Abstract
Blackleg is an infectious disease caused by Clostridium chauvoei. Cardiac blackleg has been reported in ruminants as an uncommon presentation of the disease; its pathogenesis is not understood completely. We include here a literature review of cardiac blackleg and a description of 2 cases in 12–15-mo-old feedlot steers in Argentina. Fourteen of 1,190 steers died suddenly over a period of 10 d. Postmortem examinations were performed on 5 of these animals. Grossly, severe, diffuse, fibrinous pericarditis and pleuritis, multifocal necrohemorrhagic myocarditis, diffuse pulmonary congestion, mild splenomegaly, and moderate congestion of meningeal vessels were observed. No significant gross lesions were observed in the skeletal muscles of any animal. Histology was performed on 2 of the steers. The main microscopic features were necrotizing myocarditis with myriad intralesional gram-positive rods with subterminal spores plus fibrinosuppurative pericarditis and pleuritis. C. chauvoei was detected by immunohistochemistry and PCR in the myocardium of both animals. These findings confirm a diagnosis of cardiac blackleg in these 2 steers and presumptively in the other affected animals.
Keywords
Blackleg is an infectious disease caused by Clostridium chauvoei, a gram-positive, spore-forming rod that affects cattle, and less frequently sheep and other domestic and wild animals.1,6,20,22,29,35,41,44,45 Although blackleg is a well-recognized disease, its pathogenesis is still not understood completely.2,58 The current theory is that C. chauvoei spores can survive in the environment for many years and that spores are ingested by grazing animals. 1 After ingestion, the spores are absorbed from the intestine and subsequently distributed via the bloodstream to various tissues, including skeletal and cardiac muscle, where they remain latent. 1 When anaerobic conditions occur in those tissues, usually associated with blunt trauma,1,3,63 the spores germinate, and the vegetative forms of C. chauvoei proliferate and produce several exotoxins that cause characteristic necrotic lesions, mainly in large skeletal and cardiac muscles.3,19 Although cases of blackleg with skeletal muscle lesions, but no cardiac muscle lesions, occur frequently,22,58 the opposite (i.e., lesions in the heart without skeletal changes) are rare, and the few reports of this condition describe individual cases.3,15,20,27,28,58 We present here a literature review of the disease and also describe 2 cases of cardiac blackleg in steers without skeletal muscle lesions.
Etiology
C. chauvoei is a highly pathogenic, anaerobic, endospore-forming gram-positive rod measuring 0.6 × 3–8 µm, with rounded ends, that appears singly, in pairs, and rarely in short chains of 3–5 cells. 11 The organism is usually motile by means of peritrichous flagella. Sporulation of C. chauvoei occurs easily; the spores are ovoid, central, or subterminal, and usually distend the cell.2,59 Spores are destroyed by the action of iodine and chlorine disinfectants, but resist boiling, as well as phenolic and quaternary ammonium disinfectants.2,59
The best growing conditions for C. chauvoei are on blood agar, pH 7.2–7.5, in strict anaerobic conditions at 37–40°C. 46 C. chauvoei colonies on blood agar are circular, raised, usually 2–4 mm in diameter, bright and semitransparent, although after 24 h of incubation they became completely opaque. Frequently, the colonies are surrounded by a variable zone of hemolysis on sheep blood agar. The hemolysis sensitivity depends on the bacterial strain and the species of animal erythrocytes present in the medium.2,24,59
Glycolysis, gluconeogenesis, and purine and pyrimidine metabolism have been identified as the main sources of energy for C. chauvoei. However, the absence of genes of the citric acid cycle and some essential amino acids suggest that the main source of these components is from host tissue damage. 19 C. chauvoei is highly susceptible to several antimicrobials, including cephalothin, clindamycin, enrofloxacin, erythromycin, penicillin, chloramphenicol, tetracycline, sulfadimidine, and gentamicin. 46
C. chauvoei produces 2 β–pore-forming toxins (β-PFTs) named C. chauvoei toxin A (CctA) and chauveolysin. The general mechanism of action of β-PFTs consists of formation of pores in the lipid cell membrane that results in the alteration of membrane permeability and subsequently cell death by necrosis or apoptosis depending on toxin concentration.42,43 CctA is a 32-kDa β-PFT protein that belongs to the Staphylococcus aureus α-hemolysin family and is considered to be the main virulence factor of C. chauvoei. This toxin has hemolytic and cytotoxic activity.19,42,43 The presence of protective CctA-neutralizing antibodies in guinea pigs experimentally vaccinated and challenged against C. chauvoei 18 and in cattle immunized with commercial blackleg vaccines containing CctA 39 demonstrates the immunogenic role of this toxin. Chauveolysin is a cholesterol-dependent cytolysin that forms large pores in cellular membranes as a product of oligomerization of 30–50 units instead of the 6–7 units described for CctA.30,42,43
In addition to CctA and chauveolysin, C. chauvoei produces other virulence factors, including hyaluronidase, sialidase, hemolysin, DNase, and flagella, which are involved in the pathogenesis of blackleg.2,18,19,30,33,42,43,51,59 C. chauvoei hyaluronidases and sialidases (neuraminidases) provoke the breakage of intercellular matrix components and cellular tight junctions, facilitating rapid spread of the microorganism in the tissues of the host.2,16,19,54,60 Two hyaluronidase genes, named nagH (hyaluronoglucosaminidase) and nagJ (β-N-acetylglucosaminidase), have been identified in the genome of C. chauvoei, but the enzymatic activity has been confirmed only for the former.16,19 The C. chauvoei nanA gene codifies the activity of sialidases. 60 The mode of action of this enzyme consists of hydrolyzing glycosylic linkages of terminal sialic residues in oligosaccharides, glycosaccharides, glycoproteins, and glycolipids located on cell membranes.19,55 C. chauvoei produces others hemolysins, named hemolysin III-superfamily, hemolysin A, and hemolysin of the XhlA type, but they seem to play a minor role in the pathogenesis of blackleg. 19 C. chauvoei DNase, or beta toxin, produces DNA degradation through cleavage of phosphodiester bonds. 2 The flagella of C. chauvoei provide motility to the bacteria and play an important role in virulence.2,19,43,51 Various epitopes on the flagella generate protective immunity in mice38,52; thus, the flagella antigens are valuable candidates for the development of vaccines against blackleg.33,38,51,52
Epidemiology
Blackleg most often affects non-vaccinated cattle in good nutritional condition, 6-mo to 2-y-old. However, sporadic cases have been described in younger calves,25,58 in a fetus, 2 and in animals >2-y-old. 14 Rarely, blackleg has been reported in vaccinated cattle.26,61
Blackleg has been described rarely in sheep.6,20 In this species, it is important to establish the differential diagnosis between C. chauvoei–associated gas gangrene of exogenous pathogenesis and blackleg of endogenous pathogenesis, particularly when the portal of entry of the infection cannot be identified. 2 Infections by C. chauvoei have also been described in other animal species, including horses, 22 elephants, 45 chickens, 44 deer, 5 mink, 35 and ostriches. 36 The pathogenesis of these infections in species other than cattle and sheep remains mostly undetermined. Cats, dogs, rabbits, and humans are believed to be resistant to infection by C. chauvoei. 13
C. chauvoei is ubiquitous in soil, silage, freshwater, pasture, and feces of healthy animals.9,23,58 Spore-contaminated soil from carcasses and feces of infected animals seems to be the main source of infection for animals. Although it has not been proved that C. chauvoei multiplies in soil, its spores can persist in soil for long periods,10,23 and their survival is favored by certain environmental condition, including high humidity and temperature. The presence of large quantities of organic matter in soil, 58 soil type, 23 soil excavations, 10 flooding events, 31 and annual rainfall 54 have been associated with the occurrence of blackleg, underlining the importance of these predisposing factors in the dissemination of C. chauvoei spores. 61 Most blackleg cases occur in animals grazing on contaminated pastures, generally when they are moved to a new pasture after high rainfalls,2,54 and less frequently in housed animals, most likely associated with ingestion of spore-contaminated feed. 21 The association between rainfall and the occurrence of blackleg in grazing animals is supported by the fact that heavy rainfalls create anaerobic soil conditions that favor the multiplication of the bacteria and the subsequent wide dissemination of spores. 54
Pathogenesis
The classical model of the pathogenesis of blackleg begins when animals ingest spores from contaminated soil or feed. Spores or vegetative cells reach the intestine, where the bacteria undergo one or more replication cycles, and are subsequently absorbed and distributed via the bloodstream to various tissues, including skeletal and cardiac muscle.2,14,32,59 In these tissues, spores are phagocytized by macrophages, 41 in the cytoplasm of which they remain latent for months to years, until the oxygen tension decreases. It has been hypothesized that, in addition to a traumatic event, stress associated with movement of animals or excessive exercise can increase cortisol and catecholamine levels,2,25,58 creating an anaerobic environment for C. chauvoei multiplication and the production of toxins that cause severe myonecrosis, toxemia, and shock.2,14,58,59 Because the disease is not associated with skin or mucosal wounds, it is considered an endogenous infection.2,14,58,59
Blackleg must be differentiated from gas gangrene (formerly malignant edema), an exogenous infection in which the causative clostridia enter through skin or mucosal wounds. 50 Although the pathogenesis of blackleg described above is widely accepted, it does not explain why clusters of cases occur in certain circumstances, such as cases occurring every year in the same geographic area, or after the movement of animals to a new pasture, or in housed animals. In those situations, the main factor proposed is the unusual consumption of large numbers of C. chauvoei spores, which leads to septicemia. 2
Another issue that remains unclear about the pathogenesis of blackleg is the predilection of the bacteria for targeted organs. Most cases of blackleg have simultaneous lesions in skeletal muscles and heart. However, in rare occasions, only the heart is affected.2,3,59
It has been suggested that there is variability in the virulence of C. chauvoei strains within one host, between different hosts, and in repeated outbreaks on the same farm or pastures. 53 If the former holds true, it is possible that the causes of unusually virulent outbreaks of blackleg are associated with genetic changes in C. chauvoei strains. However, analysis of genomics of strains from cases affecting skeletal muscles alone and strains from cases in which the heart, sublingual muscles, and diaphragm were affected showed highly conserved strain genomes from both groups of cases. 65
Abreu et al.2,3 and Uzal et al. 59 have postulated that, when both the skeletal muscles and heart are affected, lesions in the latter could be secondary to the skeletal muscle lesions as a consequence of C. chauvoei septicemia. In those cases, it has been suggested that toxemia and/or bacteremia-associated hypoxia could have acted as a predisposing factor for the cardiac and/or skeletal muscle lesions.
Cardiac blackleg had been reported in sheep and cattle,20,25,58 but its pathogenesis has not been explained successfully. It was suggested that the main predisposing factor for anaerobiosis in the heart is associated with stress, or preexisting myocardial injury caused by toxins such as gossypol or ionophores, or nutritional deficiencies, including selenium and vitamin E.2,3,59 This, however, remains speculative.
Clinical signs
The clinical manifestations of blackleg are often not observed; given the acute course of blackleg, animals are frequently found dead. 14 However, in some subacute cases, when death usually occurs within 48 h, clinical signs may include depression, anorexia, lethargy, anorexia, lameness, and reluctance to move.1,2 Very few affected animals survive for >48 h.21,49 When superficial muscles are affected, swelling and crepitation may be perceived, mainly in the rear legs.1,2 Clinical signs associated with heart lesions include brisket edema, increased diffuse lung sounds, dyspnea, and pericardial friction rubs as a result of accumulation of fluid, but these signs are rarely observed.1,2,48
Gross and microscopic pathology
Animals that die of blackleg are usually in very good nutritional condition. Significant lesions are observed most frequently in skeletal muscles and less frequently in the myocardium. Rarely, other tissues, such as the thymus, liver, intestine, meninges, and brain are affected.15,26,37 It has been estimated that only 3% of the cases of this disease have cardiac lesions in the absence of skeletal muscle damage.1,3,27 Lesions caused by C. chauvoei usually involve more than one group of muscles of the pelvic and pectoral girdles, but any striated muscle, including the myocardium can be affected. 14 Although some references indicate that cardiac lesions associated with blackleg are uncommon,3,14,58 a 2016 retrospective study 1 showed the opposite; most cases analyzed had lesions in both the heart and skeletal muscles. The author of one study 3 suggested that it is possible that in at least some cases, once the lesions are found in large skeletal muscles, the carcass is not investigated further, and potential lesions in the heart and other skeletal muscles such as the diaphragm are overlooked.
On external inspection, a poorly circumscribed area of swelling may be visible over the affected muscle areas in which the skin is stretched and usually dark, and crepitation caused by the subcutaneous accumulation of serosanguineous fluid and emphysema is detectable by palpation.1,3,14 The affected muscle in the center of the lesion is dry, friable, dark-red to black, and has many small cavities caused by gas bubbles. Toward the periphery of the lesion, the affected muscle is edematous.1,3,14 The odor emanating from the affected tissues is sweet and reminiscent of rancid butter, associated with bacterial production of butyric acid.3,14,21,49 In cases in which the heart is affected, the main lesions consist of focally extensive dark areas of hemorrhage, necrosis, and occasional emphysema that can affect any area of the myocardium. Pericarditis is frequently present,3,37 and the 2 layers of the pericardial sac often are adherent by variable amounts of fibrin. In addition, the presence of excess serosanguineous fluid in the pericardial sac is common.1,3 Other occasional lesions include fibrinohemorrhagic pleuritis, affecting mainly the pleura adjacent to the heart, pulmonary congestion, and edema.1,3,14 Necrotizing enteritis and glossitis have been described rarely. 26
Microscopically, lesions of blackleg are similar in cardiac and skeletal muscle. The affected muscle fibers of both tissues have degenerative or necrotic changes characterized by hypereosinophilia, swelling, vacuolation, fragmentation with loss of cross striations, and presence of contraction bands. Initially, the interstitium and fragmented fibers may be infiltrated by neutrophils, and in later stages of the infection there is also infiltration of macrophages, plasma cells, and lymphocytes.1,14 However, leukocytic infiltration is not a prominent feature in most blackleg cases.1,3 The interstitium is expanded by hemorrhage and proteinaceous edema fluid mixed with large numbers of gram-positive rods, some of them with subterminal spores, singly or in clusters. Large empty vacuoles created by gas bubbles are usually evident in the interstitium of skeletal muscle, but rarely in the myocardium. Frequently, vessels contain fibrin thrombi, and fibrinoid necrosis with intramural infiltration of neutrophils can be seen in arteries and arterioles.1,3 The areas of fibrinosuppurative pericarditis and pleuritis may have vascular changes similar to those described in muscle.
Diagnosis
A definitive diagnosis of blackleg is based on clinical signs, compatible gross and microscopic findings, and detection of C. chauvoei in the affected tissues. The identification of the bacteria can be accomplished through anaerobic culture, immunodetection by a fluorescent antibody test (FAT) or immunohistochemistry (IHC), and/or PCR.1,3,7,8,12,63 Microbial isolation is not always possible because of the difficulties in submitting samples to the laboratory, and also because not all diagnostic laboratories around the world have facilities available for anaerobic culture.3,63 In addition, postmortem contaminants, including other fast-growing anaerobes such as C. septicum, may interfere with the isolation of C. chauvoei. 1
FAT and IHC are techniques of great value when fresh tissues are not available for microbiologic culture. 58 Both methods are simple, rapid, and have similar sensitivity and specificity.7,8 FAT is faster and less expensive than IHC, and it has the advantage that it can be performed on smears or on tissues fixed in formalin for short periods of time.1,8 IHC is particularly useful for retrospective studies by detecting the organism within lesions. 7
PCR is a very useful method for the identification of C. chauvoei in clinical samples, 9 pure cultures, or in formalin-fixed, paraffin-embedded (FFPE) tissues. 58 In a 2021 study, 17 a PCR assay was used to detect C. chauvoei in filter paper impregnated with tissue samples of suspected blackleg cases, demonstrating that the procedure is reliable, practical for field conditions, and cost-effective. In addition to conventional PCR, a multiplex real-time PCR has enabled the simultaneous identification of C. chauvoei and C. septicum. 34 A recombinant C. chauvoei flagellin ELISA was developed for the detection of the microorganism with high specificity; however, this test has not yet been used for routine testing and further studies are required to evaluate its diagnostic potential. 56
Prevention and treatment
Because blackleg causes acute disease and frequently sudden death, antimicrobials are not generally used to treat the disease. 25 Vaccination is the most efficient preventive measure to control blackleg worldwide.57,62 Initial vaccination against blackleg is recommended at 2-mo-old, followed by a booster dose 1 mo later, and then annually or every 6 mo depending on the epidemiologic conditions in the area.1,4 However, although vaccination against blackleg is anecdotally believed to be 100% effective, scientific evidence showing its efficacy in protecting cattle challenged against C. chauvoei or in cattle naturally exposed is scant. 57 The main factors that could affect the efficacy of blackleg vaccines have been suggested to be inadequate vaccine management of the herd and the differential expression of toxins that depend on the vaccine manufacturing process. 64 Commercial C. chauvoei vaccines consist of formalin-inactivated whole cultures. However, it is not clear which antigens in those vaccines provide the most protection.39,62 It has been believed that flagella were the main antigens that conferred protective immunity against blackleg,51,52 but studies on this topic revealed that CctA is the major virulence factor of C. chauvoei, making this toxin a valuable candidate for future blackleg vaccines.18,39
Case report
A cluster of cases occurred in 12–15-mo-old Angus-Holstein crossbred steers in a feedlot located in Trenque Lauquen, Buenos Aires province, Argentina, during October 2017. The affected herd consisted of 1,190 steers that had been purchased from 2 different farms 7 mo before the outbreak started. On arrival, the steers were treated with doramectin and vaccinated twice with a commercial clostridial aluminum hydroxide vaccine including antigens of C. chauvoei, C. septicum and C. perfringens types C and D (Vacuna anti clostridial triple tradicional; Centro de Diagnóstico Veterinario, Argentina), and with an oil-adjuvanted commercial vaccine including formalin-inactivated cultures of bovine herpesvirus 1 (Bovine alphaherpesvirus 1), bovine viral diarrhea virus (Pestivirus), bovine parainfluenza 3 virus (Bovine respirovirus 3), and bovine respiratory syncytial virus (Bovine orthopneumovirus), and antigens of Pasteurella multocida, Mannheimia haemolytica, and Histophilus somni (Vacuna viral respiratoria; Centro de Diagnóstico Veterinario, Argentina). The animals were fed a ration composed of corn, sunflower expeller, and a multi-mineral mixture (including selenium, vitamin E, copper, and monensin). Two months after arrival, there was an outbreak of respiratory disease and all the animals in the herd were treated with chlortetracycline and sulfamethazine preparations in the ration. The most severely affected individuals were treated subcutaneously with tilmicosin.
During the first 10 d of October 2017, 14 steers were found dead with no premonitory clinical signs observed. All affected steers had been bought from the same farm but were located in different pens since their arrival to the ranch where the deaths occurred. Three of these animals were autopsied by a practitioner, who observed macroscopic changes consistent with myocardial necrosis, suppurative pericarditis and pleuritis, but no skeletal muscle lesions. No samples for histology or other ancillary tests were collected from these 3 animals. E. Odriozola autopsied 2 additional steers (steers 1 and 2) 7 h (steer 1) and a few minutes (steer 2), respectively, after death. These 2 steers were fully skinned, and most skeletal muscles of the body were bread-loafed and examined. This included, but was not limited to, epaxial and hypaxial muscles, including neck and vertebral (semispinalis, trapezius, serratus, longissimus, psoas), thoracic (pectoralis, latissimus, cleidobrachialis, trapezius), and all 4 limb (deltoid, triceps, brachialis, tensor fasciae, lateral vastus, gluteobiceps, semitendinosus, semimembranosus, and others) muscle groups. In addition, tongue and masseter muscles were examined. All 5 animals subjected to postmortem examination were in excellent nutritional condition, with abundant fat reserves and well fleshed.
The main gross lesions were observed in the heart of both steers and consisted of multiple 5-mm to 10-cm diameter dark red foci compromising ~30% (steer 1) and 70% (steer 2) of the myocardium (Fig. 1). There were ~20 mL of serosanguineous fluid with fibrin strands in the pericardial sac. The pericardium and epicardium were diffusely congested, adhered to each other, and covered by a thick layer of fibrin. Moderate cerebral meningeal congestion was observed in both steers. In addition, steer 1 had diffuse pulmonary congestion and edema, mild splenomegaly, and ~20 mL of serosanguineous fluid in the abdominal cavity. Steer 2 had 3–4-mm diameter, red, round areas in the diaphragmatic lobe of the right lung, fibrous adhesions between the visceral and parietal pleura of the cranial lobe of the right lung, abomasal mucosal ecchymoses, and thymic hemorrhage. No other significant gross abnormalities were observed in either carcass. In particular, detailed examination of the skeletal muscles did not reveal gross abnormalities.
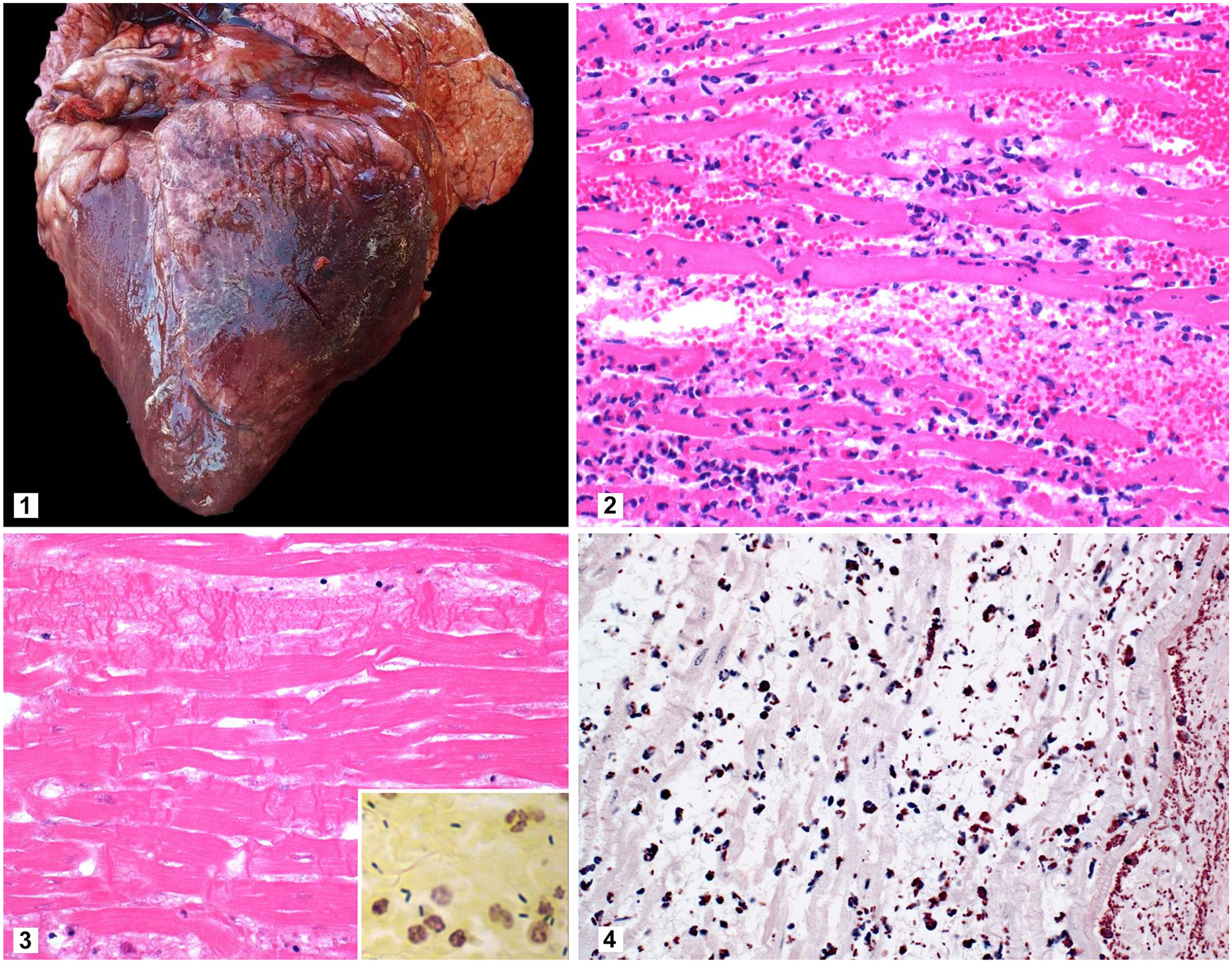
Heart of a steer with blackleg.
Samples of heart, liver, spleen, kidney, skeletal muscles (vertebral and from muscles from each of the 4 limbs), lung, abomasum, thymus, mediastinal lymph node, small intestine, colon, and brain from steers 1 and 2 were fixed in 10% neutral-buffered formalin, processed routinely, and 4–5-µm sections stained with H&E for histologic examination. Microscopically, both steers had severe, focally extensive necrohemorrhagic myocarditis. In these areas, the myofibers were separated by proteinaceous edema, hemorrhage, large gas bubbles, and mild inflammatory infiltrates composed of neutrophils, lymphocytes, plasma cells, and macrophages (Fig. 2). Necrosis of the myofibers was characterized by hypereosinophilia, loss of cross-striations, contraction bands, and fragmentation (Fig. 3). Myriad intralesional rods (~4 × 2 µm) with rounded ends, many of which had subterminal spores, were also present. These bacteria were observed singly or arranged in small groups in the interstitium of the myocardium, admixed with edema and the mixed inflammatory cell infiltrate (Fig. 3). Small groups of rods were also within the cytoplasm of necrotic myofibers and surrounding arterioles. The epicardium was thickened by a layer of fibrin mixed with moderate numbers of neutrophils, macrophages, lymphocytes, and plasma cells. In addition, fibrinoid necrosis and neutrophilic infiltrates were common in arterioles.
The lungs of both animals had moderate, diffuse neutrophilic, lymphoplasmacytic, and histiocytic infiltrates in the interstitium. No other significant microscopic lesions were observed in steer 1. Additionally, in steer 2, scarce neutrophils were scattered within the hepatic sinusoids, and the renal interstitium had discrete foci of neutrophils, lymphocytes, plasma cells, and macrophages. The abomasal mucosa had petechial hemorrhages. The thymus had diffuse and severe interstitial edema, neutrophilic inflammatory infiltrate, emphysema, and severe multifocal hemorrhages. The spleen was diffusely congested, and neutrophilic infiltrates surrounded marginal zones. The mediastinal lymph nodes had multifocal hemorrhage in germinal centers and moderate numbers of neutrophils within medullary sinuses.
Sections of heart from steers 1 and 2 were Gram-stained and processed for IHC using polyclonal anti–C. chauvoei, anti–C. septicum, anti–C. novyi, and anti–Paeniclostridium sordellii antibodies as described previously. 3 The great majority of the rods observed in H&E sections of the myocardium of both animals were gram-positive (Fig. 3, inset) and were positive for expression of C. chauvoei antigen in IHC (Fig. 4). All of the other IHC stains for different Clostridium species performed were negative.
Samples of heart, lung, spleen, and brain from both animals were collected aseptically and inoculated onto 7% bovine blood Columbia agar plates and MacConkey agar plates, before being incubated aerobically or in 5% CO2 at 37°C for 48 h. No attempt was made to culture anaerobic microorganisms because facilities for anaerobic incubation were not available. No bacterial pathogens were isolated from any of the tissues cultured under aerobic or microaerophilic conditions.
Scrolls (10-µm thick) were prepared from the FFPE myocardium samples using a new microtome blade for each animal. These scrolls were processed for DNA extraction (QIAamp DNA FFPE tissue kit; Qiagen). The extracted DNA was used as template for PCR to amplify the flagellin (fliC) genes of 5 histotoxic clostridial species including C. septicum, C. novyi type A, C. novyi type B, C. chauvoei, and C. haemolyticum, as described previously.40,47 Only a band of ~535 bp, consistent with the expected amplification size of the C. chauvoei fliC gene, was detected on the gel loaded with myocardial DNA from both steers.
The selenium concentration was measured in liver from both steers by a hydride vapor generation inductively coupled plasma spectrometer (Thermo ICAP 6500; Thermo Fisher). Normal selenium hepatic concentrations were found in both steers.
A presumptive diagnosis of cardiac blackleg was established based on gross (n = 5) and microscopic findings (n = 2). The diagnosis was confirmed in steers 1 and 2 by positive IHC and PCR for the C. chauvoei fliC gene.
Our cases occurred during a warm and rainy spring, in agreement with reports that describe a high correlation between rainy seasons and the occurrence of blackleg.1,55 All affected animals were <2-y-old and had optimal nutritional condition. These factors have been described as predisposing factors for blackleg, but their implication in the epidemiology of the disease is not understood completely.2,14,26,58,59
In the 2 confirmed and 3 presumptive cases presented here, lesions were observed in the heart but not in skeletal muscle. Although it is possible that subtle lesions in the skeletal muscle were overlooked, this is unlikely, given that thorough autopsies were performed. In addition, 2 of these autopsies were performed by an experienced pathologist who, being aware of the results of the previous 3 autopsies of this cluster of deaths, paid special attention to the examination of all skeletal muscle groups.
Several predisposing factors have been proposed in the pathogenesis of cardiac blackleg, including stress associated with husbandry practices and the presence of previous myocardial lesions.1,2,58 In addition, ionophores or gossypol poisoning and nutritional deficiencies (vitamin E and/or selenium) could create optimal conditions for spore germination and contribute to cardiac blackleg.1,2,58 In the 2 animals of this cluster in which selenium was measured, the concentration of this mineral was within the RI. The ration did not contain gossypol. Although the ration contained ionophores, we could not determine if this substance was a predisposing factor given that, at the time the animals were autopsied, no samples of the original ration were available to determine ionophore concentration. Vitamin E was not measured, but it is unlikely to have been a contributing factor because deficiency of this vitamin is infrequent in feedlot cattle. The microscopic lesions in the myocardium were acute and monophasic, suggesting that they were a consequence of C. chauvoei infection and not associated with any previous toxic event. We cannot rule out, however, that a sublethal intoxication was the predisposing factor for the blackleg lesions to develop.
Although in our study the number of affected animals with cardiac blackleg was higher than in previous reports,25,58 at the herd level, only 14 of 1,190 (1.17%) animals died. The low incidence of blackleg at herd level was probably associated with adequate vaccination against C. chauvoei in most of the animals, which usually is sufficient to prevent the disease.2,57 Surprisingly, all of the animals involved in this cluster of cases had been vaccinated against blackleg.
Our diagnosis of cardiac blackleg was based on compatible clinical presentation (sudden death), gross and microscopic postmortem findings, and the identification of C. chauvoei in the heart by PCR and IHC. Blackleg is a well-known disease, which remains a major cause of economic losses around the world.27,55 Although clinical signs and lesions are easily recognizable by practitioners, cardiac blackleg is infrequent and may be overlooked during autopsy. Therefore, this disease should be considered as a differential diagnosis in cases of sudden death in young cattle.
Footnotes
Acknowledgements
We thank Dr. Carlos Arbesú for providing the complete case history and Ms. Lilian Lischinsky for histologic technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support from INTA (PNSA1115054, RIST.I111) and CAHFS for their research and/or authorship of this article.
