Abstract
A 7-y-old mixed-breed male dog was presented with a history of generalized lymphadenopathy. Fine-needle aspirates of the enlarged peripheral lymph nodes were suggestive of lymphoma. Histologic examination of a retromandibular lymph node was suggestive of high-grade, medium large-cell lymphoma. Immunohistochemistry revealed concurrent expression of CD3 and CD20. The co-localization of the 2 antigens was confirmed by immunofluorescence. PCR for antigen receptor gene rearrangements (PARR) detected clonal rearrangements for both T-cell receptor gamma and B-cell receptor. The final diagnosis was CD3-CD20–positive anaplastic lymphoma with cross-lineage rearrangement.
A 7-y-old mixed-breed male dog was presented to the Clinica Veterinaria San Rocco (Arezzo, Italy) for generalized lymphadenopathy, mainly affecting retromandibular and prescapular lymph nodes. Physical examination revealed a normal body condition score and moderately to severely enlarged peripheral lymph nodes. No significant abnormalities were observed in a complete blood count apart from activated lymphocytes in blood smear examination. Serum biochemical analysis identified a mild increase in activities of alanine transaminase (ALT; 229 U/L, RI: 15–64 U/L) and alkaline phosphatase (ALP; 125 U/L, RI: 12–120 U/L), and increased total bilirubin concentration (6.67 µmol/L, RI: 1.37–5.13 µmol/L). Additionally, moderate increases were detected in activities in amylase (2,520 U/L, RI: 360–1100 U/L), lipase (1,315 U/L, RI: 23–527 U/L) and 1,2-o-dilauryl-rac-glycero-3-glutaric acid-(6′-methylresorufin) ester (DGGR) lipase (447 U/L, RI: 6–120 U/L).
Fine-needle aspirates obtained from 4 enlarged peripheral lymph nodes revealed a prevalent population of medium-to-large round cells, characterized by pale cytoplasm and irregular nuclei with fine chromatin and inconspicuous nucleoli. Numerous mitotic figures were observed. In addition, small mature lymphocytes, non-degenerate neutrophils, and a few macrophages and plasma cells were identified intermingled with the neoplastic cells. The cytologic features were suggestive of high-grade lymphoma of presumed T-cell phenotype.
To obtain a histologic diagnosis according to the WHO classification, 13 a retromandibular node was removed surgically. Histologically, a dense, highly infiltrative, nonencapsulated, moderately well-differentiated, round-cell neoplasm effaced the entire node. Neoplastic cells were intermediate to large-sized (1.5–3 times the size of erythrocytes) with distinct cell borders and moderate amounts of eosinophilic cytoplasm, and exhibited cleaved or oval-shaped nuclei with prominent indentations and granular or coarse chromatin. Nucleoli were rarely present, and anisocytosis and anisokaryosis were marked. A total of 95–105 mitoses per 2.37 mm2 (ten 400× fields) was counted, including occasional bizarre mitoses. Numerous individual neoplastic cells were apoptotic, and rare eosinophils and macrophages were observed (Fig. 1A, B). The capsule was thin, and the peripheral sinuses were compressed in association with neoplastic infiltration of the perinodal fat. Based on these observations, a diagnosis of high-grade medium large-cell lymphoma was given.
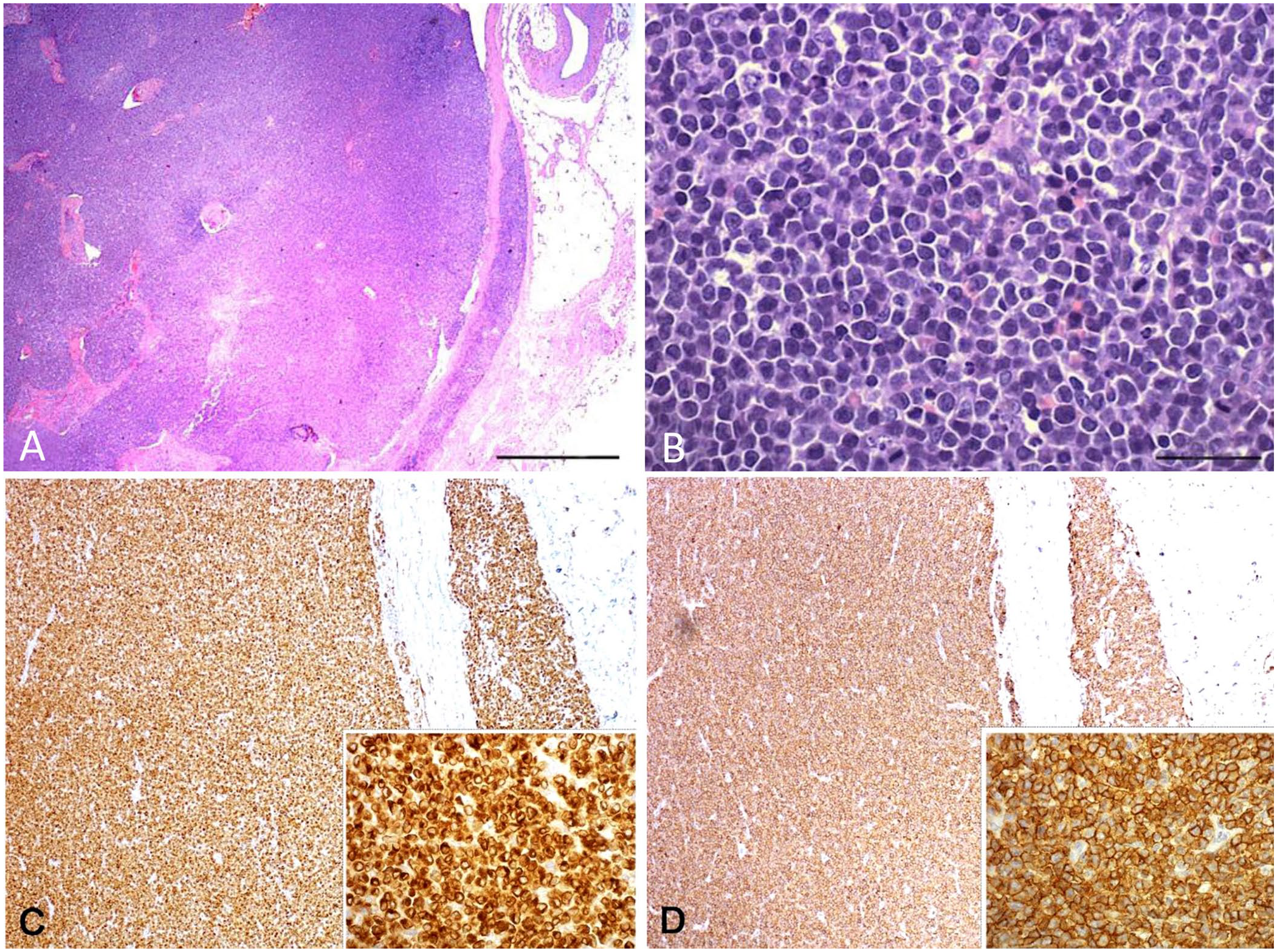
Nodal lymphoma in a dog.
A panel of 3 antibodies was used to phenotype the tumor, including anti-CD3, anti-CD20, and anti-Pax5. 1 Neoplastic cells had diffuse and intense cytoplasmic staining for CD3 (Fig. 1C) and membrane staining for CD20 (Fig. 1D); Pax5 staining was negative.
Given the apparent co-expression of the 2 markers in the neoplastic cells, double immunofluorescence staining was performed. Anti-CD3 (1:50) and anti-CD20 (1:400) antibodies were applied as a mixture overnight at 4°C. Alexa Fluor 488–conjugated goat anti-mouse IgG and Alexa Fluor 594–conjugated goat anti-rabbit IgG (1:500; Thermo Fisher Scientific) were selected as secondary antibodies and incubated for 60 min at room temperature. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (Vector Laboratories), and antigen localization was evaluated (AF6000 LX; Leica). CD3 and CD20 co-localization was confirmed in the neoplastic cells; positivity was detectable in the cytoplasm for CD3 and membrane for CD20 (Fig. 2).
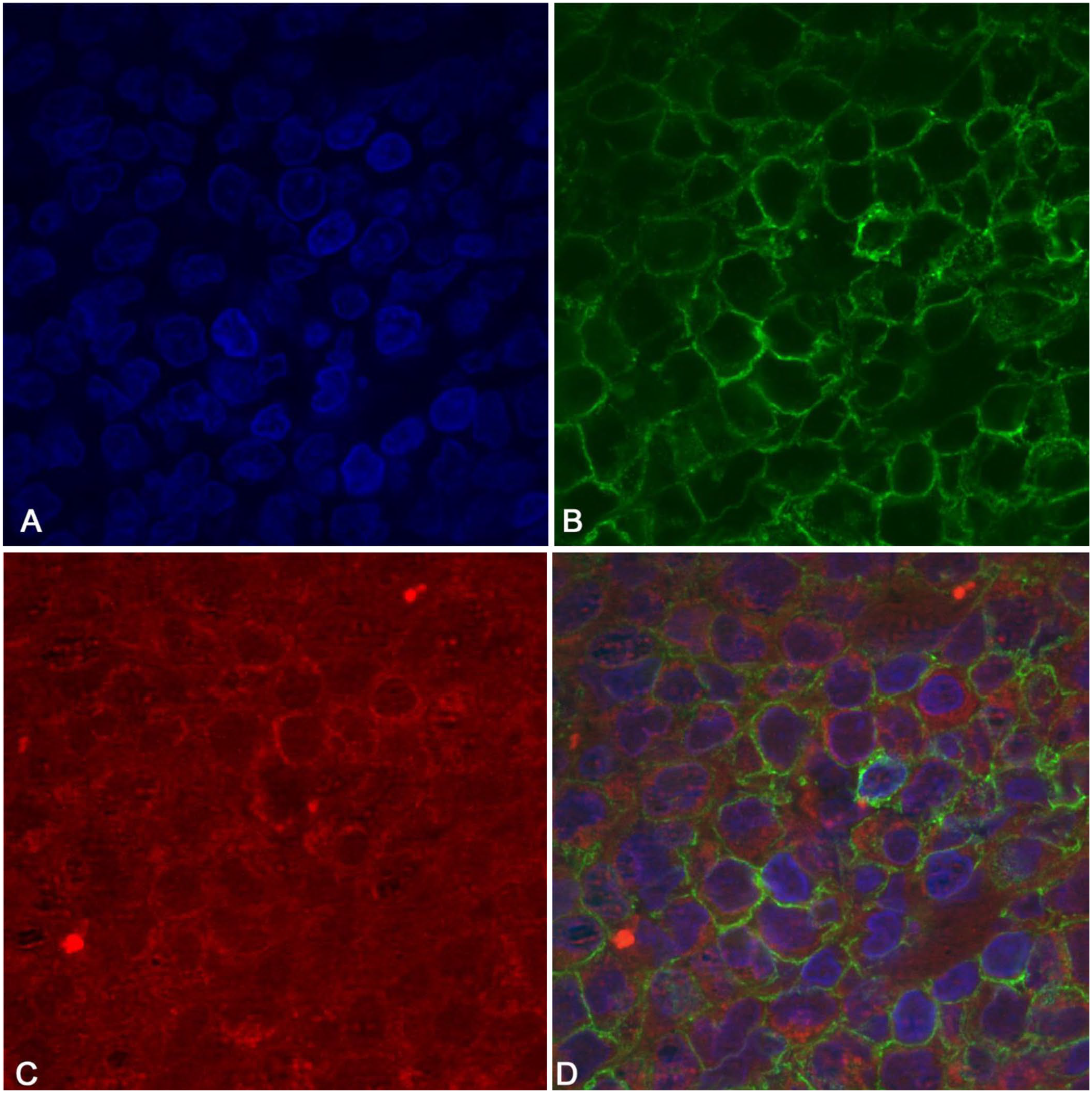
Nuclear counterstain by DAPI (
Given that immunohistochemical results did not reveal a definitive tumor phenotype, PCR for antigen receptor rearrangement (PARR) was proposed (Suppl. materials and methods). The clonality test of the lymphoid population detected clonal rearrangements of a similar amplitude of both T-cell receptor gamma locus (TCRγ) and B-cell receptor (BCR) major and minor genes (Suppl Fig. 1).
Based on histologic and immunohistochemical findings along with PARR results, the final diagnosis was CD3+ CD20+ anaplastic lymphoma. A few days after the lymphadenectomy, the dog was presented to the emergency clinic because of dysorexia, polyuria, and a mild hemorrhagic peritoneal effusion identified with ultrasound examination and abdominocentesis. The owner refused chemotherapy, and palliative therapy with prednisone (2 mg/kg q24 h PO) was prescribed. Three months later, the dog was in stable condition, and despite the owner-reported polyuria, polydipsia, and polyphagia, the daily dosage of prednisone was not reduced.
Co-expression of CD3 and CD20 is rarely observed in canine lymphoma and is described only in cutaneous epitheliotropic T-cell lymphomas 4 and intestinal T-cell lymphomas.7,9 Also, one dog has been reported with CD3-CD20 peripheral T-cell lymphoma infiltrating the heart and peripheral nerves. 8 All the described cases had a clonal TCRγ gene rearrangement assessed by PARR, and a diagnosis was given of T-cell lymphoma expressing aberrant CD20 immunophenotype. Conversely, B-cell lymphomas with aberrant CD3 expression have not been reported in dogs.
To further investigate the cell of origin, PARR was performed, but a definitive lineage was not assessed because concomitant TCRγ and BCR clonal peaks were detected. The WHO system of classification of canine lymphomas that relies on aspects such as topography, cell type, and immunophenotype does not include nodal lymphomas with CD3 and CD20 co-expression, therefore we could not arrive at a definitive diagnosis.
Constitutively, somatic V(D)J rearrangements occur in the premature development of lymphoid progenitors. Indeed, TCR gene rearrangement generates a diversity of T lymphocytes by V(D)J recombination; Ig genes are rearranged in B cells using the same enzyme machinery. Two hypotheses can explain the cross-lineage rearrangement in our case. The first supports a possible origin of this lymphoma from an immature subset of lymphocytes that harbor both aberrations.2,6 In human medicine, this deregulation is described in B-cell acute lymphoblastic leukemia (ALL), but the reasons for cross-lineage TCR rearrangements in malignant B precursors are unknown.5,12 Unfortunately, flow cytometry analysis of the lymph node, peripheral blood, and bone marrow was not performed in our case, therefore we could not definitively rule out ALL. However, blasts were not identified in blood smear examination, and the overall survival of the dog was much longer compared to the median overall survival of canine ALL. 3
The second hypothesis supports an unusual origin of this lymphoma from T cells expressing CD20. In humans, this subset refers to a small T-cell population accounting for 3–5% of total T lymphocytes. These cells transcribe both CD3 and CD20 and are mainly detected in peripheral blood and bone marrow. 10 However, no description of a similar subset is reported in veterinary medicine. Finally, biclonal rearrangements have been reported in dogs with acute myeloid leukemia (AML), but both the immunophenotype of the neoplastic cells and the absence of blasts in blood and cavity allowed us to exclude AML in our case. 11 Also, blasts were still not present at the last recheck 3 mo after the diagnosis.
Supplemental Material
Supplemental_material – Supplemental material for CD3-CD20–positive nodal lymphoma with cross-lineage rearrangement in a dog
Supplemental material, Supplemental_material for CD3-CD20–positive nodal lymphoma with cross-lineage rearrangement in a dog by Arturo Nicoletti, Luca Aresu, Michele Marino, Maria Massaro, Eugenio Martignani, Elisa Caporali, Silvia Capuccini, Ugo Bonfanti and Cecilia Gola in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
