Abstract
The effects of filter paper strip (FPS) storage time and temperature on antibody detection are poorly understood despite widespread use in wildlife research. We collected sera and FPSs from 23 wild turkeys (Meleagris gallopavo) and 20 northern bobwhites (Colinus virginianus) experimentally infected with West Nile virus (WNV) to compare FPS storage methods with WNV plaque reduction neutralization test (PRNT) sensitivity. FPS storage methods included: immediate elution after drying, and storage at −20°C for 3 mo, −20°C for 6 mo, room temperature (RT) for 3 mo, and RT for 6 mo prior to elution. FPS eluates and sera were co-titrated to determine endpoint antibody titers, which were compared between FPS sera eluted immediately and sera, and among FPS eluates that underwent different storage conditions. Antibody titers were ~4-fold less in FPS sera eluted immediately versus sera, and dropped more frequently below PRNT detection threshold in northern bobwhites, which had ~10-fold lower serum antibody titers than wild turkeys. Antibody titers were lower in FPS samples stored at RT and for 6 mo. WNV serologic surveys may result in falsely low seroprevalence estimates if FPSs are stored at RT for ≥ 3 mo before elution.
Collection of blood samples for pathogen surveillance in free-ranging wildlife poses logistic challenges, including the need for highly trained personnel, maintenance of sample viability including centrifugation and refrigeration or freezing in remote locations, and limited blood volume collection in small-bodied animals. Blood sample collection utilizing Nobuto filter paper strips (FPSs; Toyo Roshi Kaisha) is often used instead of blood collection tubes in wildlife pathogen surveillance because FPS sample collection and storage logistics allow for expanded sampling opportunities, particularly in hunter-harvested carcasses.8,15,19
Despite widespread use of FPSs in game bird serosurveys, there is a paucity of research evaluating the effects of storage duration and method on the ability to detect antibodies in Galliformes, particularly for West Nile virus (WNV; Flaviviridae, Flavivirus). Although FPS manufacturers claim that long-term storage at ambient temperature does not affect test results, 1 researchers have determined that, in some situations, delayed sampling after harvest and other variation in sample collection and storage prior to testing may affect sample quality and test sensitivity.3,8 Thus, the use of FPSs may result in falsely low antibody titers compared to serum samples collected simultaneously, and this difference may be sufficient to fail to detect antibodies, resulting in false-negative results. The utility of FPSs in wildlife disease research needs to be validated for each pathogen, assay, and species of interest. 6
We aimed to better understand the utility of FPSs in WNV serosurveys by determining the efficacy of blood collected onto FPSs compared with gold standard sera, using samples collected from wild turkeys (Meleagris gallopavo) and northern bobwhites (Colinus virginianus) experimentally inoculated with WNV. We determined if various FPS storage conditions before elution, corresponding to typical FPS storage conditions after collection, influence the ability to detect anti-WNV antibodies in these 2 galliform species.
We collected blood samples from the jugular vein of 23 wild turkeys and 20 northern bobwhites, which had been inoculated subcutaneously with 106 plaque-forming units (PFU) of WNV, at 14 and 15 d post–WNV inoculation (i.e., known seropositive birds), respectively.10,11 Blood was placed into serum separator tubes, and the absorption area of 10 Nobuto FPSs per bird was completely saturated with 100 µL of blood via syringe. FPSs air-dried overnight at room temperature (RT) 1 were then stored (2 FPSs per bird per storage condition) in individual, resealable plastic bags in the dark until elution. Serum separator tubes were centrifuged10,11 within 4 h of collection; sera were heat inactivated at 56°C for 30 min and frozen at −80°C until testing. Animal husbandry, experimentation, handling, and sampling were conducted under Institutional Animal Care and Use Committee approval (University of Georgia; protocol A2018 09-020-Y1-A0).
FPSs from each bird were divided haphazardly into 5 storage conditions: 1) FPSs in which the sera were eluted 1 d after collection (hereafter, referred to as eluted immediately); 2) FPSs stored at RT (19–20°C) for 3 mo; 3) FPSs stored at RT for 6 mo; 4) FPSs stored at −20°C for 3 mo; and 5) FPSs stored at −20°C for 6 mo before elution. The latter 4 conditions were representative of typical FPS storage conditions after sample collection and did not include desiccant packs.8,19 At the predetermined elution times, sera were eluted from FPSs, heat-inactivated, centrifuged, and stored at −20°C until testing.1,19 For each FPS eluate, the blood-saturated portions of 2 FPSs soaked in 800 µL of Dulbecco PBS (DPBS) solution (MilliporeSigma) for 1 h prior to FPS removal and heat inactivation. 1 Sera and FPS eluates were co-tested for neutralizing antibodies to WNV by a plaque reduction neutralization test (PRNT).2,10 The starting PRNT dilutions (and limit of detection [LOD]) for the sera and eluates were 1:10 and 1:20, respectively; samples were serially diluted 2-fold to determine endpoint antibody titers.10,11
We calculated PRNT sensitivity estimates and 95% Clopper–Pearson exact CIs for each storage condition relative to serum PRNT results for both species. For each species, antibody titers from serum and FPS serum eluted immediately were compared by linear mixed models or Tobit censored regression with robust SEs to account for repeated measurements on the same bird. Antibody titers of FPSs stored at RT and −20°C for 3 and 6 mo were compared by Tobit censored regression, which accounted for samples with antibody titers below the LOD (i.e., left-censored). Generalized estimating equation (GEE) binomial regression using robust SEs and an exchangeable working correlation structure was used to compare the proportion of FPSs stored at RT and −20°C for 3 and 6 mo that had detectable antibody titers. Antibody titers were log2-transformed for descriptive and statistical analyses. Antibody titer was the response variable; storage time and temperature were fixed categorical variables. To control for individual antibody titer variation, antibody titer from FPS serum eluted immediately was a covariate in the analyses that compared the 4 FPS storage conditions. Bird ID was a random effect for linear mixed models and a clustering variable for Tobit censored and GEE binomial regression. Post-hoc pairwise comparisons were performed using the Tukey procedure to limit the family-wise type I error probability to 5% over comparisons. Analyses were completed in R version 4.0.2, 17 with significance level α = 0.05.
Test sensitivity estimates for the wild turkey samples were > 90.0% for all storage conditions except for FPSs stored at RT for 6 mo. Conversely, test sensitivity estimates for all northern bobwhite FPS storage conditions were ≤ 70.0% (Table 1). Antibody titers from FPS sera eluted immediately were 2.0 log2 lower than those detected in paired sera in wild turkeys (95% CI: 1.5–2.6; p < 0.001) and were 2.2 log2 lower in northern bobwhites (95% CI: 1.5–2.9, p < 0.001; Table 2).
Plaque reduction neutralization test (PRNT) sensitivity estimates (%; relative to serum [i.e., gold standard] PRNT results) for the detection of antibodies to West Nile virus (WNV) and 95% Clopper–Pearson exact CIs for serum and all Nobuto filter paper strip (FPS) storage conditions (before elution) for samples collected at 14 or 15 d post-inoculation from experimentally WNV-inoculated wild turkeys (Meleagris gallopavo) and northern bobwhites (Colinus virginianus).
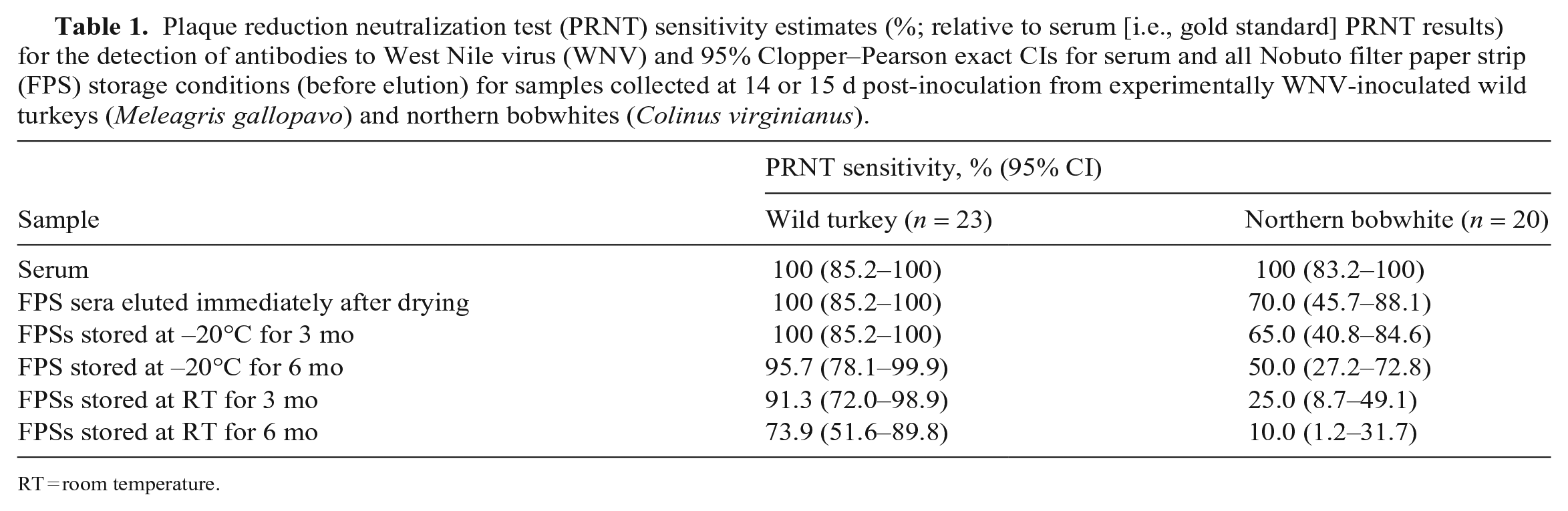
RT = room temperature.
Estimated marginal mean log2 anti–West Nile virus (WNV) antibody titers (via plaque reduction neutralization test) and the mean difference between paired serum (i.e., gold standard) and Nobuto filter paper strip (FPS) sera eluted immediately after drying collected at 14 or 15 d post-inoculation from experimentally WNV-inoculated wild turkeys and northern bobwhites.

There was an interaction between time and temperature on antibody titers from FPSs stored at RT or −20°C for 3 or 6 mo in wild turkeys (p = 0.003), indicating that antibody titers over time were dependent on temperature. Antibody titers were similar for FPSs stored at −20°C for 3 or 6 mo (p = 0.990) but differed for all other storage conditions (p < 0.01). Antibody titers from FPSs stored at RT were lower than those stored at −20°C, and those stored at RT for 6 mo had the lowest antibody titers (Fig. 1A).
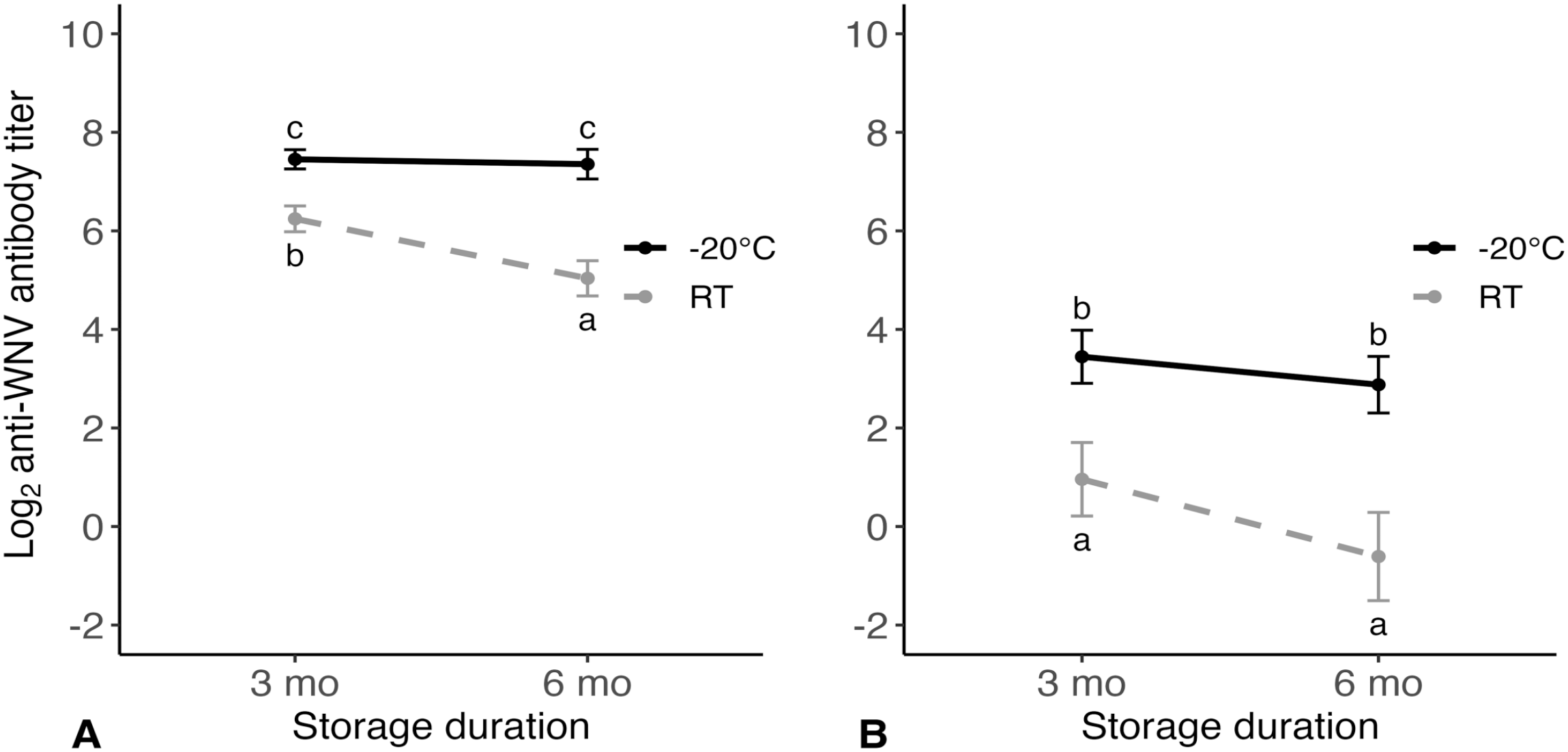
Estimated marginal mean log2 anti–West Nile virus (WNV) antibody titers (via plaque reduction neutralization test) and SEs from Nobuto filter paper strips that underwent 4 different storage conditions (before elution) after being collected at 14 or 15 d post-inoculation from experimentally WNV-inoculated (
Antibody titers from FPSs from northern bobwhites were affected by temperature (p < 0.001) and time (p = 0.027), but there was no interaction (p = 0.084). Antibody titers for FPSs stored at RT were ~3.0 log2 lower than those stored at −20°C (95% CI: 1.8–4.1; p < 0.001) when averaged over time. When averaged over temperature, antibody titers for those stored for 6 mo were ~1.1 log2 lower than those stored for 3 mo (95% CI: 0.1–2.0; p = 0.027). If the effect of time was evaluated separately for each temperature, there was no reduction in antibody titers between 3 and 6 mo for FPSs stored at RT (p = 0.057) or −20°C (p = 0.652; Fig. 1B).
The probability that FPSs from a wild turkey would test positive was affected by temperature (p = 0.018) and time (p = 0.028). Estimates failed to converge when the interaction term between temperature and time was included; thus, this term was excluded from our analysis. Predicted probabilities of a positive result for all storage conditions were > 0.9 except for FPSs stored at RT for 6 mo (Fig. 2A). The probability that FPSs from a northern bobwhite would test positive was affected by temperature (p < 0.001) and time (p = 0.025); no interaction was observed in this species (p = 0.445). The probability of detecting antibody titers was 0.4 lower for FPSs stored at RT for 3 mo compared to those stored at −20°C for 3 mo (95% CI: 0.1–0.7; p = 0.002). The probability of detecting antibody titers was lower for FPSs stored at RT for 6 mo compared to those stored at −20°C for 3 mo (p < 0.001) and −20°C for 6 mo (p = 0.002; Fig. 2B).
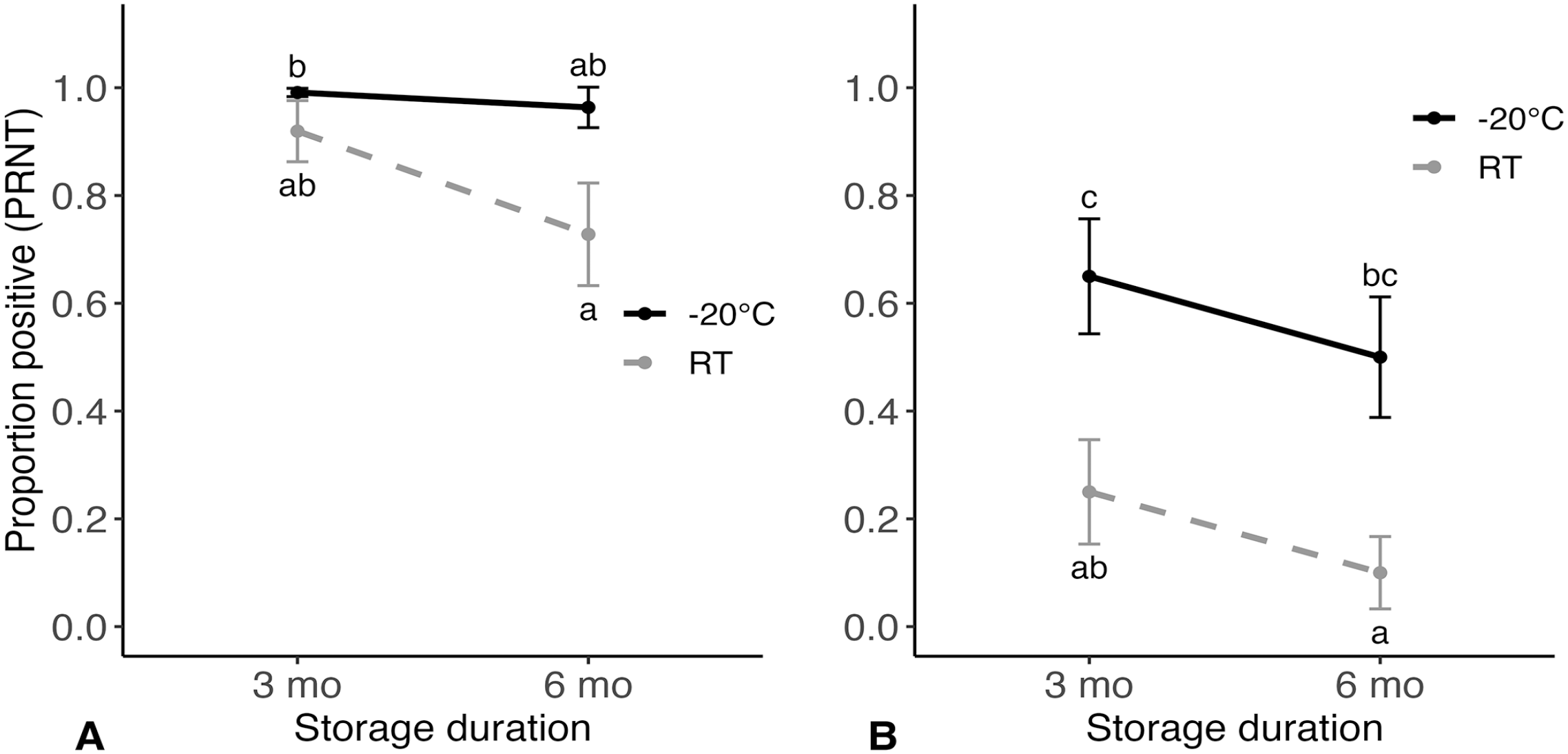
Predicted probabilities of samples that had detectable anti–West Nile virus (WNV) antibodies (i.e., tested positive via plaque reduction neutralization test [PRNT]) and SEs from Nobuto filter paper strips (FPSs) that underwent 4 different storage conditions (before elution) after being collected at 14 or 15 d post-inoculation from experimentally WNV-inoculated (
Our results suggest that the utility of FPSs for detecting anti-WNV antibodies by PRNT may be dependent on FPS storage temperature and duration and species. Antibody titers from FPS sera eluted immediately were ~2 log2 lower compared to serum samples in wild turkeys and northern bobwhites, which may be sufficient to result in false-negative results in some individuals. If the aim of testing FPSs for anti-WNV antibodies is to collect seroprevalence data (i.e., positive or negative results), then storing FPSs at −20°C for up to 6 mo or at RT for up to 3 mo may yield similar results in wild turkeys. However, wild turkey seroprevalence estimates may be falsely low if FPSs are stored at RT for 6 mo before elution. The proportion of northern bobwhite FPSs that tested positive remained low regardless of storage condition, although antibody titer decay was less in FPSs stored at −20°C and those eluted at 3 mo. In some cases, this decline in PRNT sensitivity should prompt consideration of other sampling methods. Our ability to compare WNV seroprevalence estimates among different species may be confounded by varied PRNT sensitivity estimates related to FPS storage, as observed in our study.
The differences between wild turkey and northern bobwhite results may partially be the result of general interspecies variation in serum antibody titers and potentially time to peak titer development, which also may vary within a species. 13 Wild turkey serum antibody titers were ~10-fold higher than those of northern bobwhites in our study. Consequently, northern bobwhite antibody titers more frequently dropped below the PRNT detection threshold, resulting in false-negative results. The apparent decline in detectable antibody titers over time from FPSs has been observed previously,3,6,7 suggesting that antibodies degrade while in the FPS matrix. In some instances, freezing FPSs before elution may help retain antibody titers. Serum anti-WNV antibody titers also vary among and within avian species12,13,18; reasons for this variation likely are multifactorial but may partially be attributable to varied host susceptibility to disease and immune responses.16,18 The observed differences between 2 galliform species emphasize the need to evaluate FPS usage for each species, pathogen, and assay (with inclusion of positive controls) for accurate results interpretation.
Our findings are consistent with previous work evaluating the utility of FPSs for WNV serology in wildlife. Antibody titer detectability was similarly lower in FPS sera eluted within 1 or 2 d of drying as compared to sera in WNV-inoculated ruffed grouse (Bonasa umbellus). 14 Consistently positive WNV test results were observed in FPSs collected from WNV-inoculated partridge (Alectoris chukar × A. rufa) that were stored at RT up to 3 mo and at −20°C up to 6 mo before elution. 8 Similar to our wild turkey results, the proportion of partridge samples that had detectable antibodies declined in FPSs stored at RT for 6 mo. 8
We selected FPS storage conditions that reflect those reported frequently in studies that utilize hunter-harvested FPSs for wildlife pathogen testing.8,19 A few previous studies included desiccants with FPSs to minimize humidity; however, the influence of desiccants on the utility of FPSs in conjunction with storage conditions was not fully evaluated.3–6 It is unclear if the use of desiccants would have affected our results, in which FPSs were fully saturated with clean whole blood. FPSs contaminated with gastrointestinal contents or bacteria, which can degrade antibodies, may yield false-negative results. Therefore, hunters and researchers should be provided with clear instructions on collection techniques using FPSs. 9
The results of our study highlight the efficacy of FPSs in WNV serologic testing in 2 galliforms, while shedding light on the influence of FPS storage conditions on anti-WNV antibody detection by PRNT. Based on our results, FPSs may be a good alternative to whole blood collection in wild turkeys. In the northern bobwhite, a species that generally may have low serum anti-WNV antibody titers, caution should be exercised when using FPSs in WNV serosurveys because of decreased test sensitivity, even in FPS sera eluted immediately after drying. Ultimately, freezing FPSs and eluting samples before 3 mo of storage may minimize false-negative results.
Footnotes
Acknowledgements
We thank Kayla G. Adcock and Charbel E. Gerges (Southeastern Cooperative Wildlife Disease Study, University of Georgia) for laboratory assistance. We thank the Southeastern Cooperative Wildlife Disease Study (SCWDS; University of Georgia) member state wildlife agencies for continued support through the Federal Aid to Wildlife Restoration Act (50 Stat. 917), including the states of Alabama, Arkansas, Florida, Georgia, Kansas, Kentucky, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Oklahoma, South Carolina, Tennessee, Virginia, and West Virginia. Further support was provided by SCWDS federal wildlife agency partners, including the U.S. Fish and Wildlife Service–National Wildlife Refuge System and the U.S. Geological Survey–Ecosystems Mission Area.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was funded by the Multistate Conservation Grant Program (grant F19AP00089), a program supported with funds from the Wildlife and Sport Fish Restoration Program and jointly managed by the Association of Fish and Wildlife Agencies and the U.S. Fish and Wildlife Service, 2019. Additional funding was provided by the member state wildlife management agencies of the Southeastern Cooperative Wildlife Disease Study (University of Georgia) through the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
