Abstract
Cerebellar granule cell layer conglutination is a tissue artifact associated with postmortem autolysis that causes cerebellar granule cell changes once thought to be caused by degeneration and necrosis. Granule cell layer conglutination has been reported mainly in humans and cattle and rarely in other animal species, but its frequency remains vastly unknown in veterinary medicine, mostly because this postmortem change is typically not recorded in autopsy reports. Pathology trainees should be aware of autolytic tissue changes that may mimic pathologic changes in the CNS, particularly when those changes are highly selective for a specific cell population within the cerebellar cortex. Here we provide a brief historical perspective on the evolution of cerebellar granule cell layer conglutination from “enzootic cerebellar necrosis,” a presumed necrotic lesion affecting granule neurons in humans and cattle, to a tissue change associated with postmortem autolysis and increased tissue acidity in the cerebellum. We also provide an update on the animal species in which cerebellar granule cell layer conglutination has been observed during our diagnostic pathology routine.
Keywords
From July 1971–July 1976, 16 diagnoses of “enzootic cerebellar necrosis” were recorded in cattle autopsied at the Laboratory of Veterinary Pathology (Laboratório de Patologia Veterinária [LPV]; Federal University of Santa Maria, Rio Grande do Sul, Brazil). Most cases were autopsied because of spontaneous death or euthanasia following neurologic clinical signs (Barros CSL, pers. comm., 2024 Feb 21). The reported changes consisted of “necrosis of the granule cell layer” of the cerebellar cortex. No cause for the apparent cerebellar changes was established at the time. These 16 cases were retrieved from the LPV autopsy archives in 2004 by one of the authors (DR Rissi) when working on a retrospective investigation of the most common diseases of cattle, which was subsequently published by a colleague as a retrospective study describing tumors in cattle. 5 After enquiring about the nature of the mysterious “enzootic cerebellar necrosis” diagnosed in the 1970s, pathology trainees at the LPV were informed by their advisor and one of the authors (CSL Barros) that the necrotic changes were in fact part of postmortem autolysis described in cattle and humans and referred to as cerebellar granule cell layer conglutination.
During the late 1950s, similar histologic changes attributed to ischemic necrosis of the cerebellar granule cell layer were referred to as “Kiriyoi disease,” an endemic and idiopathic disorder of young calves that occurred in the mountainous areas of the Chugoku-Sanin region in Japan.4,11 Neurologic clinical signs consisted mainly of ataxia, somnolence, nystagmus, and tremors, but less-specific clinical signs such as hypothermia and weakness were also described. 4 Further studies confirmed similar cerebellar changes that were attributed to degeneration and loss of granule cells throughout the cerebellar cortex with preservation of other cerebellar cortical layers, nuclei, and white matter.4,11 Pathologic findings also revealed that many affected cattle had lesions in extraneural tissues, indicating that other nutritional or infectious processes might have been present and perhaps implicated in the development of Kiriyoi disease. 4
Kiriyoi disease and similar cerebellar changes in cattle described by German pathologists in 1968 2 served as the basis for the diagnosis of “enzootic cerebellar necrosis” in the 16 cases diagnosed at the LPV. However, H&E-stained slides of cerebellum from these LPV cases were shared with Dr. Stuart Young (1926–2003) at Colorado State University (CSU; Fort Collins, CO, USA) by one of the authors (CSL Barros) in 1977. After histologic examination of the tissues, Dr. Young concluded that the observed tissue changes in cases of “enzootic cerebellar necrosis” should be attributed to postmortem autolysis of the cerebellum rather than degeneration and necrosis, as highlighted by other authors by the late 1970s. 1
Before then, “acute selective necrosis of the granular layer” attributed to acute ischemic necrosis of the granule cell layer was described in humans who had died of diabetes, uremia, or other disorders leading to coma, but who had no cerebellar clinical signs.1,6 Mirroring findings of Kiriyoi disease, histologic changes consisted of “severe nuclear changes in the granules which presented themselves as washed-out with blurred outlines” with preservation of neighboring cell populations. 6 After subsequent investigations demonstrated that similar tissue changes could be reproduced in cerebellar samples incubated at 37–40°C for 24 h and that their severity could be reduced significantly by sectioning the cerebellum before formalin-fixation, acute selective necrosis of the granular layer was deemed a postmortem phenomenon associated with autolysis due to elevated body temperature after death, insufficient or delayed formalin-fixation, and particularly low tissue pH levels. 1 The reason that the cerebellar granule cell layer is particularly prone to postmortem autolysis remains undetermined, but it is thought that the acidic environment associated with autolysis facilitates the release of proteolytic lysosomal enzymes such as naphthylamidase, which is selectively localized in the granular layer, with subsequent breakdown of granule cells. 1 Because of its high susceptibility to autolysis, the cerebellar granule cell layer has been referred to as the “pancreas of the brain.” 9
Today, cerebellar granule cell layer conglutination is a relatively well-known tissue artifact associated with postmortem autolysis that leads to the cerebellar granule cell changes once thought to be caused by necrosis.1,10 Cerebellar conglutination has been reported in humans (in which it is also referred to as état glacé = frozen state) and cattle or other Bovidae,3,8 including sheep (Rissi DR, pers. comm., 2024 Feb 21). These changes are also rarely seen in dogs (Rissi DR, pers. comm., 2024 Feb 21) and horses (Howerth EW, pers. comm., 2024 Feb 21); however, we were unable to locate any non-bovid cases in our laboratory files. In humans, cerebellar conglutination reportedly occurs in ~35% of autopsy specimens. 1 Descriptions of cerebellar conglutination are sporadic in the veterinary literature and its frequency remains vastly unknown,7,10 mostly because this postmortem change is typically not recorded in the autopsy report.
From 2000–2007, the LPV was the national reference laboratory dedicated to the surveillance of transmissible spongiform encephalopathies in Brazil. During that period, tissue changes associated with neurologic disease or autolysis were recorded in all examined bovine brains, with cerebellar conglutination described in 36% of cases regardless of the final diagnosis. 7 From January–December 2023, 41 bovine autopsies in individuals >4-mo-old were conducted at the Athens Veterinary Diagnostic Laboratory (University of Georgia, Athens, GA, USA). Evaluation of archived H&E-stained slides and autopsy reports revealed that cerebellum was available in 22 cases and that cerebellar conglutination was present in 13 cases (59%). The reported degree of postmortem autolysis in these 13 cases was mild (7 cases), moderate (4 cases), and advanced (2 cases). The postmortem interval was available from clinical forms and autopsy reports in 7 of 13 cases and consisted of 1–2 d for mild and moderate autolysis and 2–3 d for advanced autolysis.
As with many postmortem artifacts in the CNS (and outside the CNS), it is important that pathology trainees are aware of this autolytic tissue change so that it is not confused with a true pathologic change. Gross changes associated with cerebellar conglutination reflect tissue autolysis and/or poor formalin-fixation and are typically more evident within deeper cerebellar folia, which appear light pink and soft in contrast with the pale-yellow, firm, well-fixed areas. 7 Regardless of the animal species, histologic changes consist of overall pallor of the granule cell layer that is particularly evident at low magnification and may be more prominent within the deeper cerebellar folia (Figs. 1–6). Affected cerebellar granule neurons have a smudgy appearance, with overall dissolution of the surrounding granular layer tissues. As reported elsewhere and unlike cases of true degeneration and necrosis, no tissue response is observed within or around affected segments of the cerebellum, and all other cerebellar cell populations are preserved.
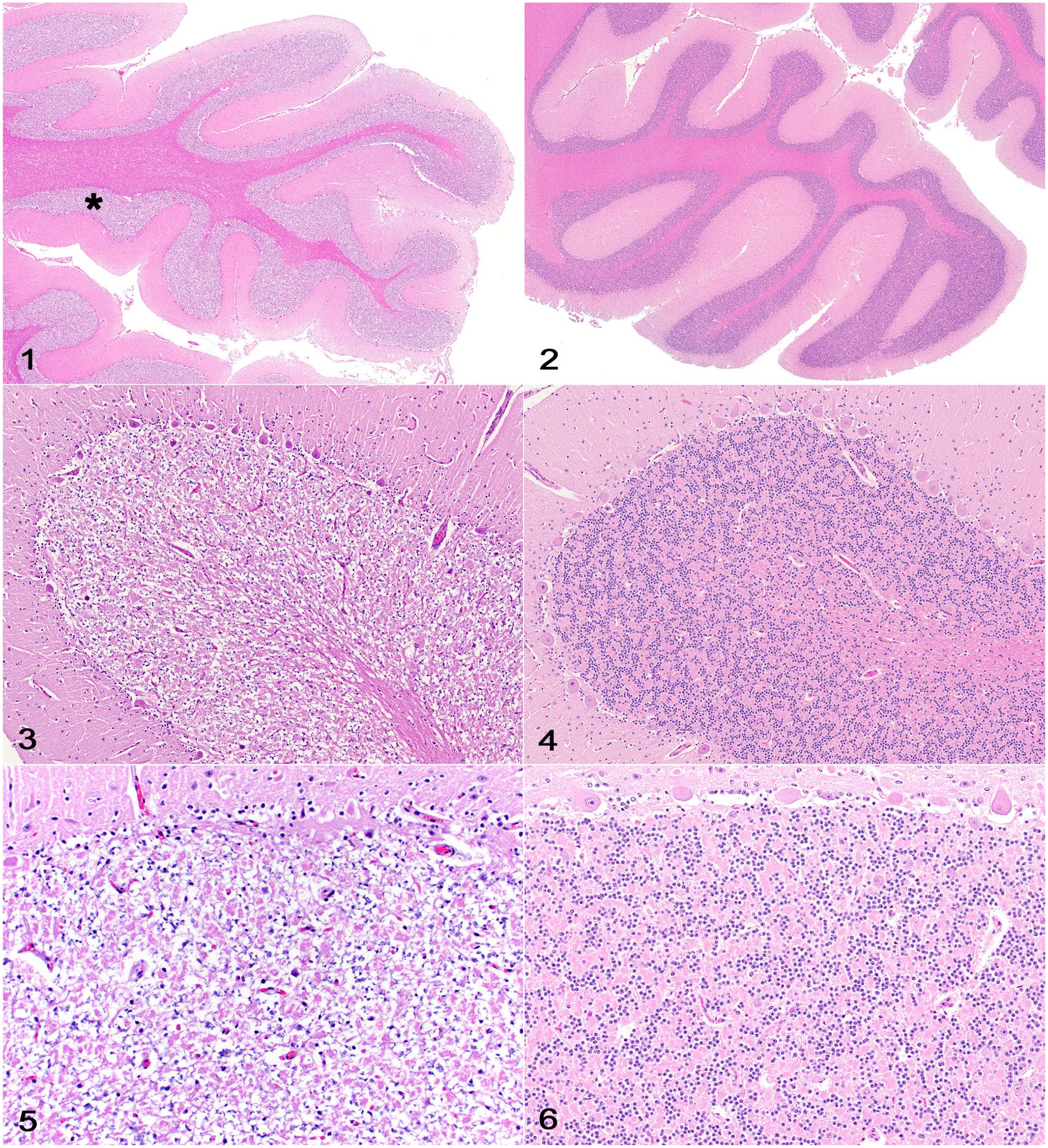
Cerebellar granule cell conglutination and control freshly fixed cerebellum (from a 5-y-old Aberdeen Angus cow with a postmortem interval of 1 d).
Footnotes
Acknowledgements
We thank Dr. Ricardo B. Lucena (Federal University of Paraíba, João Pessoa, Paraíba, Brazil) for sharing the list of diagnoses from his retrospective study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
