Abstract
CNS lesions associated with chronic otitis have not been reported in red kangaroos (Macropus rufus), to our knowledge. Here we describe an intracranial inflammatory polyp secondary to chronic otitis in a 6-y-old female red kangaroo with right auricular discharge, loss of balance, and head tilt. Autopsy highlighted a pale-yellow, firm, intracranial polypoid growth that extended from the right tympanic cavity through the internal acoustic meatus and intracranially, with compression of the right cerebellopontine angle. Anaerobic bacterial culture yielded Bacteroides pyogenes from fresh brain and a right external ear swab. Histologically, the tympanic cavity was effaced by neutrophils and macrophages surrounded by lymphocytes and plasma cells, as well as edematous fibrovascular tissue. The epithelial lining of the mucoperiosteum was hyperplastic, with epithelial pseudoglands surrounded by fibrovascular tissue. Areas of temporal bone lysis and remodeling were associated with the inflammatory changes, which occasionally surrounded adjacent nerves. Fibrovascular tissue and inflammatory cells extended from the tympanic cavity through the internal acoustic meatus and into the intracranial cavity, forming the polypoid growth observed grossly; the polyp consisted of a dense core of fibrovascular tissue with scattered clusters of neutrophils and foamy macrophages. Lymphocytes and plasma cells surrounded the leptomeningeal perivascular spaces in the brainstem, cerebellum, and occipital lobe.
Pathologic processes and infectious organisms enter the CNS via hematogenous spread, leukocyte trafficking, retrograde axonal transport through cranial nerves, or direct extension from lesions within adjacent tissues. 6 CNS lesions caused by direct extension from adjacent areas are typically associated with penetrating physical trauma to the skull or vertebrae (including gunshot wounds, dehorning in cattle and small ruminants, and tail docking) or inflammatory and neoplastic disease affecting the skull bones, nasal cavity, paranasal sinuses, middle or inner ear, or vertebrae. 6
Otitis media is a common complication of otitis externa in dogs but is uncommon in other animal species.11,12 Increased pressure caused by inflammation in the tympanic cavity can rarely lead to rupture of the tympanic membrane, with extension of exudates to the inner ear via direct migration through the eroded petrous portion of the temporal bone, round (cochlear) window membrane, oval (vestibular) window, or less likely, facial canal foramen. In addition, inflammatory infiltrates can extend into the intracranial cavity and brain. 7 Meningoencephalitis associated with middle or inner ear infections have been described infrequently in dogs and cats, 11 small ruminants, 1 cattle, 13 and New World camelids. 3 In macropods, suppurative meningoencephalitis secondary to chronic otitis has been described in a wallaby. 4 On a search of PubMed, CAB Direct, Web of Science, Scopus, and Google, we found one case of chronic otitis with secondary osteomyelitis in a red kangaroo, with no gross or histologic lesions in the CNS. 8
Here we describe an intracranial inflammatory polyp with cerebellopontine compression and leptomeningitis secondary to chronic otitis in a 6-y-old female red kangaroo (Macropus rufus) with a 3-mo history of right auricular discharge, progressive loss of balance, and head tilt to the right. Although the right ear discharge resolved after treatment with antibiotics, the neurologic clinical signs persisted, and the patient was euthanized. No neurologic evaluation was conducted.
During autopsy, a pale-yellow, firm, 8-mm diameter, intracranial polypoid growth was discovered that extended from the right tympanic cavity through the internal acoustic meatus and intracranially onto the right occipital bone (adjacent to the occipital condyle), compressing the right cerebellopontine angle (Fig. 1). Cords of thick, tortuous blood vessels were observed along the floor of the cranial vault, extending from the right optic chiasm to the polypoid growth (likely representing regional neovascularization in response to the inflammatory process). No pathologic changes were observed in the other tissues. Routine tissue samples (including brain, right external ear, and tympanic cavity with part of the petrous portion of the temporal bone, heart, lung, liver, spleen, mesenteric lymph node, gastrointestinal tract, kidney, and adrenal glands) were fixed in 10% neutral-buffered formalin. The right tympanic cavity and petrous portion of the temporal bone were subsequently demineralized in Kristensen solution (1 N sodium formate and 8 N formic acid solution). All tissues were processed routinely for histology and stained with H&E. Fresh brain tissue samples and a swab from the right external ear canal were subjected to anaerobic bacterial culture.
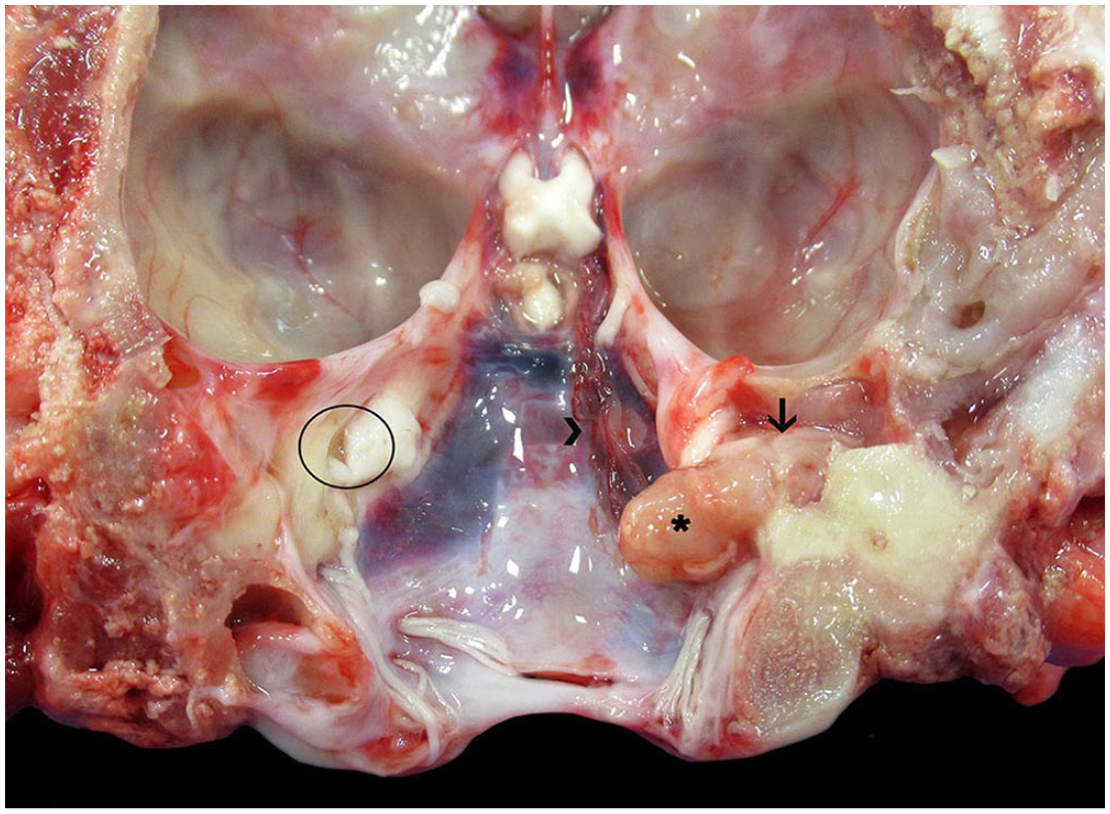
Intracranial inflammatory polyp with cerebellopontine compression secondary to chronic otitis in a kangaroo. A pale-yellow, firm, inflammatory polyp (asterisk) extends from the right tympanic cavity through the internal acoustic meatus (arrow) and onto the right occipital bone. Circle = left internal acoustic meatus. Cords of thick, tortuous blood vessels cover the dura mater along the right side of the cranial vault (arrowhead).
Histologically, the tympanic cavity was effaced by numerous neutrophils and macrophages surrounded by lymphocytes and plasma cells embedded in abundant edematous fibrovascular tissue that expanded the auricular mucoperiosteum (Fig. 2). There were extensive areas of hemorrhage and clusters of keratin flakes throughout. The epithelial lining of the mucoperiosteum was hyperplastic and often formed epithelial pseudoglands lined by goblet cells and ciliated columnar epithelial cells surrounded by fibrovascular tissue (Fig. 3). Areas of temporal bone lysis and remodeling were observed associated with the inflammatory changes, which occasionally surrounded adjacent nerves. No evidence of neuritis or nerve compression was observed. Fibrovascular tissue and inflammatory cells extended from the tympanic cavity into the internal acoustic meatus (Fig. 4) and into the cranial cavity, forming the polypoid projection observed grossly (Fig. 5). The polypoid growth consisted of a dense core of fibrovascular tissue with scattered clusters of neutrophils and foamy macrophages (Fig. 6). A moderate number of lymphocytes and plasma cells surrounded the leptomeningeal perivascular spaces in the brainstem, cerebellum, and occipital lobe (Fig. 7). The neuroparenchyma was not affected by the inflammation. Gram stain did not highlight any organisms. Anaerobic bacterial culture yielded light growth of Bacteroides pyogenes from both tissue samples. No histologic lesions were observed in the other tissues sampled.
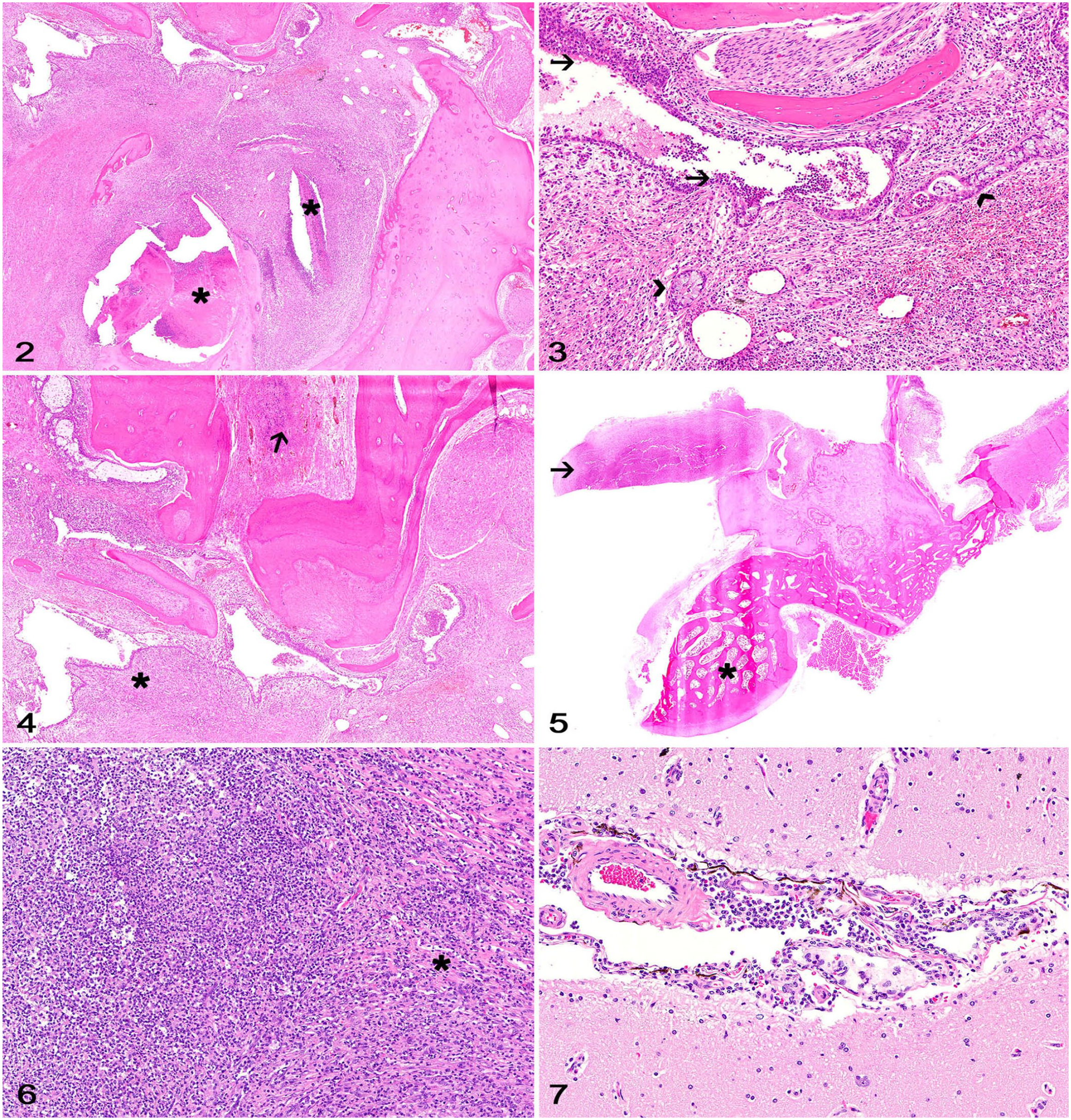
Intracranial inflammatory polyp with cerebellopontine compression secondary to chronic otitis in a kangaroo.
Pathologic changes were consistent with chronic otitis externa and media affecting the right tympanic cavity, with extension of the inflammatory infiltrates and fibrous connective tissue through the right internal acoustic meatus and into the cranial cavity. Given that the inner ear compartments could not be identified definitively on histology, the presence of otitis interna could not be confirmed. Bacterial otitis media is rare in macropods, and has been associated with B. tectus and Porphyromonas gulae infection in a captive Parma wallaby (Macropus parma) and with Klebsiella pneumoniae and Pasteurella multocida infection in a red kangaroo.4,8 Similarly, anaerobic bacterial culture of fresh brain and external ear swab samples in our case yielded light growth of B. pyogenes. Bacteroides spp. are opportunistic gram-negative anaerobic bacilli that have also been associated with chronic otitis, brain abscesses, 15 oral disease, 2 and cat or dog bite wounds in humans, 14 oral disease in cats and horses, 2 subcutaneous abscesses and pyothorax in cats, 5 and mandibular and periodontal disease in macropods.9,10 No other lesions were observed in our case, and the potential primary source of the ear infection was not identified.
Neurologic disease associated with chronic otitis in macropods has been described in a Parma wallaby and a red kangaroo,4,8 but CNS lesions were present only in the wallaby. 4 Reported gross changes consisted of suppurative exudate within the left tympanic cavity with extension to the adjacent meninges and neuroparenchyma of the caudal brainstem and medulla oblongata. 4 The neuroanatomic localization of the lesions in our case and the wallaby likely reflects the proximity of these affected CNS structures to the middle and internal ear. 4 Although there was compression of the cerebellopontine angle by the polypoid growth, with extension of the inflammation to the leptomeninges in the brainstem, cerebellum, and occipital lobe, no invasion into the neuroparenchyma was observed grossly or histologically in our kangaroo. Histologically, our findings were similar to those described in a case of chronic otitis in a red kangaroo. 8
The polyp observed in our case arose from the chronic inflammatory lesions within the tympanic cavity and extended to the intracranial cavity via the internal acoustic meatus, a canal within the petrous portion of the temporal bone that allows the passage of the vestibulocochlear nerve, facial nerve, and blood vessels from the brainstem to the inner ear and face. 12 As evidenced by the lesions in our case, neural compromise caused by the adjacent inflammation and/or by the space-occupying lesion could explain the vestibular signs developed by affected individuals even in the absence of CNS lesions. 8 The vestibular signs could be also attributed to extension of the inflammation into the compartments of the internal ear (cochlea, utriculus, sacculus, semicircular canals), but that could not be confirmed histologically.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
