Abstract
The implementation of Salmonella control programs in the pork production chain demands rapid and cost-effective methods to assess the prevalence of infection in pig herds. The objective of the present study was to develop an in-house enzyme-linked immunosorbent assay (ELISA) based on S. Typhimurium lipopolysaccharides (LPS) to measure the prevalence of infection caused by Salmonella in swine herds. Coating antigen was produced by phenol extraction of S. Typhimurium culture. After standardization of ELISA test conditions, the assay was validated by testing serum samples on different animal categories: pigs orally inoculated with S. Typhimurium and sentinel animals in contact with them, naturally infected animals, colostrum-deprived piglets, and bacterin-immunized pigs. Seroconversion was observed in inoculated pigs (7 days postinfection [DPI]) and in the sentinels (21 DPI). Nonspecific reactions were not detected in the sera of colostrum-deprived animals. Serum samples from animals immunized with Salmonella Agona, Salmonella Derby, Salmonella Panama, and Salmonella Bredeney bacterins showed marked cross-reaction with the LPS from the serovar Typhimurium. Moreover, positive results obtained with the in-house ELISA were associated with Salmonella isolation in 75 infected pig herds. Comparisons with 2 commercial kits showed a linear correlation coefficient of 0.847 between the in-house ELISA and kit A and 0.922 with kit B but a low agreement in the qualitative results. In conclusion, the newly developed in-house ELISA based on S. Typhimurium LPS can be a useful tool to determine the intensity of Salmonella sp. infection in swine herds.
Keywords
Introduction
The southern region of Brazil is the most important swine-producing area in the country, supplying almost 60% of national pork production, which reached 2.7 million tons in 2005. 1 Meat quality and food safety are important issues for the meat industry all over the world. In Brazil, ongoing sanitary and food safety surveillance programs are conducted in poultry and have been started in pig herds.
Salmonella is recognized worldwide as one of the most important human food-borne pathogens. 13,22 Although eggs are considered the most important product involved in food-borne outbreaks, consumption of contaminated pork and pork products may be responsible for up to 25% of all Salmonella infections in humans. 5,11
Serological tests have been the basis for national Salmonella control programs because the possibility of automation to accommodate large-scale screening renders them cost-effective. 2,15 An enzyme-linked immunosorbent assay (ELISA) based on a combination of the somatic lipopolysaccharide (LPS) antigens of Salmonella Choleraesuis and Salmonella Typhimurium was the first test developed to detect antibodies against Salmonella enterica in pigs. 17,18 Since then, several LPS-based ELISA tests have been proposed, and commercial kits are already available.
Studies performed in southern Brazil have reported a high prevalence of pigs carrying Salmonella at slaughter. 7 Since there is a high positive correlation between the prevalence of Salmonella carriers and carcass contamination at slaughter, 6 preharvest Salmonella control programs should be implemented in infected pig herds. In Brazil, the adoption of the ELISA test for monitoring pig herds depends on imported commercial kits, which may become too expensive to be used as part of control programs.
In southern Brazil, S. Typhimurium has been the most prevalent serovar in pigs and pork. 7 This serovar shares at least 2 LPS antigens with other highly prevalent serovars isolated in this region (Salmonella Agona, Salmonella Derby, Salmonella Bredeney, and Salmonella Panama). The aim of this study was to develop an in-house ELISA test based on the LPS of S. Typhimurium (0:1, 4, 5, 12) and to evaluate the new test in infected herds in southern Brazil.
Materials and methods
Antigen preparation
Phenol extraction of LPS was performed using an overnight tryptic soy agar culture of S. Typhimurium (strain IOC 3485) suspended in phosphate-buffered saline (PBS; pH 7.4) with 0.5% (v/v) formalin and 5 mL of 5 mM EDTA, to a final concentration of 1010 colony-forming units/milliliter (cfu/mL). The suspension was boiled for 1 hr and centrifuged at 12,000 χ g for 45 min. The supernatant was removed and filtered through a 0.22-μm filter. Extraction was carried out with an equal volume of 90% (v/v) phenol solution. The suspension was homogenized for 30 min at room temperature and centrifuged at 12,000 χ g for 45 min, and the phenolic layer was discarded. This procedure was repeated until no precipitate between the phenol and aqueous layers was detected. The aqueous upper layer was removed, dialyzed against distilled water for 3 days, and stored at −70°C.
ELISA procedure
Optimal working dilutions of the LPS antigen preparation, antibody conjugate, and serum were determined by checkerboard titration. The optimal reagent concentrations were those that gave the maximum ratio between the optical density (OD) values of wells containing a positive control serum from a pig inoculated with S. Typhimurium (strain IOC 3485) and a negative serum from a specific pathogen-free (SPF) pig. First, ELISA plates a were coated with 100 μl of antigen (1:2,000) in 0.5 M of carbonate buffer (pH 9.6) and then incubated overnight at 4°C. After this, they were kept for at least 1 hr at −70°C. Finally, the plates were washed 3 times for 3 min with PBS pH 7.4 containing 0.05% Tween 20 (PBS-T). The sera were diluted (1:400) in PBS-T with 1% bovine serum albumin (PBS-TA) and added to the plates in triplicate. After incubation for 30 min at 37°C, the plates were washed as described above. In each well, 100 μl of antipig IgG conjugated to horseradish peroxidase diluted 1:25,000 in PBS-TA was added.
The plates were incubated for 1 hr at 37°C. After washing, the color reaction was developed using 100 μl of substrate: 3.5 μl H2O2 (molecular weight 34.01), 230 μl 10 N NaOH, and 10 ml 3,3',5,5’ tetramethyl-benzidine. After 15 min at room temperature, the reaction was stopped with 50 μl of 2 M sulphuric acid. The OD at 450 nm was assessed using a plate reader. b
ELISA evaluation
To evaluate the test, two 95-day-old SPF pigs were orally inoculated with 3.5 − 108 cfu of S. Typhimurium (strain IOC 3485). Two days after inoculation, 2 sentinel SPF pigs were housed together with the inoculated pigs. Serum and fecal samples were collected weekly until 42 days post-inoculation (DPI). At postmortem, bacterial isolation was performed on intestinal contents and mesenteric lymph nodes, following previously described procedures. 16
Detection of antibodies against other Salmonella serovars
The ability of the ELISA to detect antibodies against other Salmonella serovars was evaluated using serum samples from 55-day-old pigs, which were vaccinated with 5 individual bacterins produced, using serovars Typhimurium, Agona, Derby, Bredeney, and Panama. Suspensions of each serovar, yielding an OD value of 0.32 at 540 nm, were heat inactivated (boiled for 2 hr), treated with 22% aluminum hydroxide as adjuvant, and used to inoculate 5 pigs intramuscularly twice at a 2-wk interval. Four pigs inoculated with PBS were used as negative controls. Blood was collected from all pigs on days 0 to 49 after the inoculation of the first bacterin dose, allowed to clot, and the serum extracted and tested for Salmonella antibodies using the in-house ELISA.
Cutoff point determination and test standardization
Blood samples were collected from 30 colostrum-deprived piglets, and the serum was used to assess ELISA background reaction. One hundred fifty-five serum samples from a Salmonella-free pig herd were used as negative control sera for cutoff determination. The Salmonella-free status was determined through bacteriological analysis of fecal samples. The cutoff point value was set at the average obtained for the negative control serum samples plus 4 standard deviations.
Intraplate, interplate, and day-to-day variation of the ELISA was evaluated by repeated analysis of negative and positive sera. Serum samples from negative and infected pigs were pooled according to their ELISA OD values into negative, weak positive, medium positive, and strong positive. These serum pools were tested in triplicate 56 times on 39 different days, and the median of each triplicate was determined. The averages of all triplicate medians obtained for each serum pool were 0.097 (negative), 0.259 (weak positive), 0.5535 (medium positive), and 1.145 (strong positive). These averages were defined as the standard OD values. Sample OD values were transformed to calibrated ODs using a linear regression equation for standard ODs on the actual plate versus mean reference ODs.
Evaluation of ELISA test on field conditions
The in-house ELISA was applied to serum samples collected from 75 finishing-pigs herds of southern Brazil slaughtered in 5 different abattoirs. At slaughter, blood and mesenteric lymph nodes were sampled from 24 pigs of each herd. Salmonella isolation was performed as previously described, 16 and the ELISA test was done as described above. Isolates identified as Salmonella sp. were serotyped at the Brazilian Salmonella Reference Center (Fundação Instituto Oswaldo Cruz). The association between the proportion of Salmonella isolation from a herd and the within-herd seroprevalence was modeled using a generalized estimation equations logistic regression model. This analysis was performed using the PROC GENMOD present in the commercial software SAS. c Clustering at the herd level was adjusted for by allowing an overdispersion factor to inflate the variance as previously proposed. 14
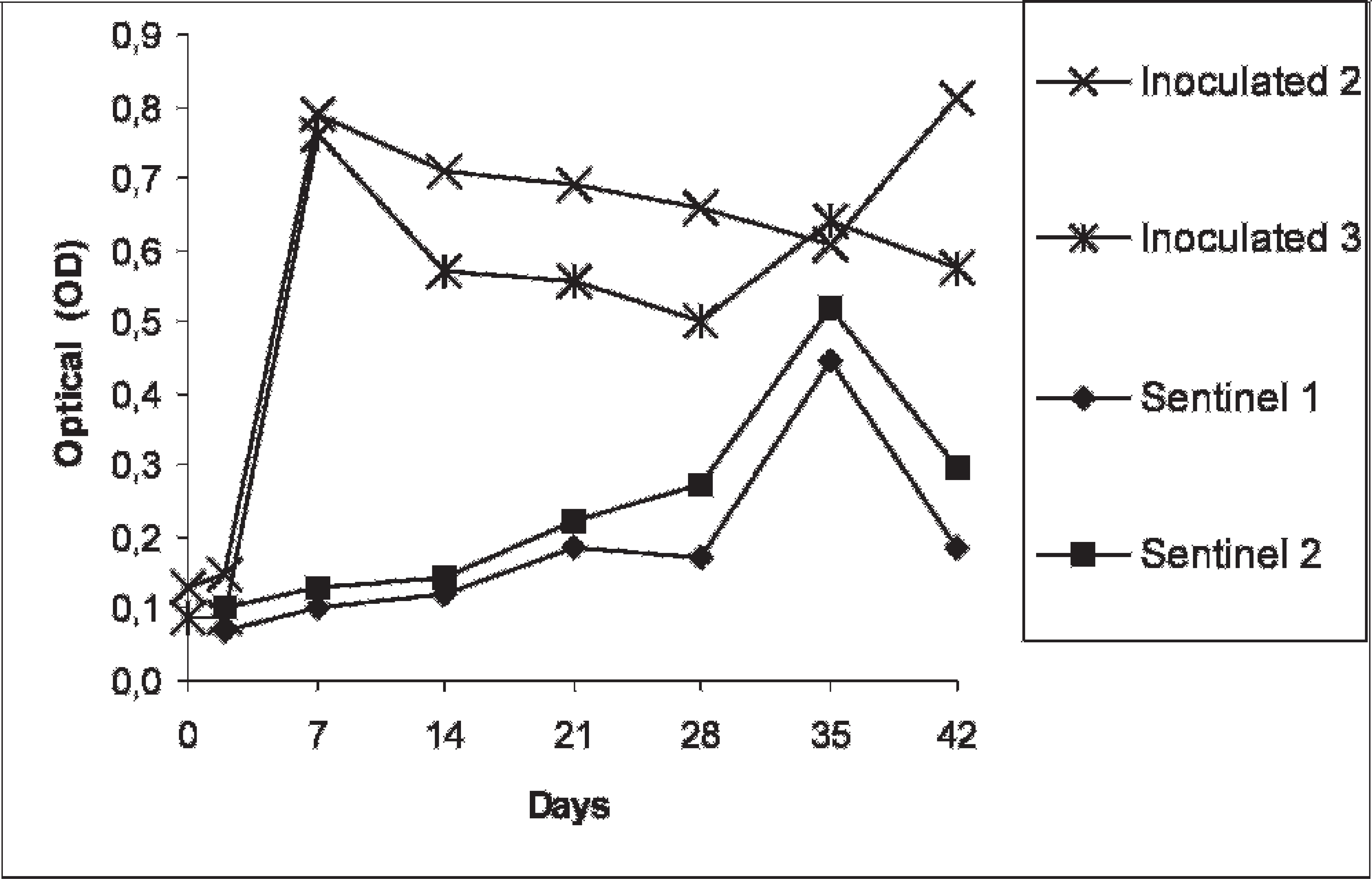
Optical densities of serum samples collected from pigs inoculated with Salmonella Typhimurium and sentinel pigs, measured in the in-house ELISA.
Comparison of the in-house ELISA with commercial kits
A panel of 273 sera from finishing pigs was tested with both the in-house ELISA and 2 commercial kits (A and B). Serum analyses using the commercial kits were performed as indicated by the manufacturers. Raw data and qualitative interpretations, using the cutoff indicated by the manufacturers, were compared with results obtained with the in-house ELISA by linear correlation coefficient and the Kappa coefficient, respectively.
Results
The in-house ELISA with purified LPS antigen from S. Typhimurium proved to be able to detect antibodies against Salmonella in pigs.
Pigs orally inoculated with S. Typhimurium showed no clinical signs during the experiment. Salmonella was isolated from the feces of both pigs after inoculation. One week postinoculation, the excretion became intermittent. At necropsy (42 DPI), Salmonella was isolated from the feces of 1 pig. The OD level prior to inoculation was <0.12 (Fig. 1). A marked increase of the OD values was observed at 7 DPI, and high OD values were maintained in this group throughout the experimental period (i.e., up to 42 DPI). Sentinel pigs had positive fecal cultures 2 weeks after housing with the inoculated pigs. One pig excreted Salmonella during the entire sampling period, while the other sentinel pig showed intermittent shedding. At necropsy, Salmonella was isolated from the feces and lymph nodes of both animals. A steady increase in OD values started at 21 DPI, reaching a maximum value at 35 DPI. After 42 DPI, the OD values began to decline.
The preinoculation OD level of pigs inoculated with different Salmonella bacterins was also <0.1 (Fig. 2). Increasing OD was observed in all pigs; however, there was a marked difference within each group and among groups in the time interval and level of increase in absorbance. The control group presented OD values <0.1 during the entire monitoring period.
The OD values of the serum samples collected from colostrum-deprived piglets ranged from 0.001 to 0.022 (0.012 ± 0.006), showing that sera without antibodies have no effect on ELISA background values. Serum samples obtained from Salmonella-negative pigs showed a mean OD of 0.065 (±0.026). The ELISA cutoff point was calculated as the OD's mean (0.065) of the negative population plus 4 standard deviations (0.1037). According to this criterion, serum OD values higher than 0.169 were statistically excluded from the negative population, and this value was used as a cutoff.
The analysis of OD results obtained in inoculated pigs, using the set cutoff, demonstrated that all animals orally inoculated with S. Typhimurium as well as sentinel animals seroconverted. Evaluation of this cutoff was also investigated in animals vaccinated with bacterins produced with different Salmonella serovars. Most pigs vaccinated with S. Typhimurium, S. Agona, and S. Bredeney became seropositive after 21 days (Fig. 2). One pig inoculated with S. Typhimurium did not become seropositive, while 2 pigs vaccinated with S. Typhimurium and S. Bredeney, respectively, seroconverted but presented OD values very close to the cutoff point. Among pigs inoculated with S. Panama and S. Derby, the seroconversion was observed only after 28 days. Most animals vaccinated with S. Panama bacterins showed OD values very close to the cutoff point during the entire monitoring period (Fig. 2).
Salmonella was isolated from 74 of 75 pig herds sampled. Prevalence of Salmonella isolation ranged from 20% to 100% of sampled animals. Eight hundred eighty-five isolates, representing 19 serovars and 8 serogroups, were isolated from slaughtered pigs (Table 1). The most common serovars isolated were Panama and Typhimurium, and together, serogroups B and D1 represented 86.6% of all isolates.
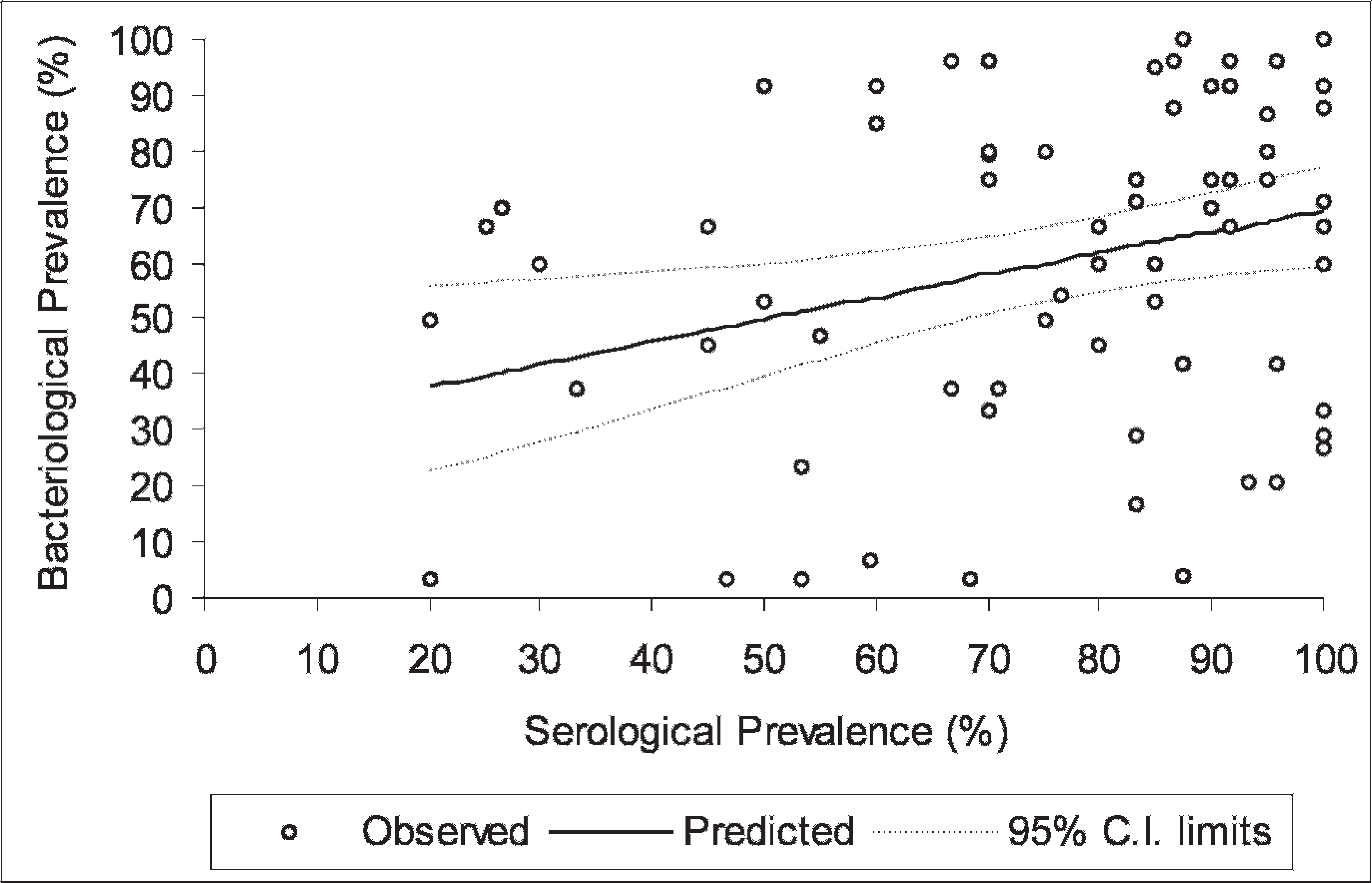
Two of these herds had no seropositive pigs when tested with the in-house ELISA, but Salmonella was isolated from 1 of them at slaughter. The association between Salmonella isolation at slaughter and within-herd seroprevalence is demonstrated in Fig. 3. Most herds presented a high within-herd seroprevalence (>0.50) as well as a high prevalence of Salmonella isolation at slaughter (>0.50). Increasing seroprevalence was significantly associated with bacteriological prevalence (P = 0.0108). The odds of positive bacteriological isolation increased 1.5 times for each 25% increase in seroprevalence. The logistic regression equation found for the predicted means was logit(y/n) = −0.8172 + 0.0162 − seroprevalence, where y is the number of pigs with positive Salmonella isolation in mesenteric lymph nodes and n the number of pigs submitted for bacteriological analysis.
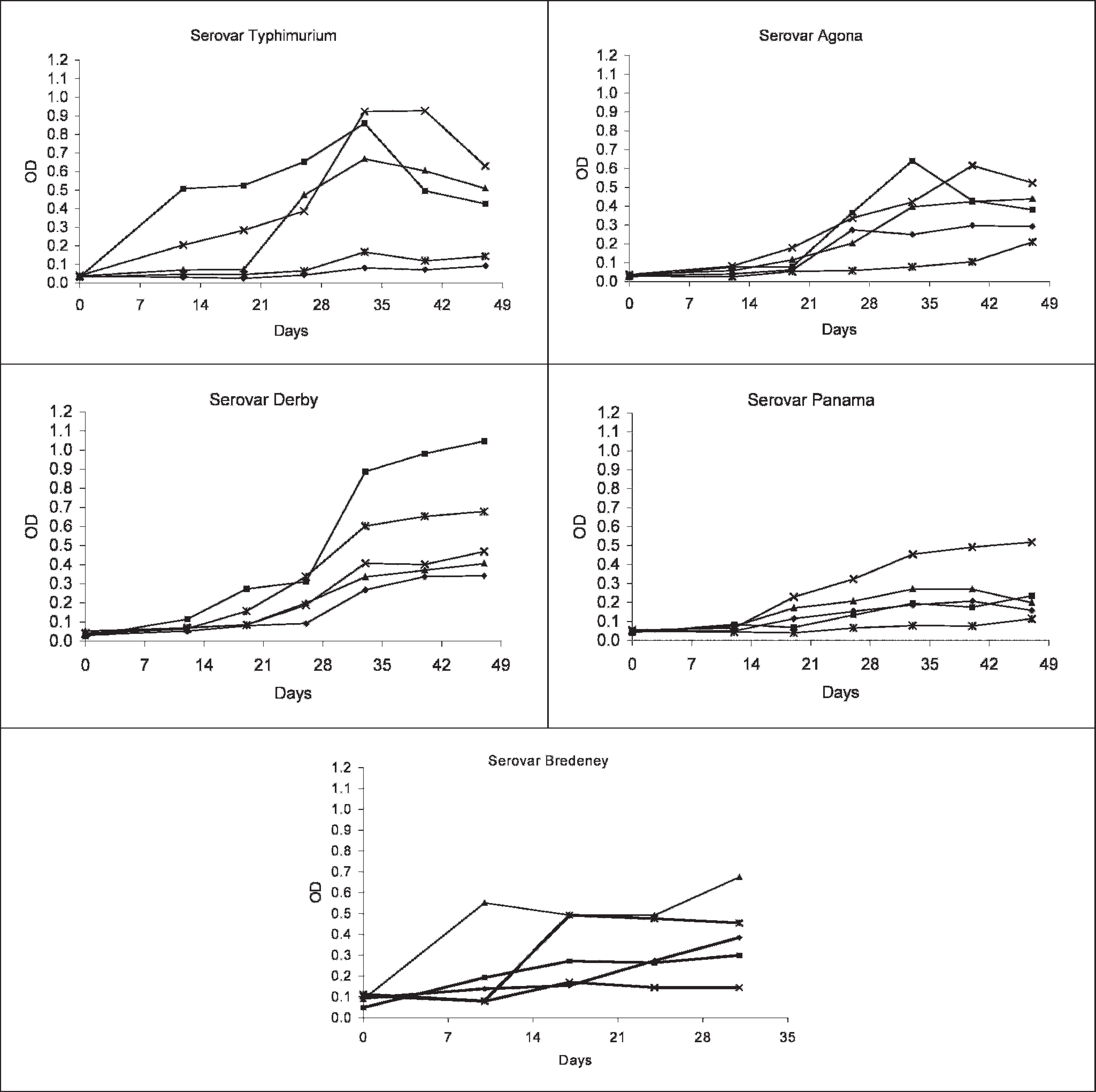
Optical densities of serum samples collected from pigs vaccinated with S. Typhimurium, S. Agona, S. Panama, and S. Bredeney, measured in the in-house ELISA.
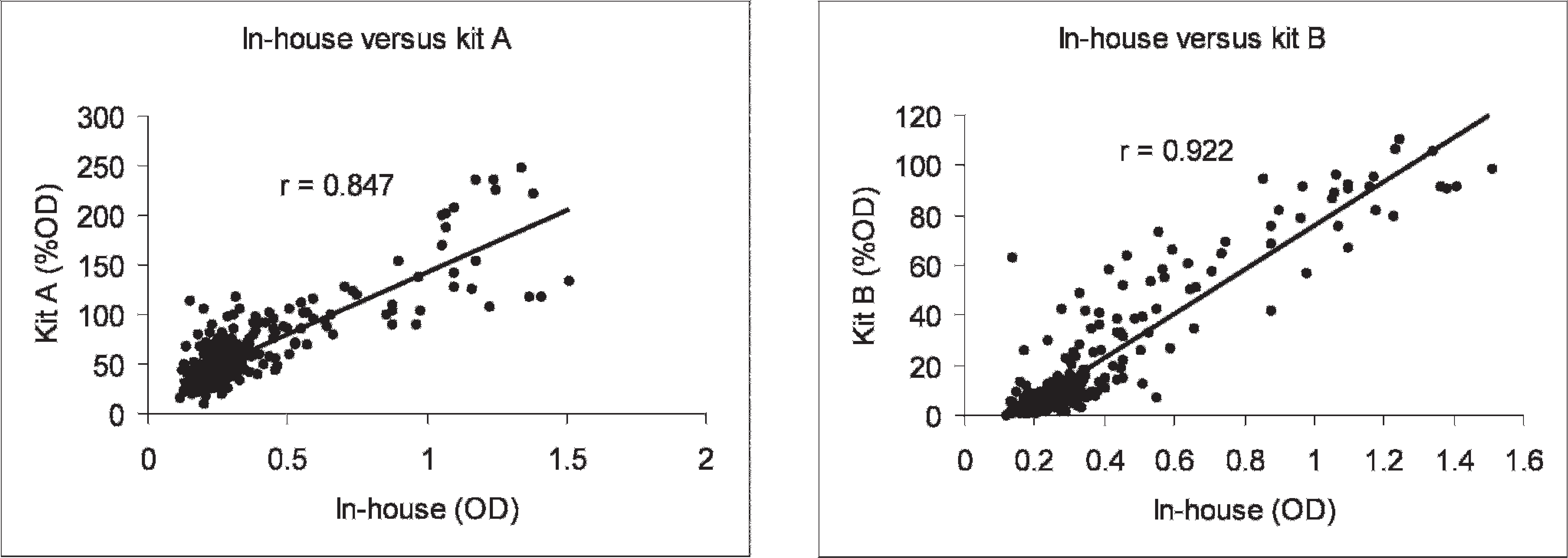
The comparison of OD values from a panel of sera tested with the in-house ELISA showed a good correlation with those OD values obtained using commercial kits (Fig. 4). A higher correlation coefficient (r = 0.922) was observed with kit B than with kit A (r = 0.847). However, when the qualitative interpretation attained with the tests was compared, a low agreement was observed between the in-house ELISA and kit A and B, with a kappa of 0.175 and 0.105, respectively.
Discussion
Infection caused by S. enterica in pigs results in shedding and seroconversion, usually without clinical signs. 9 Salmonella control programs aim to reduce the number of pork-related human cases of salmonellosis and have been based mainly on serology. 21 The Danish mix-ELISA test and commercial kits include serovars Typhimurium and Choleraesuis to detect antibodies against Salmonella belonging to serogroup B, C1, C2, and D, which represent the most isolated Salmonella serovars in Europe and the United States. However, the spectra of serovars can vary among countries. 14 For this reason, an in-house ELISA adding serogroups D1 and E1 to the coat antigen had to be developed to detect infection on French pig farms. 20
Distribution of Salmonella serovars isolated from 75 pig herds in southern Brazil.
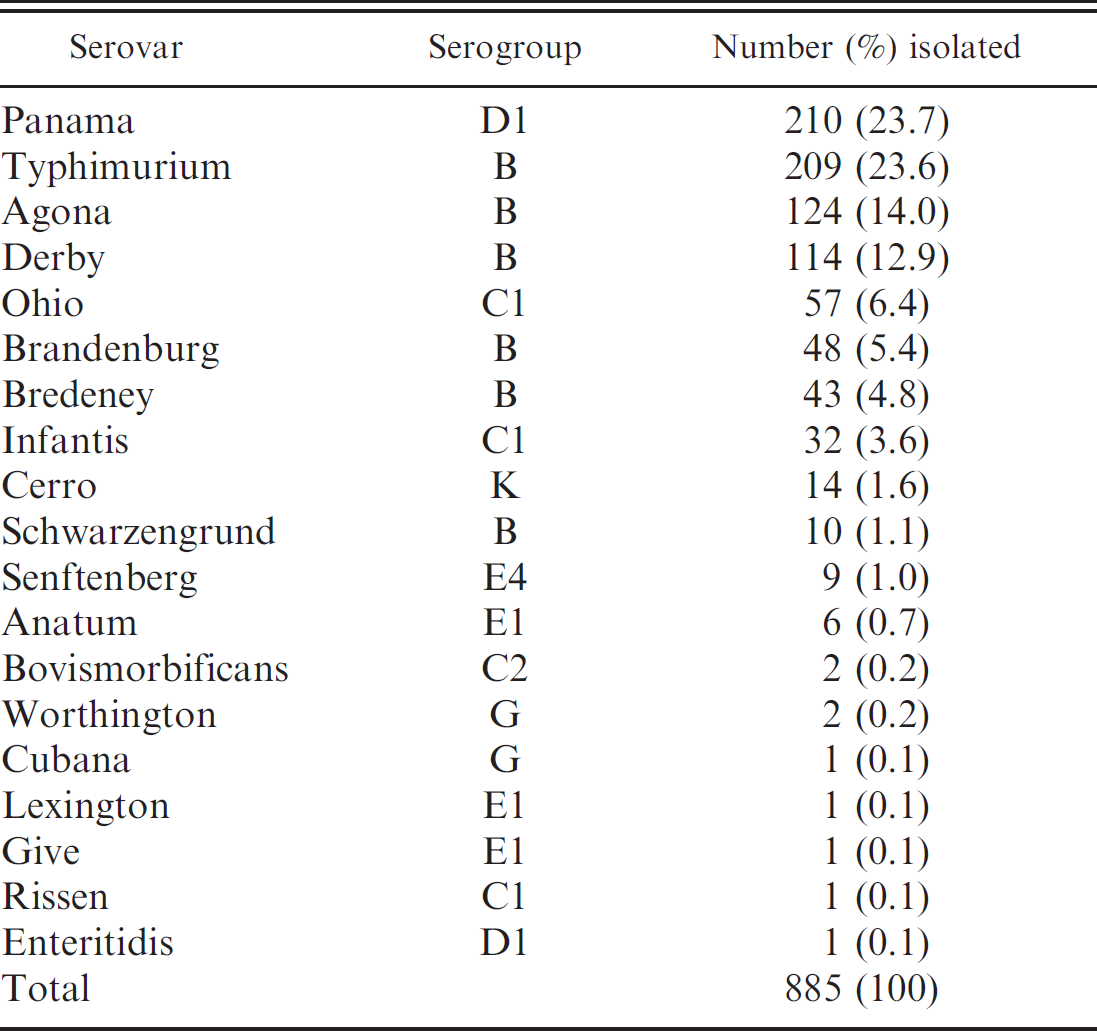
In previous studies conducted in southern Brazil, the most prevalent serovars were Typhimurium, Agona, Derby, Bredeney, and Panama. 7,10 In the current study, isolates belonging to these serovars were also prevalent (Table 1); thus, serogroups B and Dl seem to be the most important in southern Brazil. For this reason, it was decided to develop an in-house ELISA test including only S. Typhimurium LPS since strains of serogroups B and Dl share at least 1 common somatic antigen (0:12) with S. Typhimurium, and a single LPS-coated ELISA is simpler to develop than a mixed ELISA, resulting in a more cost-effective test.
Determination of the cutoff point is always a critical step in the standardization of an ELISA because a continuous variable (i.e., OD) must be transformed into a dichotomous response (i.e., positive or negative). With the chosen cutoff in the current study, all pigs orally exposed to S. Typhimurium and almost all animals inoculated with Salmonella bacterins were classified as seropositive. One pig inoculated with the S. Typhimurium bacterin did not seroconvert, in agreement with earlier observations that individual differences regarding the seroconversion period and response intensity can occur, even when animals are challenged with homologous serovars. 18,25
The correlation between the within-herd seroprevalence (in-house ELISA) and the within-herd bacteriological prevalence of Salmonella sp. in 75 finishing-pigs herds, the curve of predicted values, and 95% confidence limits at the cutoff adopted for the test.
In concordance with earlier studies, an increase in OD up to 21 DPI 18 was observed. In the present study, pigs seroconverted and had positive fecal cultures in the first week after the oral inoculation of S. Typhimurium, probably associated with the high infectious dose (108 cfu) that was administered. The contamination of the environment by the shedding pigs resulted in fecal-oral transmission to sentinel pigs that seroconverted at day 21 after exposure. The time lag before seroconversion and the gradual increase in the OD values of sentinel pigs reflect more closely a within-herd transmission, as previously observed. 24 All animals had high OD values throughout the experiment period, which may have been caused by further Salmonella reinfection of the pigs. Even considering that Salmonella was excreted intermittently, there was at least 1 positive pig on fecal culture on all sampling days, indicating that Salmonella was present in the environment. At necropsy, Salmonella was isolated from fecal samples and lymph nodes of all sentinel pigs, showing that these animals became carriers after being housed with excreting pigs. In the inoculated group, all animals were seropositive at necropsy, but only 1 pig was positive on fecal culture. The discrepancy between serology and isolation in inoculated pigs has already been reported 18 and may be related to spontaneous recovery from infection or from false-negative results on isolation.
Correlation between results of the in-house ELISA and 2 commercial LPS-ELISA kits using 273 serum samples from finishing pigs.
Differences were also seen in the time interval needed for seroconversion and intensity of the immune response among pigs inoculated with Salmonella bacterins belonging to serogroup B and D1. Variation could be observed among serovars, with Panama presenting a greater time lag before seroconversion and lower OD values. In a previous study, 25 2 ELISA kits used to detect antibodies against S. Panama performed poorly. However, in this case, most pigs were negative on bacteriological examination as well and may not have been infected by S. Panama. In the present study, the in-house ELISA detected seroconversion in vaccinated pigs, and the within-herd seroprevalence was high in herds naturally infected with S. Panama. This serovar was isolated from slaughter pigs belonging to 32 finishing herds tested with the in-house ELISA. Detection of seroreactors in herds infected with Salmonella serovars, which were not included in the adopted ELISA test, was also reported. 14 In this case, the detection of seropositive animals was associated with the concomitant infection of these herds with multiple serovars. Herds with mixed serovars infections were also found in the study and may explain the detection of seropositivity in herds with isolation of serogroups C, E, G, and K, which were not detectable by the in-house ELISA. With regard to S. Panama, multiple infections were also observed in 26 herds, but in 6 herds, S. Panama was the only serovar isolated. The within-herd seroprevalence in these herds was high (60%–100%), demonstrating that the in-house ELISA is feasible for screening S. Panama–infected herds.
The correlation between serological and bacteriological results at herd level depends on the prevalence of Salmonella, sampling method, and sensitivity and specificity of the tests. Depending on the experimental design, the correlation between bacteriological and serological evaluation in finishing pigs can be variable. 11,12,14,15,19,21 Recent exposure, either preharvest or during transport and lairage, can result in positive bacteriological tests in a seronegative herd. On the other hand, after seroconversion, some infected seropositive pigs become carriers and intermittent shedders and can be negative on isolation at sampling time. In this study, most herds showed a high prevalence of Salmonella isolation at slaughter, and a tendency for high within-herd seroprevalence was detected, demonstrating that pigs may have been infected on the farm and continued to be carriers at slaughter. This result is in accordance with previous studies, which indicated that even when the correlation between bacteriological and serological results is moderate, an increasing probability of Salmonella isolation in herds with high seroprevalence is found. 14 Thus, serology has been proposed as a monitoring tool, indicating exposure to Salmonella during production, while bacteriological testing is a way to confirm and locate a current infection. 15
To implement a surveillance program, the cutoff point of the ELISA must be continually assessed and adapted to the test purposes, allowing adjustments in sensitivity and specificity, 4 and it should also be adapted according to the development of the Salmonella control program in which it is used.
The cutoff point set for the in-house ELISA resulted in a high within-herd seroprevalence in herds with a high Salmonella isolation. Only 1 herd showed a false-negative result in the in-house ELISA, since Salmonella was isolated from pigs of this herd at slaughter. Considering that serogroups B and D were isolated and should have been detected by the in-house ELISA, the false-negative result was probably associated with a recent infection (at the end of the finishing phase, transport, or lairage).
A comparison of results obtained with a panel of sera tested in parallel with 2 commercial ELISA kits and the in-house ELISA demonstrated a high correlation between the raw data but a low agreement in the qualitative interpretation (positive/negative). This discrepancy reflects a different cutoff adopted by each test, higher for the commercial kits than for the in-house ELISA, and showed that the comparison of tests with different cutoff points may be difficult. A ring trial of ELISAs was conducted for Salmonella-antibody detection and demonstrated the influence of the cutoff on the results of a commercial ELISA test applied to a panel of positive sera. 23 In this study, the adoption of the cutoff at 10% OD resulted in a higher number of positive sera, while with the cutoff at 40% OD, the test showed the poorest performance of all tests included in the trial. In contrast, the cutoff value at which the combination of sensitivity and specificity are maximal may not always be optimal, and the economic and public health implications of mis-classification of a herd need to be considered in the planning of a control program. 14 Moreover, the cutoff point should be adjusted together with the evolution of the control program. 21
The cutoff at 40% was used during the implementation of the Danish Salmonella control program in 1995, and thus, more false-negative results were accepted. Five years later, the program was evaluated, and the cutoff point was reduced to 20% OD, increasing the sensitivity of the test and thus the prevalence of positive samples. 2 A 40% OD cutoff has also been proposed to start Salmonella control programs in other countries 8,12 and is adopted by the commercial ELISA kits for the qualitative interpretation of sample results.
Despite being lower than the cutoff adopted by the commercial ELISAs, the cutoff set for the in-house ELISA showed low false-negative results and a good performance in detecting infected herds. It must be considered that the cutoff of 40% was set in Denmark, where the estimated true prevalence of infected slaughtered pigs was 12.4% at the start of the control program. 3 Thus, a high cutoff produced fewer positive samples, including false-positive results, because of the exposure to and recovery from Salmonella infection. In contrast to the Danish situation, a higher prevalence of Salmonella-positive slaughtered pigs 7 and herds has been found in southern Brazil, so a cutoff at 40% may result in a high level of false-negative samples and a poorer correlation between serological results and Salmonella detection. Thus, a cutoff is proposed that is able to detect most infected herds to determine the risk factors for Salmonella transmission on the herd level and to propose intervention strategies on farms in southern Brazil.
In conclusion, the in-house ELISA based on the LPS-antigen of S. Typhimurium is able to detect Salmonella-infected pig herds in southern Brazil and can be adopted in monitoring programs in the region.
Footnotes
a.
Dynex Immulon 2 HB, Unitech Universal Tecnology Co., Medley, FL.
b.
Titertek-Multiscan, ICN Flow Ltd., Flow Laboratories, Inc., McLean, VA.
c.
System for Microsoft Windows, release 9.1.3, SAS Institute Inc., Cary, NC.
