Abstract
Immunophenotyping of canine large-cell lymphoma (LCL) for B-cell and T-cell surface antigens is commonly performed to better predict the clinical outcome. Expression of surface antigen CD3 is associated with T-cell malignancies; surface antigen CD20 is expressed on B cells. However, a small subset of canine LCLs expresses both CD3 and CD20 (CD3+/CD20+); this form of lymphoma remains poorly defined at the molecular level. In a retrospective study, we aimed to better characterize immunophenotypic properties and antigen receptor clonality of CD3+/CD20+ LCL. We selected formalin-fixed, paraffin-embedded tissues from 10 cases of CD3+/CD20+ LCL and breed-matched controls of peripheral large T-cell lymphoma (PTCL) and diffuse large B-cell lymphoma (DLBCL). Using PCR for antigen receptor rearrangement (PARR), we identified monoclonal T-cell receptor gamma (TCRγ) rearrangements in all CD3+/CD20+ cases. Three of 10 cases had monoclonal rearrangements in the immunoglobulin heavy chain (IgH), supportive of cross-lineage rearrangement. There was no significant difference in the frequency of antigen receptor rearrangement between CD3+/CD20+ and PTCL cases. In comparison with DLBCL, CD3+/CD20+ LCL had TCRγ rearrangement more frequently and IgH rearrangement less frequently, respectively. Immunolabeling of the B-cell marker PAX5 occurred less frequently in all CD3+/CD20+ LCL cases compared to the DLBCL controls. Immunolabeling for BCL-2 was robust, regardless of immunophenotype. Nuclear Ki67 positivity was variable in CD3+/CD20+ cases, indicating a heterogeneity in proliferation. Overall, cases of canine CD3+/CD20+ LCL had properties similar to PTCL, suggesting a similar histogenesis of these 2 subsets.
Canine non-Hodgkin lymphoma refers to a diverse group of hematologic neoplasms, with aggressive, large-cell lymphomas (LCL) representing the most prevalent subset. 30 Diagnostic recommendations provided by the World Health Organization assume a specific cellular origin for these cancers. 28 Specifically, most lymphomas arise from either CD3+ T lymphocytes or CD20+ B lymphocytes. Diffuse large B-cell lymphoma (DLBCL) and peripheral large T-cell lymphoma (PTCL) remain the most common subtypes in dogs. 30 Immunophenotyping is clinically important because high-grade T-cell lymphomas have the shortest survival times, and T-cell neoplasms have been reported to have decreased response rates to chemotherapy.14,27 Therefore, immunohistochemistry (IHC) for B- (CD20 and PAX5) and T-lymphocyte (CD3) antigens is often recommended in the diagnostic workup of lymphoma.
Although immunophenotyping allows for either a B- or T-cell classification for most LCL cases, a small subset of canine lymphomas expresses both CD3 and CD20. Conversely, null LCL has been reported in dogs and lacks expression of both CD3 and CD20. 19 The CD3+/CD20+ phenotype has been described in a small number of case reports spanning a few different types of lymphoma, including enteropathy-associated T-cell lymphoma, anaplastic lymphoma, and cutaneous epitheliotropic T-cell lymphoma.2,9,15 –17,26 Despite these publications, understanding of CD3+/CD20+ LCL in dogs remains extremely limited.
We compared 10 CD3+/CD20+ LCLs, which affected mostly peripheral lymph nodes, to breed-matched cases of DLBCL and PTCL. More specifically, we determined clonality in all cases as well as provided immunolabeling properties for paired box gene 5 (PAX5), B-cell lymphoma 2 (BCL-2), and Ki67. We found that CD3+/CD20+ lymphomas are similar to PTCL with respect to antigen receptor rearrangement and PAX5 expression. Ki67 nuclear positivity was variable in our case series, indicating a difference in the proliferative activity of CD3+/CD20+ LCL.
Materials and methods
Case selection
We selected formalin-fixed, paraffin-embedded (FFPE) tissues retrospectively from the Michigan State University Veterinary Diagnostic Laboratory (IACUC exempt; MSU-VDL, Lansing, MI, USA). Cases of LCL, as determined by a board-certified anatomic pathologist (B.K. Harrington), that immunolabeled positive for both CD3 and CD20 met the criteria for inclusion in the study. DLBCL and PTCL case controls were selected based on breed similarity to the CD3+/CD20+ LCL group. Seven of 10 cases had exact breed matches. For the 3 unmatched patients, we selected Labrador Retriever controls for the Newfoundland and Shi Tzu controls for the Dachshund based on reports on the genetic similarities between these breeds,3,18 and a mixed-breed control for the Beagle (Tables 1, 2; Fig. 1).
Demographics of biopsy and autopsy submissions of canine lymphoma cases at the Michigan State University Veterinary Diagnostic Laboratory.
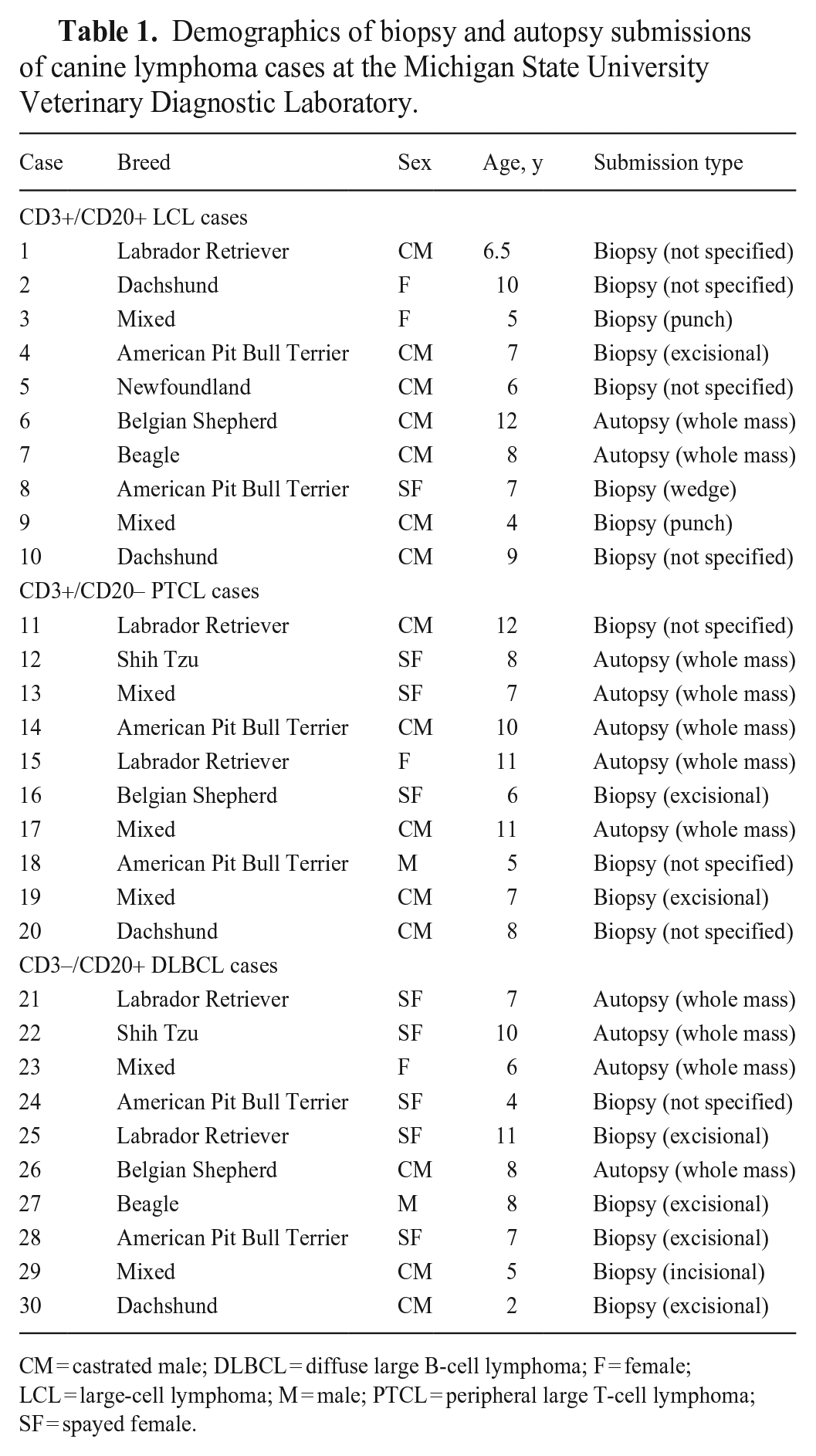
CM = castrated male; DLBCL = diffuse large B-cell lymphoma; F = female; LCL = large-cell lymphoma; M = male; PTCL = peripheral large T-cell lymphoma; SF = spayed female.
Nodal and/or extranodal involvement of biopsy and autopsy submissions of canine lymphoma cases at the Michigan State University Veterinary Diagnostic Laboratory.

DLBCL = diffuse large B-cell lymphoma; Gen. lymphadenopathy = generalized lymphadenopathy; LCL = large-cell lymphoma; LN = lymph node; MALT = mucosal-associated lymphoid tissue; PTCL = peripheral large T-cell lymphoma.
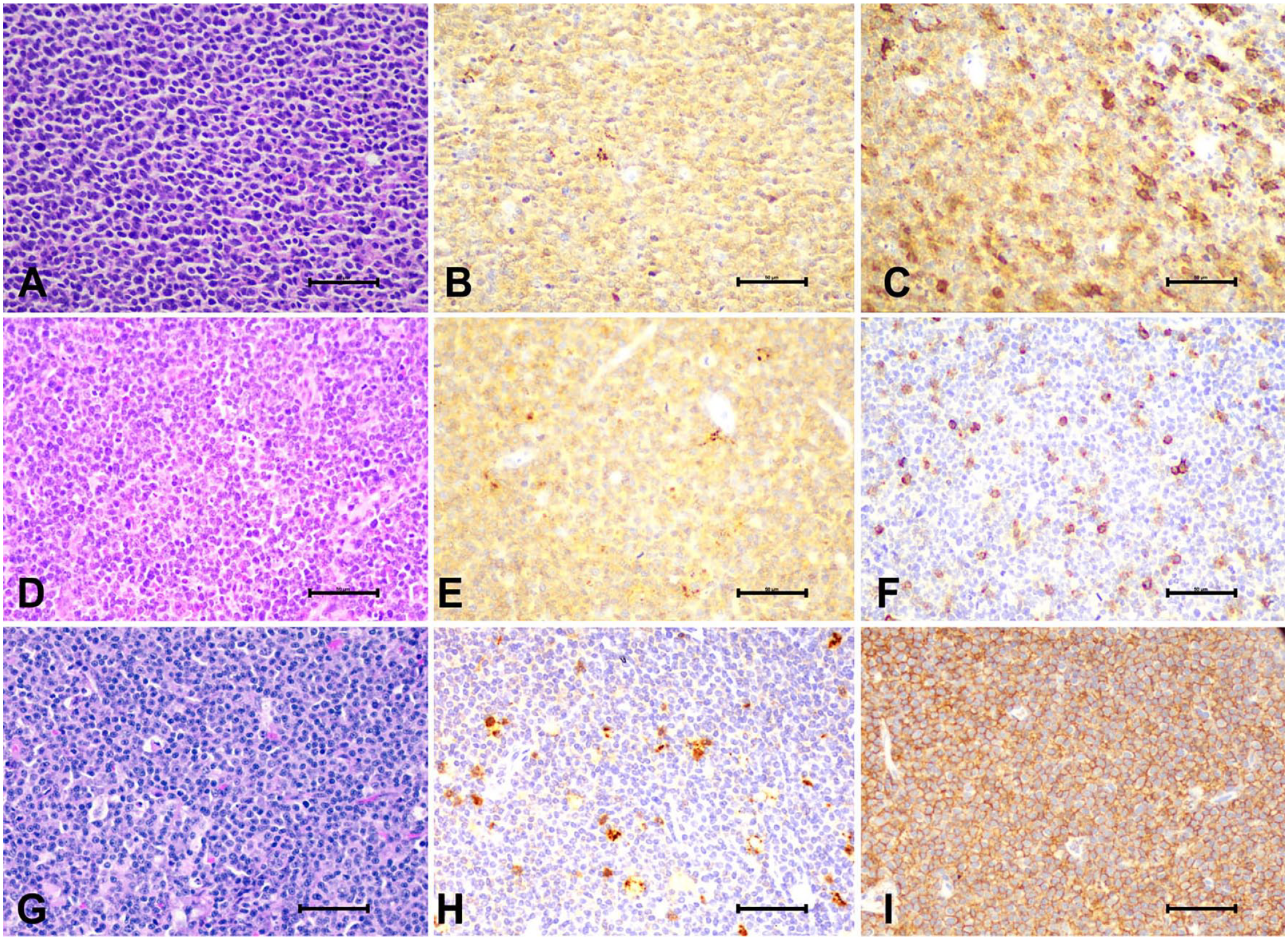
Representative H&E stains and CD3 and CD20 immunolabeling images from CD3+/CD20+ large-cell lymphoma (LCL) and control cases. Case 10, CD3+/CD20+ LCL:
Parr
DNA was extracted from FFPE tissues (QIAamp DNA FFPE tissue kit; Qiagen) according to the manufacturer’s instructions. IgH and TCRγ primer sets were designed based on previous publications (Table 3).4,11 The negative reaction control was performed by using the DNA FFPE extraction kit with nuclease-free water. In the positive control condition, previously documented DNA from a canine lymphoma sample unrelated to our study was used. PCR was performed (Type-it mutation PCR kit; Qiagen) with 20–100 ng of DNA from each case. For amplification of the TCRγ CDR3 region, a forward and reverse primer “cocktail” of the 6 listed sets in equal parts was made as described previously. 11 PCR was conducted in a T100 thermocycler (Bio-Rad). Denaturing, annealing, and extension were allowed to proceed for 35 cycles. The annealing temperature for the TCRγ region was 64°C for 90 s; B-cell amplification was at 60°C for 30 s. The subsequent PCR product was then detected, separated by capillary electrophoresis, and analyzed (QIAxcel advanced instrument; Qiagen) using a high-resolution pre-cast gel cartridge. DNA intensity peaks were obtained (QIAxcel ScreenGel software; Qiagen).
Sequences of primers used in PCR for antigen receptor rearrangements to determine clonality in either the IgH or TCRγ gene in canine lymphoma cases.

IgH = immunoglobulin heavy chain; TCRγ = T-cell receptor gamma.
Immunohistochemistry
All tissues were fixed in 10% neutral-buffered formalin for 12–24 h and processed at the MSU-VDL. Slides were deparaffinized and re-hydrated using standard laboratory methods. IHC was conducted with the following parameters, as described previously5,12,24,25 (Table 4). Antigen retrieval was performed with CC1 (Cell conditioning I, Discovery Ultra; Ventana) at 95°C for 4 min, or ER1 (Bond epitope retrieval solution I; Leica) at 95°C for 20 min. A Bond-Max automated IHC stainer was used for CD3, CD20, and Ki67 with the Bond polymer detection system (Vision Biosystems; Leica) and 3,3′-diaminobenzidine (DAB) as the chromogen. The Discovery Ultra automated staining system was used for BCL-2 and PAX5 with the Discovery UltraMap alkaline phosphatase system and Discovery Red (Roche). Sections of canine lymph node tissue were used as a positive control for all antibodies. The antibody used for BCL-2 has been established in primary colorectal follicular canine lymphoma, marginal zone lymphoma, and mantle cell lymphoma.20,25
Primary antibody source, dilution, antigen retrieval method, and detection method employed in immunohistochemistry of canine lymphoma cases.
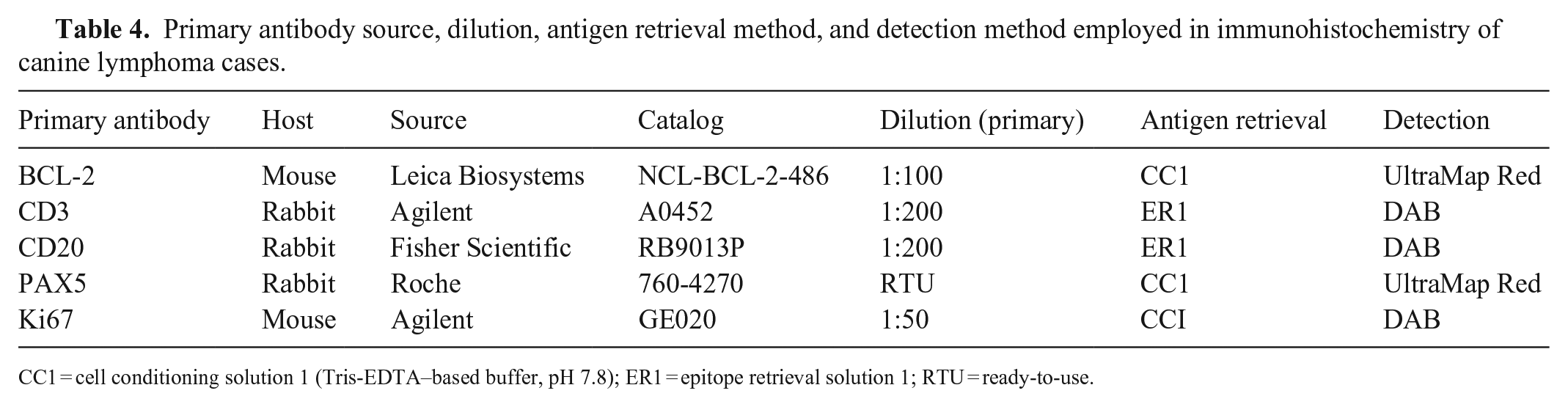
CC1 = cell conditioning solution 1 (Tris-EDTA–based buffer, pH 7.8); ER1 = epitope retrieval solution 1; RTU = ready-to-use.
Ki67 quantitation
Nuclear Ki67 positivity was quantified using the QuPath (v.0.4.3) positive cell detection feature. 1 Positive cells were determined using the optical density sum and standard settings assuming default DAB and hematoxylin staining parameters.
Statistical analysis
Fisher exact tests were performed in Prism v.9.1.2 (GraphPad) between groups for comparisons involving positive or negative variables.
Results
Antigen receptor clonality in CD3+/CD20+ LCL
To ascertain antigen receptor clonality for each case, we performed PARR using primers specific to TCRγ and IgH genes (Fig. 2). Monoclonal TCRγ rearrangements occurred in all CD3+/CD20+ cases. Three of 10 cases also had rearrangements in the IgH, supportive of cross-lineage rearrangement (Table 5). Interestingly, 5 cases of PTCL and 2 cases of DLBCL also had cross-lineage rearrangement. CD3+/CD20+ LCLs had TCRγ rearrangement more frequently and IgH rearrangement less frequently than DLBCL cases (
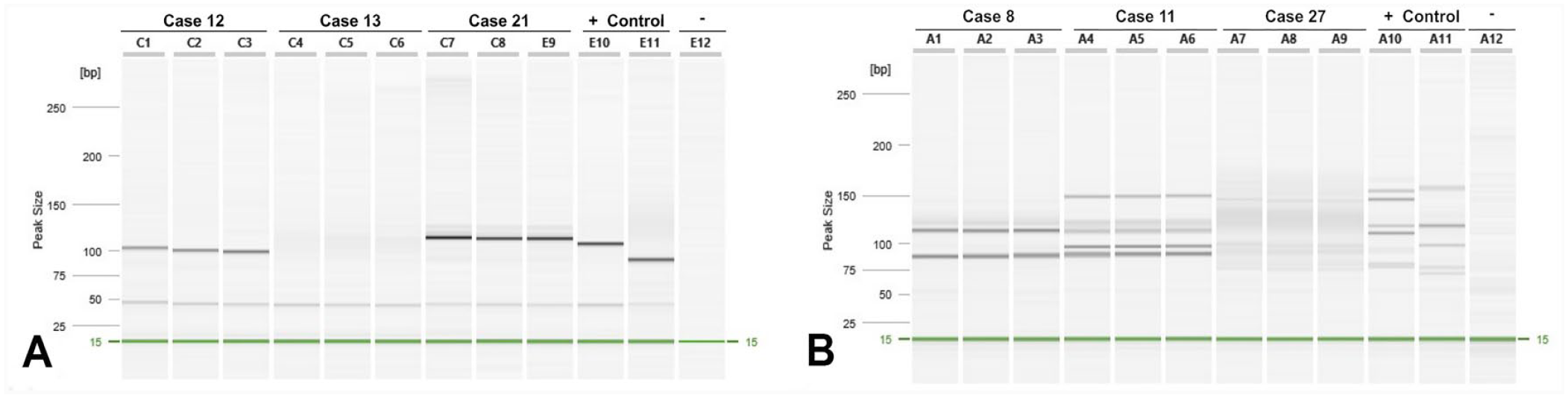
Antigen receptor clonality in CD3+/CD20+ canine large-cell lymphoma.
Frequency of antigen receptor rearrangements in CD3+/CD20+ large-cell lymphoma (LCL), diffuse large B-cell lymphoma (DLBCL), and peripheral large T-cell lymphoma (PTCL) cases.

Neither immunoglobulin heavy chain (IgH) nor T-cell receptor gamma (TCRγ) were amplified for one case of PTCL.
Summary of Fisher exact tests comparing CD3+/CD20+ large-cell lymphoma (LCL), diffuse large B-cell lymphoma (DLBCL ), and peripheral large T-cell lymphoma (PTCL) for TCRγ or IgH rearrangements (*

IgH = immunoglobulin heavy chain; TCRγ = T-cell receptor gamma.
Immunophenotyping of CD3+/CD20+ LCL
To further characterize our CD3+/CD20+ LCL cases, we performed IHC for PAX5 and BCL-2 (Fig. 3). PAX5 is a nuclear transcription factor that regulates the differentiation and development of B cells and is, therefore, used commonly as a B-cell marker.
29
In all CD3+/CD20+ cases, we observed negative PAX5 immunolabeling. This was unlike our DLBCL group, which immunolabeled for PAX5 in 90% of cases. PAX5 was only positive in 10% of PTCL cases. We observed no statistical difference in the PAX5 immunolabeling between CD3+/CD20+ LCLs and PTCLs (
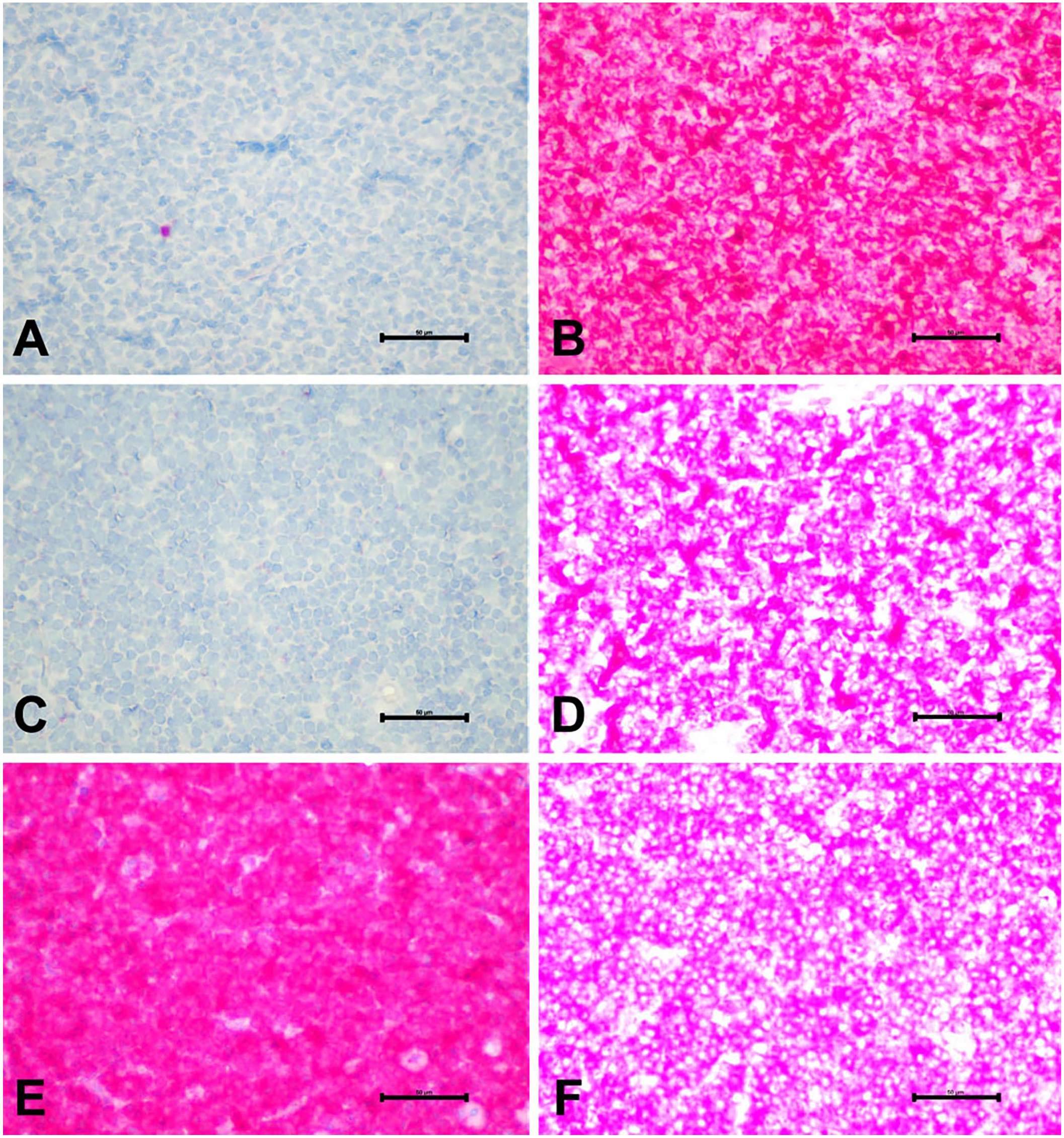
Expression of PAX5 and BCL-2 in CD3+/CD20+ canine large-cell lymphoma (LCL). Representative images of immunolabeling of PAX5 and BCL-2. Case 10, CD3+/CD20+ LCL:
Summary of Fisher exact tests comparing PAX5 or BCL-2 immunolabeling in CD3+/CD20+ large-cell lymphoma (LCL), diffuse large B-cell lymphoma (DLBCL), or peripheral large T-cell lymphoma (PTCL) where indicated (*
Next, we immunolabeled for BCL-2, an anti-apoptotic protein commonly overexpressed and therapeutically targeted in numerous cancer types.
7
BCL-2 has also been reported to be expressed ubiquitously in canine DLBCLs.
6
Moreover, a high BCL-2:BAX ratio has been associated more commonly with T-cell lymphomas, which may explain the poor clinical outcome in T-cell lymphomas.
13
Robust immunolabeling for BCL-2 occurred in all cases, and no difference was observed among groups (
High Ki67 positivity has been associated with a shorter survival time in dogs with DLBCL treated with chemotherapy.
23
Differences in proliferation markers, such as Ki67, and the apoptotic activity of the neoplastic cells, can account for variations in the proliferative activity of tumors and subsequent clinical outcome.
21
Therefore, we were interested in the Ki67 immunolabeling index of our CD3+/CD20+ LCL cases (Fig. 4A–D). Nuclear positivity was variable in both the PTCL and CD3+/CD20+ subsets (range: 4–99%), suggesting variability in the proliferation of these tumors. However, the average Ki67 immunolabeling index for the CD3+/CD20+ group was significantly different and higher than that of the DLBCL group (
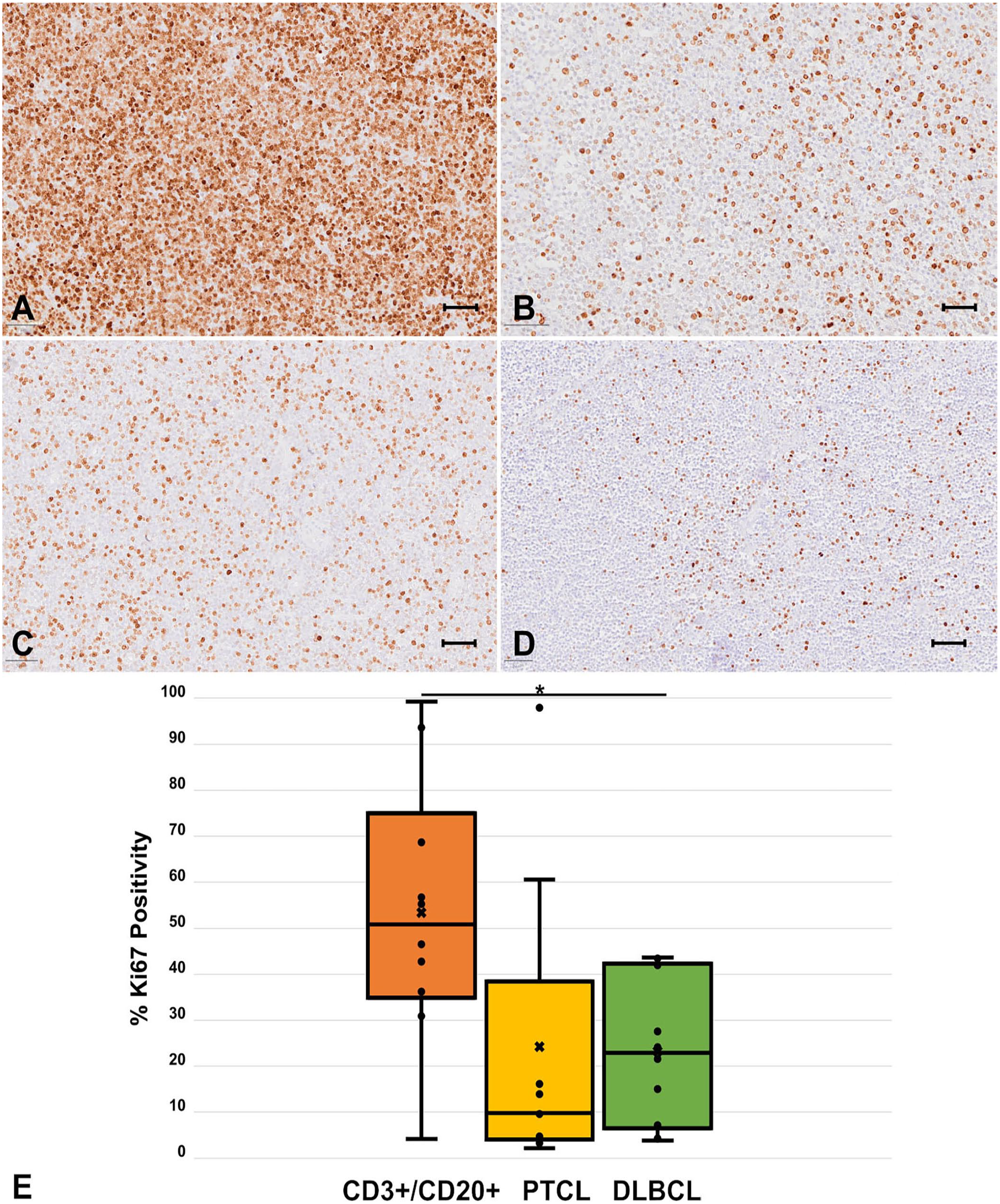
Ki67 immunolabeling index in CD3+/CD20+ canine large-cell lymphoma (LCL). Representative images of immunolabeling of Ki67 from CD3+/CD20+ lymphoma:
Average Ki67 index and variation in CD3+/CD20+ large-cell lymphoma (LCL), diffuse large B-cell lymphoma (DLBCL), and peripheral large T-cell lymphoma (PTCL).

Discussion
The frequency with which the TCRγ and IgH were rearranged differed statistically between CD3+/CD20+ LCL cases and DLBCL cases. However, there was no statistical difference in antigen receptor rearrangement frequency between CD3+/CD20+ LCL cases and PTCL cases. Furthermore, PAX5 immunolabeling patterns were similar between PTCL and CD3+/CD20+ cases but differed statistically between DLBCL and CD3+/CD20+ cases. The Ki67 immunolabeling index was highly variable within all groups, but a significant difference was noted between the CD3+/CD20+ LCL and DLBCL groups. Overall, our data highlight similarities between PTCL and CD3+/CD20+ LCL, suggesting that CD3+/CD20+ LCL cases have histogenesis similar to PTCL.
Cross-lineage rearrangement occurred frequently in our study: in 7 of our control cases and 3 CD3+/CD20+ LCL cases. V(D)J recombination in both the T-cell receptor and IgH genes has been reported in dogs, 16 although with a lower frequency than we observed. However, the true frequency with which cross-lineage rearrangement occurs in canine lymphoma is not known, given that a large-scale study has not yet been reported. Additionally, PARR is often assessed in only one antigen receptor for many diagnostic cases, further hindering our ability to retrospectively assess the frequency with which this phenomenon occurs. Other investigators have developed an improved primer set for canine PARR. 8 The sensitivity of the IgH primer set that we used is reported to be ~70%, which should be considered when interpreting our results. 8 Overall, the frequency of cross-lineage rearrangement, in addition to occasional aberrant expression of PAX5 in PTCL, bring into question the accuracy and adequacy of immunophenotyping with a single surface marker for canine lymphoma cases.
Inhibitors of the anti-apoptotic protein BCL-2 are under investigation in a wide variety of human cancers and have shown success in some forms of non-Hodgkin lymphoma. 22 BCL-2 expression levels are not prognostic in determining a response to CHOP chemotherapy, but BH3 mimetics have had in vitro efficacy activity against canine T-cell neoplastic cells.6,10 We observed no differences among groups in the immunolabeling of the therapeutic target BCL-2 and noted that BCL-2 had robust expression regardless of lymphoma subtype. Our data support the exploration of BCL-2 inhibitors in canine LCL patients regardless of immunophenotype.
Limitations of our study include the small sample size and retrospective approach. Despite these shortcomings, we performed a robust statistical analysis, identifying clear similarities and differences among lymphoma subtypes. Future studies will be directed at determining prognostic differences among these patients and evaluating additional prognostic markers and therapeutic targets.
Footnotes
Acknowledgements
We thank all of the individuals in the MSU-VDL histology laboratory for their assistance with our project. Our work was presented as a student poster at the 2021 American College of Veterinary Pathologists Annual Meeting (Virtual: Oct 30–Nov 2, 2021).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Morris Animal Foundation Veterinary Student Scholar Award.
