Abstract
Feline meningiomas typically arise on the surface of the brain but can occur in the third ventricle. Meningiomas in the fourth ventricle have not been reported in cats, to our knowledge. Here, we describe the clinical and pathologic findings of meningioma in the fourth ventricle in 2 cats. Both affected patients (1 castrated male, 1 spayed female) were 15-y-old. Case 2 had neurologic signs, including absent menace response, head tilt, and ataxia. Grossly, both tumors were well-demarcated and firm, expanded the fourth ventricle, and compressed the adjacent neuroparenchyma. Cytology of the ventricular mass in case 1 revealed a spindle-cell neoplasm. Based on histology, the neoplasms were classified as WHO grade 1 fibrous meningioma (case 1) and transitional meningioma (case 2). Immunohistochemistry revealed diffuse, robust cytoplasmic E-cadherin and vimentin immunolabeling in both cases and multifocal, weak cytoplasmic pan-cytokeratin AE1/AE3 immunolabeling in case 2. Our findings indicate that meningiomas should be included as a differential diagnosis for tumors of the fourth ventricle in cats.
Meningiomas are the most commonly diagnosed CNS tumors in cats, accounting for up to 60% of all intracranial and 85% of all primary feline brain neoplasms. 21 Nearly 75% of feline meningiomas grow along the external surface of the brain, primarily in the supratentorial region, 21 and originate from arachnoid cells covering the meningeal arachnoid villi. 14 Meningiomas may compress, but rarely invade, the surrounding neuroparenchyma. 21 Approximately 20% of feline meningiomas arise from the third ventricle and presumably originate from the tela choroidea. 21 Although meningiomas can rarely occur in the fourth ventricle in humans and dogs,18,20 our search of PubMed, Web of Science, Scopus, and Google revealed no meningiomas of the fourth ventricle in cats. Here, we describe the clinical and pathologic findings of meningiomas in the fourth ventricle in 2 cats.
We searched for cases of meningiomas in the fourth ventricle of cats in the autopsy archives of the Department of Veterinary Pathology (College of Veterinary Medicine, Universidade Federal do Rio Grande do Sul, Porto Alegre, RS, Brazil) and the Athens Veterinary Diagnostic Laboratory (College of Veterinary Medicine, University of Georgia, Athens, GA, USA) between January 2007 and May 2024. The keywords “feline”, “cat”, “meningioma”, and “4th ventricle” were used in the search. Clinical forms and autopsy reports were reviewed for case selection. Cases without formalin-fixed paraffin-embedded archived tissue blocks were excluded.
We selected and included 2 cases in our study (Table 1). The affected cats (1 castrated male, 1 spayed female) were 15-y-old. One patient (case 1) was found dead at home without any history of clinical signs; the cause of death was not determined. The second cat (case 2) had progressive neurologic signs characterized by an absent menace response, head tilt, and ataxia for 3 mo. Because of the poor prognosis associated with the progressive clinical signs, the cat was euthanized.
Signalment, neurologic signs, gross neuropathology, diagnosis, and immunohistochemical features of meningiomas in the fourth ventricle in 2 cats.

CM = castrated male; DSH = domestic short hair; E-cad = E-cadherin; IHC = immunohistochemistry; PCK = pan-cytokeratin; SF = spayed female; Vim = vimentin; + = positive immunolabeling; – = negative immunolabeling.
This cat died suddenly.
Gross changes were observed in both cats and consisted of well-demarcated, spherical-to-elongated, white-to-red, firm masses that expanded the fourth ventricle (Fig. 1) and compressed the cerebellum, midbrain, pons, and medulla oblongata. The main gross differential diagnosis was ependymoma. 11 Imprint smear samples from the ventricular mass in case 1 were collected during autopsy and stained with aqueous Romanowsky stain for cytology. The smears were highly cellular, with individual or aggregated spindle cells (Fig. 2) along the margins of branching capillaries on a background of blood. The cells had wispy borders and a moderate amount of light-blue cytoplasm that frequently contained clear vacuoles and had fine long processes. Nuclei were single and round-to-oval, with finely stippled chromatin and one nucleolus. There was marked anisocytosis and anisokaryosis, frequent karyomegaly, and occasional binucleate cells. Mitoses were not identified. Cytologic interpretation was consistent with a spindle-cell neoplasm, possibly primary to the brain.
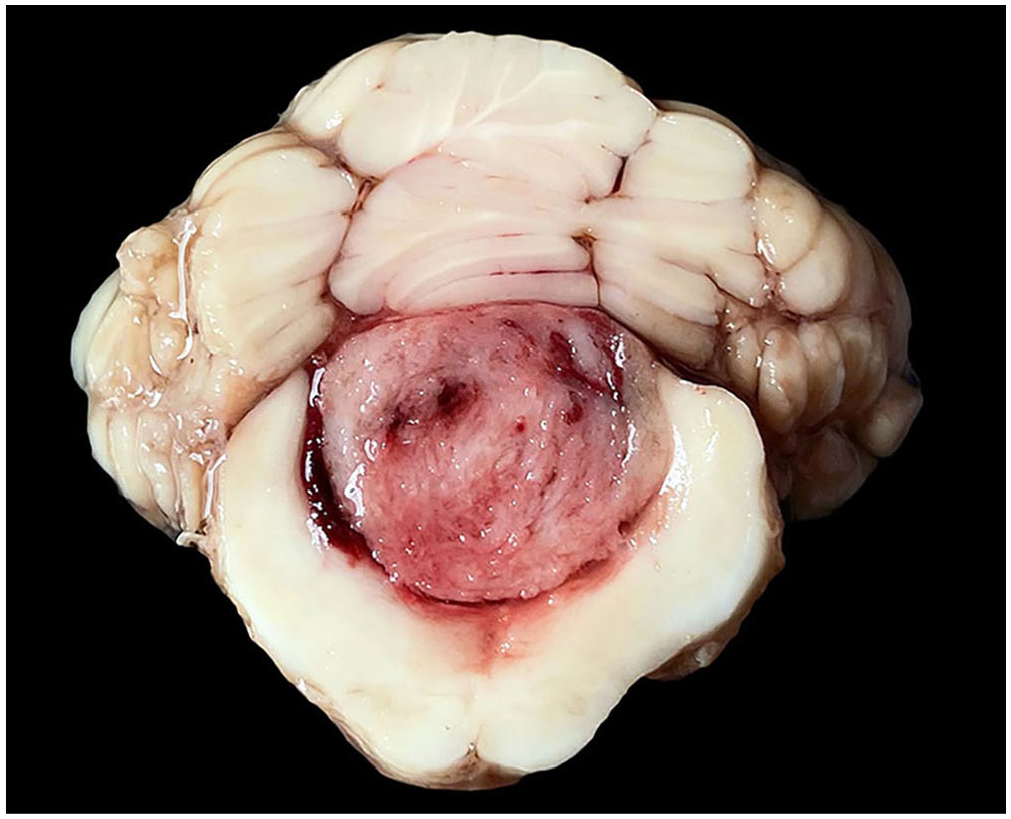
Meningioma in the fourth ventricle, case 1. A white-to-red mass expands and occludes the fourth ventricle. There is compression of the cerebellum and medulla oblongata.
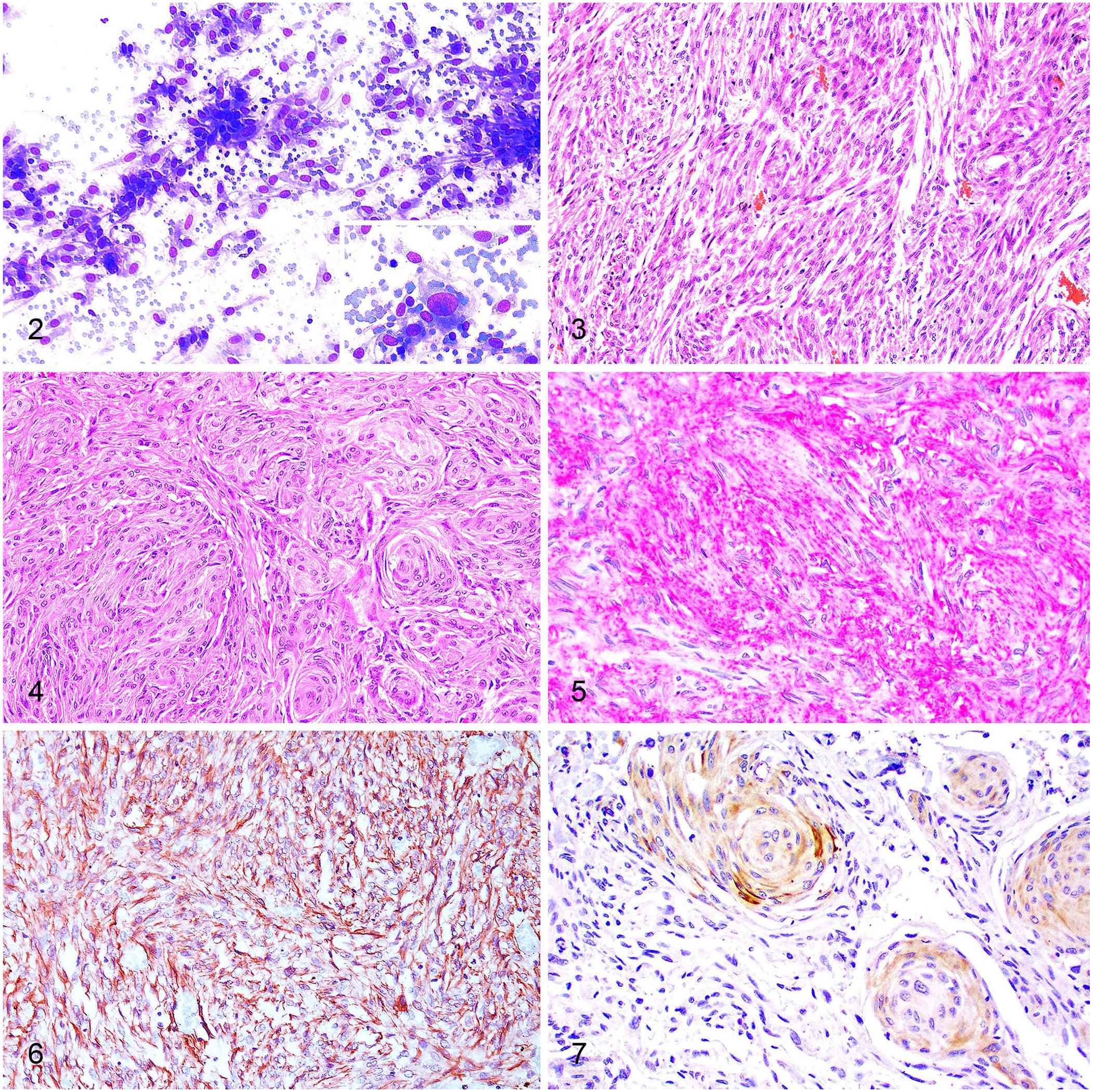
Meningiomas in the fourth ventricle in cats.
Histologic evaluation of archived H&E-stained tissue sections was consistent with a WHO grade 1 fibrous meningioma (case 1) and a WHO grade 1 transitional meningioma (case 2). Briefly, the fibrous meningioma (case 1) was composed of interweaving bundles and fascicles of elongate meningothelial cells supported by a fine fibrovascular stroma (Fig. 3). Neoplastic cells had a small amount of eosinophilic cytoplasm and oval nuclei with finely stippled chromatin and one nucleolus. There was moderate pleomorphism and 2 mitoses in 2.37 mm2 (10 FN22/40× fields). The transitional meningioma (case 2) consisted of closely apposed lobules of meningothelial cells and fascicles of elongate cells supported by a fine fibrovascular stroma (Fig. 4). Neoplastic meningothelial cells often formed scattered concentric whorls. Cells had moderate amounts of eosinophilic polygonal cytoplasm and round-to-oval nuclei with finely stippled to vesicular chromatin and one nucleolus. There was minimal pleomorphism and no mitoses were observed in 2.37 mm2.
In both cases, the meningothelial neoplastic proliferation was non-infiltrative but compressed the neuroparenchyma near the fourth ventricle, including the midbrain, cerebellum, pons, and medulla oblongata. Focally extensive hemorrhage was observed within the tumor and in the mesencephalic aqueduct of case 1.
Immunohistochemistry (IHC) for E-cadherin, vimentin, and pan-cytokeratin AE1/AE3 was performed (Suppl. Table 1). Both cases had diffuse, robust, membranous, and cytoplasmic immunolabeling for E-cadherin (Fig. 5) and cytoplasmic immunolabeling for vimentin (Fig. 6). Multifocal, weak cytoplasmic immunolabeling for pan-cytokeratin was observed in case 2, particularly in areas with concentric whorls (Fig. 7). No pan-cytokeratin immunolabeling was observed in case 1. These IHC findings support the diagnosis of meningioma. Furthermore, one cat (case 1) was also diagnosed with concurrent chronic kidney disease (CKD). The second cat (case 2) had thyroid hyperplasia, cardiac hypertrophy, CKD, and parathyroid hyperplasia.
Intraventricular meningiomas (IVMs) presumably originate from the tela choroidea, a fibrovascular tissue structure that gives rise to the choroid plexus. 9 In humans, IVMs are uncommon and located in the lateral ventricles (88%), fourth ventricle (8%), and the third ventricle (3%). 13 This distribution is thought to reflect the extent of the choroid plexus within the ventricles. 9 In domestic animals, the choroid plexus tissue is scant and thin in the lateral and third ventricles, and more robust and lobulated in the fourth ventricle. 4 Most feline IVMs arise from the third ventricle, 21 and cases of IVMs in dogs have been described in the lateral and fourth ventricles.12,20 Because IVMs are rarely reported in veterinary medicine, the frequency of tumors within specific ventricles remains undetermined.
Cats with meningiomas have insidious and progressive neurologic deficits, typically reflecting the location of the tumor. 6 In our study, case 1 had no clinical signs and case 2 had clinical signs consistent with lesions in the brainstem areas compressed by the tumor. Because these tumors are slow-growing and intraventricular, clinically silent cases may be expected until tumors reach considerable sizes. 2 Furthermore, determining the precise location of the intracranial tumors based on neurologic examination can be challenging due to secondary effects of the mass. 1 Most human patients with meningiomas in the fourth ventricle develop clinical signs related to internal hydrocephalus and intracranial hypertension resulting from obstruction of the flow of CSF. 9 Neither of our cats had evidence of hydrocephalus.
Intraoperative cytology of feline CNS tumors can provide an accurate and rapid diagnosis. 22 Cytologic findings of meningiomas are heterogeneous and normally reflect their morphology and cellular arrangement, such as epithelioid cells forming clusters (meningothelial subtype) or elongate cells forming fascicles (fibrous subtype). 17 The transitional subtype can have both meningothelial and fibrous patterns. 17 Despite variations, certain typical features such as whorls, psammoma bodies, and intranuclear cytoplasmic invaginations (pseudoinclusions) can facilitate cytologic recognition during routine evaluation.17,22 Although these distinctive features of meningiomas were not observed in the autopsy smears from the mass in the fourth ventricle of case 1, fibrous meningioma was listed as a differential diagnosis because of the presence of neoplastic elongate cells.
Meningiomas can have a wide range of histologic variations, which reflect the mesodermal and neural crest origin of arachnoid cells. 14 In humans, IVMs are predominantly fibrous tumors (40%). 13 Data from large case series of feline meningioma indicate that most cases are WHO grade 1 transitional meningiomas (44%), followed by fibrous (17%), meningothelial (15%), and psammomatous (16%) tumors.3,5,10,15,19 Rare subtypes include WHO grade 1 angiomatous, WHO grade 3 papillary, and WHO grade 3 anaplastic meningiomas.5,7,16 In cats, given that only our 2 cases have been reported, it remains unknown whether IVMs tend to be of a particular WHO grade or tumor subtype.8,23 Several markers are useful to support the diagnosis of IVM, including vimentin, pan-cytokeratin, E-cadherin, CD34, claudin-1, glial fibrillary acidic protein, glucose transporter 1, laminin, N-cadherin, and S100.16,19 However, immunolabeling may vary greatly among tumors, and no validated IHC panel has been standardized to confirm the diagnosis. In our cases, positivity for vimentin and E-cadherin supported the diagnosis of meningioma.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241312895 – Supplemental material for Meningioma in the fourth ventricle in 2 cats
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241312895 for Meningioma in the fourth ventricle in 2 cats by Igor R. Santos, Milena C. Paz, Marcele B. Bandinelli, Rochana R. Fett, Daniel R. Rissi, Renato S. Sousa, Shannon Swist and Saulo P. Pavarini in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Financial support was provided by the National Council of Scientific and Technological Development (CNPq) and the Brazilian Federal Agency for the Support and Evaluation of Graduate Education (CAPES)–Finance code 001.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
