Abstract
Hamartomas are benign tumor-like lesions composed of disorganized growth of mature mesenchymal or epithelial tissues indigenous to the organ involved. Sporadically observed in ruminants, vascular, fibrous, nasal, and pulmonary hamartomas have been reported in calves; pulmonary and cutaneous forms have been reported in sheep. A full-term elk calf found dead had a large intrathoracic mass replacing the left caudal lung lobe and compressing other thoracic organs. Histologically, cross- and tangential sections of bronchi were separated by collagenous mesenchyme and irregularly shaped canaliculi and saccules resembling terminal bronchioles. Rarely present were regions in which saccules, lined by simple cuboidal epithelium, transitioned into attenuated epithelium lining fully developed alveoli. These findings are consistent with a pulmonary hamartoma. To our knowledge, pulmonary hamartoma has not been reported previously in a non-domestic ruminant.
Hamartomas are defined as benign tumor-like lesions composed of disorganized growth of mature mesenchymal or epithelial tissues indigenous to the organ involved. 9 They are noninvasive and confined to a limited anatomic field. 14 A hamartoma differs from a choristoma, which is a benign tumor-like lesion composed of normal tissue in an abnormal location. 2 Once thought of as developmental malformations and not true neoplasms, clonal chromosomal aberrations acquired through somatic mutations have been demonstrated in some human hypothalamic hamartomas.11,17 Hamartomas are observed sporadically in ruminants. 19 In calves, vascular, fibrous, nasal, and pulmonary hamartomas have been reported.9,14,16,18,20,24 Pulmonary and cutaneous forms have been reported in sheep.1,13 Glioneuronal hamartomas have been reported in goats. 26 When present as congenital lesions, hamartomas may be accompanied by other defects, 2 the result of errors in embryogenesis.12,16
Historically, the terminology associated with pulmonary hamartomas in humans and animals has been confusing, with numerous synonymous or similar conditions, 22 such as accessory lungs, congenital adenomatoid malformation, bronchial hypoplasia, and adenomatoid hamartoma.16,23 In domestic animals, pulmonary hamartomas have been described as being of cartilaginous, vascular, bronchiolar, or alveolar origin. 21 In cattle, hamartomas are often observed as a congenital abnormality, and have been observed in both aborted and full-term calves.14,16,23 In such cases, the majority of affected calves are born 6 mo to 4 wk before the expected date of birth. 16
In humans, pulmonary hamartomas are the most common benign tumors of the lung and are generally recognized as incidental findings during routine radiographic studies. They have been described as small, 2–5-cm masses in the peripheral parts of the lung,2,8 and are found in ~0.25% of all patients at autopsy. 8 Larger pulmonary hamartomas in humans and animals are often confluent with residual pulmonary tissue at the hilus and replace a portion of the lung. 16 A commonly reported complication of large pulmonary hamartomas is cardiovascular malfunction resulting from external compression of the heart, caudal vena cava, pulmonary artery, or aorta. 23
An elk (Cervus elaphus canadensis) calf was found dead in a pasture containing a group of 13 mature (3-y-old), bred, captive elk cows. External examination revealed no abnormalities, and the hair coat and skin were normal for a full-term calf. The right lung lobes and left cranial lobe were present but were deep-red and atelectatic, consistent with uninflated fetal lung, suggesting that the calf was either stillborn or unable to breathe at birth. Confluent with the left cranial lung lobe, transitioning from the atelectatic left cranial lobe, and in place of the left caudal lung lobe, there was a large (19 × 10 × 8 cm), irregular, firm, heavy, expansile mass covered by opaque parietal pleura. The mass occupied >50% of the thoracic cavity, and compressed the right lungs, left cranial lung lobe, heart, and mediastinum. There was no evidence of invasion of adjacent lung lobes. On cut surface of the mass, cross-sections of variably sized airways surrounded by cartilage (bronchi) were separated by red atelectatic lung tissue and connective tissue (Fig. 1).
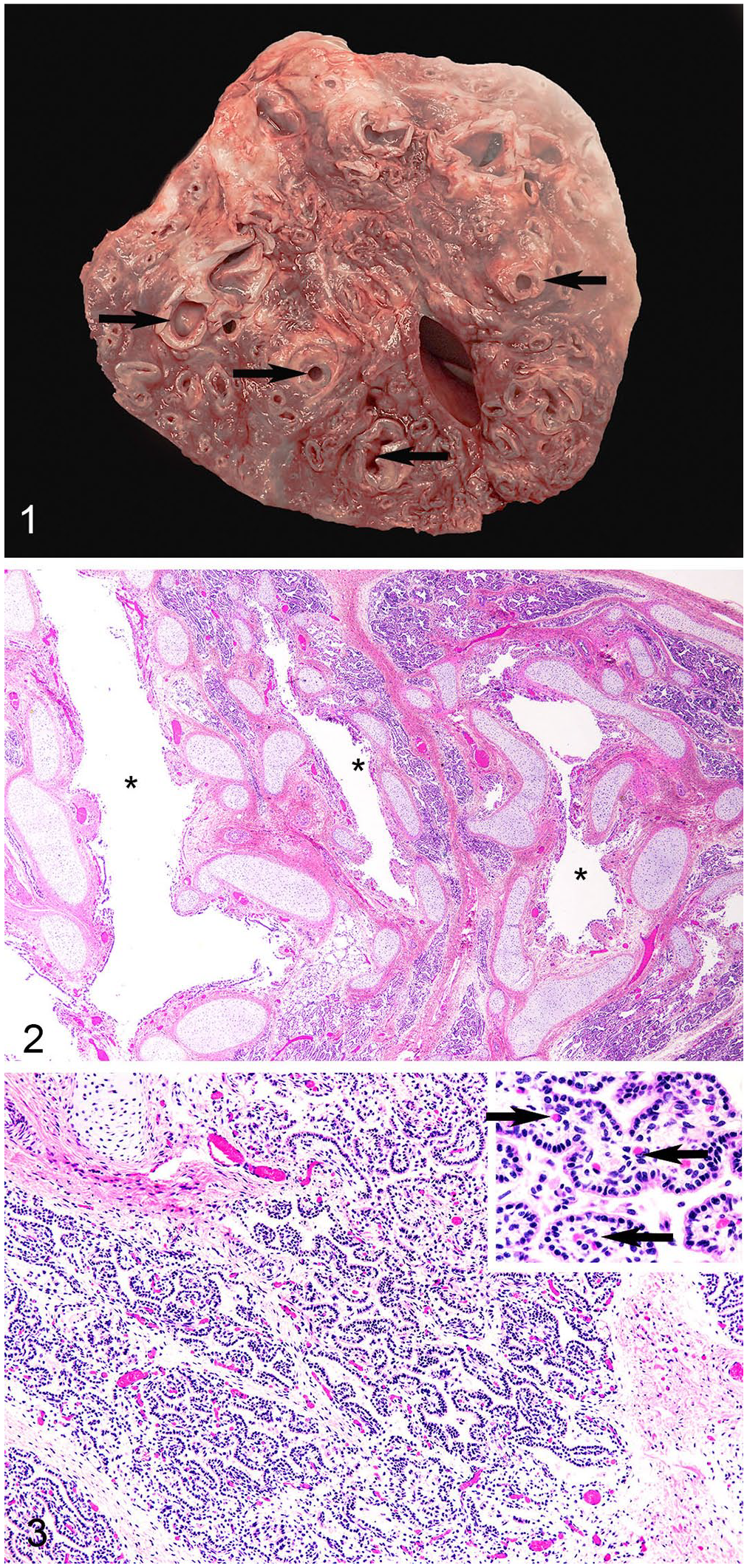
Pulmonary hamartoma in an elk calf.
Microscopically, cross-sections and tangential profiles of haphazardly arranged bronchi (Fig. 2) in the mass were lined by simple columnar-to-pseudostratified ciliated epithelial cells. Bronchi were separated by a moderately cellular collagenous mesenchymal matrix with well-developed vasculature. Within the collagenous matrix were many irregularly shaped canaliculi and saccules resembling terminal bronchioles (Fig. 3), lined by non-ciliated, simple cuboidal epithelium, most with basal nuclei and abundant eosinophilic cytoplasm. Subjacent to this epithelial layer was moderately cellular stroma containing numerous capillaries, venules, and arterioles. Rarely present were regions in which saccules, lined by simple cuboidal epithelium, transitioned into attenuated epithelium lining fully developed alveoli.
During development, from conception to parturition, mammalian lungs pass through 5 recognized phases: embryonic, pseudoglandular, canalicular, saccular, and alveolar. 4 In cattle and sheep, the alveolar phase appears at ~230 and 120 d of gestation, respectively. 4 In spite of the full-term status of this elk calf, which was 240–260 d, the pulmonary hamartoma contained areas consistent with canalicular, saccular, and alveolar developmental phases.
The heart was noticeably firm. A cross-section midway between the apex and coronary sulci revealed proportionate thickening of both ventricular free walls and the ventricular septum. Multifocally, within hypertrophic cardiac muscle were small, irregular streaks of pallor. There was slight dilation of both right and left atria. Compression of the aorta and pulmonary artery by the large hamartoma likely resulted in an increase in pressures experienced by the ventricles with resultant compensatory cardiac hypertrophy. Microscopically, cardiomyocytes in various regions were thin, with vacuolated eosinophilic cytoplasm and small hyperchromatic nuclei. Elsewhere, cardiomyocytes were large, with abundant eosinophilic fibrillar cytoplasm and large vesicular nuclei. Within the interstitial adipose tissue and separating some myofibers were infiltrates of low-to-moderate numbers of lymphocytes and polymorphonuclear leukocytes. Grossly, the liver had an accentuated lobular pattern. Microscopically, there was centrilobular congestion and hemorrhage. Centrilobular hepatocytes were either necrotic or degenerate with vacuolated cytoplasm and condensed nuclei consistent with chronic passive congestion.
Few studies of morbidity and mortality among farmed elk have been conducted.6,7,10,15 In one survey of farmed elk submissions to pathology laboratories in Canada and the United States over a 10-y period, neoplasia was listed as a primary diagnosis in 13 of 1,077 (1.2%) submissions; congenital anomalies were not listed. 25 In other published reports, neither neoplasia nor congenital anomalies have been recorded as causes of morbidity or mortality in elk. 25 Reported congenital anomalies in the closely related red deer (Cervus elaphus) have included deformed limbs, dicephalism, microphthalmia, and dermoid cysts.3,5 In our case, the large size of the pulmonary hamartoma resulted in compression of the heart and resultant cardiovascular malfunction and was the likely cause of death.
Footnotes
Acknowledgements
We thank the National Animal Disease Center’s Animal Research Unit personnel, Caitlyn Ehrlich, Jacob Fritz, Jonathan Gardner, Kolby Stallman, Derek Vermeer, and Dr. Rebecca Cox, for their support with animal care and husbandry. A special thanks to Kolby Stallman for his efforts in animal care and during elk calving season.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We received financial support from USDA CRIS 3625-32000-234 and CRIS 3625-32000-235. Our research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. USDA is an equal opportunity provider and employer.
