Abstract
We estimated the diagnostic sensitivity (DSe) and specificity (DSp) of an immunohistochemistry (IHC) protocol compared to the direct fluorescent antibody test (DFAT), which is the gold standard test for rabies diagnosis. We obtained brain samples from 199 domestic and wild animal cases (100 DFAT-negative, 99 DFAT-positive), by convenience sampling from 2 government-accredited rabies virus (RABV) testing laboratories in South Africa, between February 2015 and August 2017. Tissues that had been stored at 4–8°C for several days to weeks at the 2 accredited laboratories were formalin-fixed and paraffin-embedded. Nighty-eight cases tested IHC-positive using a polyclonal anti-RABV nucleoprotein antibody and a polymer detection system. The overall DSe and DSp for the RABV IHC test were 98% (95% CI: 93–100%) and 99% (95% CI: 95–100%), respectively. Domestic dogs accounted for 41 of 98 RABV IHC–positive cases, with the remainder in 4 domestic cats, 25 livestock, and 28 wildlife. Herpestidae species, including 7 meerkats and 9 other mongoose species, were the most frequently infected wild carnivores, followed by 11 jackals. Three cases in domestic dogs had discordant test results; 2 cases were IHC–/DFAT+ and 1 case was IHC+/DFAT–. Considering the implications of a false-negative rabies diagnosis, participating in regular inter-laboratory comparisons is vital, and a secondary or confirmatory method, such as IHC, should be performed on all submitted specimens, particularly negative cases with human contact history.
Rabies is an acute progressive encephalitis most often caused by infection of the nervous system with Lyssavirus rabies (RABV; Rhabdoviridae, Lyssavirus).8,35 Rabies occurs on every continent except Antarctica and is one of the most feared zoonotic diseases, with a case-fatality rate that approaches 100% if post-exposure prophylaxis is delayed or not administered before the onset of clinical signs. 10 Although the reservoirs for RABV are carnivores and bats predominantly, all mammalian species are susceptible. 15
Two variants of RABV, designated the canid and mongoose biotypes, are present in southern Africa. 34 The domestic dog is the principal reservoir and vector of the canid RABV biotype.34,37 However, many wildlife species are susceptible to the canid RABV biotype infection including the side-striped jackal (Canis adustus), black-backed jackal (Canis mesomelas), bat-eared fox (Otocyon megalotis), African wild dog (Lycaon pictus), honey badger (syn. ratel; Mellivora capensis), hyrax (Procavia capensis), aardwolf (Proteles cristatus), brown hyena (Hyaena brunnea), and several antelope species.7,14,30
The mongoose RABV biotype (previously referred to as viverrid RABV biotype) is associated mostly with members of the Herpestidae family (mongooses, meerkats) but can also occur in some Viverridae (i.e., genets, civets). 23 The yellow mongoose (Cynictis penicillata) is the main vector for the mongoose RABV biotype. 23 Rabies-related lyssaviruses (i.e., Duvenhage, Lagos bat, Matlo, Mokola) have also been identified in southern Africa and can cause clinical disease indistinguishable from RABV infection. 4 Although infections with these related viruses are rarely associated with human or animal cases, they might nonetheless contribute to the public health burden of rabies. 22
There are no pathognomonic clinical signs or postmortem lesions associated with rabies in animals. Historically, the diagnosis relied on the clinical case history and histologic detection of Negri bodies (2–5 µm, round-to-oval, eosinophilic intracytoplasmic inclusion bodies) in the neuronal perikarya and proximal dendrites in pyramidal cells of the hippocampus and Purkinje cells of the cerebellum. 24 However, given its low sensitivity, histology is no longer regarded as a suitable test for RABV and has been discontinued. 10 The diagnosis of RABV in animal tissues relies on laboratory testing of samples from the CNS using methods recommended by the World Organisation for Animal Health (WOAH), founded as the Office International des Epizooties (OIE). 38 These methods include the direct fluorescent antibody test (DFAT), the direct rapid immunohistochemistry (IHC) test (dRIT), and conventional nucleic acid detection–based assays, such as conventional reverse-transcription PCR (RT-PCR). 38 Virus isolation in neuroblastoma cells is recommended in the case of inconclusive results; serologic testing is not recommended for antemortem diagnosis because of late seroconversion and the high mortality rate of infected animals.
The DFAT gives reliable results on fresh samples within 1–2 h in 95–99% of cases. In the DFAT, fluorescein isothiocyanate–labeled polyclonal or monoclonal anti-RABV antibody facilitates the detection of RABV nucleocapsid protein. 38 Preparations of polyclonal anti-RABV antibody conjugates are preferred for tests on brain tissues given that conserved antigenic sites on the nucleocapsid proteins permit identification of all lyssaviruses. 38 Evaluated brain regions should preferably include a composite of the brainstem and the cerebellum.3,28 Cerebral cortex and hippocampus should also be included, if possible, but can yield false-negative results if these are the only brain regions available for testing. A limitation of the DFAT is that samples must be fresh or preserved in 50% glycerol–PBS and refrigerated to slow putrefaction.1,19 Negative DFAT results might be invalid because of deterioration in the quality of viral antigen in samples that are not refrigerated and tested within 24–48 h. 2 Other challenges with the DFAT include the need for a fluorescence microscope, suitable infrastructure, and waste disposal protocols. 6 Additionally, if formalin-fixed tissues are the only available specimens given a lack of dependable refrigerated shipment, samples must first be digested with proteinase K to expose the target antigen, which increases the likelihood of obtaining false-negative results when using the DFAT. 36
The dRIT for RABV is as sensitive and specific as DFAT and can be performed on fresh, frozen, or glycerol-preserved samples in a shorter time than required routinely for DFAT.6,17,29 Touch impression smears of the brain regions described for DFAT are tested using biotinylated polyclonal or monoclonal antibodies that detect RABV nucleocapsid protein. The choice of antibody preparations depends on the lyssavirus variants specific to the geographic area of focus. 6 Biotinylated antibodies have the advantage that results can be evaluated using a light microscope, thus allowing testing at local point-of-care facilities with follow-up testing done at central laboratories. 29
A few studies have reported that IHC might be a suitable alternative to DFAT or dRIT, if only formalin-fixed, paraffin-embedded (FFPE) brain samples from natural cases are available for testing.9,12,16,32,39 The nucleoprotein of RABV is resistant to autolysis, routine formalin-fixation, paraffin-embedding, as well as harsh antigen retrieval methods and can be detected with variable success depending on the affinity and reactivity of either polyclonal or monoclonal antibodies.9,26,39 Prior freezing of tissues also does not affect IHC test results. 12 An added advantage of having FFPE tissues available is that RABV-negative cases can be investigated further using histochemical stains such as H&E, and alternative diagnoses might be made. 32
In South Africa, the WOAH (OIE) Rabies Reference Laboratory at the Agricultural Research Centre–Onderstepoort Veterinary Research (ARC-OVR) and the Allerton Provincial Veterinary Laboratory (APVL) are accredited by the Department of Agriculture, Land Reform and Rural Development to perform the DFAT for rabies diagnosis. Although refrigerated transport of samples preserved in 50% glycerol–PBS has been encouraged for many decades in South Africa, occasionally a rabies diagnosis is not suspected at the time of sampling and only formalin-fixed samples are available. 5 The Faculty of Veterinary Science at the University of Pretoria (FVS) therefore developed IHC protocols in 1994, utilizing a streptavidin–biotin complex (ABC) system and a peroxidase–anti-peroxidase (PAP) system with monoclonal and polyclonal antibodies against RABV nucleocapsid protein. 16 The sample size in that study was too small to determine the sensitivity or specificity of the method. However, the techniques successfully identified southern African canid and mongoose RABV biotypes in a domestic dog, black-backed jackal, bat-eared fox, and yellow mongoose. Since then, suspected rabies cases have been tested regularly using both DFAT and IHC at the ARC-OVR and the FVS. A polymer-based IHC protocol has replaced the ABC and PAP systems given the reported higher antigen detectability of this technology, 25 but the protocol has not been validated by the FVS.
Our aim was to estimate the diagnostic sensitivity (DSe) and diagnostic specificity (DSp) of a polymer-based IHC protocol using a polyclonal rabbit anti-RABV nucleoprotein antibody for domestic and wild animal species of South Africa. In addition, we investigated the effects of severe autolysis and prolonged formalin-fixation on the interpretation of IHC results.
Materials and methods
Case selection
Exact binomial methods were used to estimate the sample size of rabies-positive as well as rabies-negative cases required. 11 A sample size of 200 (100 DFAT-positive, 100 DFAT-negative) was estimated based on the assumed sensitivity and specificity of IHC against RABV antigen of 0.95, with the desired estimate of these proportions ±0.05 at the 95% level of confidence. Samples were collected between February 2015 and August 2017 as per approval of the Animal Ethics Committee of the University of Pretoria (clearance certificate V091-14).
We originally identified 239 cases at the WOAH (OIE) Rabies Reference Laboratory of the ARC-OVR and the APVL for possible inclusion in our study. From this collection, cases were excluded when tissues from the brain were absent, or data were incomplete. We investigated 99 DFAT+ and 100 DFAT– samples from domestic and wild animals. When available, sections of brainstem, cerebrum, cerebellum, and hippocampus were collected from each case. Absence of any of the brain regions was recorded.
Samples that had been stored at 4–8°C for several days to weeks at the 2 accredited rabies laboratories were transferred to 10% neutral-buffered formalin and fixed for a minimum of 24 h, as recommended previously. 25 Samples that had already been formalin-fixed at the 2 laboratories were also paraffin-embedded. Therefore, duration of formalin-fixation for cases varied considerably, ranging from 1 d to 12 mo. As a result, the DFAT had frequently been performed on less-degraded tissue compared to the formalin-fixed tissue.
Negative tissue controls for IHC
Negative control tissues for IHC were obtained from 13 cases in the archives of the FVS. Inclusion criteria were that brain tissue from a variety of different regions of the brain was available. Additionally, Negri body–like inclusions were present in H&E-stained sections (i.e., bovine viral diarrhea viral inclusions, Lafora-like bodies, confronting cisternae) or the animals must have had neurologic signs before death from other non-rabies etiologies (e.g., canine distemper, cerebral babesiosis, malignant catarrhal fever, heartwater, toxoplasmosis, neosporosis, histophilosis, cerebral cortical necrosis, brain abscess, encephalomalacia).
Test protocols
DFATs had been conducted at the ARC-OVR and the APVL using a standard operating procedure (SOP). Briefly, impression smears were prepared from a composite brain sample, fixed in 100% cold acetone, air dried, stained with fluorescein isothiocyanate–labeled polyclonal anti-lyssavirus antibody conjugate, and examined for specific fluorescence using an inverted fluorescence microscope. 38 Antibodies were produced at the WOAH (OIE) Rabies Reference Laboratory of the ARC-OVR by immunizing goats with purified ribonucleoprotein (RNP) antigens obtained from 2 lyssavirus species (RABV laboratory strain SAG-2; Mokola virus [MOKV, 229/97]) as described previously. 6
Embedding, sectioning, and H&E-staining of formalin-fixed tissues were done according to the SOPs of the FVS. IHC was performed on 4-µm thick sections at the same laboratory, using a polyclonal rabbit anti-lyssavirus RNP antibody (Ministry of Agriculture, Food and Rural Affairs, Agricultural Research Institute of Ontario, Canada, lot 9/2/88). Briefly, the method included blocking endogenous peroxidases with 3% hydrogen peroxide, microwave antigen retrieval in Tris–EDTA buffer (pH 9) for 21 min, and incubation with the anti-RABV primary antibody at 1:500 dilution for 1 h. This was followed by detection with rabbit anti-mouse secondary antibody (F0232; Dako) and a polymer detection system (EnVision rabbit/mouse peroxidase, K5007; Dako), NovaRED peroxidase substrate (SK-4800; Vector), and hematoxylin counterstain.
Additionally, to confirm that immunoreactivity observed in our study was specific to RABV antigen and not nonspecific antibody binding to brain tissue, sections from 10 randomly selected DFAT–/IHC+ rabies cases were tested sequentially with antibodies to Toxoplasma gondii (negative reagent control). IHC for toxoplasmosis is performed frequently at the FVS and, comparable to RABV, T. gondii antigens are also present in the brain. Additionally, the anti–T. gondii antibody is a polyclonal rabbit (USDA Agricultural Research Service, Animal Parasitic Diseases Laboratory, Beltsville, MD, USA). The method uses the same secondary antibody and EnVision rabbit/mouse peroxidase polymer detection system as described for the RABV IHC. The protocol only differs in the antigen retrieval step, in which slides are bathed in pronase (protease from Streptomyces griseus, type XIV, P5147-5G; MilliporeSigma) in an incubator at 40°C for 30 min.
Sections that were tested with IHC were randomized separately from the H&E-stained sections to ensure that the observer was anonymized to specimen origin and other test results. Slides were examined by light microscopy, and anti-RABV RNP labeling was recorded as positive or negative. In the case of a positive result, and where brain regions were identifiable, immunolabeling in the brainstem, cerebellum, cerebrum, and hippocampus were each semi-quantitatively graded as low-, intermediate-, or high-grade. Low-grade labeling was defined as positive labeling present in the neuroparenchyma in <10% of each of the brain regions examined. Intermediate-grade labeling was defined as 10–25% positive labeling. High-grade labeling was defined as >25% positive labeling. Repeatability of the IHC results was determined by randomly selecting 20 DFAT+ and 20 DFAT– cases. Selected cases were randomized again and anonymized for a second reviewer who recorded positive or negative labeling.
H&E-stained sections were examined to determine the levels of autolysis, check for the presence of Negri bodies, and to record microscopic lesions suggestive of rabies encephalitis or other causes of neurologic disease. Autolysis was recorded for each case as mild, moderate, or severe. Mild autolysis was defined as cases in which all of the anatomic regions of the brain were readily identifiable on histologic examination. In the case of moderate autolysis, anatomic detail was still identifiable, and RBCs were intact, but the nuclei of neurons showed degradation, and putrefactive bacteria could be present. In the case of severe autolysis, anatomic detail and individual neurons were not identifiable, RBCs were lysed, and many putrefactive bacteria and/or fungi were present.
Statistical analysis
DSe and DSp of IHC were estimated as the proportion of cases with DFAT results that tested positive or negative on IHC, respectively. Mid-P exact CIs were calculated for DSe and DSp using statistical freeware (http://www.OpenEpi.com).
Results
Descriptive results
We included 199 specimens from 34 domestic and wild carnivore species in our study (Table 1). The cerebrum and brainstem were the anatomic regions sampled most frequently and were present in 182 of 199 (91.5%) and 166 of 199 (83.4%) cases, respectively. The hippocampus and cerebellum were sampled in 106 of 199 (53.3%) cases, but both areas were not present in all cases. For example, some cases had cerebrum, cerebellum, and brainstem; other cases had cerebrum, brainstem, and hippocampus. Almost a third (65 of 199) of the cases had all 4 of the brain regions available for analysis. In 7 of 199 (3.5%) cases, the tissues were too autolyzed to identify anatomic regions.
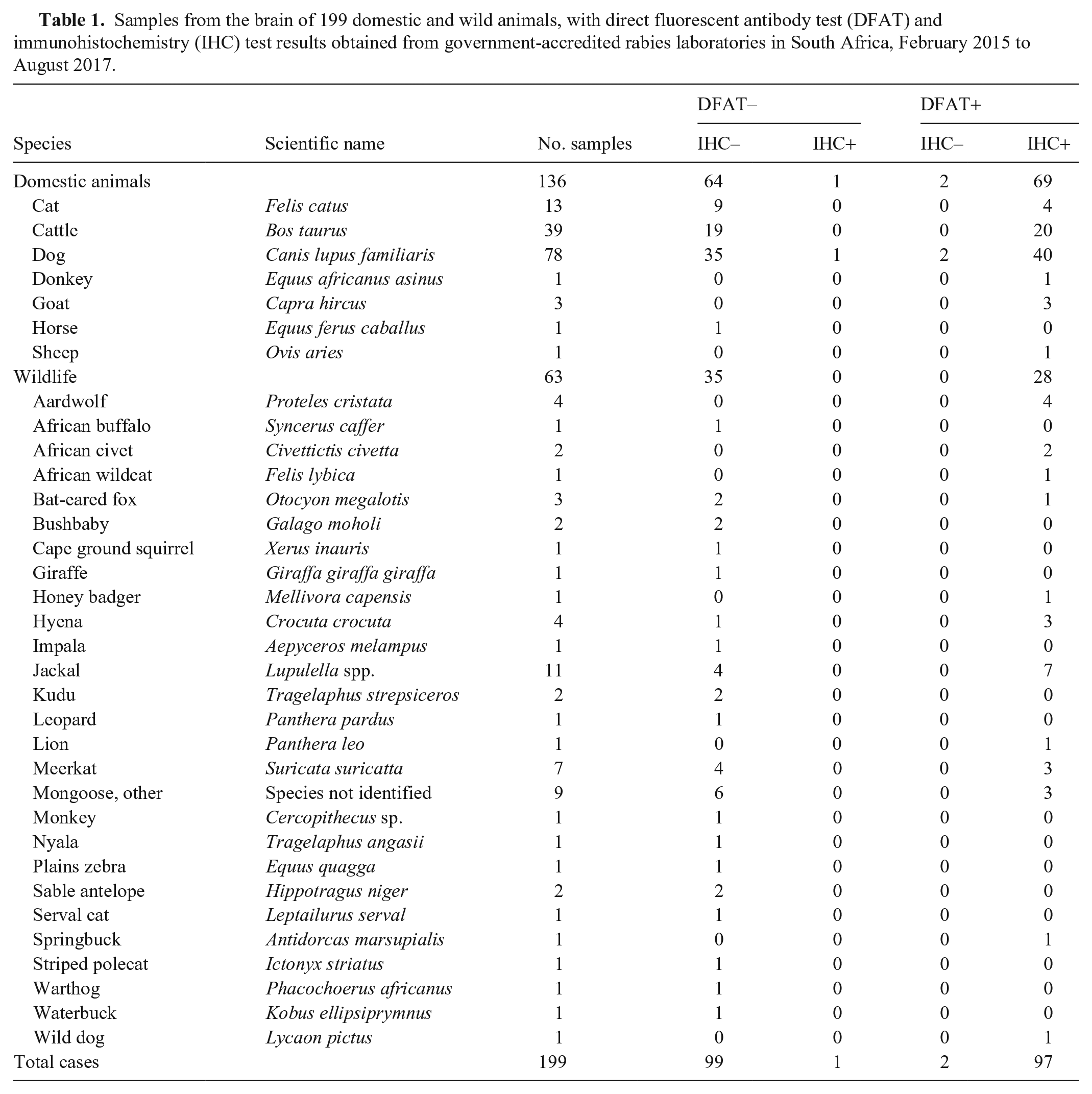
Samples from the brain of 199 domestic and wild animals, with direct fluorescent antibody test (DFAT) and immunohistochemistry (IHC) test results obtained from government-accredited rabies laboratories in South Africa, February 2015 to August 2017.
Anti-RABV RNP was visible by IHC in 98 of the 99 DFAT+ cases as stippled red-brown granules within the perikaryon, axons, and dendrites of neurons (Figs. 1, 2). Negri bodies were typically large homogeneous aggregates in the perikaryon; dust-like granules (“RABV dust”) in the neuronal processes and axons gave the neuroparenchyma a fine granular appearance (Fig. 1).
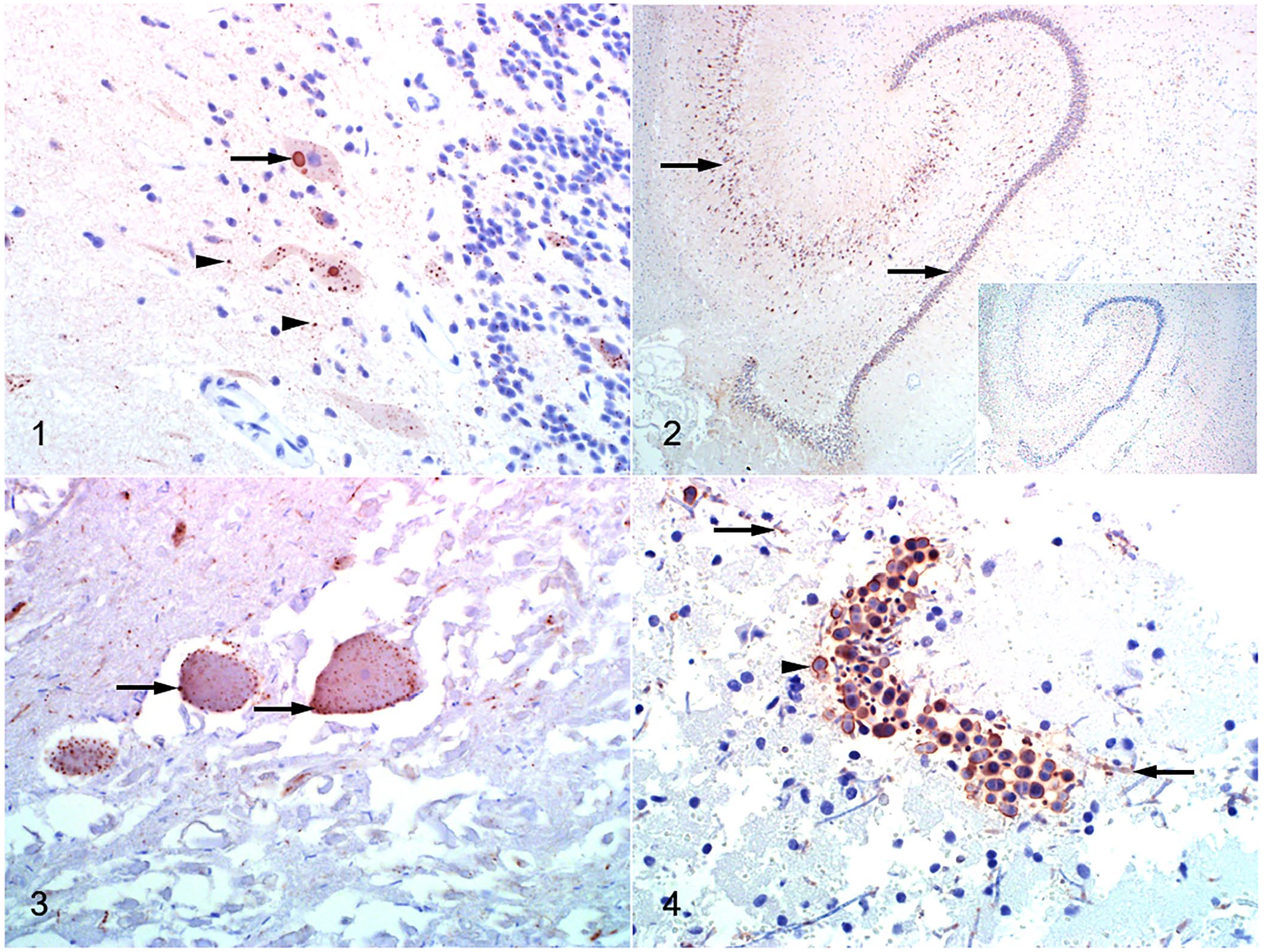
Immunohistochemistry in various brain sections using a polyclonal rabbit anti-lyssavirus ribonucleoprotein (RNP) antibody, the EnVision polymer detection system, NovaRED chromogen, and hematoxylin counterstaining.
The level of grading of each anatomic region of the brain was scored in 91 of 98 IHC+ cases (Table 2). This excluded the 7 cases that were too autolyzed to recognize anatomic regions. Generally, positive labeling was present diffusely throughout all of the available anatomic regions (Fig. 2) and was characterized as a very distinctive, sharply demarcated reaction product. Brainstem tested positive in 90 of 90 (100%) available specimens; intermediate- to high-grade labeling was present in 85 of 90 (94%) specimens. Cerebellum, hippocampus, and cerebrum were positive in 41 of 43 (95%), 43 of 47 (91%), and 81 of 89 (91%) specimens, respectively; low-grade labeling was most common in the cerebrum where only 6 of 89 (7%) samples tested positive.
Brain regions available for examination for each of the immunohistochemistry (IHC)-positive cases, the percentage of cases in which the specified region tested negative, and the grade assigned for identifiable brain regions for domestic and wild animals obtained from government-accredited rabies laboratories in South Africa, February 2015 to August 2017.

Low-grade labeling = positive labeling in <10%, intermediate-grade labeling = 10–25%, and high-grade labeling ≥25% positivity in the brain regions examined.
All 4 of the main brain regions were available for testing in 25 IHC+/DFAT+ cases. The brainstem was positive in all 25 cases and had high-grade labeling in 20 cases. Cerebellum, hippocampus, and cerebrum were IHC+ in 24, 22, and 21 cases, respectively. High-grade labeling was present in 18 cerebellum, 18 hippocampus, and 12 cerebrum specimens.
In H&E-stained sections, autolysis was mild or moderate in 51 of 199 (25.6%) and 87 of 199 (43.7%) specimens, respectively. Autolysis was severe in 61 of 199 (30.7%) specimens, with 2 samples liquefied. All of the severely autolyzed or liquified cases that were DFAT+ were also classified as IHC+. Tissues for 1 DFAT+ case had been in formalin for 365 d and were still IHC+. In severely autolyzed samples, RABV antigen outlined gaps where neurons had been (Fig. 3).
Background and other nonspecific labeling were present in 20 of 199 (10%) cases but were readily distinguishable from RABV-specific positive labeling. Labeling of putrefactive fungi was present on the periphery of the spores and in hyphae, and was easily distinguishable from Negri bodies (Fig. 4). In addition, phagocytized fungal debris within macrophages labeled false-positive in a case of granulomatous meningitis diagnosed on microscopic examination of H&E-stained sections.
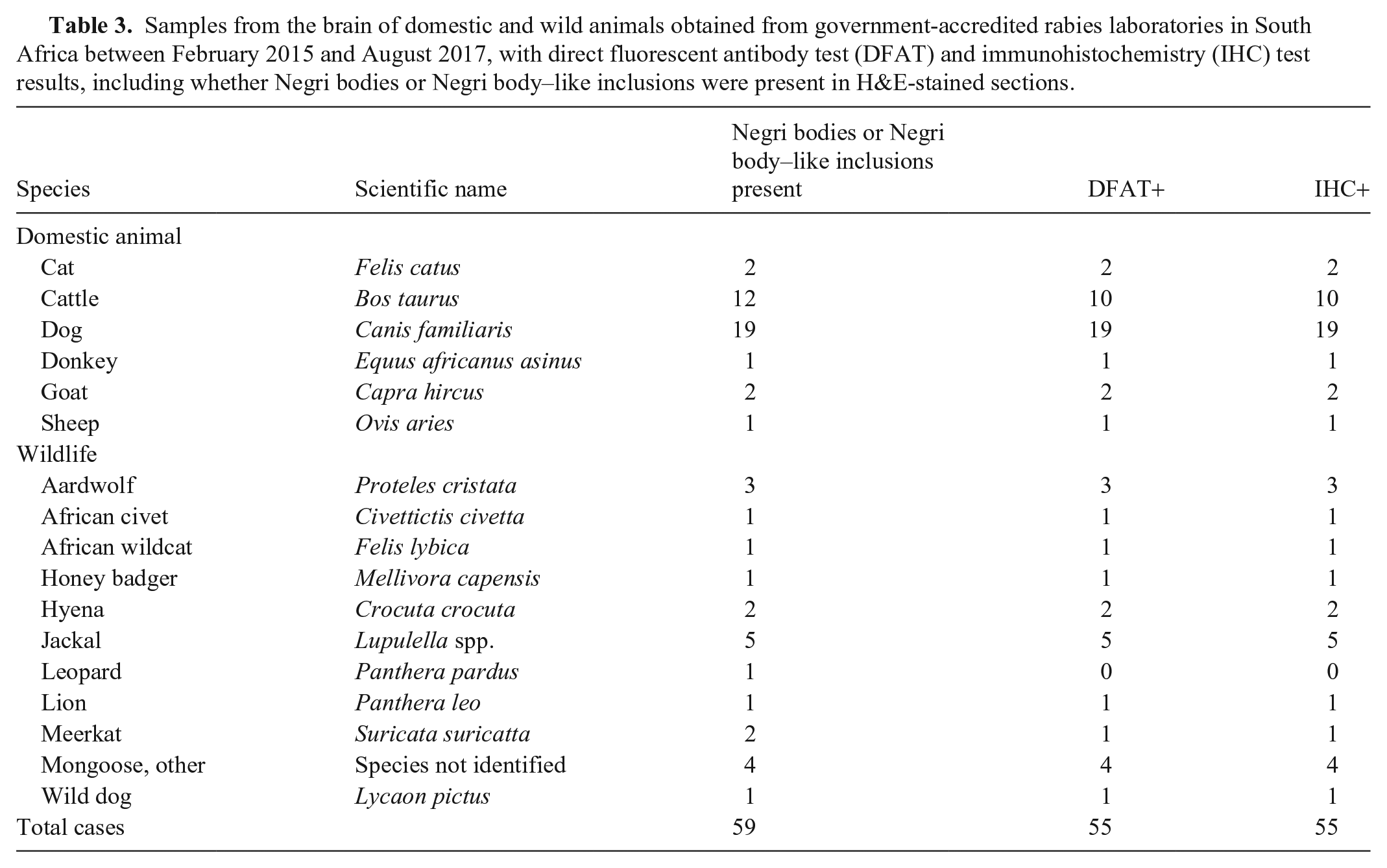
Negri bodies were observed in 59 of 99 (60%) RABV DFAT+ cases in a variety of domestic and wild species (Table 3) and were more often present in the cytoplasm of Purkinje cells of the cerebellum and in the pyramidal and larger neurons of the hippocampus. Fifteen DFAT+/IHC+ cases were too severely autolyzed to detect inclusions. Twenty of the 199 cases were classified as “uncertain” for the presence of Negri bodies, and observed structures within the neuronal perikaryon were classified as intracytoplasmic proteinaceous aggregates (Negri body–like inclusions). Of these 20 cases, 7 were DFAT+/IHC+. A non-RABV negative control case with confronting cisternae was diagnosed as RABV-positive on H&E. Thus, 1 of the 113 RABV-negative cases examined was considered to have convincing Negri bodies.
Samples from the brain of domestic and wild animals obtained from government-accredited rabies laboratories in South Africa between February 2015 and August 2017, with direct fluorescent antibody test (DFAT) and immunohistochemistry (IHC) test results, including whether Negri bodies or Negri body–like inclusions were present in H&E-stained sections.
Mononuclear cell perivascular cuffing was the microscopic lesion present most often in H&E-stained sections. However, the lesion was observed in 85 of 199 (42.7%) specimens, and only 50 of these were RABV DFAT+. Thus, of the 99 DFAT+ cases, only 50.5% had perivascular cuffing. Focal or multifocal microglial proliferation (also referred to as glial nodules or Babès nodules) was observed in 31 of 199 (15.6%) specimens, of which only 19 were DFAT+ for RABV. Forty-six of the 99 RABV DFAT+ cases did not have perivascular cuffing or glial nodules. A further 23 of these 46 cases also did not have Negri bodies that could be detected on H&E-stained sections. Therefore, 23 of 99 (23.2%) DFAT+ cases had no lesions or inclusions that would be indicative of possible RABV infection.
Discordant cases
In 3 cases, the IHC and DFAT results were not correlated, and all 3 cases were from domestic dogs. Two cases were DFAT+/IHC–; 1 case was DFAT–/IHC+. For the DFAT–/IHC+ case, cerebrum, cerebellum, and brainstem were available for testing, and typical Negri body–like inclusions were IHC+ in the large neurons of the brainstem. Microscopically, mononuclear perivascular cuffing and glial nodules were prominent, but Negri bodies could not be identified.
For 1 of the 2 DFAT+/IHC– cases, cerebrum, cerebellum, hippocampus, and brainstem were available for testing. Negri bodies and microscopic lesions were absent in H&E-stained sections. The DFAT and IHC were repeated; intense positive fluorescence was again detected on the DFAT, and the IHC result remained negative. RT-PCR targeting a partial region of the glycoprotein and the G-L intergenic region of RABV, 31 performed at the ARC-OVR, indicated the presence of a rabies lyssavirus. One possible explanation for the discrepancy might be a laboratory sample registration error, whereby the IHC test was done on an incorrectly labeled case. To exclude this possibility, a frozen archived sample was obtained from the ARC-OVR and IHC repeated a third time. The specimen was severely autolyzed, and the only structures detectible on microscopic examination were scattered neuronal nuclei, without visible cytoplasmic borders. Numerous IHC+ round-to-oval structures resembling Negri bodies were observed. However, these would have been overlooked had the reader not been biased to look for RABV antigen. The reason for the discrepancy between the DFAT and the IHC test results for this case could not be established conclusively.
In the second DFAT+/IHC– discordant case, the Botswana National Rabies Laboratory obtained a RABV DFAT+ test result. Only cerebrum and brainstem were submitted for testing, and Negri bodies and perivascular cuffing were absent in H&E-stained sections. Malacia and pyogranulomatous meningitis (including an inflammatory infiltrate in the choroid plexus) with leukostasis comprising monocytes, lymphocytes, and neutrophils were observed. CSF examined by a private veterinarian contained numerous monomorphic, rod-shaped bacteria, some of which were phagocytized by neutrophils. Yersinia pestis was isolated on bacterial culture of blood. Only formalin-fixed specimens of the cerebrum and brainstem were submitted to the ARC-OVR in South Africa and therefore the DFAT could not be repeated. The microscopic lesions observed in the brain were not typical features of RABV infection. It is possible that the biological conjugate used for the RABV DFAT cross-reacted with Y. pestis organisms given that the biological conjugate can stain bacterial proteins.
DSe and DSp of the IHC
None of the 13 non–RABV-negative tissue control cases had any RABV IHC+ labeling. Also, no labeling was observed in sequential slides incubated with antibody to T. gondii instead of anti-RABV antibody (Suppl. Figs. 1, 2). Hence, immunoreactivity to brain tissue of the reagents used in anti-RABV IHC was not detected.
The DSe and DSp of the RABV IHC for all species combined were 98% (95% CI: 93%, 100%) and 99% (95% CI: 95%, 100%), respectively. The DSe and DSp of IHC in domestic dogs were 95% (95% CI: 85%, 99%) and 97% (95% CI: 87%, 100%), respectively. Apart from the evaluated dog cases, the DSe and DSp of the RABV IHC were both estimated as 100% in all other evaluated species.
Regarding repeatability, results for all 40 cases examined by the second investigator were in perfect agreement with those of the primary investigator.
Discussion
To our knowledge, we evaluated RABV IHC in more cases (99 DFAT+, 100 DFAT–) and in a wider range of species (34) than reported previously.9,12,16,32,39 Samples from 78 domestic dogs and 39 cattle were the most frequent submissions, but our study also included 63 specimens from wild animal species. A large proportion of samples from wildlife were meerkats, other unidentified mongoose species, and jackals. Mongoose rabies is endemic in South Africa and occurs widely on the interior plateau of the country, with spillover of infection to a variety of both domestic and wild animals.23,33 Meerkats are also often inadvisably kept as pets and must be euthanized to exclude the possibility of rabies if humans are bitten. The sizable number of submitted specimens from jackals were obtained during a rabies outbreak in this species in the Muldersdrift area of Gauteng Province in 2016.
The DSe and DSp of IHC relative to the DFAT using the EnVision polymer detection system were nearly perfect (98% and 99%, respectively). Therefore, the test performed well against the DFAT, one of the primary tests for RABV, even though specimens were often more autolyzed or degraded by the time our study was performed. Three cases of domestic dogs had discordant results. For a DFAT–/IHC+ case, the DFAT was repeated, and fluorescence was weak positive on the repeat test. A similar case, not included in our study, was reported at the FVS in October 2017 in which a hyena tested DFAT–/IHC+ for RABV. Microscopically, lesions typically associated with rabies were present in both cases and included mild-to-moderate mononuclear perivascular cuffing and glial nodules. This finding suggests that IHC using a polymer detection system might have higher analytical sensitivity than the DFAT. Specimens submitted for RABV testing should ideally be subjected to a confirmatory test, such as virus isolation in cell culture, as part of the continued validation of laboratory tests.
Previous IHC studies used the PAP or ABC detection systems.9,12,16,25,39 However, sample numbers were small in most of these studies, and DFAT– cases were not included, which precludes determination of DSp. In one study, rabies was diagnosed in 100 of 187 naturally infected animals using the DFAT, the PAP system, mouse inoculation, and cell culture. 39 In 2 cases, a positive diagnosis was only made using the DFAT, which was considered false-positive, given that sparse fluorescence was detected in only 1 of 4 investigated brain regions, and all other test methods were negative. There were no false-positive results with the PAP technique, but 2 false-negative results were recorded that were attributed to irregular distribution of viral antigen, or too little antigen detectable using the PAP system. Corresponding to our results, severe autolysis and prolonged storage in formalin did not hinder interpretation of IHC results.20,39 Results in our study suggest that the EnVision polymer detection system might be more sensitive than the PAP system given that at least 2 DFAT– cases were likely truly positive for RABV. However, inter-laboratory comparisons of rabies diagnosis using similar IHC and DFAT protocols must be organized to determine if the results obtained in our study are repeatable. The discordant cases observed also underscore the need for continuous validation of RABV detection techniques against other methods and the importance of sustained inter-laboratory comparisons of rabies diagnosis to ensure continuous quality improvement.26,27
In our study, the anatomic brain region of choice for IHC was the brainstem, which tested positive more often than cerebellum, hippocampus, or cerebrum. All 4 of the main brain regions were only available in 25 cases, and brainstem tested positive by IHC and DFAT in all. This result agrees with a study in which the brainstem was always positive by DFAT in RABV-infected brains, and antigen was often most abundant in the thalamus. 3 Cerebellum, hippocampus, and cerebrum tested negative in 1 of 25 (4%), 3 of 25 (12%), and 4 of 25 (16%) cases, respectively, in our study. Similar results were obtained in studies in which the cerebellum, hippocampus, and different parts of the cerebrum (frontal, temporal, caudal, unidentified lobes) were negative in, respectively, 4.5, 4.9, and 3.9–11.1% of positive brains. 3 We therefore strongly recommend that specimens must minimally include the brainstem for rabies diagnosis for both DFAT and IHC testing.
An important caveat regarding IHC is that, although IHC is very sensitive and reliable in the demonstration of RABV RNP, formalin-fixation for at least 24 h is recommended, which can delay rabies post-exposure prophylaxis in human contact cases. 26 Fixation time can be reduced to a few hours if formalin-fixed samples are 2–4-mm thick and are subjected to oven or microwave heating.5,26 However, even with accelerated fixation, results cannot be made available on the same day as sample submission, as is possible with the DFAT or dRIT. A significant benefit of transporting rabies-suspect samples in formalin is the preservation of tissues for extended periods of time, which is important in cases in which samples are sent from remote rural areas in the absence of a reliable cold chain. 5 Formalin-fixed samples are also safe to handle by unvaccinated staff. IHC has the added benefits of combining positive labeling with the target cells (perikaryon of neurons), anatomic brain region (brainstem and cerebellum especially), and microscopic lesions typical for rabies, if present. If the IHC result is negative, alternative differential diagnoses can be investigated using the same specimens, and additional IHC tests can be requested. One drawback is that formalin masks antigens, making it difficult to isolate the virus in biological systems such as laboratory animals or cell culture. In formalin-fixed tissues, RNA is also degraded, making conventional RT-PCR methodologies unreliable. However, a RT-PCR assay has been performed successfully on formalinized brain tissue, and it was possible to confirm RABV infection and determine the phylogenetic relationships among RABV variants from different geographic areas. 5
In our study, we reported that IHC detects viral antigen within the perikaryon, axons, and dendrites of neurons in the CNS. However, in cases with abundant dust-like granules (“RABV dust”) viral antigen appeared to be extracellular. Previous research in laboratory animals has shown that, in the CNS, RABV primarily exhibits neurotropism, and that terminally viral antigen is often extremely widespread within nearly all neurons in the brain. 21 Additionally, RABV does not cause cytolysis, and nearly all virus is formed by budding on internal membranes of neurons, thereby trapping viral antigens within neurons.18,21 Virus transits between contiguous neuronal cell processes and perikarya, and significant amounts of viral antigen are not released into the extracellular space. 13 Possibly, cell lysis occurs in some species terminally, or postmortem autolysis compromises the integrity of axons and dendrites causing viral antigen to diffuse into the extracellular spaces. Ultrastructural studies beyond the scope of this paper would be necessary for confirmation.
A limitation of our study was that submitted specimens were a convenience sample of brain tissues that had been tested previously by DFAT. Technologists at the ARC-OVR rabies laboratory transferred as much material as they could, from as many of the 4 anatomic regions as could be identified, to formalin for sectioning and embedding at the FVS. Because the DFAT took precedence over the IHC, following South African government protocols for rabies diagnosis, a proportion of the samples that included hippocampus or cerebellum for the DFAT did not have these regions available for IHC. In addition, apart from domestic dogs and cattle, only small numbers, or even a single sample per species, were available for testing, suggesting the need to improve surveillance and specimen submissions in the country. Quality of tissue specimens also varied greatly, as a result of putrefaction or degradation, and specific anatomic areas of the brain critical for definitive rabies diagnosis (i.e., brainstem and cerebellum) were missing in some cases.
Despite these limitations, our results suggest that IHC using the EnVision polymer detection system is an accurate and repeatable method for rabies diagnosis in domestic and wild species that is especially well suited in situations when cold chain maintenance is problematic. Additionally, a diagnosis of rabies should not rely on one method alone; continuous validation using other methods should be performed. Thus, in clinical cases that are highly suspicious for RABV infection (especially in which there has been close human contact), a negative DFAT result should be supplemented by another RABV test, such as dRIT or IHC, to inform contact management.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387231154537 – Supplemental material for Diagnostic sensitivity and specificity of immunohistochemistry for the detection of rabies virus in domestic and wild animals in South Africa
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231154537 for Diagnostic sensitivity and specificity of immunohistochemistry for the detection of rabies virus in domestic and wild animals in South Africa by Drienie D. Claassen, Lieza Odendaal, Claude T. Sabeta, Geoffrey T. Fosgate, Debrah K. Mohale, June H. Williams and Sarah J. Clift in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Rephima Phaswane, Naomi Timmerman, and Xolani Mtshali for sectioning the slides and performing the IHC methods, and all of the field officials involved in rabies control in South Africa who assisted with sample collection for routine tests.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our funding was provided by the Free State Department of Agriculture and Rural Development and the South African Department of Agriculture, Fisheries and Forestry.
Supplemental material
Supplemental material is available online. Additional material in support of the results presented in this article is also available from the authors upon request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
