Abstract
Currently, the factors/toxins responsible for Clostridium perfringens-associated avian enteritis are not well understood. To assess whether specific C. perfringens' toxinotypes are associated with avian enteritis, the isolates of C. perfringens from 31 cases of avian necrotic or ulcerative enteritis submitted between 1997 and 2005 were selected for retrospective analysis using multiplex PCR. C. perfringens was isolated from chickens, turkeys, quail, and psittacines. The toxinotypes of isolates from diseased birds were compared against the toxinotype of 19 C. perfringens isolates from avian cases with no evidence of clostridial enteritis. All C. perfringens isolates were classified as type A regardless of species or disease history. Although many isolates (from all avian groups) had the gene encoding the C. perfirngens beta2 toxin, only 54% produced the toxin in vitro when measured using Western blot analysis. Surprisingly, a large number of healthy birds (90%) carried CPB2-producing isolates, whereas over half of the cpb2-positive isolates from diseased birds failed to produce CPB2. These data from this investigation do not suggest a causal relationship between beta2 toxin and necrotic enteritis in birds.
Clostridium perfringens is divided into 5 types: A, B, C, D, and E on the basis of the production of 4 major toxins: alpha (α), beta (β), epsilon (ε), and iota (ι). 1 , 21 Type A isolates, the focus of this study, produce α, but not β, ε, or ι toxins. In addition, all types of C. perfringens can produce 2 other major toxins, enterotoxin (CPE) and the recently discovered C. perfringens beta2 toxin (CPB2). CPE and CPB2 also have been associated with clostridial gastrointestinal diseases in domestic animals. 4 , 14 , 27 Clostridium perfringens can cause necrotic enteritis and cholangiohepatitis in many avian species. 12 , 25 However, it is not clear whether CPE and CPB2 are involved in C. perfringens-associated avian enteric diseases. Consequently, the goal of this study was to assess the presence and production of CPE and CPB2 by C. perfringens isolated from healthy and diseased bird species.
Thirty-one isolates of C. perfringens, from cases submitted to the California Animal Health and Food Safety Laboratory (CAHFS) between 1997 and 2005, were selected for this retrospective study: 10 from commercial chickens (5 from broiler flocks and 5 from layer flocks), 10 from meat turkeys, 4 from quail, and 7 from different species of psittacines. The main criteria for selecting the isolates were: 1) primary diagnosis of necrotic or ulcerative enteritis in the bird and 2) C. perfringens as the main isolate from intestine or liver. Selected cases from chickens and turkeys only had necrotic enteritis, while quail and psittacines had necrotic or ulcerative enteritis and necrotic hepatitis associated with C. perfringens.
In addition, intestine from avian cases with no evidence of gastrointestinal disease submitted to CAHFS between November 2004 and April 2006 were cultured anaerobically. From poultry cases (chicken and turkeys), ceca were primarily cultured for C. perfringens. From psittacines, distal small intestine and colon samples were cultured as these birds do not have ceca. Isolation of C. perfringens was attempted in a total of 56 chickens, 51 turkeys, and 58 psittacines. No quails were submitted during this period. Clostridium perfringens was only isolated from a small number of birds that were not suffering from enteric disease (6 chickens, 9 turkeys, and 4 psittacines).
The C. perfringens isolates used in this study were from birds located in California. Eighty percent of the chicken isolates and all the turkey and quail isolates were from the San Joaquin Valley of Central California, whereas only 20% of the chicken isolates were from the California's Deserts (South East). The psittacines were from all areas of California and one psittacine (PH4) with normal intestines was from Illinois. In addition, all the isolates were from birds located on different farms or were recovered in different years, except 3 quail isolates (QC2, QC3, and QC4) that were from the same farm and collected within a 4-month period.
Clostridium perfringens isolates were obtained by plating samples on Brucella 5% sheep blood agar a or in phenylethyl alcohol sheep blood (PEA) agar a or by inoculation in chopped meat broth a . Samples were incubated anaerobically at 37°C and examined daily, up to 5 days. Pure colonies were obtained by plating subcultures on Brucella agar and incubating them anaerobically for 24 hours. The identity of the isolates was confirmed by Gram stain, colony morphology, aerotolerance in chocolate agar plate (at 37°C in 5–10% CO2 for 24 hours), double zone of beta hemolysis, and reverse CAMP reaction (using C. perfringens ATCC 13124 as positive control).
The identity of these isolates was further confirmed by multiplex PCR as previously described. 11 , 18 Briefly, a single colony from solid medium was resuspended in 200 μl of HPLC grade water in a 1.5 ml microcentrifuge tube and heated at 98°C for 20 minutes before centrifugation for 5 minutes in a microcentrifuge at 3,000 × g. The supernatants were retained and used as DNA template.
The sequences of the oligonucleotide primers used to amplify portions of the major toxin genes of C. perfringens have been previously described. 11 Each 50-μl reaction contained 10X assay buffer (100 mM Tris-HCl, pH 8.3 [at 25°C]; 500 mM KCl; 15 mM MgCl2;0.01% [w/v] gelatin), 0.12 mM dNTPs, 0.5 μM of each cpa primer, 0.36 μM of each cpb primer, 0.44 μM of each etx primer, 0.52 μM of each iA primer, 0.34 μM of each cpe primer, 0.36 μM of each cpb2 primer, and 5 U of Taq DNA polymerase. Ten microliters of supernatant from boiled cells was added to each 50-μl PCR reaction and overlaid with 2 drops of mineral oil before processing in a MJ Research thermal cycler for 35 cycles (1 minute at 94°C, 1 minute at 55°C, 1 minute at 72°C). PCR products were identified by UV transillumination, following electrophoresis through a 2% agarose gel with 0.28 μg/ml of ethidium bromide. A molecular weight marker (1 kb DNA Ladder b ) was added to the agarose gels before electrophoresis.
Results of PCR toxin typing are shown in Tables 1 and 2. All isolates had the α-toxin gene (cpa). Sixty-six percent of isolates from both sick and healthy chickens, turkeys, and quail also had the CPB2 toxin gene (cpb2). Only 36% of the isolates from psittacines had the cpb2 gene, and one of these isolates also had the enterotoxin (cpe) gene. Furthermore, for each avian group the percentages of the cpb2-positive isolates were similar between birds with necrotic enteritis and those with normal intestine.
For CPB2 Western blot analysis, Clostridium perfringens colonies grown on oxygen reduced blood agar plates were resuspended in 10 ml of FTG media c and grown for 20 hours at 37°C. A 100 μl aliquot of the FTG culture was then used to inoculate a 10-ml tube of TGY (3% tryptic soy broth c , 2% glucose d , 1% yeast extract c , and 0.1% L-cysteine d ). Those TGY tubes were incubated at 37°C until the bacterial cultures reached an OD600 nm of 1.5 (monitored using a Bio-Rad Smart Spec™ 3000 e ). Cultures were then centrifuged at 4°C for 20 min at 9,000 × g to pellet the bacteria. Culture supernatants were retained and concentrated 20-fold using Amicon® Ultra centrifugal filter devices f as directed by the manufacturer. The concentrated supernatants were then mixed with 2X SDS-PAGE loading buffer, boiled for 5 minutes, and loaded onto a 12% SDS-PAGE gel. Following SDS-PAGE, the proteins were electrophoretically transferred to nitrocellulose membrane for Western blot analysis.
Western blot analysis was performed as previously described. 27 Briefly, blots were blocked with Blotto and then probed with a rabbit polyclonal anti-CPB2 antibody g . After probing with the primary antibody, the blot was washed with Tween20 e (0.01%) TBS 3 times and then probed with a horse radish peroxidase-conjugated anti-rabbit IgG-antibody d . Blots were developed with Supersignal® West Pico chemiluminescent substrate h and the results were recorded on autoradiographic film.
Clostridium perfringens isolate B2 (a human GI disease isolate previously shown to produce cpb210) and purified CPB2 were used as positive controls. Supernatant from ATCC 3624 (a cpb2-negative C. perfringens type A isolate) was used as a CPB2 negative control. All isolates were analyzed in triplicate experiments starting from different colonies.
CPB2 Western blot analysis of culture supernatants from bacteria grown in TGY media indicated that 18 (54.54%) cpb2-positive isolates were able to produce CPB2 toxin (Table 1). Expression of the toxin occurred more commonly in isolates from healthy intestines (4/4 chicken, 4/5 turkey, and 1/1 psittacine isolates) than those from diseased intestine (1/9 chicken, 4/9 turkey, 1/3 quail, and 2/3 psittacine isolates). CPB2-positive supernatants were identified by the presence of a single immunoreactive band of approximately 27 kD. This experimental molecular weight matches closely with previous reports. 5 , 13 , 27
Clostridium perfringens type A, and to a lesser extent type C, have been isolated from cases of enteritis caused by C. perfringens in birds. 23 Apparently, types B, D and E do not play a role in poultry disease. 23 In the present study, all isolates of C. perfringens belonged to type A.
The gene encoding the CPB2 toxin was detected in most chicken (75%), turkey (74%), and quail (75%) cases. Recent studies of the CPB2 toxin strongly suggest an association between this toxin and clostridial gastrointestinal diseases in some domestic animals, including typhlocolitis in horses and necrotic enteritis in piglets. 14 , 27 Recently, the cpb2 gene has been found in a few isolates from broilers with C. perfringens-associated hepatitis. 9 In the present study, the gene for the CPB2 toxin was detected in most C. perfringens isolates from chickens with acute necrotic enteritis with no obvious liver involvement as well as from those chickens with no clostridial enteritis. Also, the percentage of cpb2-positive C. perfringens strains in chickens is higher than that reported by other workers. 5 , 9 This difference may be owing to geographic location. Furthermore, this is the first report of detection of this gene in C. perfringens isolated from other avian species.
Presence of toxin genes and expression of cpb2 gene in vitro in Clostridium perfringens isolates from diseased birds.
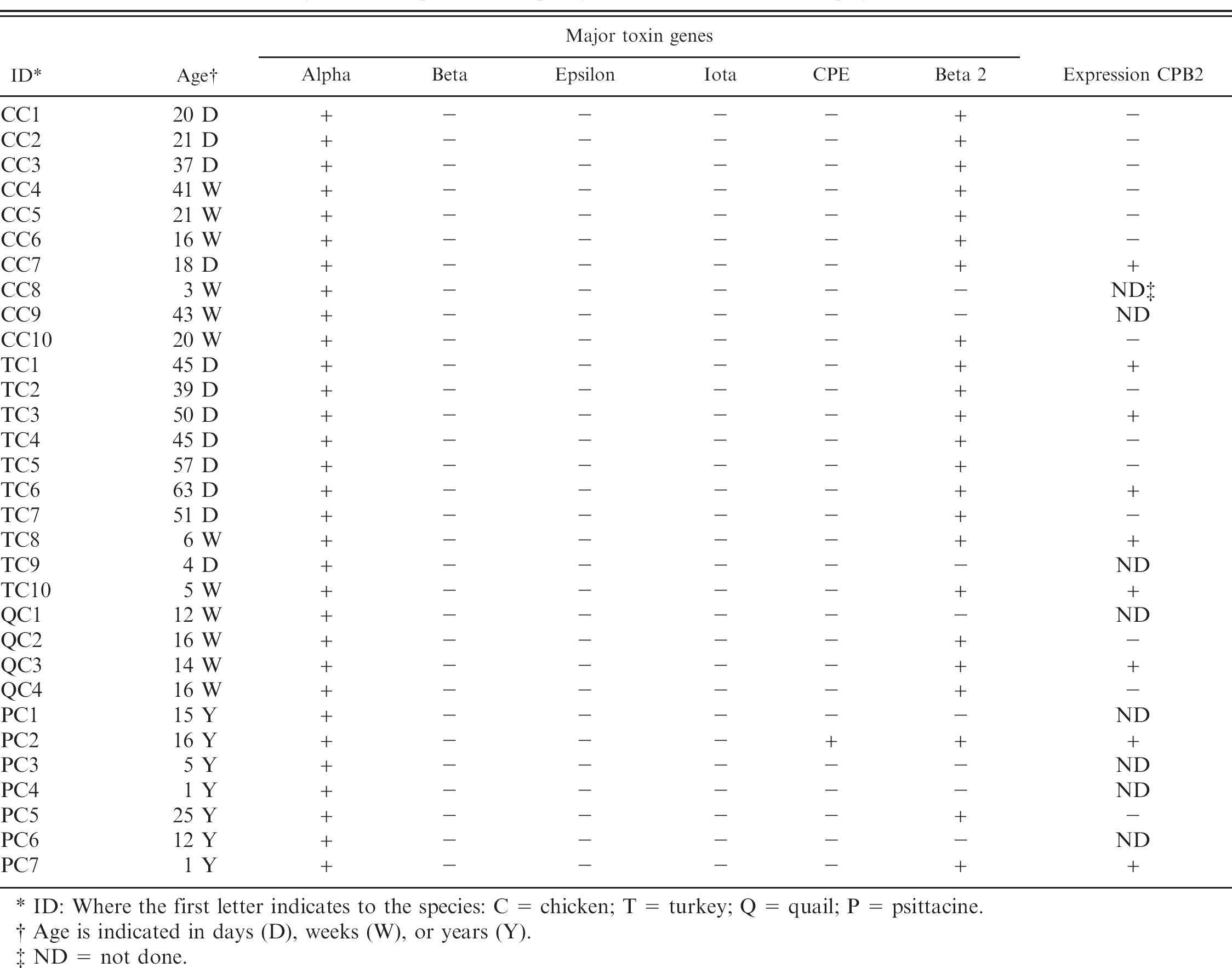
ID: Where the first letter indicates to the species: C = chicken; T = turkey; Q = quail; P = psittacine.
Age is indicated in days (D), weeks (W), or years (Y).
ND = not done.
Even though most isolates were PCR-positive for cpb2, in vitro expression of the CPB2-toxin occurred in only 54.5% of all the isolates that were cpb2-positive by PCR, regardless of species. Even more interesting is the fact that the in vitro expression of the CPB2-toxin occurred more frequently in isolates from unaffected intestines (90%) than those from necrotic enteritis (38.5%). Bueschel et al. 5 obtained about 40% of C. perfringens avian isolates that had the cpb2 gene and produced the protein in vitro.
However, the authors did not indicate either the type of birds the isolates came from or the disease condition in the birds. The fact that the correlation between the presence of the cpb2 gene and its expression in vitro was low in isolates from birds with necrotic enteritis and high in isolates from unaffected intestines, suggests that the CPB2 toxin probably does not play an important role in the pathogenesis of necrotic enteritis in birds. However, it is possible that an unknown factor that is not present under the in vitro conditions used in this study could trigger CPB2 toxin production in vivo by those isolates failing to produce CPB2 in vitro. 24 It also remains possible that unknown preexisting conditions within the avian intestines are required for CPB2 to cause disease.
The failure of isolates to produce CPB2 in vitro is not entirely understood. Similar to other C. perfringens toxins, including alpha toxin, cpb2 expression is known to be positively regulated by the bacterial 2-component regulator, VirR/VirS, through a RNA intermediate known as VR-RNA. 19 Although the VirR/VirS proteins 6 , 7 and the genes controlled by this regulator have been well characterized, 20 the identity of the signaling molecule and the mechanism of VR-RNA regulation have not been solved. However, the regulation story for cpb2 seems to be even more complicated than VirR/VirS regulation alone.
Presence of toxin genes and expression of cpb2 gene in vitro in Clostridium perfringens isolates from healthy birds.

Where the first letter indicates to the species: C = chicken; T = turkey; Q = quail; P = psittacine.
Age is indicated in days (D), weeks (W), or years (Y).
ND = not done.
In C. perfringens strains of porcine origin the production of CPB2-toxin is constitutive in vitro, 5 whereas in cpb2-positive equine disease isolates the onset of CPB2-production seems to be dependent on particular inducers, such as aminoglycoside antibiotics. 19 In this recent study, treatment of cpb2-positive C. perfringens with the aminoglycosides gentamicin or streptomycin was able to induce production of CPB2 in a representative equine strain by inducing translational slippage. 24 These results provide evidence for the role of certain chemicals, such as antibiotics affecting ribosomal function, in the induction of toxin production in clostridia. Translational slippage would provide a second layer of regulation (translational level) occurring after VirR/S mediated induction of gene expression (transcriptional level). Finally, isolates from different species also have been identified that fail to express cpb2 message in vitro. 16 , 26 Unfortunately, a mechanism explaining the lack of gene expression in these studies was not determined.
Owing to the problems associated with in vitro measurements of CPB2, it is important to stress that our study was performed in vitro and that these conditions may not portray the same situation present in the intestine of birds. However, the aim of our study to correlate in vitro production of CPB2 with disease is validated by the success of similar approaches used for other species such as swine, where in vitro CPB2 production was found to correlate very strongly with disease. 5 , 27 While the measurement of CPB2 toxin production in vivo would be ideal (based on the in vitro problems), currently no systems are available to evaluate production of CPB2 in vivo. Subsequently, methods to detect CPB2 toxin pre-formed in the intestine should be developed.
Only one isolate, from a psittacine, had the cpe gene encoding C. perfringens enterotoxin. Presence of the cpe gene in C. perfringens type A from poultry has been estimated to be as low as 10%, 22 but has not been reported in psittacines. Enterotoxin has been detected in chickens with necrotic enteritis, 8 but the role of this toxin in the etiology of this disease has not been determined. In humans and other animal species, CPE is responsible for alterations of intestinal permeability leading to watery diarrhea 4 and in one report it was associated with necrotic colitis in human patients. 3 It is possible that CPE was responsible for the necrotizing enteritis observed in a psittacine.
Lastly, it is well known that C. perfringens is a natural inhabitant of the soil and intestinal tract of warm-blooded animals and humans, 4 , 17 , 23 but C. perfringens was isolated only from 10–15% of healthy birds. The standard approach in the US poultry industry to protect against clostridial enteritis has been through the use of in feed subtherapeutic antibiotics and/or antibiotic ionophores. In the poultry samples, it is possible that the use of these drugs may have inhibited the growth of these bacteria. 2 , 17 Furthermore, the incidence of necrotic enteritis associated with C. perfringens in poultry has increased in countries that stopped using antibiotic growth promoters. 23 Clostridium is found in large numbers in the ceca, but in low numbers in other parts of the digestive tract of normal animals, including birds. 15 Consistent with these findings, in psittacines (which do not have ceca or have only vestigial remnants of ceca) C. perfringens is rarely found. 12
In summary, most of the C. perfringens isolates from avian origin were cpb2 positive; but only a few isolates from birds with necrotic enteritis expressed CPB2 toxin in vitro. Based on our results with a limited number of samples, this toxin probably does not play an important role in necrotic enteritis in birds, as the expression of the CPB2 toxin in vitro was very common in isolates from unaffected intestines. In this study we did not investigate transcriptional or translational regulation of CPB2 under different conditions. Therefore, to determine more conclusively the role of the CPB2 toxin in clostridial enteritis in birds, the expression of this toxin should be investigated in the presence of different intestinal conditions that might influence transcription and/or translation, such as several pharmaceuticals. Also, the presence of CPB2-toxin itself should be investigated in intestinal contents of sick and healthy birds.
Acknowledgements. The authors would like to thank Karen Tonooka for all her technical support. This research was partly supported by National Institute of Allergy and Infectious Diseases (Grant AI056177-03).
Footnotes
a.
PRAS media, Anaerobe Systems, Morgan Hill, CA.
b.
Gibco BRL-Life Technologies, Gaithersburg, MD.
c.
Difco, Detroit, MI.
d.
Sigma, St. Louis, MO.
e.
Bio-Rad, Hercules, CA.
f.
Millipore, Bedford, MA.
g.
Kind gift of Michel Popoff, Pasteur Institute, Paris, France
h.
Pierce, Rockford, IL.
