Abstract
Thyrotropin-releasing hormone (TRH) stimulation can be used as a test of thyroid function and pituitary thyrotropin (thyroid-stimulating hormone, TSH) reserve, but optimal stimulation testing protocols in cats are unreported. We randomly divided 6 healthy young adult cats into 3 groups of 2 and administered 3 different intravenous doses of TRH (0.01, 0.05, 0.1 mg/kg) at weekly intervals in our crossover study. Serum TSH and thyroxine (T4) concentrations were measured using chemiluminescent immunoassay before, and at 30 and 60 min after, TRH administration. All cats were monitored for 4 h post-TRH administration for side effects. All 3 TRH doses induced significant TSH (0.01 mg/kg, p = 0.001; 0.05 mg/kg, p = 0.002; 0.1 mg/kg, p = 0.006) and total T4 (0.01 mg/kg, p = 0.008; 0.05 mg/kg, p = 0.006; 0.1 mg/kg, p = 0.001) responses. Lower TRH doses (0.01 and 0.05 mg/kg) caused fewer side effects (1 of 6 cats) than did the highest dose (3 of 6 cats), and may be safer in cats than the previously reported higher dose (0.1 mg/kg) of TRH. Our results do not support the use of maropitant to prevent side effects of a TRH stimulation test in cats.
Thyrotropin-releasing hormone (TRH) stimulation can be used as a test of thyroid function and pituitary thyrotropin (thyroid-stimulating hormone, TSH) reserve.2,7,9,16,20 Normally, TRH from the hypothalamus stimulates release of thyroid-stimulating hormone (TSH) from the pars distalis, which in turn increases synthesis and release of thyroid hormones from the thyroid gland. Therefore, administration of exogenous TRH to an individual (cat, dog, or human) with normal pituitary and thyroid function is expected to increase TSH secretion and, thereby, also increase serum total thyroxine (T4) concentration.4,11,19 In human patients, TRH stimulation testing has been used as a test for mild hyperthyroidism, as well as for primary and central hypothyroidism.2,7,9,16,20
A TRH stimulation testing protocol was developed in cats and proposed as a test for mild or occult hyperthyroidism 13 ; the serum total T4 concentration was measured before and 4 h after the IV administration of 0.1 mg/kg TRH. In clinically normal cats, the administration of TRH caused a 2-fold increase in serum total T4 concentrations, whereas cats with hyperthyroidism had a blunted or totally absent serum total T4 response to TRH. This lack of total T4 response to TRH stimulation was likely caused by chronically suppressed TSH secretion in cats with hyperthyroidism. 15 However, serum TSH concentrations were not measured in that study. 13 Only a decade later was the serum canine TSH (cTSH) chemiluminescent assay validated to measure serum TSH in cats.13,21,24
Although use of the original TRH stimulation testing protocol could be helpful in differentiating clinically normal euthyroid cats from cats with hyperthyroidism, the test has drawbacks that make it less useful for clinical use. The primary problem is that the test cannot always differentiate between cats with hyperthyroidism and those with nonthyroidal illness, with some cats having serum total T4 responses in the borderline or equivocal range.13,18 Given that TRH directly stimulates TSH secretion (only indirectly stimulating total T4 secretion induced by the increased circulating TSH), adding serum TSH measurements as an endpoint for the test might improve its diagnostic accuracy. Second, use of a 4-h test makes it less practical, given that it requires admission to the hospital for at least half a day. As well, the TRH dose (0.1 mg/kg) used in previous studies was commonly associated with adverse effects (e.g., tachypnea, drooling, vomiting, defecation, lethargy) that developed almost immediately after TRH injection.13,18 Although all of the adverse side effects associated with TRH administration resolved by the end of the 4-h test period, the severity of these side effects in some cats made the test unacceptable for most owners and veterinarians.13,18
Another indication for TRH stimulation testing is for the diagnosis of central hypothyroidism,6,7,12,20 which likely is underdiagnosed in cats that have undergone pituitary radiotherapy or hypophysectomy for acromegaly or hyperadrenocorticism.3,25 In one study evaluating stereotactic radiation in cats with acromegaly, the reported prevalence of hypothyroidism post-radiation therapy was 14%; however, the diagnosis of hypothyroidism was based on low serum total T4 concentrations alone. 25 Hypothyroidism in these cats was reported to develop after 4–48 mo. 25 Confirming central hypothyroidism can be challenging, given that affected cats have deficient TSH secretion, but with concentrations that overlap with the serum TSH concentrations in normal cats. For example, in one study, up to a third of normal cats also had low, undetectable serum cTSH concentrations. 15 In our searches of Google, PubMed, CAB Direct, Web of Science, and Scopus, using search terms “TRH stimulation test” and “cats,” we found no reports of such a study. A valid protocol for such TRH testing is needed to reliably diagnose iatrogenic secondary hypothyroidism in cats after pituitary radiotherapy or hypophysectomy.
Our primary objective was to determine the dose of TRH that maximizes increases in TSH and total T4 concentrations while minimizing side effects in healthy young adult cats before continuing studies on healthy and diseased older, client-owned cats. Our hypothesis was that TRH administered at doses < 0.1 mg/kg would adequately stimulate TSH production in healthy young adult cats and cause minimal side effects. Our secondary objective was to determine whether administration of maropitant, an anti-emetic, could decrease side effects of TRH administration.
Materials and methods
We included in our study 6 young adult (mean: 2.2 years; range: 2.2–2.2) domestic shorthaired cats (3 castrated males, 3 intact females) from a research facility. Median body weight of the cats was 6.5 kg (IQR: 3.50–6.70; range: 3.20–8.90), with a median body condition score (BCS) of 4.5 of 9 (IQR: 4.00–5.75; range: 4.00–8.00). All cats were considered healthy based on results of physical examination, blood pressure measurements, CBC, serum chemistry profile, urinalysis, and baseline serum total T4 and TSH concentrations; however, 2 of the cats had increased BCSs. No cats were euthanized as part of our study. Our study was approved by the IACUC (protocol 170.069) at the research facility (High Quality Research, Fort Collins, CO, USA).
The cats were sedated with 0.3 mg/kg butorphanol (Torbugesic; Zoetis) and 2 mg/kg alfaxalone (Alfaxan; Jurox) administered IM, and a jugular catheter was placed for blood sampling. Blood (2 mL) was collected prior to IV administration of TRH and at both 30 and 60 min after each dose of TRH.
Different doses (0.01, 0.05, 0.1 mg/kg) of TRH (Protirelin; Wedgewood Pharmacy) were administered IV. All cats were monitored for 4-h post-TRH administration for side effects. Cats were randomly divided into 3 groups of 2 (2 males, 2 females, and 1 male and 1 female), and administered a different dose of TRH at different weeks. All sampling was performed between 9:00 and 11:00 am (Table 1). There was a 1-wk washout period between doses. On week 4, all 6 cats received an injection of maropitant (1 mg/kg IV; Cerenia, Pfizer), and blood was collected 1 h later and before IV administration of TRH (0.01 mg/kg, IV). Additional blood was collected at 30 and 60 min post-TRH administration.
A different dose of thyrotropin-releasing hormone was administered to each group of cats each week in our crossover study.
Serum TSH (Immulite canine TSH; Siemens) and total T4 concentrations (Immulite total T4; Siemens) were measured by chemiluminescent immunoassay (CLIA; Immulite 2000 XPi, Siemens) as described previously.13,22,25 Assay performances were validated prior to our study.
Nonparametric statistical tests were used to evaluate the TRH stimulation results. Results are given as the median and interquartile range (25th–75th percentile, IQR). The Friedman test was used to compare serum total T4 and TSH results after TRH administration to baseline values. The Wilcoxon test was used to compare serum total T4 and TSH results between the maropitant pretreatment and no-maropitant study. Analyses were performed using Prism v.9.5.1 (GraphPad). For all analyses, statistical significance was set at p ≤ 0.05.
Results
The median serum total T4 concentration in the cats prior to our study was 26.6 nmol/L (IQR: 23.5–30.3 nmol/L), and the median serum TSH concentration of the cats prior to the study was 0.073 ng/mL (IQR: 0.054–0.078 ng/mL).
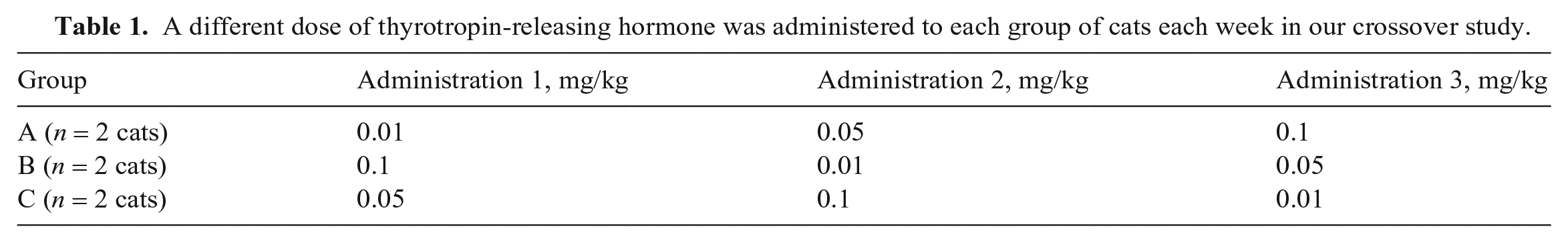
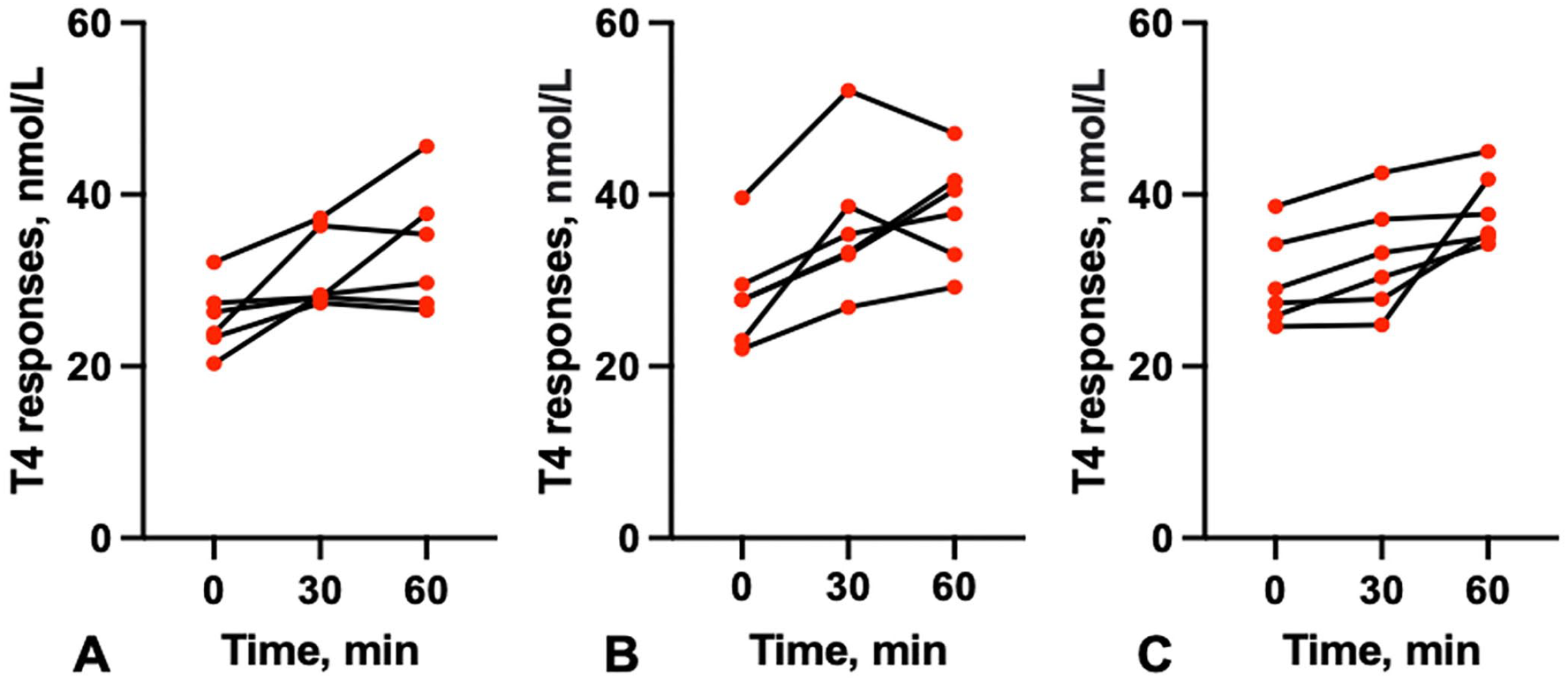
All 3 TRH doses induced significant total T4 (0.01 mg/kg, p = 0.008; 0.05 mg/kg, p = 0.006; 0.1 mg/kg, p = 0.001; Fig. 1, Table 2) and TSH (0.01 mg/kg, p = 0.001; 0.05 mg/kg, p = 0.002; 0.1 mg/kg, p = 0.006) responses (Fig. 2; Table 2). After administration of 0.01 mg/kg of TRH, all cats had higher TSH responses at 30 min compared to 60 min post-TRH administration. The same pattern was seen in 5 of the 6 cats after administration of 0.05 mg/kg TRH, and in 4 of 6 cats after administration of 0.1 mg/kg TRH; the remaining cats had higher TSH concentrations at 60 min compared to 30 min. The TRH dose of 0.01 mg/kg induced a mean 17-fold (range: 6–49-fold) increase in TSH concentrations at 30 min and a mean 13-fold (range: 5–36-fold) increase at 60 min, which was the highest fold increase from the 3 doses. The TRH dose of 0.05 mg/kg induced a mean 16-fold (range: 4–43-fold) increase in TSH concentrations at 30 min and a mean 12-fold (range: 3–27-fold) increase at 60 min. The TRH dose of 0.1 mg/kg induced a mean 13-fold (range: 5–33-fold) increase in TSH concentrations at 30 min, and a mean 11-fold (range: 7–20-fold) increase at 60 min.
Total thyroxine (T4) response in healthy cats after intravenous thyrotropin-releasing hormone (TRH) administration at different concentrations:
The median and interquartile range (IQR) of total thyroxine (T4) and thyroid-stimulating hormone (TSH) concentrations in 6 cats following the administration of different doses of thyrotropin-releasing hormone (TRH).
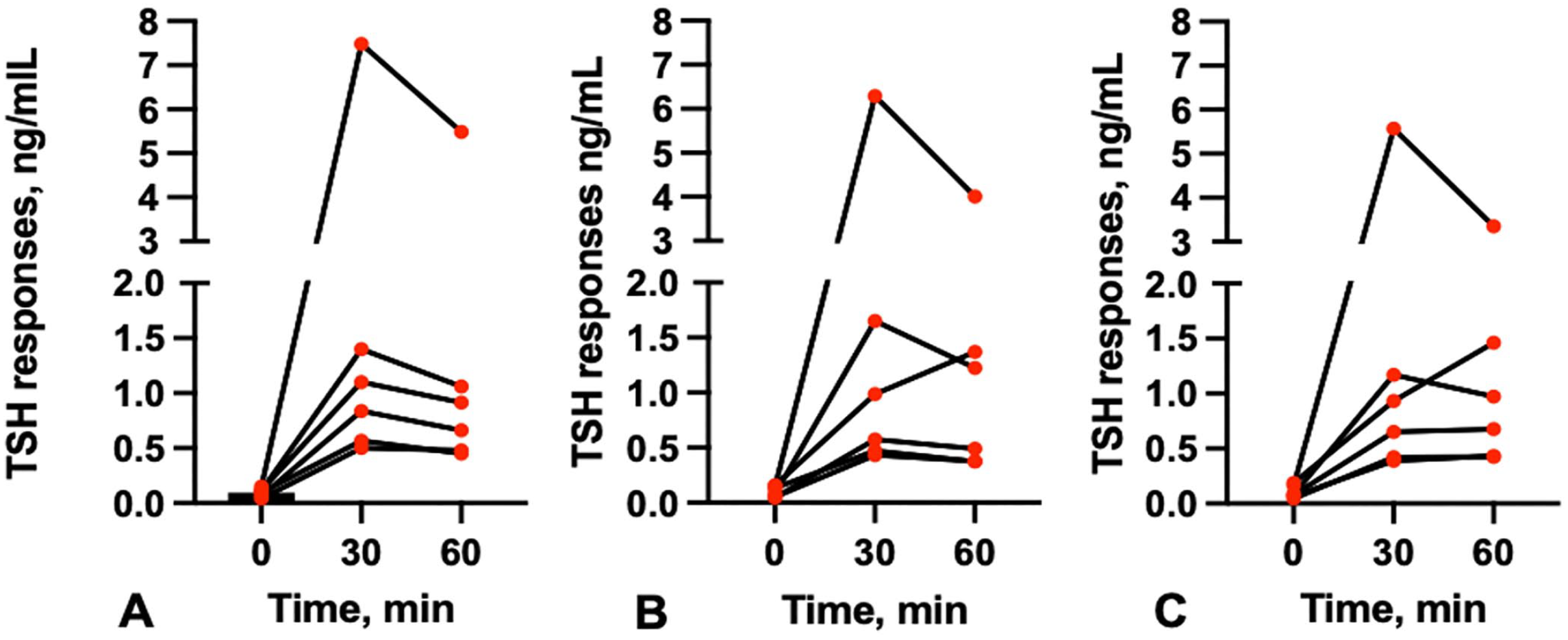
Thyroid-stimulating hormone (TSH) response in healthy cats after intravenous TRH administration at different concentrations:
The only side effect noted after TRH administration was drooling. Administration of the lower TRH doses (0.01 and 0.05 mg/kg) caused drooling in fewer cats (1 of 6) than did the highest concentration (0.1 mg/kg; 3 of 6), but these results were not significantly different. None of the cats vomited or panted.
The serum TSH concentrations after administration of 0.01 mg/kg TRH were statistically different at 60 min but not at 0 or 30 min between the maropitant pretreatment and the no-maropitant experiments (0 min: p = 0.094; 30 min: p = 0.219; 60 min: p = 0.031; Table 2). There was no statistical difference in the serum total T4 concentrations after administration of 0.01 mg/kg TRH between the maropitant pretreatment and the no-maropitant experiments (0 min: p = 0.063; 30 min: p = 0. 844; 60 min: p = 0.563).
Side effects noted in the cats pretreated with maropitant were subjectively more severe than in the experiments performed without maropitant. In the maropitant pretreatment experiment, 1 cat defecated and had transient drooling and 1 cat appeared nauseous and had transient tachypnea.
Discussion
In clinically normal dogs, it has been well-documented that TRH doses < 0.1 mg/kg will stimulate TSH concentration adequately, with few or no adverse effects. 1 To our knowledge, the effect of TRH stimulation test on both serum total T4 and TSH in healthy cats has not been reported previously.
In healthy cats with normal thyroid function and cats with nonthyroidal disease, 14 the administration of 0.1 mg/kg TRH increased serum total T4 concentrations by > 50% 4-h post-IV administration; however, serum total T4 concentrations only increased in 11.4% of hyperthyroid cats. All 3 doses of TRH that we used induced significant increases in serum total T4 and TSH concentrations. In a 2007 study of a TRH stimulation test (0.002–10.0 mg/kg) in cats and dogs, increasing the dose of TRH increased the duration, but not the magnitude, of total T4 stimulation. 25 In our study, the TRH dose of 0.01 mg/kg did not induce any less TSH response at 30 min than at 60 min post-administration of higher doses (0.05 and 0.1 mg/kg) of TRH.
In humans, the normal response to TRH administration is that TSH is higher at 30 min than at 60 min post-TRH administration. 6 We saw this pattern in all 6 cats after administration of 0.01 mg/kg TRH; however, not in all cats with higher doses of TRH (1 cat after administration of 0.05 mg/kg TRH, and 2 cats after administration of 0.1 mg/kg TRH, had higher TSH values at 60 min than at 30 min). Our results suggest that using the lower dose of TRH might be more useful in cats; however, studies in clinically ill cats are needed to evaluate the TRH response at this dose. We saw the highest fold increase in TSH at the lowest TRH dose (0.01 mg/kg), which induced a mean 17-fold increase in TSH levels at 30 min and a mean 13-fold increase at 60 min. There was high variability in the fold increase between the cats, which suggests that TRH response may be very individual in cats; further studies are needed in healthy cats to set RIs for cats. However, a minimum 3-fold increase at 30 min and 4-fold increase at 60 min in TSH was seen in the cats of our study at any of the TRH doses.
A study of changes in total T4, free T4, and T3 concentrations in dogs after TRH reported the peak mean concentrations at 4 h without a significant difference between different TRH doses (0.1–0.6 mg/kg). 17 We did not measure serum total T4 concentrations after 1 h, and therefore the peak serum total T4 concentrations at previously reported 4-h post-TRH administration could not be evaluated post-different TRH doses and is one of the limitations of our study.
TRH administration has been reported to cause transient tachycardia, tachypnea, panting, vomiting, drooling, defecation, urination, miosis, and lethargy.8,10,14,18 In our study, the lower TRH doses (0.01 and 0.05 mg/kg) caused drooling in fewer cats than the highest concentration (0.1 mg/kg). Premedication with maropitant did not influence the TSH concentrations after TRH stimulation, but might influence the total T4 response; maropitant premedication appeared subjectively to cause more side effects, including defecation, drooling, nausea, and tachypnea, compared to our experiment with untreated cats. Following premedication with maropitant, total T4 concentrations no longer changed statistically between sampling times (p = 0.184), which could be the result of the premedication of maropitant or, as in previous reports, the peak of total T4 increase is at 4-h post-TRH administration; hence, there might not have been enough time to measure the elevation of total T4 despite us seeing a statistical difference in the first week. Our results do not support the use of maropitant to prevent side effects of TRH stimulation test in cats.
Limitations to our study include the facts that there are currently no RIs for the TRH stimulation test in healthy cats, and we performed the protocols described here in only 6 cats, which might not show all inherent differences that occur naturally within and between individuals. For example, 1 cat in our study had a much greater increase in serum TSH concentration after all 3 doses of TRH than did the other 5 cats, which likely represents individual variation. Additional differences might also occur in individual cats depending on diet and other environmental factors. However, these possible confounding factors should have been minimized given that the cats in our study were from a research colony and were all housed together and fed the same diet.
Although the feline TSH concentration is measured most commonly by the CLIA that we used,13,22,25 some cats with hyperthyroidism and some euthyroid cats do not have detectable TSH concentrations, likely because the limit of detection (LOD) of the assay is 0.03 ng/mL.5,23 In our study, all of the healthy cats had TSH concentrations above the LOD; however, data should now be collected from older healthy cats and cats with hyperthyroidism to gather information on the utility of the TRH stimulation protocol described herein.
Footnotes
Acknowledgements
We thank Dr. Mark Peterson for advice on study design and assisting with manuscript preparation. Our work was presented in part at the 2022 Am Coll Vet Intern Med Hybrid Forum.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
