Abstract
Evaluating stress in shelter and institutionally owned cats is important to help guide improvements in their welfare. Welfare assessments often focus on behavior metrics and physiologic measurements, such as systemic cortisol levels. The gold standard for measuring acute stress is serum cortisol; measuring cortisol in feces and urine gives reliable time-integrated assessments of acute stress. Monitoring chronic stress requires using a matrix that accumulates cortisol over time, such as hair or nails. Hair was collected from 29 cats representing 2 populations: cats from a local shelter and cats owned by a university. Cortisol was extracted from the hair using a method established for extracting cortisol from bovine hair. We measured hair cortisol concentrations with a commercial ELISA that is marketed for human saliva. The mean cortisol concentration was 140 pg/mg for the shelter cats and 98 pg/mg for the university-owned cats. We found no significant difference in hair cortisol concentrations between the 2 groups (p = 0.793). The intra- and inter-assay CVs for the ELISA were 9.3% and 8.4%, respectively. Observed:expected ratios for spiking recovery and dilutional parallelism were 87.7 ± 25.8% and 99.7 ± 37.5%, respectively. Measurement of cortisol in hair samples may provide a noninvasive method to monitor chronic stress and acclimation in cats that live in confinement for prolonged periods.
Cortisol is a primary stress hormone and the gold standard for assessing stress in most mammals. 10 High levels of cortisol are indicative of stress or a high state of arousal.1,6,10 Cortisol is released from the adrenal glands into the bloodstream where it can be measured in serum samples. 10 There is clear evidence of species-related differences in both glucocorticoid excretion and metabolism. 25 Serum cortisol reflects short-term or acute stress levels. Cortisol concentrates in the saliva, urine, feces, and hair, where time-integrated levels can be measured.1,6,10,20 Saliva cortisol can be minimally invasive to collect and, similar to serum cortisol, reflects the circulating cortisol concentration at the time of collection.10,12 Urine cortisol indicates the cortisol concentration over a few hours; feces cortisol is an indicator of blood cortisol concentrations over 24–36 h.1,11,23 Cortisol concentration in the hair and nails reflects long-term or chronic circulating concentrations of cortisol that have accumulated over weeks to months.1,10,18–20
Evaluating stress in shelter and institutionally owned cats is important to help guide improvements in their welfare and environment. Chronic stress in cats may not be readily apparent on typical behavioral assessments; however, behavioral problems are often secondary to chronic stress and are a leading cause of cats being surrendered to a shelter or developing stereotypy in long-term confinement.2,17,21 Procedures to recognize, measure, and monitor chronic stress are not readily available or validated for use in cats. 9
There are several published methods for extracting cortisol from different substrates1,18,20 in a variety of species. The cortisol molecule is stable and can be measured regardless of the species and type of sample collected. 10 The level of cortisol in the specific substrate will vary, impacting the ability of an assay to measure cortisol concentrations. 10 Cortisol can be measured through a variety of immunologic assays, including ELISA, radioimmunoassay (RIA), and chemiluminescent immunoassay.1,6,10,16,20 An ELISA is a solid-phase labeled immunoassay that is highly sensitive in detecting and quantifying substances. RIA is similar to ELISA in that it is a very sensitive immunoassay; however, RIA uses radioactive isotopes to detect and quantify substances, which may limit its applicability outside a laboratory setting. ELISA and RIA have been used to measure hair cortisol concentrations in dogs and cats.1,6,9,10,15,24,25 Although there are many methods for measuring cortisol, few are validated for use in cats. Assay results cannot be extrapolated between species, substrates, or extraction techniques.7,22,24
Measuring cortisol concentrations in hair to monitor chronic stress in cats is a relatively new technique that has promising applications.1,9,18 Similar to feces, the amount of cortisol extracted from the hair is partially dependent on the extraction technique. Unlike other matrices, cortisol is not continuously incorporated into the hair shaft as diffusion occurs only during the growth phase. 18 Hair has a seasonal growth pattern, and the amount of growth and cortisol uptake will differ in the various types of hair present on a cat’s body. 9 Cats with a dense hair coat will have a different cortisol profile than cats with a fine, sparse coat.
Some species have notable differences in hair cortisol concentrations based on coat color or body location.5,8,9,20 In cows, black hair is associated with higher cortisol concentrations than white hair,21,22 and areas where hair is thicker (such as the tail switch) have higher hair cortisol concentrations than areas where hair is sparse. 22 In contrast, white dog hair has higher levels of cortisol than black dog hair. 6 Intact female cats have higher hair cortisol concentrations than spayed female cats. 13 One study in cats noted a difference in hair cortisol concentration based on location but not coat color 9 ; however, that finding has not been replicated. Given the known variability, reference values may need to be developed for different hair types to allow for more accurate interpretation of cortisol concentrations.
Feline hair cortisol concentrations have been reported in 6 studies; 4 used RIA and 2 used an ELISA marketed for saliva cortisol. The hair cortisol concentrations reported in the literature were 0.02–76.3 pg/mg.1,9,13,15 Hair cortisol concentration can vary based on numerous factors, including, but not limited to: the type of hair, the sample collection method, the extraction technique, and the assay used to measure cortisol. The measured cortisol concentration should be carefully interpreted using RIs from the same assays and extraction protocols because results cannot be extrapolated accurately across all assays and/or protocols.
Our aims were to extract cortisol from feline hair and assess the utility of an ELISA for measuring cortisol in this substrate. We selected hair as the substrate for our study because it can be obtained noninvasively and can potentially be used in a shelter or other group-housing situation to monitor longer-term stress in cats.
Materials and methods
Animals
Hair samples were collected from 2 groups of cats representing 2 populations. The first group consisted of 8 healthy, spayed female, domestic shorthair (DSH) cats, 4–7-y-old, which were institutionally owned by Texas A&M University for ≥4 y. The coat color diversity in this group ranged from solid black to orange and white, and included patterns such as tabby, tortoiseshell, and calico. Cats were maintained on a 12-h light/dark cycle with exposure to natural diurnal light cycles in 244 cm (8 ft) × 305 cm (10 ft) enclosures. Cats were either pair-housed or single-housed and were provided with species-appropriate enrichment. The second group consisted of 21 healthy, 1–9-y-old cats that were available for adoption at a local animal shelter. Cats in the second group were of both sexes, intact and neutered, and included DSH, domestic medium-hair (DMH), and domestic longhair (DLH). The coat color diversity was greater in the second group than with cats in the first group in that all of the solid colors and at least one representative of each coat pattern were available to be sampled. Cats in the second group were surrendered from their owners, seized by animal control, or brought in as strays and had resided at the shelter for ≥72 h before sample collection. Shelter cage sizes and light cycles varied by housing status and room location.
The institutionally owned cats were on a research study approved by the Texas A&M University Institutional Animal Care and Use Committee (TAMU-IACUC). Our validation study was conducted within a larger welfare study that was approved by the TAMU-IACUC; however, the hair samples were discards collected during routine husbandry and grooming procedures, which did not require IACUC approval.
Sample collection
Hair was collected from the institutionally owned cats during routine grooming by care staff. Hair was collected from the shelter cats during socialization by shelter staff and volunteers. Cats in both groups were brushed (Furminator; Spectrum) over their dorsum, from the neck to the lumbosacral region for 10–15 min. Hair was collected once per cat during the summer when cats were more likely to be shedding. The volume of hair collected depended on the coat thickness of the individual cat; however, the goal was to collect enough hair to perform one cortisol extraction per cat. Upon collection, hair samples were placed in a snack-size plastic bag (Ziploc; S.C. Johnson). All hair samples were stored in an opaque plastic container in a dark, climate-controlled location until cortisol extraction. Cortisol was extracted within 4 mo of hair collection.
Hair cortisol extraction
The modifications of the cortisol extraction method included use of a ball mill instead of mincing the hair.18,20 Hair samples were washed with 4 mL of lukewarm tap water to remove dust and debris and allowed to dry overnight. Samples were then washed twice with 5 mL of isopropanol (Ricca Chemical), blotted dry, and allowed to air dry for 5 d at room temperature. Hair (~250 mg) was placed in a ball mill (MM 200; Retsch) and was finely ground for 240–270 s at 30 Hz; 20 mg of the ground hair was placed in a 5-mL glass scintillation vial (MilliporeSigma), and 1 mL of methanol (MilliporeSigma) was added. Samples were sonicated (Fisher) for 30 min and then incubated at 50°C on an orbital shaker (Fisher) at 100 rpm for 18 h. Samples were then centrifuged for 5 min at 7,000 × g. The supernatant was aspirated and placed in a 2-mL microcentrifuge tube (Eppendorf), which was placed under an air stream for up to 1 h until the methanol had evaporated. Samples were then reconstituted with 100 μL of assay diluent (I-3002; Salimetrics) for the ELISA. The hair cortisol extraction process time was 7 d.
Cortisol concentration measurement
Cortisol concentrations in samples were measured with an ELISA (Salivary cortisol assay kit, I-3002; Salimetrics). We selected this ELISA kit because it has been used to measure hair cortisol in cows and cats.9,20 We ran 25 μL of each sample, in duplicate, on an ELISA plate according to the manufacturer’s instructions. Cortisol concentrations were expressed in μg/dL, and then transformed into pg/mg of hair. Standards (0.012–3.0 μg/dL), assay diluent, and reagents provided with the kit were used for each assay. Internal high (0.9 μg/dL) and low (0.1 μg/dL) QC samples were run in duplicate with each assay. According to the manufacturer, the acceptable ranges for the controls were 0.724–1.207 μg/dL (high) and 0.069–0.127 μg/dL (low). The minimum and maximum concentrations of cortisol that can be detected by the assay, according to the manufacturer, are 0.007 μg/dL and 3.0 μg/dL, respectively. The ELISA was completed in ~3 h, depending on the number of samples run.
Validation of the ELISA
The hair cortisol extraction for each cat was relatively low, 100 μL total, so the volume available for validation was limited. Hence, we pooled samples containing similar amounts of cortisol, as determined by the cortisol concentration measurement. To determine the intra-assay variability, 11 hair cortisol extract samples from cats from both groups, with cortisol concentrations of 20–160 pg/mg, were evaluated. All 11 samples were combined into a single pool, which was then run 10 times on the same plate. The x̄ and SD were calculated, and the %CV was determined using the formula: %CV = (SD/x̄) × 100%.
To evaluate inter-assay variability, 13 hair cortisol extracts coming from both groups were divided into 3 pooled samples. Extracts with individual values of ~40 pg/mg were pooled as sample 1, extracts with individual values of ~70 pg/mg were pooled as sample 2, and extracts with individual values of ~90 pg/mg were pooled as sample 3. The 3 samples were run, in duplicate, on 5 different plates from the same lot number over 5 different days. The x̄, SD, and CV were calculated as described above. The target acceptable CV for the intra- and inter-assay variability was set at ≤15%. 3
To determine recovery, hair cortisol extract samples from 4 cats were divided into 2 aliquots. The samples were then spiked with either a high (1,088 pg/mg) or low (100 pg/mg) amount of cortisol. The “high” cortisol was 2 times greater than the highest hair cortisol concentration from the sample group. The “low” cortisol was the median hair cortisol concentration of the shelter cat group. Samples were run in duplicate, and the mean of the 2 values was reported. The observed:expected (O:E) ratios were calculated. Recovery was assessed by plotting the mean of the concentration in the dilution against the expected concentration. The target acceptance criteria for the O:E ratio for dilutional parallelism and spike recovery was set at 80–120%. 3
Linearity was assessed through dilution of 3 hair cortisol extract samples from each of 2 cats with similar cortisol concentrations. Samples 1–3 were created and used for this analysis. Each sample was serially diluted with assay diluent at 1 in 2, 1 in 4, and 1 in 8, and the O:E ratio was calculated. Linearity was assessed by plotting the mean of the concentration in the dilution against the expected concentration.
Statistical analysis
The hair cortisol concentrations for the 21 shelter cats and 8 colony cats were evaluated for normality; concentrations from the shelter cats were not normally distributed. Hair cortisol concentrations for the shelter cats and colony cats were compared using the Mann–Whitney U test. All calculations were performed using Prism v.5 (GraphPad; Dotmatics). Statistical significance was set at p ≤ 0.05.
Results
Validation of the ELISA
The intra-assay CV for the 3 pooled samples was 9.3%, with a mean cortisol concentration of 55 pg/mg (range: 45–65 pg/mg) and a SD between wells of 5 pg/mg. The inter-assay CVs for the 3 pooled samples were 6.4–9.5%, with cortisol concentrations of 41–88 pg/mg (Table 1). The O:E ratios for recovery were 72.6–98.4% (x̄ = 87.7%, SD = 8.8%; Table 2). The O:E ratios for dilutional parallelism was 81.5–119% (x̄ = 99.7%, SD = 13.3%; Table 3). Most samples (7 of 8) for the recovery assessments were acceptable and within the guidelines recommended for bioanalytical validation.3,4 One sample that was spiked with the lower amount of cortisol fell below the recommended guidelines at 72.6%.
Inter-assay variability of 3 feline hair samples run in duplicate on 5 different plates at 5 different times.

Recovery of cortisol from 8 feline hair samples spiked with a high or low concentration of cortisol and then tested with the ELISA.
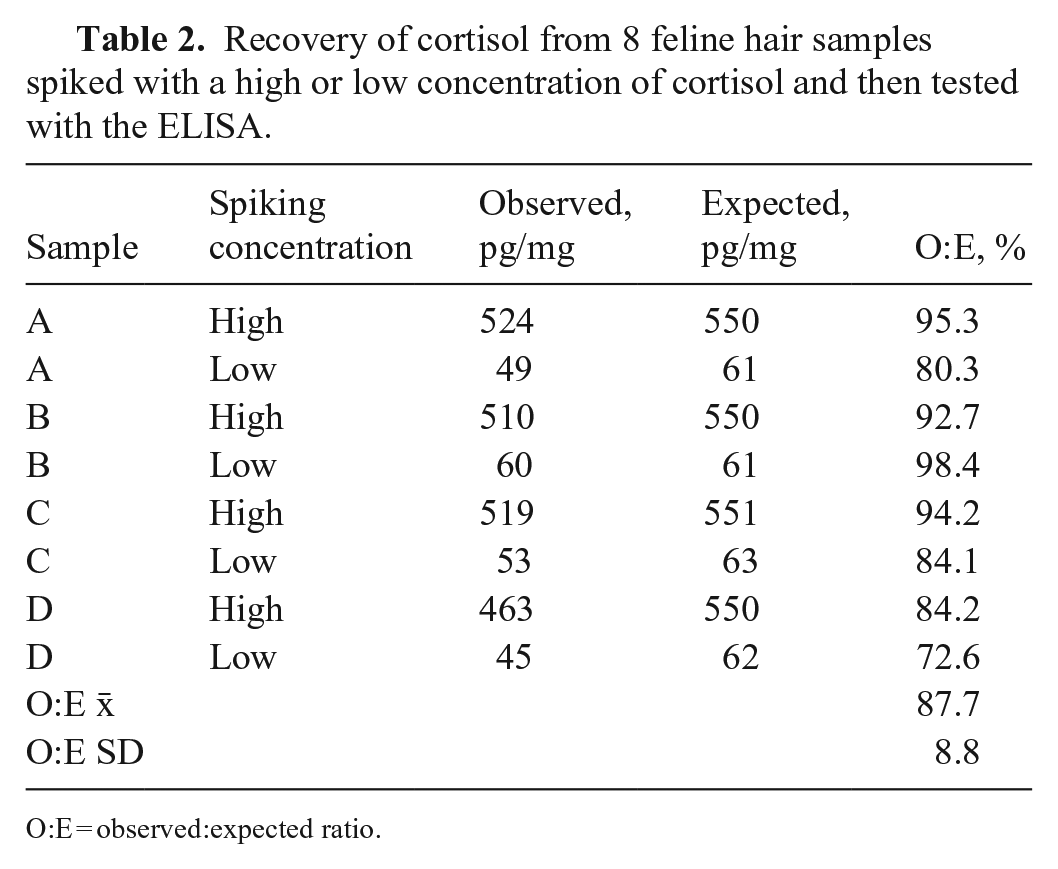
O:E = observed:expected ratio.
Dilutional parallelism of cortisol in 3 feline hair samples that were serially diluted 1 in 2, 1 in 4, and 1 in 8 in assay diluent.
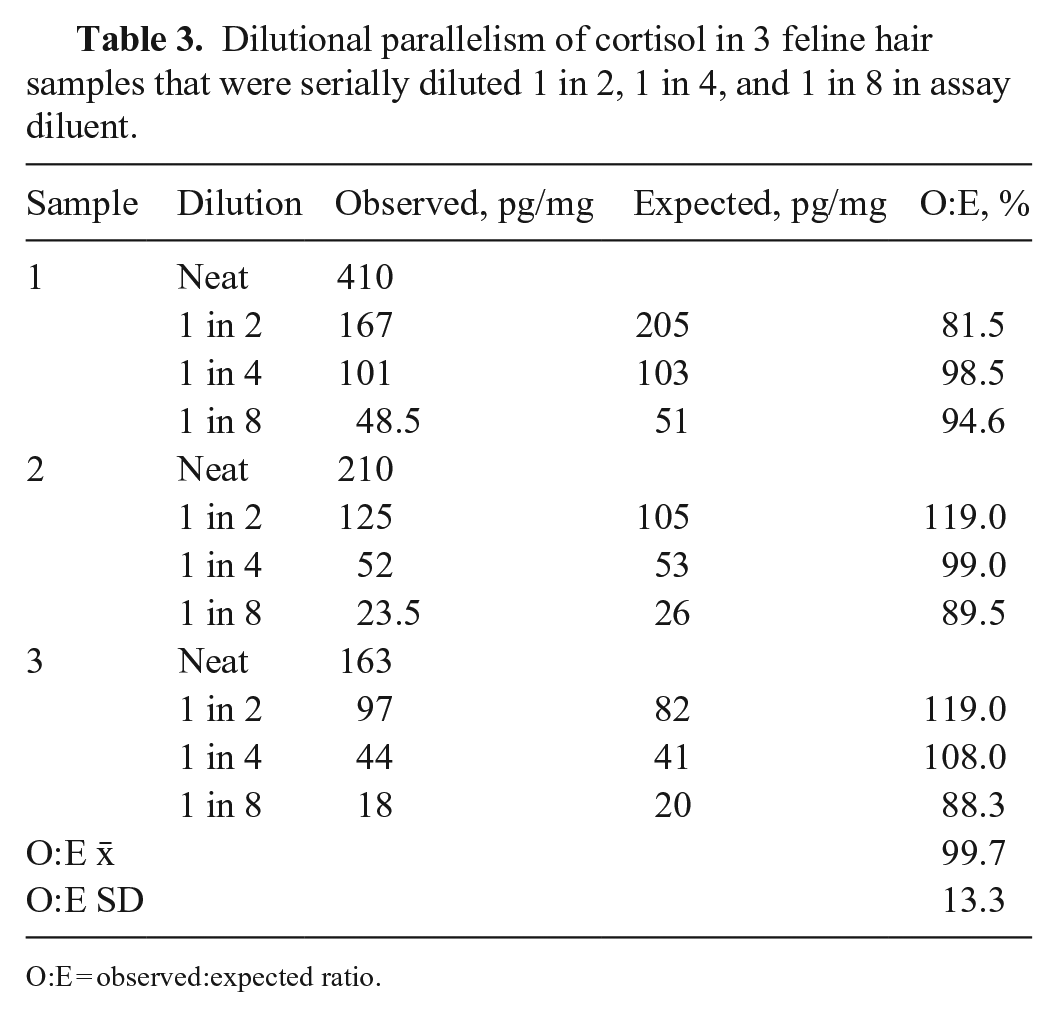
O:E = observed:expected ratio.
Comparison of hair cortisol concentrations between shelter and colony cats
Hair cortisol concentrations for shelter cats were 7–550 pg/mg (x̄ = 140 pg/mg; median = 100 pg/mg; SD = 149 pg/mg). Hair cortisol concentrations for colony cats were 12–213 pg/mg (x̄ = 98 pg/mg; median = 76 pg/mg; SD = 79 pg/mg). There was no significant difference in hair cortisol concentrations between the 2 sample groups (p = 0.793; Fig. 1).
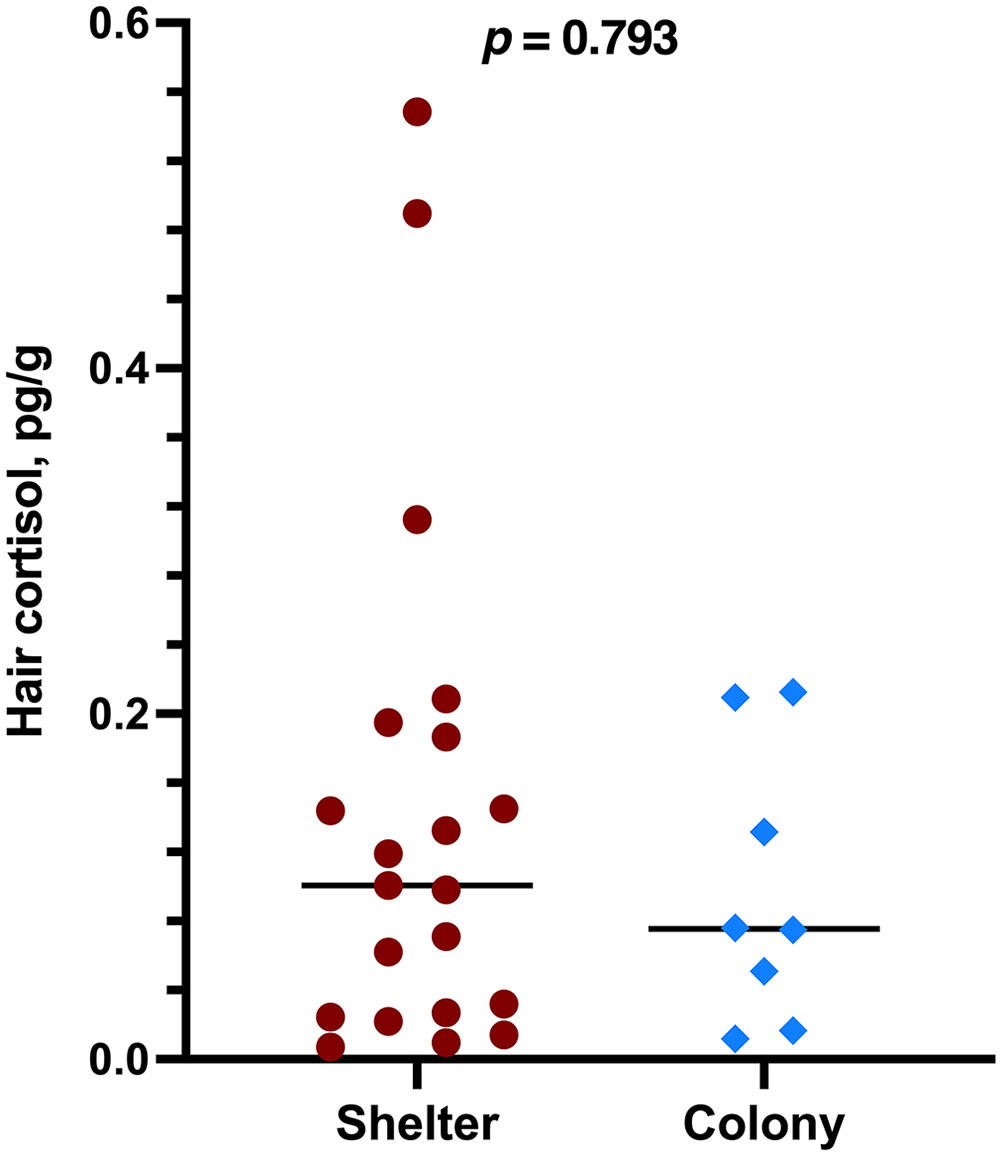
Cortisol concentrations in hair samples from 21 shelter cats and 8 colony cats were not significantly different (Mann–Whitney U test; p = 0.793).
Discussion
We were able to successfully extract cortisol from feline hair and assessed some analytical parameters of the ELISA using hair cortisol extract to establish the suitability of this assay for measuring cortisol in feline hair samples. To ensure that the cortisol assay was appropriate for use on feline hair samples, we assessed the ELISA for precision, linearity, and recovery. The intra-assay and inter-assay precision were acceptable with CVs <15% for all samples, which is within the guidelines recommended for bioanalytical validation.3,4 The linearity assessments were acceptable and also within the guidelines recommended for bioanalytical validation of an assay.3,4
Hair in our study was collected via brushing, not shaving, because brushing would be acceptable in a shelter situation where minimal physical alterations are important. Most studies that examine hair cortisol will trim or shave the hair, collecting a defined range of hair length, therefore reflecting cortisol concentrations over a known range of time.1,20 By using brushing to collect hair, the entire hair shaft, and therefore the period of time over which cortisol was assessed may have been more variable than previous studies.9,25 The uptake of cortisol in the hair shaft occurs only during the growth phase, and the amount of cortisol will depend on the cat’s stress level during that time. If the cat is in a low-stress period, they will have a minimal amount of cortisol in the hair shaft. By incorporating the entire shaft in the analysis, the periods of high and low stress are averaged. We included hair from the entire body; therefore, differences in location could not be evaluated. As with the previous study 9 that also used this ELISA to evaluate feline hair cortisol concentrations, we did not evaluate for differences in cortisol concentrations between coat colors or patterns as the sample size for each category was too small.
There was no statistical difference between the hair cortisol concentrations of the shelter cats and the institutionally owned colony cats. Samples collected within the cats’ first few weeks of arrival at the shelter would reflect stress during the time prior to shelter confinement. Some shelter cats had relatively high cortisol concentrations, reflecting a history of chronic stress; however, we had expected that they would have different levels of cortisol given their diverse history. The colony cats had resided in a stable environment at a university for ≥4 y and their history was well documented; their acute and chronic stress concentrations were presumed to be low.
Our study has limitations. A formal repeatability study with the extraction technique to evaluate the precision of that process was not completed. Therefore, we were only able to measure the imprecision associated with the assay using cortisol extract. Additional hair collections would be needed to validate the hair sampling and the extraction method. Quantifying the imprecision of the extraction process will be an important step before further validation or clinical investigation of the assay. This would help quantify the global imprecision of the technique, combining cortisol extraction imprecision and cortisol measurement imprecision. We also did not complete a full analytic validation of the ELISA. Our hair cortisol extract samples were of limited quantity, and we pooled samples with similar cortisol concentrations to assess validation parameters. With the small sample size, the number of repeats to determine the inter-assay variability was low, which may underestimate the imprecision of the assay. Additionally, the dilution study was performed in duplicate, which may have underestimated the imprecision of the method. Additional hair cortisol extractions from similar populations would allow for sufficient sample volume to perform a complete validation of the assay, including more replications of each sample.
We acknowledge that the groups were not of equal size as there was a limited number of institutionally owned cats. We did not use additional metrics to measure stress, such as behavioral measurement, although that would be a good future study to compare acute and chronic behaviors to the physiologic response. We collected hair via brushing as most cats were available for adoption and hair collection needed to be as inconspicuous as possible. Future studies to evaluate if body location influences cortisol concentration will require shaving a small patch of hair from a few locations to make appropriate comparisons.
There are few references for concentrations of feline hair cortisol in the literature,9,13,14,25 and hair was collected via shaving or trimming. Many animals are kept in long-term captive environments where shaving hair is not an option but recovering hair from an enclosure or a brush is possible. Having an accurate and reliable assay to noninvasively monitor chronic stress via hair will be beneficial to caregivers tasked with improving the welfare of cats.
Footnotes
Acknowledgements
We thank Robynne Gomez, Sarah Read, and the Gastrointestinal Laboratory staff at Texas A&M University for their assistance. We also thank the Aggieland Humane Society for allowing us to conduct our study with their animals and all of their assistance with this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
