Abstract
Canine minute virus (CnMV) is a member of the Bocavirus genus, together with Bovine parvovirus (BPV), which emerged as a new pathogen of dogs in 1967. The genus Bocavirus gained more recognition after the recent identification of Human bocavirus in pooled specimens from the respiratory tract of children. The cell culture host range of CnMV appears to be restricted to the Walter Reed canine cell (WRCC) line, although there is a report that indicates susceptibility of the Madin–Darby canine kidney (MDCK) cell line. In order to study the susceptibility of different cell lines to CnMV, the replication in cell cultures of canine, bovine, and human origin and the interaction of the virus with freshly isolated canine peripheral blood mononuclear cells were evaluated. The in vitro host range was unexpectedly wide. As shown by indirect fluorescent antibody and polymerase chain reaction assays, CnMV replicates efficiently in the A72 and MDCK canine cell lines. Bovine and human cell lines support the replication of CnMV less efficiently. In contrast, canine mononuclear blood cells are permissive to replication of CnMV in vitro. The present study revealed the ability of CnMV to replicate in continuous cell lines of different origin and, partially, in canine mononuclear cells.
Keywords
Canine minute virus (CnMV) and Bovine parvovirus (BPV) are members of the genus Bocavirus within the Parvoviridae family. The Bocavirus genus includes small, nonenveloped, autonomously replicating, single-stranded DNA viruses with an icosahedral capsid. Canine minute virus and BPV, whose molecular structure revealed a 43% sequence homology, differ from the other parvoviruses in the middle portion of the genome where they contain a third open reading frame encoding for NP1, a highly phosphorylated nonstructural protein of unknown function. A virus associated with lower respiratory tract infections in human beings was identified in 2005 and named Human bocavirus (HBoV) because of its similarity to BPV and CnMV. Phylogenetic analysis shows that HBoV groups in one clade with CnMV and BPV, sharing with these 2 animal bocaviruses an amino acid sequence identity of 44.1% and 42.6%, respectively. 14
Canine minute virus, isolated in 1967 from the feces of a healthy German Shepherd Dog in Germany, 2 causes disease of the gastrointestinal and respiratory tracts in young puppies and reproductive disorders in female dogs. The natural pathogenicity of CnMV for dogs is incompletely known because only a limited number of cases have been reported,6,10,12,15–17,20 although serological tests suggest that the virus is highly prevalent in the dog population.2,6,11,15,18,20
The dog is the only known host of CnMV, and the cell culture host range is believed to be highly restricted. The susceptibility of different cell lines to CnMV replication in vitro has not been systematically studied. The Walter Reed canine cell (WRCC) line has been reported to support the growth of CnMV. 2 A previous study 5 found limited replication in certain cell types of primary fetal canine lung, but not in cultures of canine fetal kidney or skin, and more recently, it was reported 15 that CnMV grew in Madin–Darby canine kidney (MDCK) cells. In order to study the susceptibility of cell cultures to infection with CnMV, the replication of CnMV in different cell lines of canine, bovine, and human origin was tested. Additionally, the susceptibility of freshly isolated canine peripheral blood mononuclear cells (PBMCs) was evaluated.
The CnMV strain (c/9871) used in the current study was originally isolated in Italy in 1998 from a puppy that developed a fatal CnMV infection. 17 The virus was cultivated in WRCC cell line a grown in Dulbecco minimal essential medium (DMEM) supplemented with 10% fetal calf serum (FCS). The stock virus used throughout the study was at the fourth passage and had an infectivity titer of 104.5 TCID50/50 µl.
The A72 canine fibroma cell line was established from a tumor surgically removed from a female 8-year-old Golden Retriever dog. 3 The MDCK cell line was derived from the kidney of a normal Cocker Spaniel. 9 The human rectal tumor (HRT-18) cell line was derived from human ileocecal colorectal adenocarcinoma, 19 while the Madin–Darby bovine kidney (MDBK) cell line was derived from the kidney of an apparently normal adult steer. 13
The donors for monocytes were healthy dogs that tested negative both for anti–CnMV-specific antibodies by an indirect fluorescent antibody test (IFAT) 18 and for CnMV antigen by virus isolation on WRCC and by polymerase chain reaction (PCR). 7 Unless indicated otherwise, each experiment was performed with freshly prepared monocytes from a single donor. For the isolation of monocytes, 5 ml of venous blood were collected into heparinized glass tubes. Peripheral blood mononuclear cells were isolated by centrifugation on density gradient. 4 Briefly, blood was diluted with an equal volume of medium (DMEM), layered over the gradient, b using a ratio of 2:1 (2 parts of diluted blood and one part of gradient), and centrifuged for 20 min at 800 × g at room temperature. The cells on the interphase of the gradient were collected, washed with DMEM to reduce the density of the solution, and centrifuged at 800 × g for 10 min to pellet the PBMCs. The cells were adjusted to a concentration of approximately 1 × 106 cells/ml in DMEM containing 15% (v/v) FCS and dispensed into 6-well culture plates (3 ml/well) containing sterile round glass coverslips. According to the manufacturer, more than 90% of the cells should be monocytes, and contamination with lymphoid cells should range between 2% and 13%. Monocytes convert to macrophages after a period in culture medium. In the present study, the adherent cells are referred to as monocytes, with the understanding that these cell preparations are contaminated to some degree by lymphoid cells.
On the day monocytes were harvested, each well of cells was incubated for 45 min at 37°C with the stock virus at a concentration of 100 TCID50/50 μl. The cells were washed twice, and then fresh DMEM supplemented with 10% FCS and antibiotics was added. Mock infected monocytes were used as negative controls.
Attempts to isolate CnMV were performed following concanavalin A (conA) stimulation, considering that conA is able to induce DNA and RNA synthesis in cultured monocytes. Each well of the culture plate was incubated with 5 µg/ml of conA for 24 hr and then infected with 100 TCID50/50 μl of the virus.
One hundred TCID50/50 μl of stock virus was inoculated onto A72, MDCK, HRT-18, and MDBK cells in plastic cell culture flasks immediately after trypsinization and incubated at 37°C in a 5% CO2 incubator. The virus was also inoculated onto WRCC cultures as a positive control, and mock infected WRCCs were used as negative controls. CnMV-infected cells and mock infected cells were used for various assays.
Monocytes and cell cultures were observed daily. After 3 days of incubation, the cells were frozen and thawed 3 times, and the lysates were used for subsequent passages until the 10th passage. Virus growth in infected and uninfected monocytes and in A72, MDCK, HRT-18, MDBK, and WRCC cell lines was monitored by PCR and by IFAT using a CnMV-positive dog serum, a as described previously. 17 Additionally, virus titers were determined by IFAT.
Cell culture titration of c/9871 on monocytes and on A72, MDCK, HRT-18, and MDBK cell lines was performed in triplicate. Data were evaluated using a spreadsheet program, c and standard deviations were calculated. In order to determine the effective susceptibility of monocytes and the 4 cell lines employed to CnMV replication, control samples were tested by PCR, and the end point at which the virus is no longer detected from culture media containing virus without cells was determined. The test verified the titer at which the virus is detectable in the absence of replicating cells.
The growth curve of CnMV was determined on monocytes cultures, with WRCC as a control. One milliliter of virus suspension containing 100 TCID50/50 μl was added to the monocytes pellets. After 30 min incubation at 37°C, the cells were suspended (2 × 107 cells/ml) in DMEM and distributed in 12-well plates (1.5 ml/well). Immediately after distribution (time 0), the supernatant fluid from 1 well was collected and stored at −70°C, and the plate was incubated at 37°C in 5% CO2. After 3, 6, 9, 12, 24, 36, and 48 hr of incubation, and at each subsequent interval of 24 hr until 144 hr, the supernatant fluids from individual wells were separately collected and stored at −70°C. Each sample was then titrated in WRCC by means of PCR. Data were evaluated using the spreadsheet program, c and standard deviations were calculated.
Virus titrations were performed in 96-well microtiter plates. Ten-fold dilutions of virus were prepared in quadruplicate in DMEM and mixed with 50 µl of a suspension containing 200,000 WRCC. Plates were incubated at 37°C for 5 days in a humidified CO2 atmosphere and then examined by IFAT.
Conventional PCR was performed using DNA polymerase d and primer pair 268/276, which amplifies a fragment of the VP2 protein-encoding gene of CnMV. 7 The primers 268 (5’-ACGGTGGTTGTCCAGACG-3’) and 276 (5’-GGCATT CCTTCCAGGAAC-3’) are located at nt position 3933-3950 and 4946-5013 of the CnMV genome, respectively, and yield a 1,064-bp product. Viral DNA was extracted using a commercial kit e according to manufacturer’s instructions.
The canine monocytes inoculated with CnMV showed both nuclear and cytoplasmic fluorescence 48 hr postinfection, whereas the un-inoculated monocytes did not. The cultured monocytes gave positive results until the fifth passage when tested by IFAT and until the eighth passage when tested by PCR. Monocytes cultures were tested for infectivity at various postinfection intervals, and CnMV growth was determined. CnMV growth was also measured on WRCC (Fig. 1). Viral titers dropped significantly 6 hr postinfection. Starting from 9 hr postinfection, the cultured cells showed an increase in virus titer throughout the observation period, indicating that the monocytes supported the replication of CnMV. Monocytes cultures after conA treatment had a similar pattern of viral susceptibility to that seen for monocytes cultured without stimulation, and viral antigens were detected from CnMV-inoculated monocytes until the sixth passage by means of IFAT (data not shown).
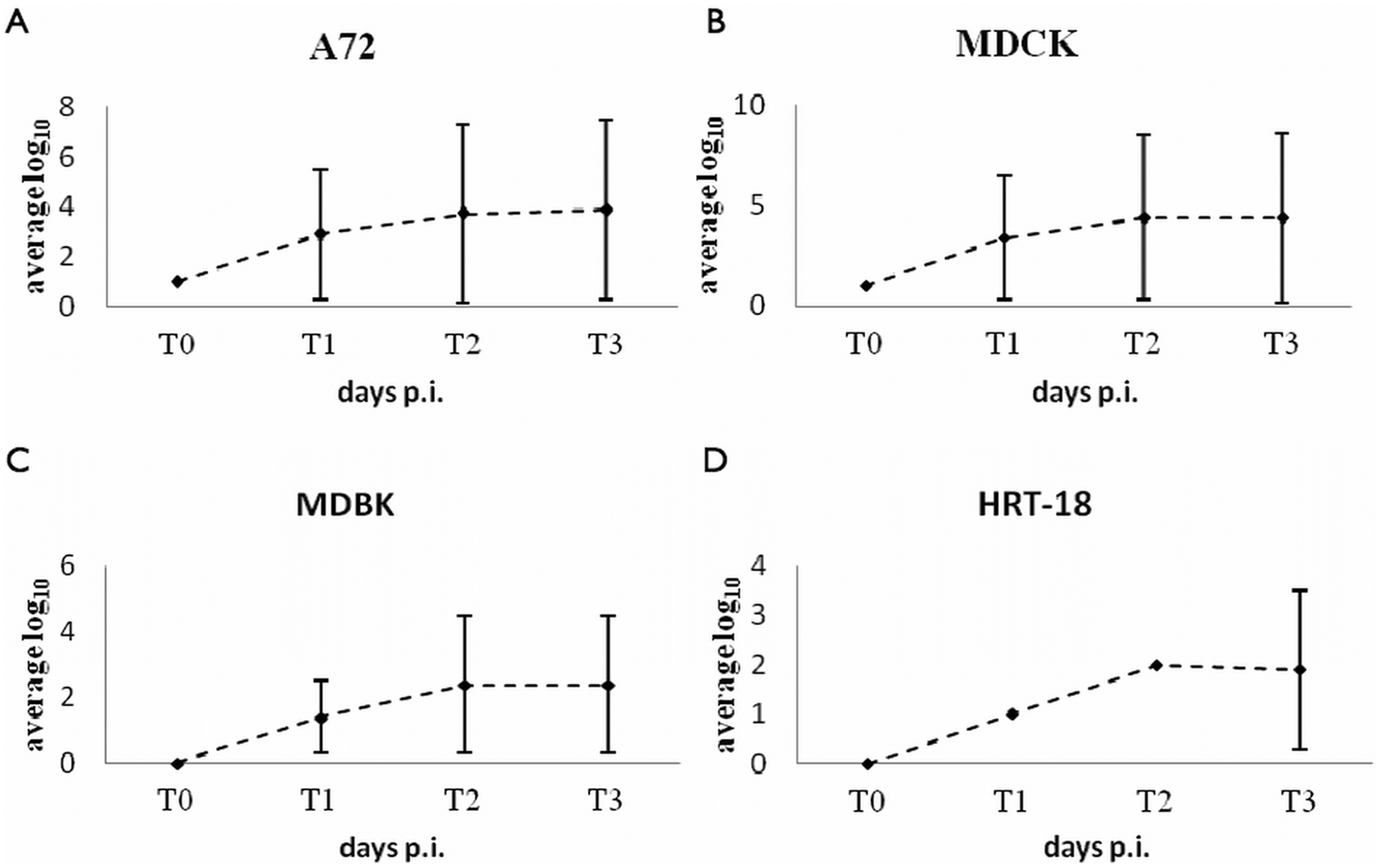
Replication of Canine minute virus (CnMV) in A72 (
When infected cultures of A72, MDCK, MDBK, and HRT-18 were assayed by IFAT and PCR, CnMV was shown to replicate efficiently in canine cell lines A72 and MDCK to similar titers (Fig. 2). DNA could be demonstrated in extracts of canine cells collected 2 days after inoculation until passage 10. The amount of infectious virus as determined by IFAT and PCR in MDBK and HRT-18 cells was lower than in A72 and MDCK cells. Although the data suggest that both these cell lines supported replication of CnMV, growth in human cells was less efficient than in bovine cell line. A faint DNA band was seen in extracts of CnMV-inoculated HRT-18 cells, at the 10th passage, indicating a very low level of DNA replication. Control samples tested by PCR to verify the presence of the virus in the absence of replicating cells revealed that the virus was no longer detected after dilution 10−6.
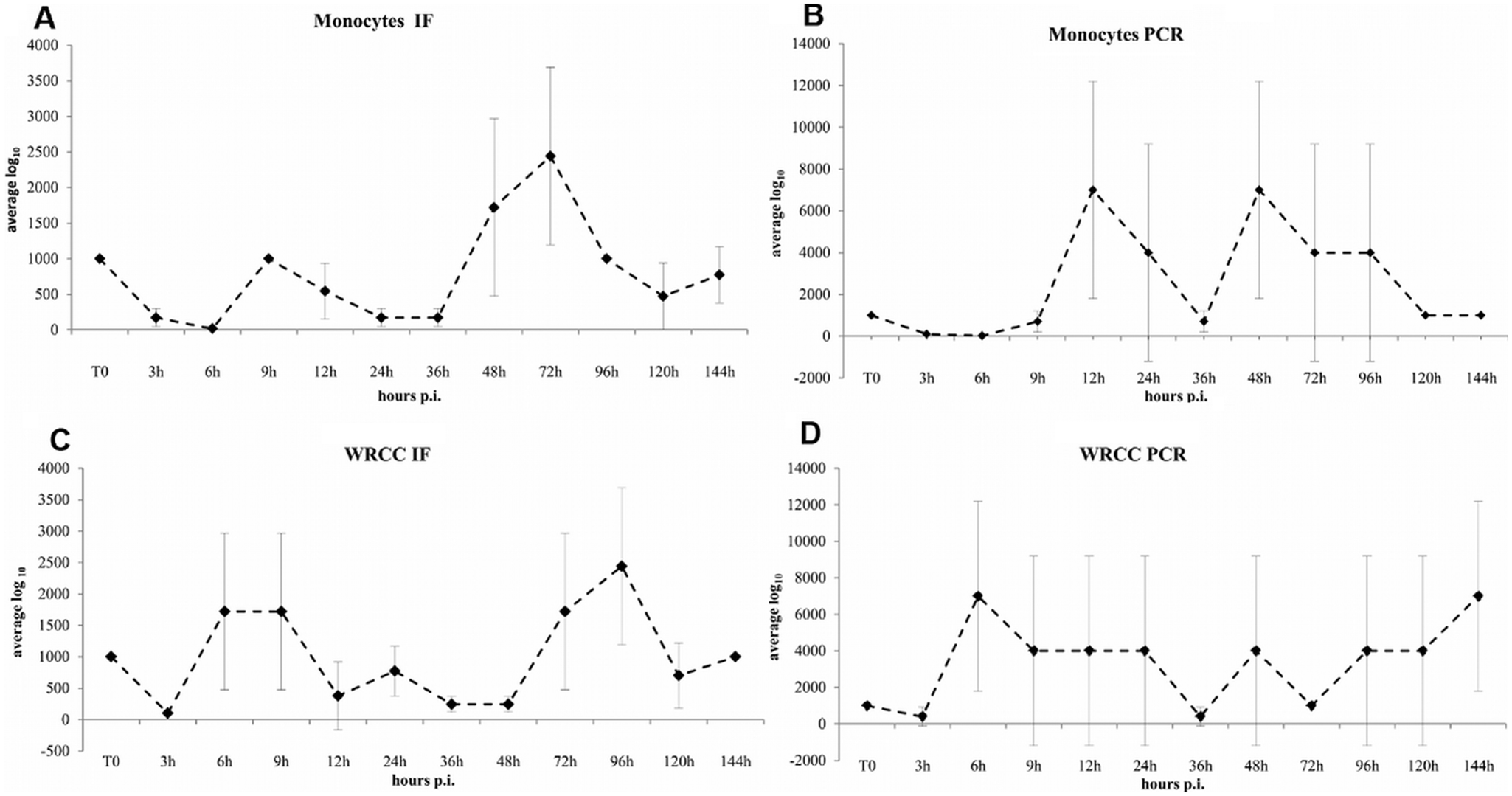
Growth of Canine minute virus (CnMV) in monocytes (
To the authors’ knowledge, there are no previous reports concerning the ability of CnMV to replicate into canine mononuclear cells, and little is known about the in vitro host range of the virus. Historically, WRCC was considered the only cell line susceptible to this virus, although a previous study 15 suggested that CnMV replicates to some degree in MDCK cells. Detailed knowledge of the host range of CnMV is important for understanding the evolution of this bocavirus, and the present study was undertaken to obtain data on the ability of CnMV to replicate in canine mononuclear blood cells as well as in continuous cell lines of canine, bovine, and human origin. The study was performed with a CnMV strain that was isolated from dogs in Italy. Whether different strains might have resulted in different findings was not determined.
The pathogenesis of CnMV has not been studied extensively. In infected puppies, lymph nodes, thymus, and spleen show evident signs of inflammation with edema, necrosis, and infiltration of inflammatory cells. A previous study hypothesized that CnMV may cause the impairment of phagocytosis and the killing of polymorphonuclear and monocytes cells. 7 That hypothesis is supported by the observation in the present study that the monocytes cultures are permissive to CnMV replication in vitro, albeit to a lower degree than permissive canine continuous cell lines.
As parvoviruses infect only actively dividing host cells, it was hypothesized that conA stimulation would enhance CnMV replication in monocytes cultures. The fact that conA stimulation did not enhance monocytes susceptibility to CnMV replication makes it difficult to understand the mechanism of the observed partial virus growth monocytes cultures. A possible explanation might be that cultured PBMCs undergo alterations such that they resemble mature macrophages, 8 which are not expected to be responsive to conA stimulation. However, cytological and biochemical studies have shown that monocytes, when cultivated in vitro, underwent a sequence of morphological and biochemical alterations, accompanied by an increase in the activity of lysosomal enzymes. 8 It is possible that cell-cultured monocytes somehow lose the ability to efficiently support CnMV replication. Another possible explanation is that interference may occur during in vitro replication in cultured monocytes by the generation of defective interfering particles, which went undetected by the methods employed in the current study.
The results of the present study confirm and extend a previous report 15 demonstrating the replication of CnMV to comparable degrees in different canine and in certain bovine and human cell lines. Further experiments are needed to determine whether canine monocytes can better support a productive infection under different conditions than used in the present study.
Footnotes
Acknowledgements
The authors are grateful to the researchers of the Department of Public Health and Animal Sciences—section “Infectious Diseases”—whose collaboration has been essential for the preparation of the present study.
a.
Professor L. E. Carmichael, Cornell University, Ithaca, NY.
b.
Lympholyte-H, Cedarlane, Burlington, Ontario, Canada.
c.
Microsoft Excel, Microsoft Corp., Redmond, WA.
d.
AmpliTaq Gold, Applied Biosystems, Applera Italia, Monza, Italy.
e.
QIAamp, Qiagen GmbH, Hilden, Germany.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declares that they received no financial support for their research and/or authorship of this article.
