possible predisposing cause for these pulmonary lesions. No microscopic lesions suggestive of PBFD were observed in the skin of this animal, and there are different possible reasons for this. First, lesions of PBFD in macaws are not thoroughly described, and infection with PBFDV could produce a syndrome similar to the one described in African grey parrots in which the lymphoid organs are primarily affected. 34 This hypothesis would also explain why the PCR assay on frozen tissues of this bird was negative. Another possible explanation could be that the severe infestation by lice and mites may have masked mild viral skin lesions. The mites observed microscopically were found inside the feather follicles, a location characteristic of the family Epidermoptidae, which includes parasites classified in the genera Microlichus and Myialges, both of which parasitize feather follicles. 30 Follicular changes associated with infestation by these mites might explain the mild feather loss observed in this bird and could have masked possible viral lesions in the feather follicles as well.
Some arthropods, including houseflies and some hematophagous insects, may act as potential vectors of porcine circovirus. 22 PBFDV antigen was found in the intestine of mites of the species Knemidocoptes pilae in a wild sulphur-crested cockatoo with PBFD by immunohistochemistry (IHC). 22 The bird in case 2 had mite infestation. Although the presence of PBFDV was not evaluated in these ectoparasites, their role as vector could not be excluded. Further studies are needed to assess the possible role of the virus within these arthropods in the transmission of this disease. Another possible route of transmission is vertical, although in our cases, no information was available about the parents of these birds; therefore, we cannot confirm or rule out this possibility.
Sequencing of the PBFDV associated with case 2 confirmed that the strain isolated was identical to strains isolated previously from different psittacine species and in multiple countries (Suppl. Fig. 1), confirming that PBFDV has a worldwide distribution and infects a wide range of species. However, the susceptibility to infection and disease may vary between different psittacine species. 26
Information about PBFDV in Costa Rica is scarce, although this virus has been described in captive birds.7,18 A thorough review of the literature, including PubMed, CAB, and Google Scholar did not yield any cases of PBFD in free-living great green macaws.
PBFD was first described in detail in Australia, in both captive and wild psittacines, in the early 1970s, although previous descriptions of this disease had been reported in the literature since the late 19th century. 38 This disease is caused by PBFDV, which is in the Circoviridae family with porcine circoviruses 2 and 3 (PCV2, PCV3), canine circovirus, 15 and other avian circoviruses that affect, among others, canaries, geese, pigeons, and gulls. 39 PBFDV is an ~14–16-nm, nonenveloped virus, with icosahedral symmetry and a single-stranded ambisense, circular DNA genome. Circoviruses, specifically the PCVs, were the first animal viruses in which single-stranded circular DNA was found. 38
The complete genome of PFBDV is ~1.7–2 kb. 31 Although it contains 7 open reading frames (ORFs), only 2 are relevant: ORF V1 and ORF C1. ORF V1 encodes the replication-associated protein (Rep), whose function is in rolling circle propagation, which is a highly conserved process 41 of nucleic acid replication to form circular molecules of DNA. 43 ORF C1 encodes the capsid structural protein (Cap). 6 Cap is involved in several events, including virus attachment and entry to the cell, and uncoating and assembly of new viral particles. 19 The analysis of the PBFDV from blood and feather samples of lorikeets from 3 locations in Australia established that the ORF encoding Cap has higher diversity than the ORF encoding Rep. 4
Replication of PBFDV occurs in the nucleus and is associated with the production of large intracytoplasmic and intranuclear inclusion bodies.29,43 Several studies mention that the frequent mutations in PBFDV 6 lead to evolutionary advantages, such as the ability to infect a wider variety of hosts. This evolutionary advantage, associated with the existence of different genotypes, is due to changes in the sequence of the cap gene. 28
PBFDV has been found in over 60 species of psittacines.16,18 The virus has been isolated from free-living birds in Oceania,8,38 Africa, and South East and South Central Asia. 8 It has been suggested that bird trafficking was responsible for the introduction of this virus from free-living birds elsewhere in the world into North American bird populations. 38
Differences in clinical manifestations are reported in different species. Traditionally, circoviruses have been considered to be host-specific.5,39 However, the presence of PBFDV in non-psittacine avian species has been demonstrated, 17 and the species specificity of this virus is under discussion. 12
A prevalence of 69.6% of PBFDV infection was found in 46 free-living sulphur-crested cockatoos, long-billed corellas, and little corellas in Australia. None of these birds had signs of disease, except for one corella with a fluffed appearance and closed eyes. 36 Other species of birds that have been found to be infected by this virus are included in Table 2, highlighting the importance of epidemiologic surveillance for this disease, due to its widespread distribution in many countries.8,9
Bird species affected by psittacine beak and feather disease virus and their world distribution.

In Guyana, 2 psittacines (Arini brotogeris and Arini pionus) were tested by PCR with negative results. 20 The virus was found in 53 of 269 (20%) captive psittacines in Costa Rica. 7 In this country, a pre-release study of 122 captive psittacines found a 9.8% prevalence of PBFDV. 18 Another study in Costa Rica did not find PBFDV in blood and feather samples from 9 free-living specimens of Amazona auropalliata. 2
Transmission of PBFDV occurs primarily orally, as the virus is shed in feces and can be recovered from crop washings and feather dust from infected birds. 32 PBFDV can also be transmitted by contact with contaminated surfaces or objects. 8 Vertical transmission has also been documented. 8 Some infected birds are subclinical, indicating the existence of a carrier state. 8 The incubation period ranges from 21 d to years, with some species, such as lovebirds, in which the disease often does not develop. 29
PBFDV has tropism for epithelia, especially the basal cells of feather follicles and the rhamphotheca, causing feather loss and malformations in the beak.13,29,39 The virus can also infect the intestinal epithelium, 43 thymus, and cloacal bursa, causing leukopenia and immunosuppression.13,34 PBFDV infects macrophages and induces severe lymphoid necrosis, which is probably mediated by cytokines and not directly by the virus. 39
PBFD is usually more severe in young birds, in which infection can lead to death; adults may survive with a few long-term effects such as discoloration of the contour feathers.13,29 Birds with subclinical disease can continue shedding virus for months, acting as reservoirs. 17 The main clinical sign of PBFD is anorexia, which leads to weight loss and weakness. In addition, there are several clinical manifestations that are the consequence of secondary bacterial or fungal infections, associated with the immunosuppression effects of PBFDV infection.16,29
PBFD lesions differ with age and bird species. In nestlings, PBFD induces a systemic and acute disease that affects all growing feathers and leads to death within 2 wk of disease onset. Chronic disease is generally seen in birds 8-mo- to 3-y-old and is characterized by dystrophic feathers, with thickened sheaths, blood in shafts, stress lines in vanes, and beak elongation with fractures and discoloration. 29
Acute lesions have been observed in nestling cockatoos, as opposed to chronic conditions seen in older cockatoos, lorikeets, eclectus parrots, and lovebirds. 29 Immunosuppression is mainly observed in African grey parrots; feather lesions are rarely observed in this species. The lesions most frequently observed in this species are atrophy and necrosis of the lymphoid tissue, with basophilic intracytoplasmic botryoid inclusion bodies. 34 In acute cases, affected birds may have necrotizing hepatitis with severe leukopenia and nonspecific clinical signs. 34 In adult African grey parrots that survive early infections, red zonal discoloration of the contour feathers may be observed. 29 Lesions observed in chronic cases include mainly beak deformities and progressive feather loss, although in some cases beak deformities may not be present. 16 In non-psittacine birds such as pigeons, the lesions observed usually predominate in both primary and secondary lymphoid organs, ranging from lymphofollicular hyperplasia to lymphoid necrosis, cellular depletion, and cystic atrophy. 39
Microscopic skin lesions include hydropic degeneration and necrosis of the epithelial cells of the feather follicles with heterophilic and lymphoplasmacytic inflammation of the feather pulp. Feather follicle keratin could be folded and dysplastic. 21 Similar lesions are seen in the beak. 21 Histologically, intracytoplasmic and intranuclear, 3–6-μm, basophilic inclusion bodies with a botryoid appearance are observed in both psittacine and non-psittacine birds.29,39 These inclusion bodies can be observed in the feather follicle epithelium and feather pulp, in dermal and epidermal macrophages, and in the cloacal bursa.16,29 Among the lymphoid organs, cloacal bursa, thymus, 21 bone marrow, and spleen are affected, leading to non-regenerative anemia, pancytopenia, and immunosuppression. 29
There are several differentials for PBFD, including avian polyomavirus infection, feather dysplasia, wing web dermatitis, nutritional deficiencies, endocrinopathy, and self-trauma. 29 A presumptive diagnosis of PBFD can be established based on clinical findings, and gross and microscopic lesions. Confirmation is based on detection of the virus by IHC,38,39 hemagglutination assay (HA), 25 electron microscopy, or molecular techniques such as conventional PCR, real-time PCR, 16 ISH, 27 and dot blot hybridization. 38 Hemagglutination inhibition assay (HIA) 16 and ELISA 42 are used to detect antibodies against PBFDV. To date, virus isolation has not been achieved because PBFDV does not grow in cell cultures. 39
ELISA is not very reliable, as the cross-reactivity of immunoglobulin Y (IgY) between different psittacine species is unknown. 35 HIA is a useful technique 23 because it avoids the use of species-specific secondary antibodies or highly purified antigens, 42 although it has a low sensitivity in infected birds with chronic disease. 23 IHC has been used with good results for the detection of this virus, and to study its distribution in tissues. 39
PCR has increased in popularity as a detection method, due to its high sensitivity and specificity, including its ability to detect animals with subclinical disease. 7 The Rep gene is relatively conserved, despite the high variability of PBFDV, and has been used as a target for primers P2-P4.12,44 Likewise, primers used to amplify segments of the orfC1 gene are also useful for detecting all variants of the virus in psittacines. 7 Suitable samples for PCR detection can be blood or feathers. 42 PCR results depend on the viral concentration, the tissue examined, and the stage of infection. 8
To date, there is no specific treatment for PBFD, and the only therapeutic option is supportive therapy. The use of b-(1,3/1,6)-D-glucans obtained from the oyster mushroom has been suggested because it showed efficiency in the clearance of the virus from blood in an experimental treatment carried out in psittacines. 40 However, little information is available on this treatment, which was tested in only a small number of birds. A treatment with pigeon interferon alpha was assessed in Columba livia naturally and experimentally infected with pigeon circovirus, and the virus was inhibited by this product, which is therefore considered as a potential treatment for these circovirus infections. 33
PBFDV viral particles excreted in the environment are highly resistant to adverse environmental conditions, such as high temperatures and desiccation, 22 and to disinfectants. 14 Captive animals can be kept free of disease by preventing contact with infected birds and keeping them in properly cleaned and disinfected cages. 39 Early detection of infected individuals using highly sensitive screening tests, such as PCR, provides information that should be used to isolate infected birds. The use of quarantine strategies for newly incorporated animals, as well as disinfection with peroxide-containing components, are useful tools in the prevention of PBFD. 42 Experimental vaccines have been tested, with promising results.3,24 Maternal antibodies induced by inactivated vaccines can play a protective role, 39 but as the virus cannot be cultured in vitro, the production of conventional vaccines is complex, highlighting the relevance of considering the production of subunit vaccines. 39
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387251333410 – Supplemental material for Psittacine beak and feather disease in 2 free-living great green macaws: a case report and literature review
Supplemental material, sj-pdf-1-vdi-10.1177_10406387251333410 for Psittacine beak and feather disease in 2 free-living great green macaws: a case report and literature review by Roberto W. I. Olivares, Laura G. Bass, Andrés Sáenz-Bräutigam, Janet Sandí-Carmiol, Ana M. Villada-Rosales, Gaby Dolz, Antony Solórzano-Morales, María J. Zúniga-Moya, Roxana Granados-Solano, Brittany McHale, Diego S. Zúñiga-Cortés and Francisco A. Uzal in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Escuela de Medicina y Cirugía Veterinaria San Francisco de Asís, Costa Rica, for their support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
