Abstract
A 4-year-old, male, neutered cat was referred because of recurrent episodes of dyspnea. Physical examination revealed a harsh systolic murmur, with the point of maximal intensity in the left heart base, with an intensity of 4 out of 6. Echocardiographic diagnosis was severe supravalvular pulmonary artery stenosis. A selective right ventricular angiocardiogram showed an absence of arterial blood flow to the left lung lobes. A balloon dilatation of the localized stenosis of the right pulmonary artery was attempted with cardiac catheterization. However, when the catheter was passed through the stenosis, the blood flow to the lungs temporarily completely ceased, which led to death. Postmortem examination revealed a circumscribed stenosis of both pulmonary arteries at the site of the bifurcation, where the ligamentum arteriosum was attached. Histopathology showed that the localized ridge-like stenosis at the pulmonary artery bifurcation was caused by connective tissue. The suspected cause of this congenital anomaly is the presence of ectopic ductal tissue in the wall of the pulmonary artery. When the ductus arteriosus closes at birth, pulmonary artery stenosis developed because of constriction of the ectopic ductal tissue.
A 4-year-old, 3.5-kg, male, neutered European shorthair cat was referred to the Companion Animal Clinic (Utrecht, The Netherlands) for further evaluation of a cardiac murmur and recurrent episodes of dyspnea. During the last event, an overnight hospitalization in an oxygen cage and treatment with parenteral furosemide and oral ramipril by the referring veterinarian resulted in rapid resolution of the clinical signs. As a kitten, the cat was found in the street in Spain and was brought to The Netherlands. During the first health check by the referring veterinarian, a loud cardiac murmur was auscultated, but at that time no further investigation was done. The cat was neutered under medetomidine and isoflurane anesthesia, without any complications.
At presentation, the cat was bright, alert, and responsive. No sign of dyspnea was seen. Physical examination revealed a respiratory rate of 20 breaths/min, a pulse rate of 140 beats/min, and a rectal temperature of 39.1°C. Cardiac auscultation revealed a harsh systolic murmur, with the point of maximal intensity at the left heart base. The intensity of the murmur was 4 out of 6. Before presentation, the cat had been treated with 10 mg furosemide once daily and 0.625 mg ramipril once daily for 6 days.
Thoracic radiographs (Fig. 1) showed severe generalized cardiomegaly and a distended caudal vena cava. The pulmonary vessels were within normal limits. Multiple mineralized structures were seen in the lung fields. The largest structure was approximately 1 cm × 1 cm and was located in the dorsal aspect of the left caudal lung lobe. The second structure was smaller and located in the right cranial lung lobe. A small, round, soft tissue opacity was seen just dorsal to the second and third sternebrae, most likely representing focal fat accumulation. These findings were thought to be clinically irrelevant coincidental findings.
To identify the cause of the murmur, echocardiography was performed. Echocardiography showed severe concentric and eccentric hypertrophy of the right ventricle with flattening of the interventricular septum (Fig. 2). The right atrium was severely dilated. The pulmonary valve appeared normal; however, the pulmonary trunk was uniformly dilated (12 mm; Fig. 3A). Severe pulmonary artery stenosis with a diameter of approximately 1 mm was identified 14 mm distal to the pulmonary valve. Distal to this stenosis, a severely dilated right pulmonary artery (RPA) was recognized (Fig. 4A). The left pulmonary artery (LPA) could not be visualized. Color flow Doppler examination revealed mild tricuspid valve insufficiency. Pulsed-wave Doppler examination illustrated decreased peak systolic blood flow velocity across the pulmonary valve (Fig. 3B). Continuous wave Doppler echocardiogram of the supravalvular pulmonary stenosis revealed a peak systolic blood flow velocity of at least 4.8 m/sec, which is compatible with a systolic pressure gradient of at least 90 mm Hg across the pulmonary trunk and the RPA. A trivial amount of pleural effusion and a congested liver were also detected. The diagnosis of congestive right-sided heart failure secondary to a severe supravalvular pulmonary artery stenosis was made.
The indication of balloon dilatation of the supravalvular stenosis was discussed with the owner. In addition to the possible risks and complications of general anesthesia and the cardiac catheterization with balloon dilatation, the limited worldwide experience in cats and uncertain outcome were discussed. 7 As a result of the supravalvular nature of the stenosis, a fibrotic ring was expected to be the cause of the stenosis. 6 Therefore, a stent placement after balloon dilatation was planned to reduce the chance of a restenosis, as described in the veterinary literature. 6 An interventional pediatric cardiologist (MWF) supervised and assisted with the procedure.
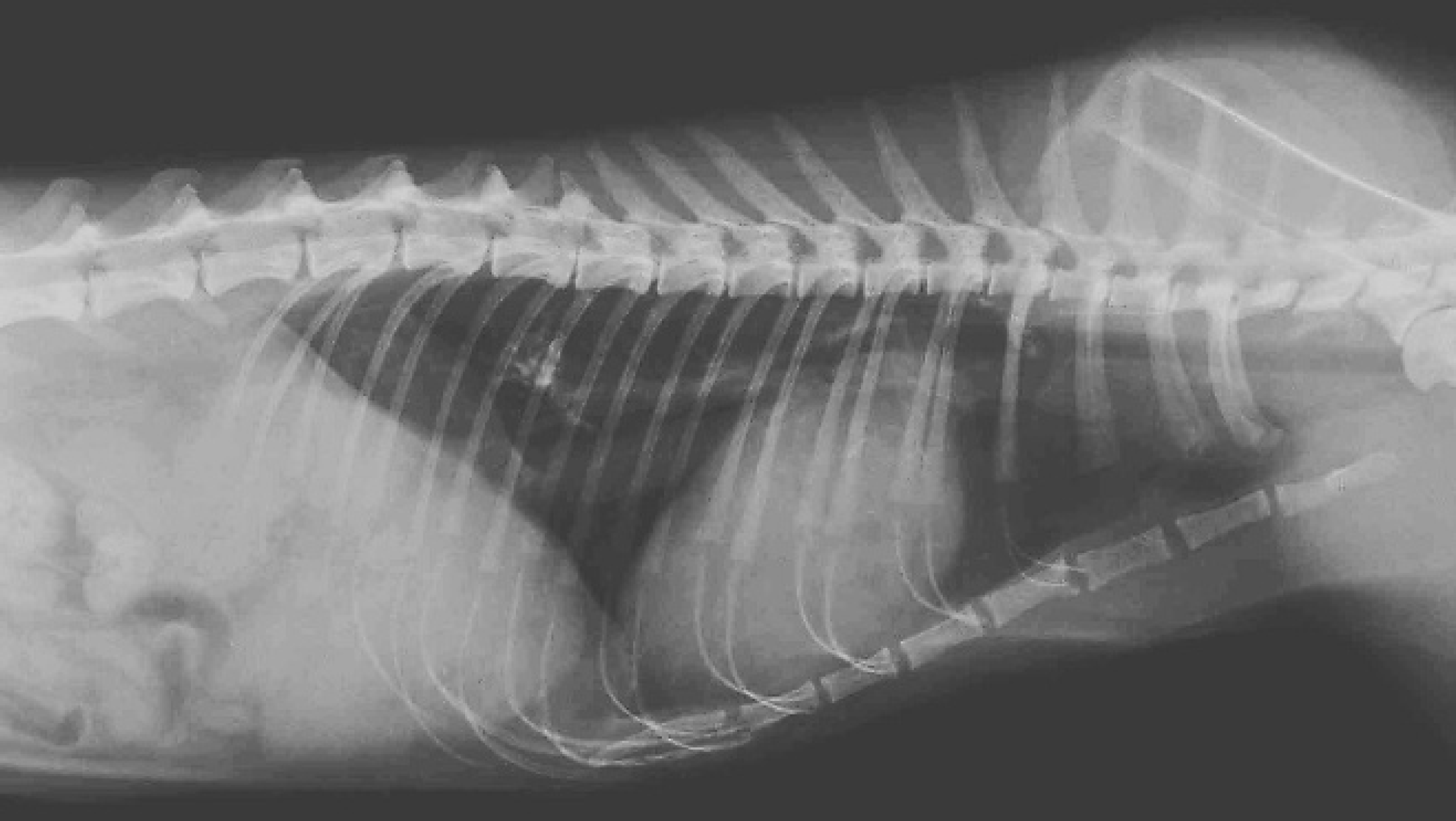
Laterolateral thoracic radiograph shows a generalized cardiomegaly. The caudal vena cava is distended, and the liver is uniformly enlarged. There is a small amount of pleural effusion. There are 2 mineralized nodular lesions in the lungs with ill-defined margins.
For the cardiac catheterization, the cat was premedicated with glycopyrrolate (35 μg intramuscularly) and methadone (0.70 mg intravenously [IV]). The anesthesia was induced with propofol (17 mg IV). After endotracheal intubation, fentanyl (IV) and sevoflurane (1.1–2 kPa inhalation) were used for anesthesia maintenance. Lactated Ringer was given (4 ml/kg/hr IV) as maintenance fluid. Pulse oximetry, noninvasive oscillometric blood pressure measurement, continuous electrocardiogram (ECG), and capnography were used to monitor the vital parameters during anesthesia. The cat was positioned in dorsal recumbency on the fluoroscopy table, and the left and right femoral veins were isolated with surgical cut-down. A 5 French introducer was secured in both femoral veins. Through the right introducer, a multifunctional end-hole catheter (4 French) with 2 side-holes was advanced to the right ventricle. The mean right atrial pressure was 16 mm Hg, and the right ventricular pressure was 68/12 mm Hg. A right ventricle angiography with 4-ml contrast medium a in the ventrodorsal projection was performed by rapid hand injection. The LPA was absent on the angiocardiogram, whereas the RPA was clearly visible. The catheter was advanced through the pulmonary valve, and the contrast study was repeated (injecting 2.5 ml contrast), which confirmed the absence of a patent LPA (Fig. 5). A straight guidewire (0.018 inch) was placed through the stenosis into the RPA. The catheter was advanced over the wire into the distal RPA. During manipulation of the catheter, a right bundle branch block and solitary ventricular premature complexes occurred on the ECG, and while the RPA stenosis was obstructed with the catheter, a sudden drop of hemoglobin oxygen saturation (SpO2 dropped from 93% to 77%) and expired CO2 (ETCO2 dropped from 5.6 to 3.8 kPa) were noted. Intravenous dopamine and 100% oxygen ventilation were started with discontinuation of sevoflurane. This was followed by the appearance of an idioventricular rhythm with very broad and aberrant QRS complexes with a regular frequency of 100/min on the ECG. Though the catheter was immediately pulled back into the right ventricle, the cardiac rhythm deteriorated into ventricular fibrillation within a couple of minutes. Despite the injection of adrenaline and lidocaine into the right ventricle and other reanimation efforts, the cat died.
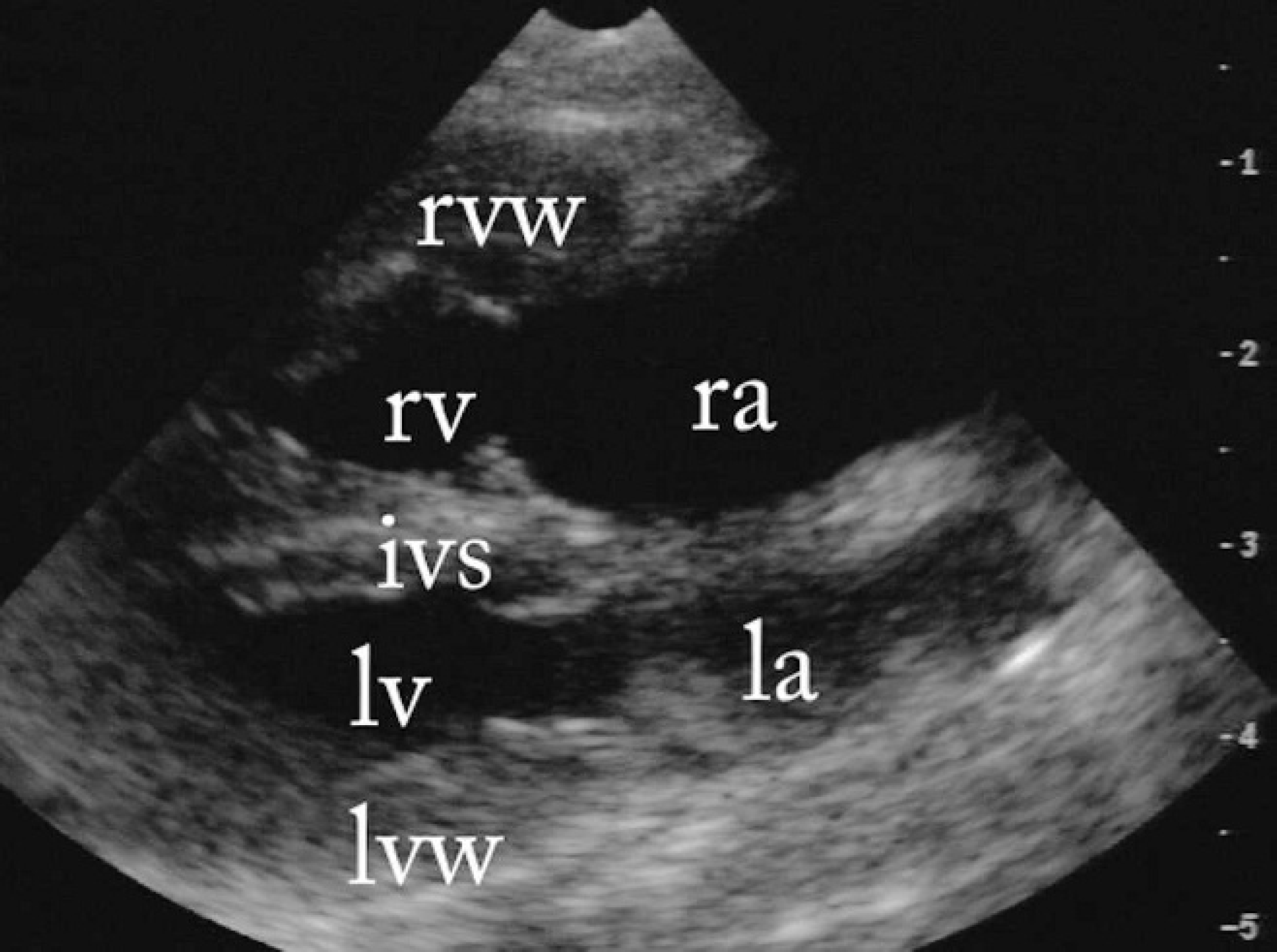
B-mode 4-chamber echocardiographic image of the heart from a right parasternal longitudinal view. The right ventricle has a thickened wall (rvw) and a dilated lumen (rv). The right atrium (ra) is markedly enlarged. lv = left ventricular lumen; la = left atrium; ivs = interventricular septum; lvw = left ventricular free wall.
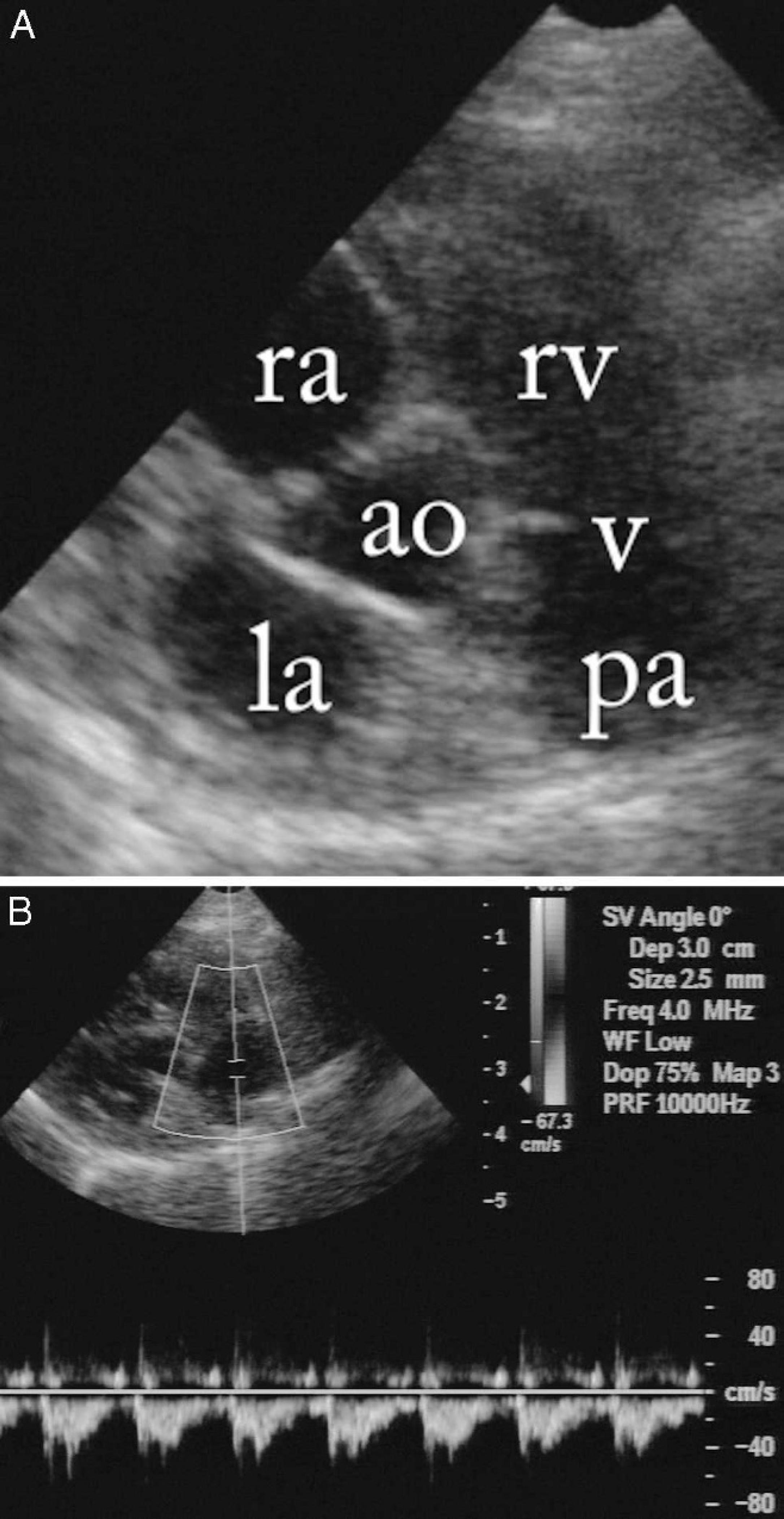
B-mode cross-section echocardiographic image of the heart base from a right parasternal short-axis view. The main pulmonary artery (pa) is uniformly dilated compared to the aorta (ao). v = pulmonary artery valves; rv = right ventricle; ra = right atrium; la = left atrium.
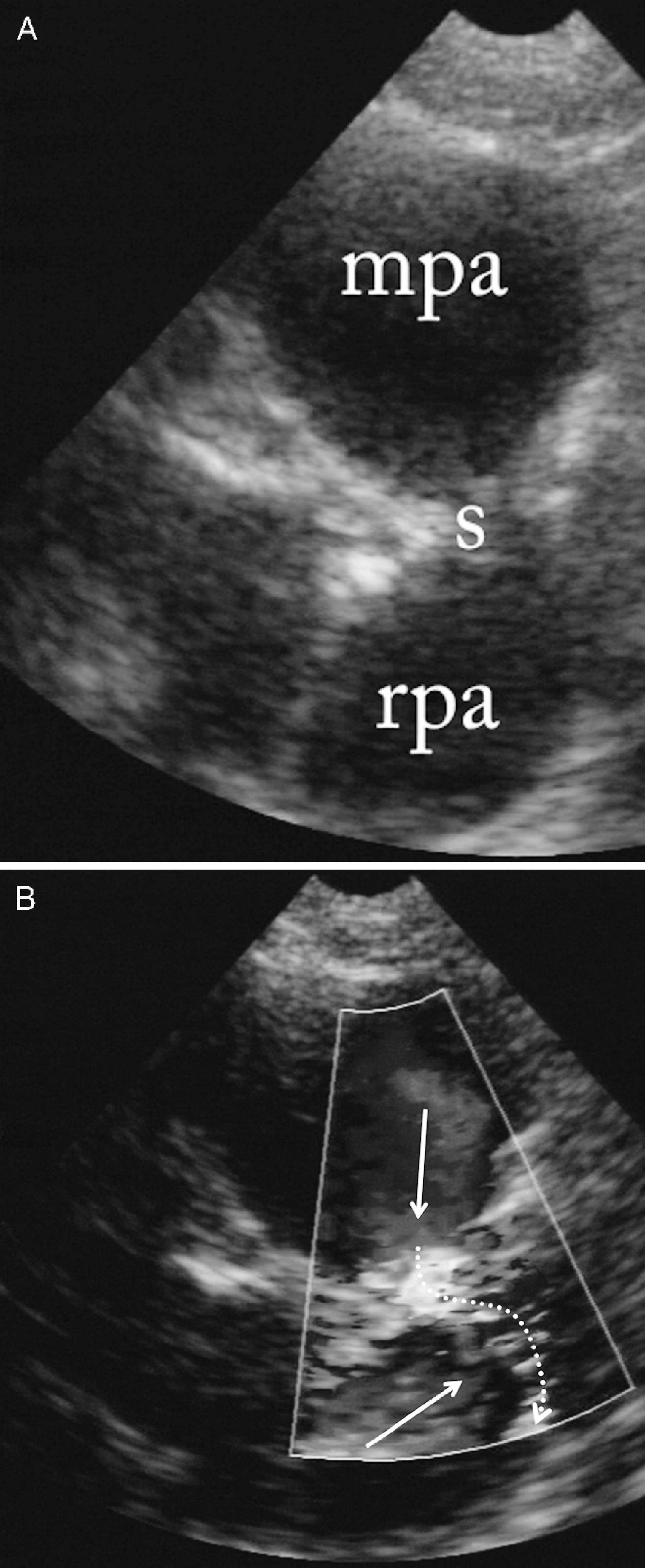
Cross-sectional echocardiographic images of the heart base from a right parasternal short-axis view.
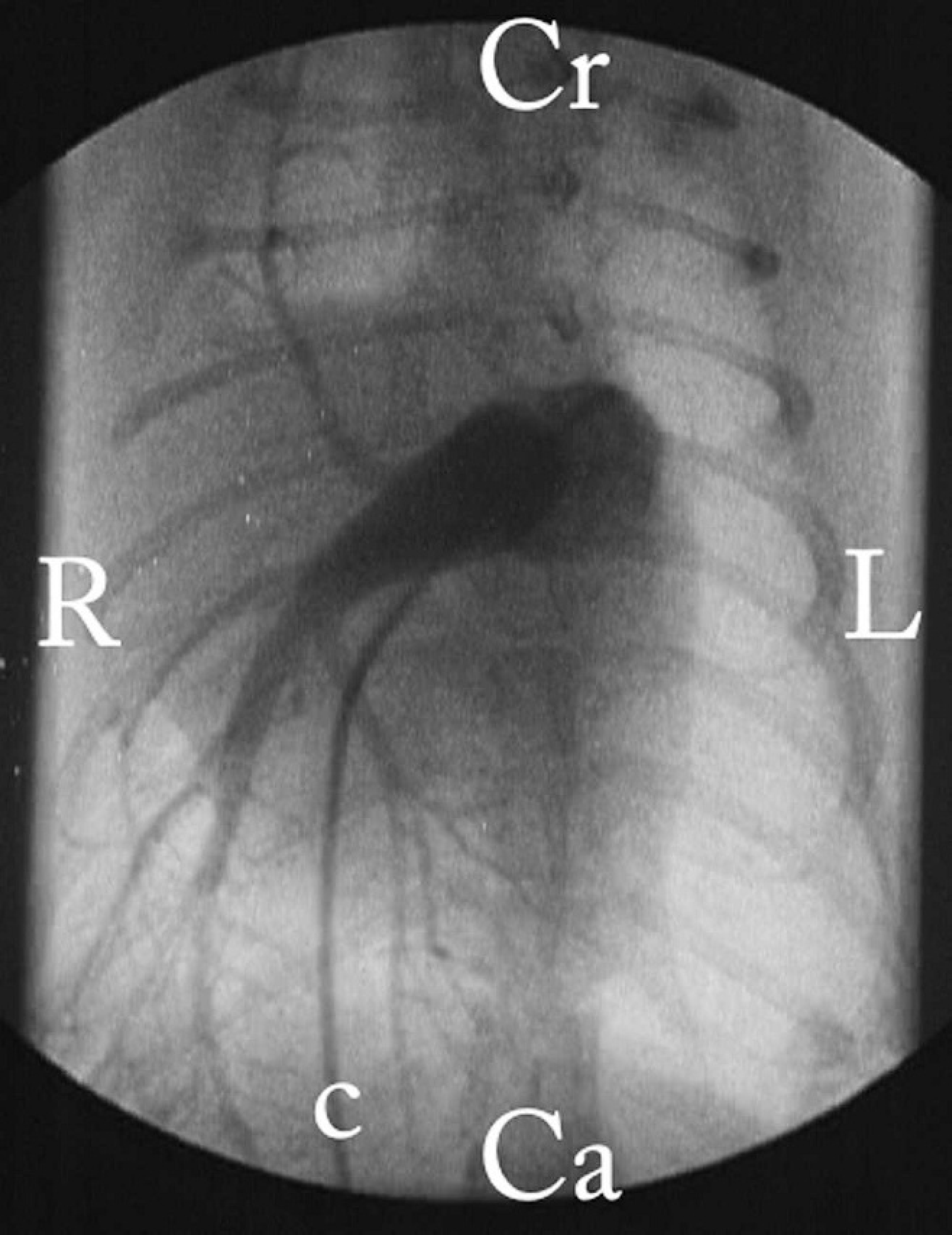
Selective angiocardiogram of the main pulmonary artery in a ventrodorsal view. The catheter (c) was inserted via a femoral vein and was advanced through the caudal vena cava, right atrium, and right ventricle into the main pulmonary artery. After the manual injection of 4 ml of iodinated contrast material, the main and the right pulmonary arteries do appear, but the left pulmonary artery does not appear on the angiogram, indicating either the absence or complete obstruction of this vessel. R = right; L = left; Cr = cranial; Ca = caudal.
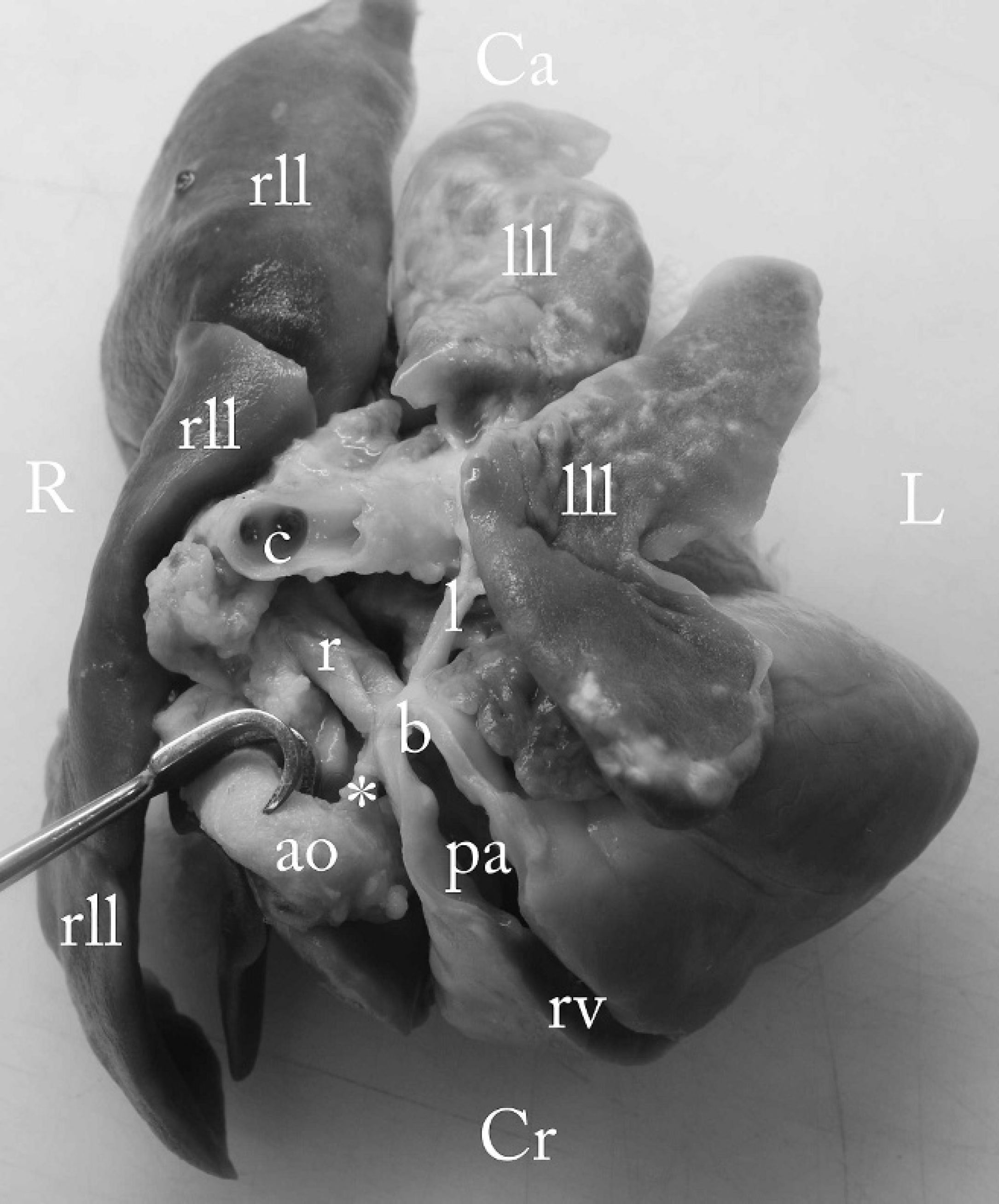
Formalin-fixed heart and lung lobes from a dorsoventral view. The main pulmonary artery (pa) and the right ventricle (rv) are opened with a straight incision. The left lung lobes (lll) are noticeably smaller when compared to the right lung lobes (rll) and contain multifocal subpleural pale yellow granules. Likewise, the left pulmonary artery (l) is much smaller than the right (r) one. By pulling the aorta (ao) to the side, the ligamentum arteriosum (*) is exposed. Note that the ligamentum arteriosum connects the aorta with the pulmonary artery exactly at the point of the bifurcation (b), where the circumscribed stenosis of both pulmonary arteries is located. R = right; L = left; Cr = cranial; Ca = caudal; c = carina, trachea.
A postmortem examination confirmed the presence of the cardiac anomalies seen on the echocardiography and angiocardiography. The RPA had a severe circumscribed stenosis by a ridge-like structure at the level of its origin, and a severe poststenotic dilation was noted (Fig. 6). The LPA was present; however, at its origin (as was the case with the RPA) a circumscribed stenosis by a ridge-like structure was found, which resulted in a subtotal atresia. The opening of the LPA was not patent. Distal to the localized stenosis, a normal diameter of the LPA was present. The localization of the stenoses of both pulmonary arteries was at the proximal part of the RPA and the LPA (i.e., at the insertion site of the ligamentum arteriosum; Fig. 6). In the wall of the main pulmonary artery, hard rounded plaques of several millimeters in diameter were noticed. The left lung lobes were about half the size of the right ones. In the left caudal lobe, a hard nodule was found.
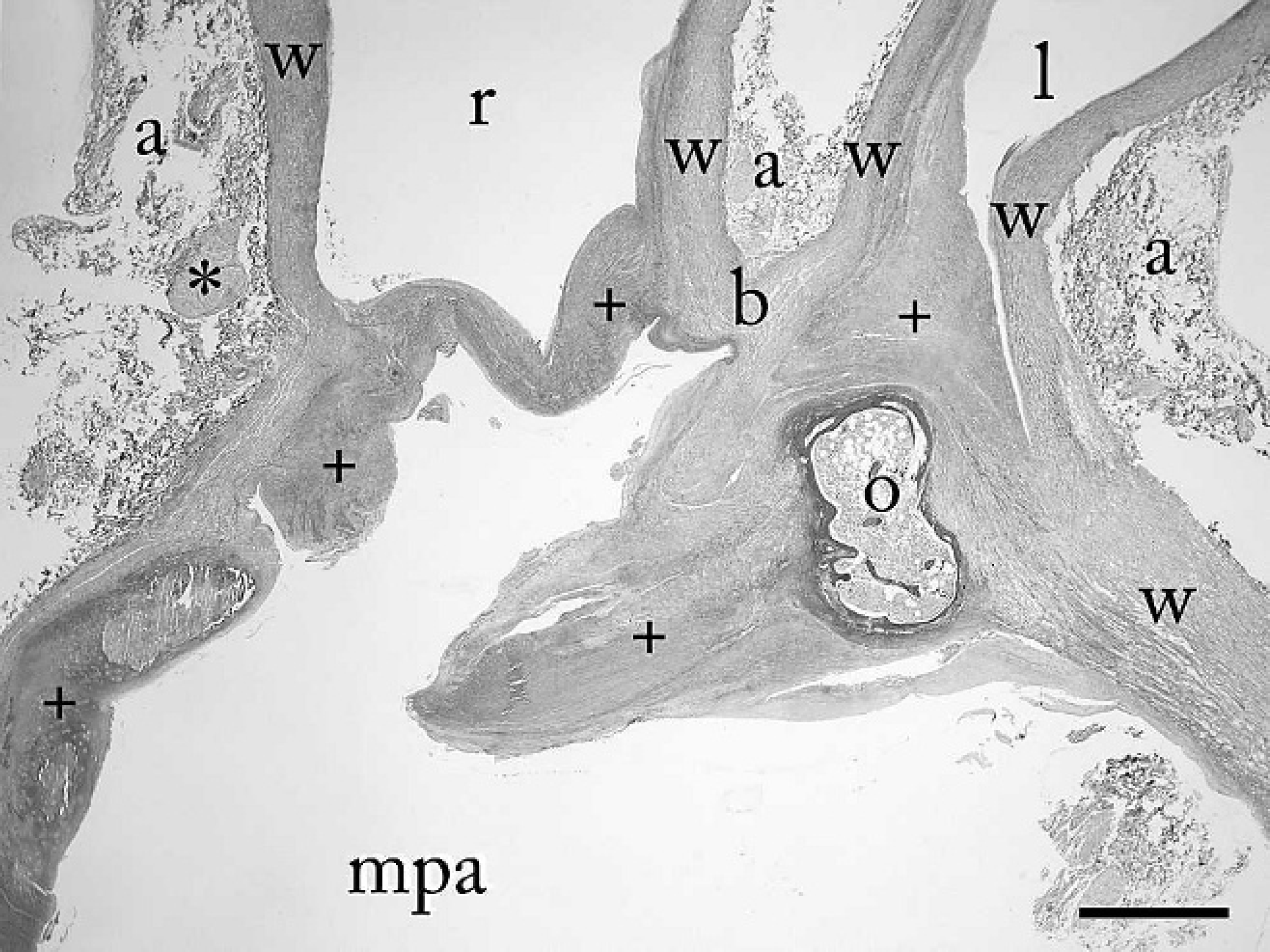
Photomicrograph of the main (mpa), left (l), and right (r) pulmonary arteries at the site of the bifurcation (b). This is also the point where the ligamentum arteriosum (*) is attached to the pulmonary artery. The position of the specimen corresponds to that of the macroscopic image (Fig. 6). The lumen of both the left and the right pulmonary arteries contain an obstructing crossing ridge of fibrous tissue (+), causing localized stenosis of both vessels. Within the fibrotic areas, islands of ossification (o) may be seen. a = adventitia; w = wall of the pulmonic arteries. Lawson elastin stain. Bar = 1 mm.
Histologic examination of the stenotic region of the pulmonary artery showed that the ridge-like stenosis of both pulmonary arteries was caused by extensive aberrant connective tissue (few fibroblasts in collagen matrix). No gross anatomical or microscopic evidence of inflammation was found in the mediastinum and adventitia. Osseous metaplasia was found in the wall of the main pulmonary artery (Fig. 7), which may have been the result of increased wall stress. The nodular processes in the lungs were granulomas, containing abundant dystrophic mineralization centered on protein and lipid deposits containing cholesterol clefts, without evident etiology.
Pulmonary artery stenosis is characterized by a circumscribed narrowing of the lumen. Coarctation is a specific form of stenosis that is caused by ectopic ductal tissue. To the authors' knowledge, no reports of a juxtaductal pulmonary coarctation in the dog or the cat have been published in the veterinary literature.
The site of a congenital pulmonary artery stenosis can be either subvalvular, valvular, or supravalvular. Pulmonary artery stenosis is in most cases a congenital anomaly, but it could also be an acquired lesion resulting from, for example, a mediastinitis. 6 Supravalvular stenosis can be localized at the level of the pulmonary artery trunk, the right or left pulmonary arteries, or the smaller pulmonary artery branches. In human beings, circumscribed stenosis of a pulmonary artery at the level of the bifurcation is known to have a specific etiology, as ductal tissue of the ductus arteriosus is extended into the wall of the pulmonary artery. 2,3 Constriction of the ductal tissue at birth not only results in ductal closure but also leads to pulmonary artery stenosis because of the ectopic ductal tissue (Skoda's theory). 4 Whether the left or the right pulmonary artery develops a stenosis depends on whether the individual has a left- or right-sided ductus arteriosus. In the case of a left-sided ductus, a narrowing of the proximal LPA develops. Embryonically, mammals have a bilateral (i.e., right and left) ductus arteriosus, together with a bilateral (i.e., right and left) aortic arch. During fetal life one, usually the right-sided ductus, along with the right aortic arch, regresses. In rare cases, both the right and the left ductus arteriosus may be persistent and remain patent. 10 Because the ductus arteriosus connects to the ipsilateral pulmonary artery, the only explanation for the presence of a bilateral coarctation of the pulmonary artery is the historical presence of right-and left-sided ectopic ductal tissue in the walls of both pulmonary arteries. Though stenosis of the right and left pulmonary arteries has been reported in the dog, 8 the above-described etiology has not been described yet in the veterinary literature. 11 Various developmental anomalies of the aortic arch have been reported in the cat, but all of them caused vascular ring anomalies. 1,9,12 According to a human classification, the anomaly of the present case report can be categorized as type IIA stenosis (i.e., a short, localized stenosis at the bifurcation). 4 There may be at least 2 reasons to assume that the stenosis of both pulmonary arteries resulted from ectopic ductus arteriosus tissue in the wall of the pulmonary artery: 1) the stenosis was juxtaductal (i.e., at the point of the bifurcation, where the ligamentum arteriosum connects to the pulmonary arteries); and 2) the LPA, the opening of which was nonpatent, had a normal lumen distal to the stenosis. This indicates that the LPA was probably patent, possibly during fetal life. 2 However, histopathologic findings of the pulmonary artery wall do not clarify this theory further, because no interruption of the elastic layer of the pulmonary artery wall by connective tissue was found described in human beings. 2,3,5 On the other hand, a possible interruption of the elastic layer of the wall by connective tissue (the remnants of the ductus) cannot be excluded, as this may be present in another histological section of the specimen. As is the case with human reports, coarctation in the present case was caused by a localized ridge. 3 Also, similar to the findings of a previous report, 3 the smaller pulmonary artery was found on the left side. The small size of the left lung lobes supports the hypothesis that LPA atresia occurred, most likely early in life, in the cat in the current case.
In conclusion, the cat in the present report was diagnosed with severe bilateral pulmonary artery coarctation with functional atresia of the left pulmonary artery. The severe stenosis was obviously caused by accessory bilateral ductal tissue. As a result of the severe pulmonary stenosis, a hypoperfusion of the pulmonary arterial tree was the most likely cause of the periods of dyspnea (so-called exertional dyspnea). 11 Only correction of the morphological vascular abnormalities can resolve this clinical sign. An interventional approach may lead to a fatal outcome when the catheter causes temporary complete occlusion of the pulmonary vascular bed. An intervention should only be attempted if enough blood can flow along the stenosis of the affected pulmonary artery and/or to the contralateral pulmonary artery once a catheter has been placed through the stenosis.
Acknowledgements
The authors wish to thank Dr. Joost Uilenreef for the anesthesiology assistance, Dr. Giora van Straten for the clinical work-up, Dr. Edoardo Auriemma for the radiographic interpretation, and Dr. Arnold Stokhof for his help during cardiac catheterization.
Footnotes
a.
Xenetix®, Guerbet, Cedex, France.
