Abstract
In male dogs, Brucella canis frequently causes epididymitis, ultimately resulting in testicular atrophy and infertility. Although B. canis predominantly affects the epididymis, the misleading term “orchitis” is still commonly used by clinicians. Of additional concern, diagnosis in dogs remains challenging because of variable sensitivity and specificity of serologic assays and fluctuations in bacteremia levels in infected dogs, reducing the sensitivity of blood culture. We describe here the histologic lesions in the scrotal contents of 8 dogs suspected of being infected with B. canis and clinically diagnosed with orchitis. We explored the possibility of using immunohistochemistry (IHC) and real-time PCR (rtPCR) in formalin-fixed, paraffin-embedded (FFPE) tissues to detect the presence of B. canis. Epididymitis of variable chronicity was identified in all 8 dogs, with only 3 also exhibiting orchitis. Using rtPCR, the presence of B. canis was identified in 4 of 8 dogs, with 3 of these 4 dogs also positive by IHC. These results suggest that rtPCR and IHC are promising techniques that can be used in FFPE tissues to detect B. canis when other detection techniques are unavailable. Additionally, accurate recognition of epididymitis rather than orchitis in suspect cases could aid in accurate diagnosis.
Bacteria of the genus Brucella are facultative intracellular bacteria and the cause of brucellosis in wild and domestic mammalian species. Several Brucella spp. are commonly reported zoonotic pathogens. 17 Canine brucellosis, caused by B. canis, remains endemic in several regions, including Central and South America and the southern United States.2,11 Although B. canis most commonly infects dogs, it may also infect humans, and thus poses a public health risk. 7 Detection of B. canis is not a straightforward procedure. The most widely available techniques for the detection of B. canis infection are serologic assays, which include the rapid slide agglutination test (RSAT), 2-mercaptoethanol RSAT (2ME-RSAT), agar gel immunodiffusion (AGID), ELISA, and immunochromatography. Although useful for screening purposes, serologic assays lack the sensitivity and specificity required to achieve a definitive diagnosis.11,14
The gold standard for detection of infected dogs is culture, with blood or semen comprising most samples submitted to laboratories.14,18 Unfortunately, blood or semen culture is frequently negative as a result of the undulant pattern of bacteremia, particularly in chronic cases, necessitating serial cultures.4,14 Culture of Brucella spp. presents additional challenges, including the need for laboratory facilities with adequate biosecurity measures (Biosafety Level 3 laboratory) and the risk of transmission to laboratory staff and clinicians. Although not as widely available as serologic assays, real-time PCR (rtPCR) is emerging as a promising detection technique for B. canis infection in dogs. However, PCR holds the same challenges as culture if blood is used for analysis. 1 The use of formalin-fixed tissues offers an opportunity for the detection of B. canis via either PCR or immunohistochemistry (IHC) in the absence of samples available for culture. To our knowledge, detection of canine brucellosis in FFPE canine male reproductive tissues has not been reported previously.
Like other Brucella species, B. canis has a tropism for the reproductive system and is a well-established cause of canine reproductive failure.11,13 B. canis invades via conjunctival, oronasal, or venereal routes. Thereafter, it is distributed to various organs, particularly the spleen, lymph nodes, and liver, to result in chronic, persistent infection.4,16 In male dogs, it can cause epididymitis, prostatitis, and orchitis, with chronic testicular and epididymal inflammation ultimately resulting in testicular atrophy and infertility. 16 The typical manifestation of brucellosis in bitches is mid- to late-term abortion followed by brown-to-yellow vaginal discharge that can persist for several weeks.4,5 In both sexes, the infection can also result in diskospondylitis.15,18 Unfortunately, the clinical signs of B. canis infection are not pathognomonic, and a subset of infected dogs may show no obvious clinical signs despite harboring and shedding the bacteria. 4 Further compounding clinical detection is the frequent description of orchitis in infected dogs without mention of epididymitis. In addition to the deficiencies in traditional detection methods and the difficulties in diagnosing brucellosis based on clinical signs, the absence of approved surveillance and prevention strategies in most countries and the lack of a protective vaccine result in significant hurdles to the control and eradication of canine brucellosis.
We aimed to accurately detect B. canis infection in male dogs by clarifying the histologic lesions of dogs clinically diagnosed with orchitis and assessing the potential of rtPCR and IHC on FFPE testicular tissue as useful tools in clinically suspected cases of canine brucellosis for which fresh tissue or serum is unavailable for testing.
Eight dogs that were presented to the University of Environmental and Applied Sciences (UDCA) veterinary school in Bogotá, Colombia were selected based on the clinical diagnosis of orchitis, with brucellosis listed as a differential diagnosis (Table 1). Enlargement of the scrotal contents was the most common clinical sign and was identified in 6 of 7 dogs. Additional clinical signs from most-to-least frequent included pyrexia (cases 1–4, 8), scrotal erythema and pain during palpation (cases 3, 4, 6–8), lameness or lumbar pain (cases 1, 5), anorexia (cases 2, 5), and enlarged popliteal lymph nodes (cases 5, 7). Only one dog (case 5) was tested for B. canis infection serologically and was identified as positive (1:200 titer) using ELISA. All dogs underwent castration. Given that samples were obtained in the course of routine clinical patient care and analyzed retrospectively for our study, specific approval by an Institutional Animal Care and Use Committee was not required as per institutional policy.
Signalment, testicular and epididymal histologic lesions, anti-Brucella immunohistochemistry (IHC), and real-time PCR (rtPCR) results, including cycle quantification (Cq) value, of dogs clinically diagnosed with orchitis.
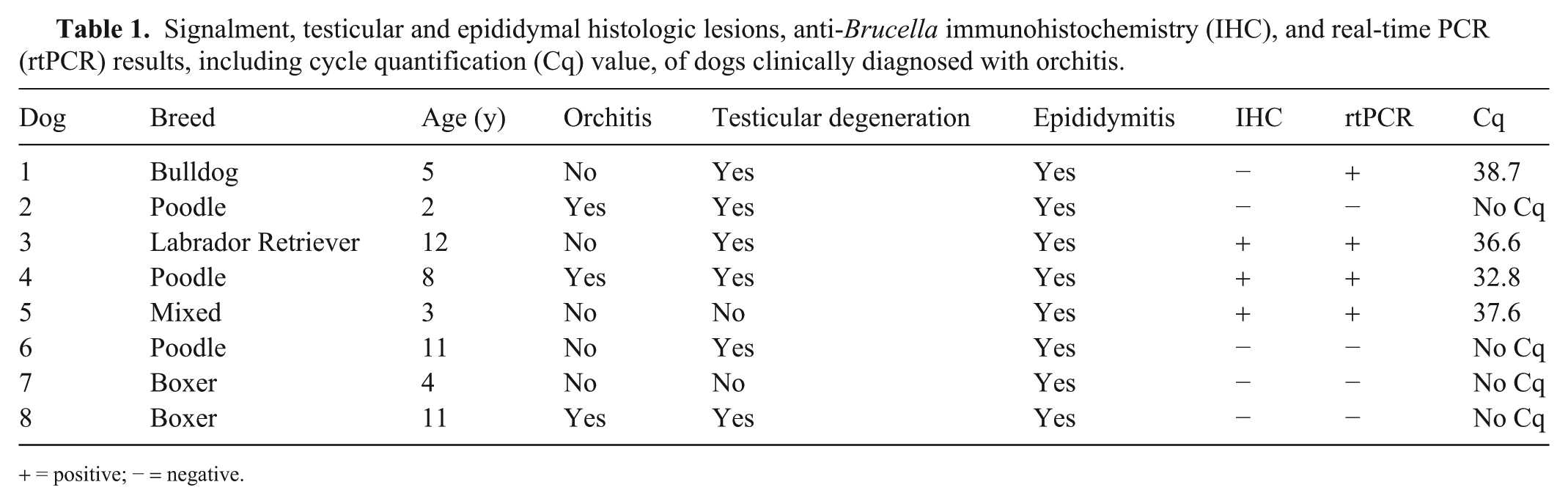
+ = positive; − = negative.
The scrotal contents from all dogs were fixed in 10% neutral-buffered formalin for 24–48 h, processed routinely, and stained with hematoxylin and eosin, followed by histologic evaluation. For all cases, the head, body, and tail of the epididymis, as well as the testicular parenchyma, tunica vaginalis, and efferent ductules, were evaluated. For case 4, the pampiniform plexus was also available for evaluation. Interestingly, despite the clinical diagnosis of orchitis, all 8 dogs had epididymitis but only 3 of the 8 dogs also had orchitis (cases 2, 4, 8). In the dogs with orchitis, inflammation occupied 25% (case 4), 50% (case 8), and 75–100% (case 2) of examined sections. Inflammatory changes displayed a range of chronicity. Acute lesions were characterized by severe necrosuppurative inflammation, frequently with complete replacement and loss of the epididymal duct (cases 1–4, 7, 8) and occasional vasculitis affecting branches of the deferential artery and infarction of the epididymis (cases 7, 8; Figs. 1, 2). No vasculitis was noted in the pampiniform plexus, although this structure was only available for evaluation for one dog (case 4). Subacute-to-chronic epididymal lesions included granulation tissue formation and fibrosis (cases 2, 3, 5, 6, 8). Testes frequently had degenerative changes within the seminiferous tubules, including loss of spermatogenic cells and formation of multinucleate spermatids (cases 1–4, 6, 8; Fig. 3). Histologic evaluation revealed that all dogs with orchitis also had epididymitis.
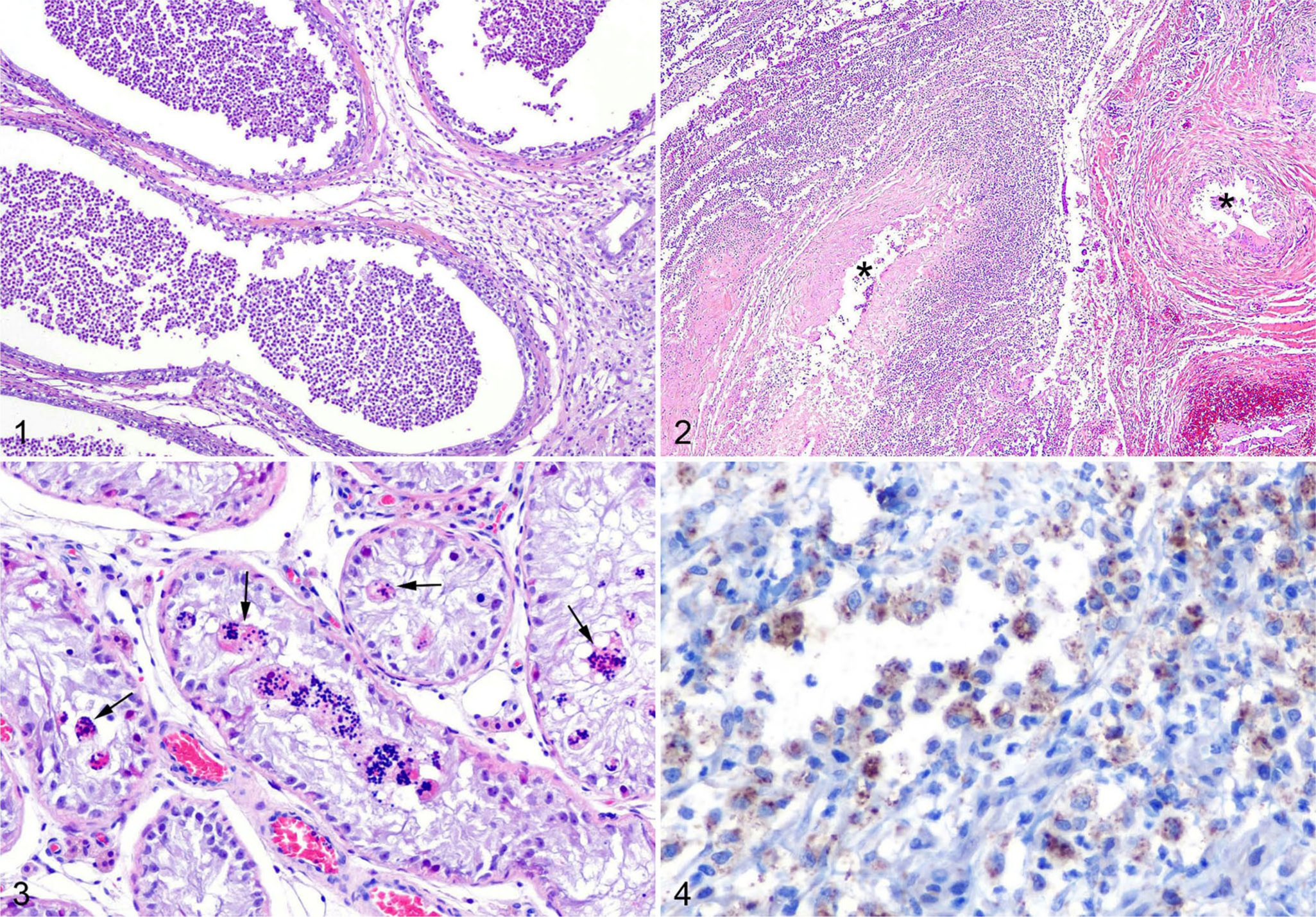
Epididymal and testicular findings in dogs clinically diagnosed with orchitis and identified as Brucella canis–positive by IHC or PCR.
Slides from FFPE testicular and epididymal tissues were prepared for IHC using a commercial kit (Vectastain Elite ABC HRP kit, peroxidase, rabbit IgG; Vector Laboratories). Following deparaffinization and rehydration, antigen retrieval was performed using citrate buffer (2100 Antigen retriever; Aptum Biologics) for 30 min and cooled for 2 h. Endogenous peroxidases were inhibited with a blocking solution (Bloxall endogenous blocking solution; Vector Laboratories) for 10 min. Nonspecific immunoreactivity was blocked with 2.5% goat serum in Tris-buffered saline + 0.05% Tween 20 (TBST) for 20 min. The primary antibody, polyclonal anti–Brucella spp. rabbit IgG (Bioss), was diluted 1:400 in TBS + 0.5% bovine serum albumin (TBSA). Slides were incubated with the primary antibody and stored at 4°C overnight. The secondary biotinylated goat anti-rabbit IgG antibody was diluted 1:200 in TBSA, and slides were incubated for 30 min at room temperature. Slides were next incubated with avidin and biotinylated hydrogen peroxidase (HRP) for 30 min diluted in TBSA. Washing for 5 min in TBST was performed between each of the preceding steps. The reaction was developed with 3,3’-diaminobenzidine (Betazoid DAB chromogen kit; Biocare Medical) for 30 s, and slides were counterstained with Gill III hematoxylin for 2 min. Slides were dehydrated and mounted under an aqueous medium. For negative controls, slides were incubated with TBSA without primary antibody. Slides from testes of healthy dogs that were presented for routine neuter with no evidence of brucellosis were included as additional negative controls. No immunopositivity was detected by substitution of the rabbit anti–Brucella spp. antibody with a rabbit antibody against a separate epitope (sheep IgG; Suppl. Fig. 1) nor was cross-reactivity with sperm antigen detected (Suppl. Fig. 2). Finally, sections of epididymis and testis from 2 dogs with epididymitis and orchitis secondary to Escherichia coli infection, as confirmed by aerobic culture, were evaluated using the same protocol. No cross-reactivity was noted in these cases (Suppl. Fig. 3).
For rtPCR, DNA was extracted from eight, 10-µm sections of each paraffin block, including from negative control samples, using a commercial kit (QIAamp DNA FFPE tissue kit; Qiagen) according to the manufacturer’s instructions. DNA concentration and purity were assessed (NanoDrop microvolume spectrophotometer; Thermo Scientific), and extracts were stored at −20°C until analyzed. All DNA extraction samples were re-labeled randomly to reduce bias. PCR runs were performed in a final reaction volume of 25 µL with the following reagents and primer concentrations: 4.5 µL of DNase-free water, 12.5 µL of PCR master mix (TaqMan universal PCR master mix; Applied Biosystems), 0.5 mM of each primer and TaqMan probe, and 5 µL of DNA template. Amplification reactions were performed (AriaMx real-time PCR instrument; Agilent Technologies) with the following cycle conditions: initial denaturation at 95°C for 10 min, followed by 45 cycles at 95°C for 15 s, and annealing and extension at 56°C for 1 min. The highly conserved insertion sequence IS711 was used as the target for the rtPCR. 12 Samples were considered positive if cycle quantification (Cq) values were < 40, or negative if no Cq values were observed. The cutoff Cq value was established based on the determination that 39 cycles are required to detect 1 genome copy of Brucella spp., with lack of amplification below 45 cycles observed using potential cross-reacting bacteria, including E. coli, Yersinia enterocolitica, Salmonella enterica subsp. enterica, Bordetella bronchiseptica, Pseudomonas aeruginosa, Agrobacterium tumefaciens, and Ochrobactrum anthropi (Suppl. Fig 4).
Brucella antigen was identified within the cytoplasm of macrophages in 3 of the 8 dogs (cases 3–5). All 3 of these dogs had epididymitis and only 1 also had orchitis (case 4; Fig 4). Importantly, Brucella antigen was only apparent within cells by IHC in areas of active inflammation. Using rtPCR to amplify the IS711 gene segment, B. canis was identified in 4 of 8 dogs (cases 1, 3–5). 12 The results of the rtPCR frequently agreed with the IHC results, apart from dog 1 in which B. canis was detectable via rtPCR but not IHC (Table 1).
Detection of canine brucellosis is a complicated process, requiring multiple tests to achieve a diagnosis. The most commonly utilized detection assays are serologic, including the RSAT, AGID, and ELISA. 11 Although these tests have reasonable specificity and are useful for screening purposes, sensitivity for the widely used RSAT is only 70%, necessitating the use of 2 different serologic tests at least 3–4 wk apart.14,16 Additionally, no serologic test is 100% accurate until 12 wk post-infection, when a strong antibody response consistently develops. 6 Culture allows for definitive detection of B. canis infection but, unfortunately, results are commonly negative when blood is submitted, given the undulant nature of bacteremia in dogs.6,11 Our use of rtPCR and IHC on FFPE tissues allowed detection of B. canis infection in male dogs diagnosed clinically with orchitis and considered to be brucellosis suspects in the absence of serologic data or fresh tissue available for culture.
Overall, these 2 detection techniques showed good concordance despite the low sample size, with IHC failing to detect B. canis in only 1 of the 4 dogs positive by rtPCR. As with culture, PCR is considered to have high specificity and has proven to be effective in detecting B. canis in the blood of infected dogs. 3 This assertion, in addition to the agreement of IHC results in 3 of 4 cases, led to the consideration of the dogs in our study that tested positively for B. canis via rtPCR as infected. Although the IHC protocol that we used did not show cross-reaction in cases of E. coli epididymitis in dogs, the polyclonal nature of the antibody allows the possibility for cross-reaction with other gram-negative organisms. It is therefore reasonable to use IHC as an adjunct test in detecting B. canis infection in male canine reproductive tissues, with greater certainty of detection based on rtPCR results. Given the small amount of DNA that was typically extracted from FFPE tissues and the number of cycles required to achieve amplification, a negative result via rtPCR in B. canis–infected dogs is a distinct possibility. Our cases that were negative by rtPCR can therefore not be interpreted as definitively negative for the presence of B. canis. Unfortunately, culture results were not available for any of our cases, and future studies should confirm the correlation of culture, rtPCR, and IHC in FFPE tissues from dogs. Nevertheless, for detection of B. canis infection when only formalin-fixed tissues are available, IHC and rtPCR offer an opportunity for pathologists faced with epididymitis in dogs to investigate the presence of B. canis infection in the absence of additional diagnostic samples.
Orchitis is a common clinical diagnosis in dogs with testicular or scrotal swelling and pain that, if left untreated, can result in tubular damage with secondary reproductive failure. 9 Veterinary clinicians frequently use the term “orchitis” to refer to inflammation of the scrotal contents as a whole, with or without epididymal involvement. As demonstrated by our study and previous work, epididymitis is the most common testicular lesion induced by Brucella canis in dogs, with all dogs in our study positively detected via IHC or rtPCR exhibiting this change.5,8 A diagnosis of orchitis in the case of a Brucella-infected dog without mention of epididymitis is therefore likely inaccurate. Orchitis in dogs with or without epididymitis should prompt the investigation of more likely causes, including the ascent of other bacterial species from the prostate gland or urethra, traumatic injury, or, less likely, fungal infection. 10
Supplemental Material
sj-pdf-1-vdi-10.1177_1040638720986883 – Supplemental material for Characterization of epididymal and testicular histologic lesions and use of immunohistochemistry and PCR on formalin-fixed tissues to detect Brucella canis in male dogs
Supplemental material, sj-pdf-1-vdi-10.1177_1040638720986883 for Characterization of epididymal and testicular histologic lesions and use of immunohistochemistry and PCR on formalin-fixed tissues to detect Brucella canis in male dogs by Andrea M. Camargo-Castañeda, Lauren W. Stranahan, John F. Edwards, Daniel G. Garcia-Gonzalez, Leonardo Roa, Lisa M. Avila-Granados, Martha E. Hensel and Angela M. Arenas-Gamboa in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
Student stipend support (MEH, LWS) was provided by the National Institutes of Health Institutional Training Grant T32 fellowship (5OD11083-7).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
