Abstract
A 12-y-old Himalayan black bear suddenly developed depression, anorexia, cough, and dyspnea and died at the Ordos Zoo, China. At autopsy, the mesenteric and cranial mediastinal lymph nodes (LNs) were enlarged; the largest cranial mediastinal LN was ~13 cm in diameter. Scattered-to-diffuse, rounded-or-oval, gray, firm 1–6-mm nodules were observed on the surfaces of the spleen, liver, lungs, and small intestine. Histologically, the enlarged cranial mediastinal and mesenteric LNs, spleen, small intestine, lungs, and liver contained dense populations of neoplastic lymphoid cells (NLCs). The NLCs were round-or-oval with small amounts of mildly eosinophilic cytoplasm and round-or-oval hyperchromatic nuclei with indistinct nucleoli; the mitotic count was 55 in 2.37 mm2. Immunohistochemically, cell membranes and the cytoplasm of NLCs were CD3+, CD79a−, CD20−, CD15−, CD30−, and CD45RA−; hence, the NLCs were derived from T lymphocytes. To our knowledge, T-cell lymphoma has not been reported previously in a Himalayan black bear.
A 12-y-old Himalayan black bear (syn. Manchurian black bear; Ursus thibetanus), a subspecies of black bear (Ursus thibetanus ussuricus) in Northeast China, moved freely with 20 other bears in a bear house at the Ordos Zoo in Ordos, China. The animal was in contact with humans only in areas where additional food was provided. On March 17, 2016, the bear suddenly appeared depressed and anorectic, and had undigested fruit skins in the feces, a cough, and hyperpnea. After 2 wk, while climbing a ladder in the playground, it experienced dyspnea, hyperpnea, and cyanotic lips. The attending veterinarian used a blow gun to administer anesthetic (xylazine hydrochloride) to the bear for inspection and treatment at the bear house. Various antibiotics (florfenicol, kanamycin, amikacin), antivirals (interferon), stomach drugs (Jianwei Xiaoshi tablet, lacidophilin tablet), and vitamin C were administered. Nevertheless, the clinical signs persisted, and the bear died 38 d after the onset of disease.
An autopsy was performed within an hour of the bear’s death, after obtaining permission from the relevant departments. The bear was well nourished, with large fat stores. The systemic subcutaneous loose connective tissue was yellow and gelatinous because of severe edema. The thoracic cavity contained a large amount of cloudy fluid. The cranial mediastinal lymph nodes (LNs) were enlarged; the largest LN was ~13 cm in diameter (Fig. 1A), with a gray-white, homogeneous, moist, and soft cut surface. A gray-white mass ~0.5 cm thick was attached to the ventral side of the trachea in the cranial mediastinum. Furthermore, the pleural surface dorsal to the heart was attached to an irregular and long 15 × 2 × 2 cm mass, with nodules on the surface. The right side of the caudal mediastinum was thickened and was covered with gray-white miliary nodules. Scattered-to-diffuse, rounded, and elliptic or irregularly rounded gray-white solid foci were observed on the surface and section of the cranial and ventral caudal lobes of the left and right lung. A few large foci were centered on small bronchi. The mesenteric LNs were gray-white and massively enlarged (Fig. 1B), with a gray-white, homogeneous, moist, and soft cut surface. The wall of the small intestine was thickened segmentally and gray-white. The liver was slightly enlarged and dark red, with many gray foci of ~1-mm diameter on the surface. The spleen was enlarged with scattered-to-diffuse, gray, miliary foci on the surface and section. Tissue samples were fixed in 10% neutral-buffered formalin and processed routinely; sections were stained with H&E for histologic examination.
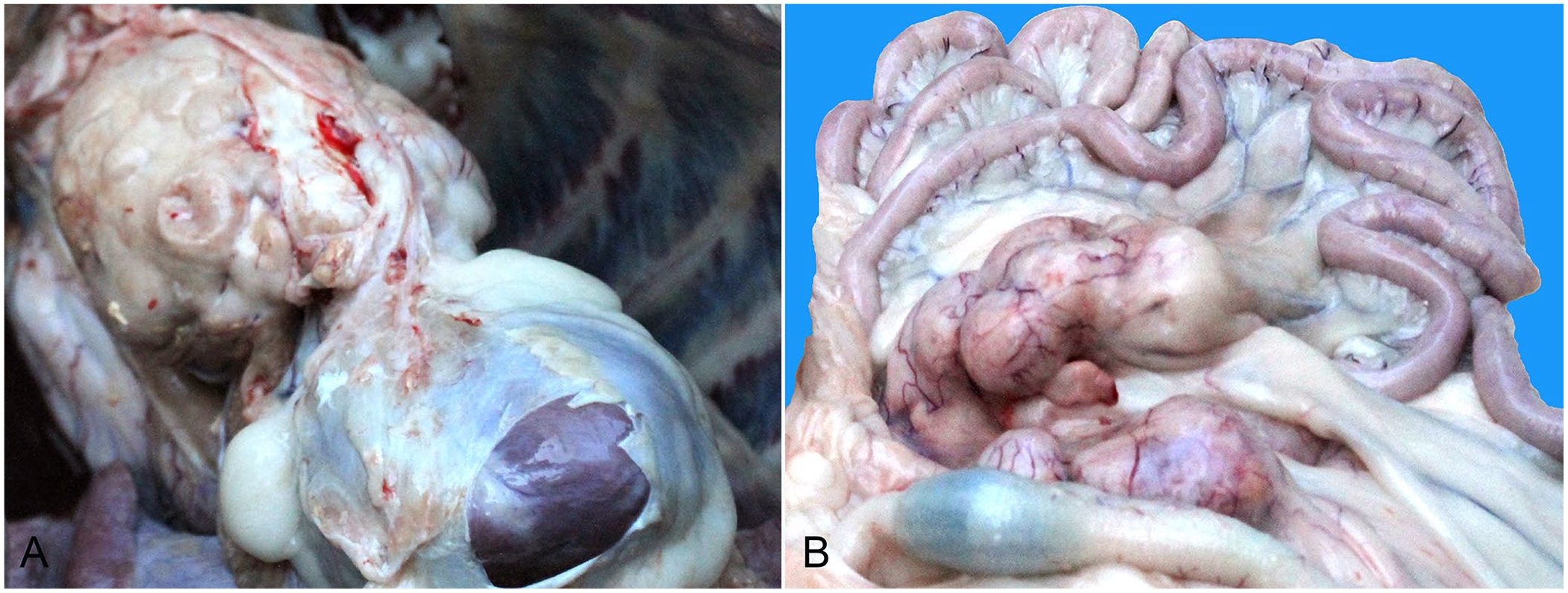
Gross features of T-cell lymphoma in a Himalayan black bear.
Histologically, the normal architecture of the enlarged cranial mediastinal and mesenteric LNs was effaced by a dense population of neoplastic lymphoid cells (NLCs; Fig. 2A). In some regions of the spleen, the normal architecture was replaced with a dense population of NLCs; moreover, reduced white pulp was observed occasionally. In other regions, the boundary between the white and red pulp was clear, and the NLCs were centered on trabeculae or scattered in the red pulp. Several macrophages containing hemosiderin, and sporadic megakaryocytes, were noted in the red pulp. In the lungs, tumors of different sizes and shapes with densely arranged NLCs surrounded the bronchioles and small vessels. Prominent folds of bronchial mucosa protruded into the lumen owing to the NLCs in the lamina propria. A dense population of NLCs was also present in the subpleural lung tissue. Nests of NLCs were present in the interlobular connective tissue of the liver. Moreover, scattered NLCs or some small nests were observed in the sinusoids and the Disse spaces that were dilated by shrinkage of hepatic cords in formalin (Fig. 2B). Several Kupffer cells contained hemosiderin in their cytoplasm. Microvesicular steatosis of the hepatocytes was evident. NLCs invaded the mucosa, submucosa, and muscle layer of the small intestine. Also, NLCs were observed in small veins within the lungs, spleen, LNs, small intestine, and liver. The NLCs were round-or-oval with a small amount of mildly eosinophilic cytoplasm and round-or-oval hyperchromatic nuclei with indistinct nucleoli; the mitotic count was 55 in 2.37 mm2 (Fig. 2C). Most cardiomyocytes had mild granular degeneration. Evident granular degeneration, sporadic necrosis of the renal tubular epithelium, and the presence of a few red homogeneous protein droplets within Bowman spaces were noted.
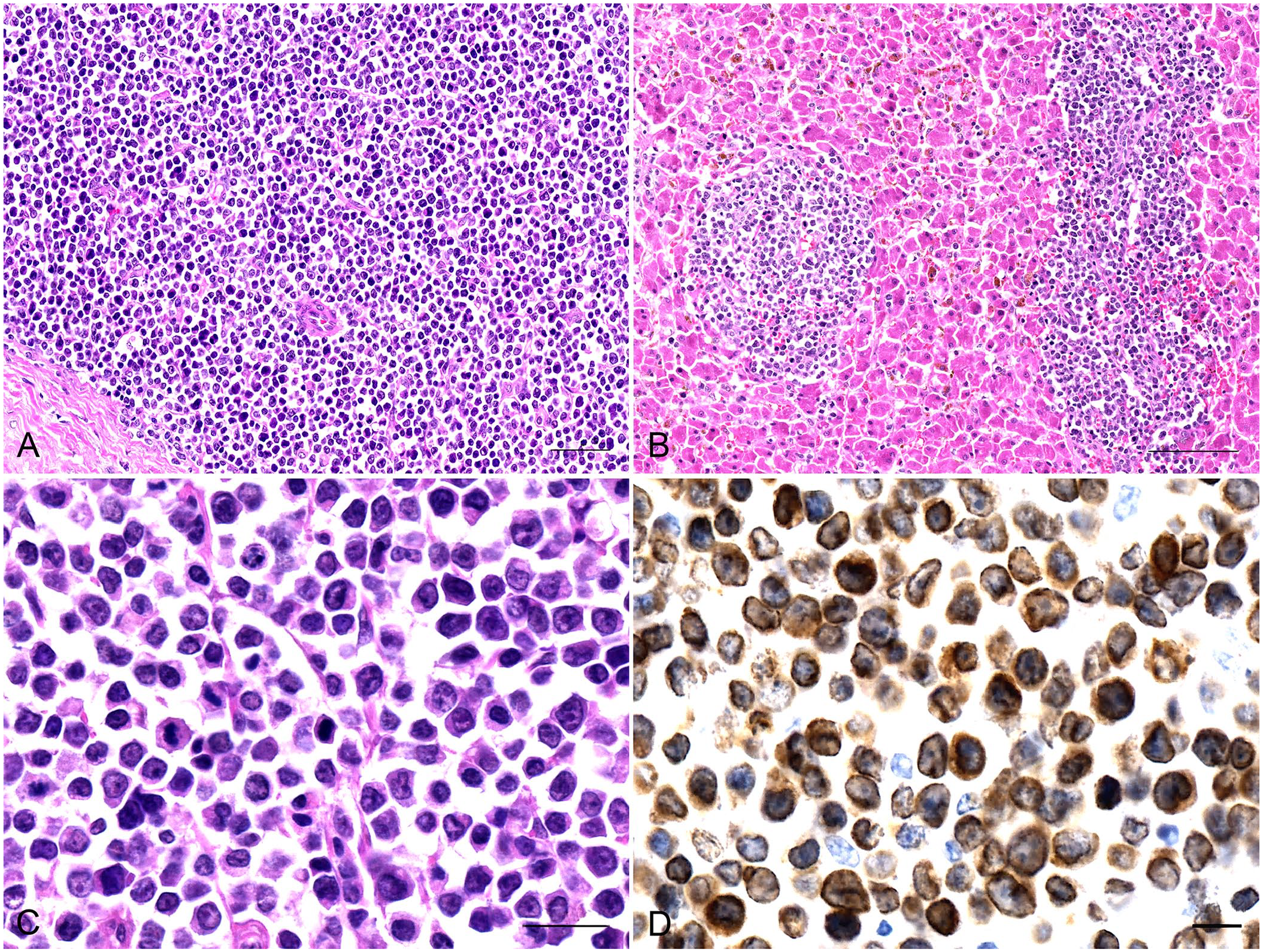
Histologic and immunohistochemical features of T-cell lymphoma in a Himalayan black bear.
Immunohistochemical (IHC) analysis with diaminobenzidine staining, and with all monoclonal antibodies (mAbs; ZSGB-BIO), showed positive reactions for the mAb CD3 (ZM-0417; Fig. 2D) on the cell membrane and in the cytoplasm of NLCs. Negative reactions were observed for the mAbs CD79a (ZA-0293), CD20 (ZM-0039), CD15 (ZM-0037), CD30 (ZM-0043), and CD45RA (ZM-0053), which indicated that the NLCs were derived from T lymphocytes.
Limited studies have been performed on naturally occurring tumors in free-ranging Himalayan black bears. The tumors recorded include biliary carcinomas,9,14 hepatic adenocarcinoma, hepatocellular carcinoma, nasal adenocarcinoma, esophageal squamous cell carcinoma, pancreatic adenoma, pancreatic adenocarcinoma, lymphosarcoma, 3 and lymphoma.3,7 Additionally, 2 cases of lymphoma have been recorded in other bears (grizzly bear, 2 brown bear 14 ). Lymphoma is one of the most common tumors in small animals, and accounts for 7–24% of all tumor cases in dogs. 13
Lymphoma can be categorized into B-cell lymphomas (BCLs), T-cell lymphomas (TCLs), and putative NK-cell lymphomas. 8 In canids, the most common type of lymphoma is BCL; TCL accounts for 17–28% of lymphomas. 10 However, in horses, TCL is more common than BCL. 8 Moreover, lymphoma has become a common and increasingly noted health problem in African lions (Panthera leo); in 1 report, 10 African lions had TCL and 1 had BCL. 5 Four cases of lymphoma in bears have been reported outside China.1–3,14 In China, one case of BCL has been documented in a black bear 7 ; however, the pathologic and immunohistochemical characteristics of TCL in bears remain unreported, to our knowledge.
The tumor cells in different types of TCL can vary markedly in size and shape.5,8,10 Precursor T-lymphoblastic lymphoma/leukemia (T-LBL/ALL) is a neoplasm of T-cell lineage composed of small-to-medium blast cells, with scant cytoplasm, finely condensed chromatin, and indistinct nucleoli. 12 CD3 is the stable specific marker of neoplasms of T-cell lineage.10,12 The features of the NLCs in our case were similar to the tumor cells of T-LBL/ALL, 12 and CD3 was positive in the cytoplasm of the NLCs. Based on our results and previous studies,4,5 our case was diagnosed as TCL. To the best of our knowledge, TCL has not been reported previously in a bear.
According to the gross histologic findings in our Himalayan black bear, NLCs most probably originated in the cranial mediastinal LNs or thymus 12 and then spread to the lungs, liver, spleen, and other organs hematogenously. The factors causing lymphoma in most animals are yet to be determined. Some studies have shown that the environment, breed, age, infection, and immune status influence the incidence of lymphoma.6,11 Information regarding lymphoma in bears is limited; furthermore, optimal treatment and strategies to prevent or control the disease are yet to be established.
Footnotes
Acknowledgements
We thank Peng Wang at Ordos Zoo for his help with the initial autopsy, and Xia Cao for optimizing and performing immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by the Inner Mongolia Agricultural University High-level Talents Research Initiation Fund Project (NDYB2019-3, NDYB2018-5), Major Science and Technology Projects of Inner Mongolia Autonomous Region (2021ZD0012), National Natural Science Foundation of China (31860698), Inner Mongolia Grassland Innovative Talent Team Project (20151031), and Program for Improving the Scientific Research Ability of Youth Teachers of Inner Mongolia Agricultural University.
