Abstract
Clostridium chauvoei causes blackleg in cattle. The disease has been reported worldwide, and although it can be prevented by vaccination, sporadic cases and occasional outbreaks still occur. We describe a case of blackleg in a 2-y-old, pregnant Gyr cow with in utero transmission to the fetus. The cow had characteristic gross and microscopic lesions of blackleg including widespread necrohemorrhagic and emphysematous skeletal and myocardial myositis, and fibrinous pericarditis. Her uterus contained a near-term, markedly emphysematous fetus with skeletal muscle and myocardial lesions similar to those seen in the dam. Histopathology of dam and fetal tissues revealed numerous gram-positive bacilli, many of them with sub-terminal spores, in multiple tissues. These bacilli were identified as C. chauvoei by immunohistochemistry. Anaerobic culture and fluorescent antibody tests performed on skeletal muscle from both the dam and fetus were positive for C. chauvoei, confirming a diagnosis of blackleg. Blackleg is a so-called endogenous infection, and the currently accepted pathogenesis involves ingestion of spores that are transported to muscle tissues where they lie dormant until anaerobiosis prompts germination. Germinating bacteria are histotoxic, producing severe, local necrosis and ultimately lethal toxemia. This model, however, has not been confirmed experimentally and also fails to explain some cases of the disease. A presumptive diagnosis of blackleg is based on clinical, gross, and histologic findings. Diagnostic confirmation necessitates the detection of C. chauvoei by culture, PCR, or immunodetection methods.
Introduction
Blackleg is an infectious disease of cattle and rarely other ruminants, produced by Clostridium chauvoei, and characterized mainly by necrohemorrhagic myositis. C. chauvoei is an anaerobic, gram-positive bacillus that persists in the soil as resistant spores.7,12,19,23,31,41 Upon germination in animal hosts, the bacteria produce potent exotoxins that cause local tissue damage and often lethal toxemia. The prevalence of many clostridial diseases of animals has decreased significantly given the use of readily available and efficacious vaccines. However, the ubiquitous distribution of C. chauvoei spores in the soil makes blackleg a continuous threat to unvaccinated cattle or animals vaccinated with poor-quality vaccines.
Blackleg occurs most frequently in animals 6–24 mo of age, and cases in animals outside that age range are very rare.7,23,41 Herein we report a case of C. chauvoei infection in a pregnant cow and its fetus. In addition, we review the literature on the main epidemiologic, clinical, microbiologic, and diagnostic features of blackleg in cattle.
Case report
In April 2015, a 25-mo-old pregnant Gyr cow was admitted to the Texas A&M University Veterinary Medical Teaching Hospital (College Station, Texas), after being found in the morning unable to stand. The cow was the only animal affected in the herd, which was composed of an unreported number of animals that were not vaccinated against clostridial diseases.
On clinical examination, the dam was recumbent. Subcutaneous and muscle emphysema was palpated in the hindquarters, and mucous membranes were congested. The fetus was unresponsive on rectal palpation and presumed dead. A chemistry profile of the dam revealed markedly elevated aspartate aminotransferase (1420 U/L; reference interval [RI]: 53–173 U/L) and creatine kinase (>16,000 U/L; RI: 55–392 U/L). A complete blood count showed leukopenia (3.8 × 109/L leukocytes; RI: 4.0–12.0 × 109/L) with a left shift (bands 0.68 × 109/L; RI: 0–0.12 × 109/L) and moderate toxic change, and lymphopenia (1.9 × 109/L; RI: 2.5–7.5 × 109/L). Frequent rods, single or in pairs, were seen in cytologic aspirates of the emphysematous hindlimb subcutaneous tissue and muscles stained with modified Wright stain. These findings, in combination with the vaccination status, led to a presumptive clinical diagnosis of blackleg. Given the poor prognosis and lack of treatment options, the cow was euthanized, and a postmortem examination was performed within 24 h of death.
At autopsy, the cow was in good nutritional condition and in a mild state of postmortem decomposition. Crepitus was palpated externally over the hindquarters and in the brisket area. Affected skeletal muscles in these areas were dry and contained multifocal-to-coalescing, dark red to black areas with various amounts of emphysema (Fig. 1). Most muscles of the hindquarters were affected by hemorrhage and edema. This change was most marked in the gastrocnemius muscles. The lesions were associated with a distinctive odor that was reminiscent of rancid butter. The epicardium was diffusely covered by a large amount of fibrin, and multifocal dark red areas were present in the myocardium (Fig. 1). The vulva, vagina, and uterine walls were markedly edematous. The uterus was taut and significantly distended by a large amount of gas. Within the uterus was a 13-kg, female fetus that was estimated to be ~217 d in gestation. The fetal subcutaneous tissues and skeletal muscles over the entire body were markedly expanded by emphysema and edema, causing distension and bulging of the overlying skin, with severe, palpable crepitus (Fig. 2). There was moderate hydrothorax with a few fibrin strands floating in the pleural fluid. Most muscles of the fetus had diffuse or multifocal dark red areas. The placenta had an estimated 25% decrease in the number of cotyledons, and scattered between remaining cotyledons were small proliferations of soft, white tissue, consistent in appearance with adventitial placentation.
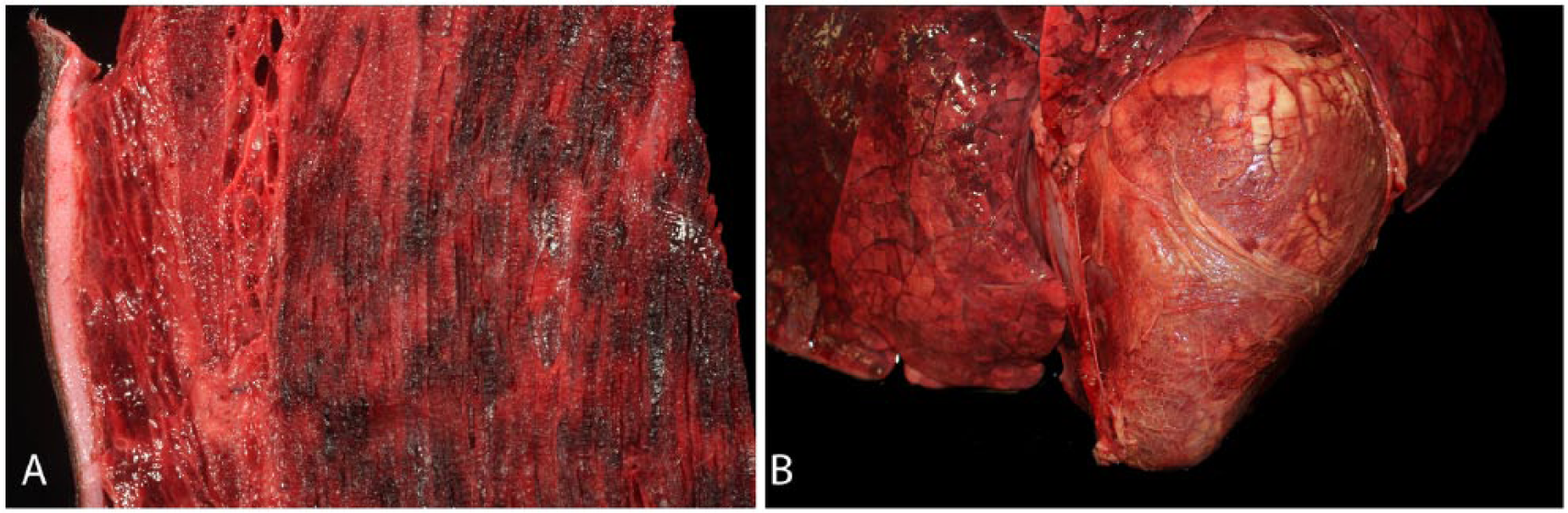
Gross lesions of Clostridium chauvoei infection in a 2-y-old pregnant Gyr cow.
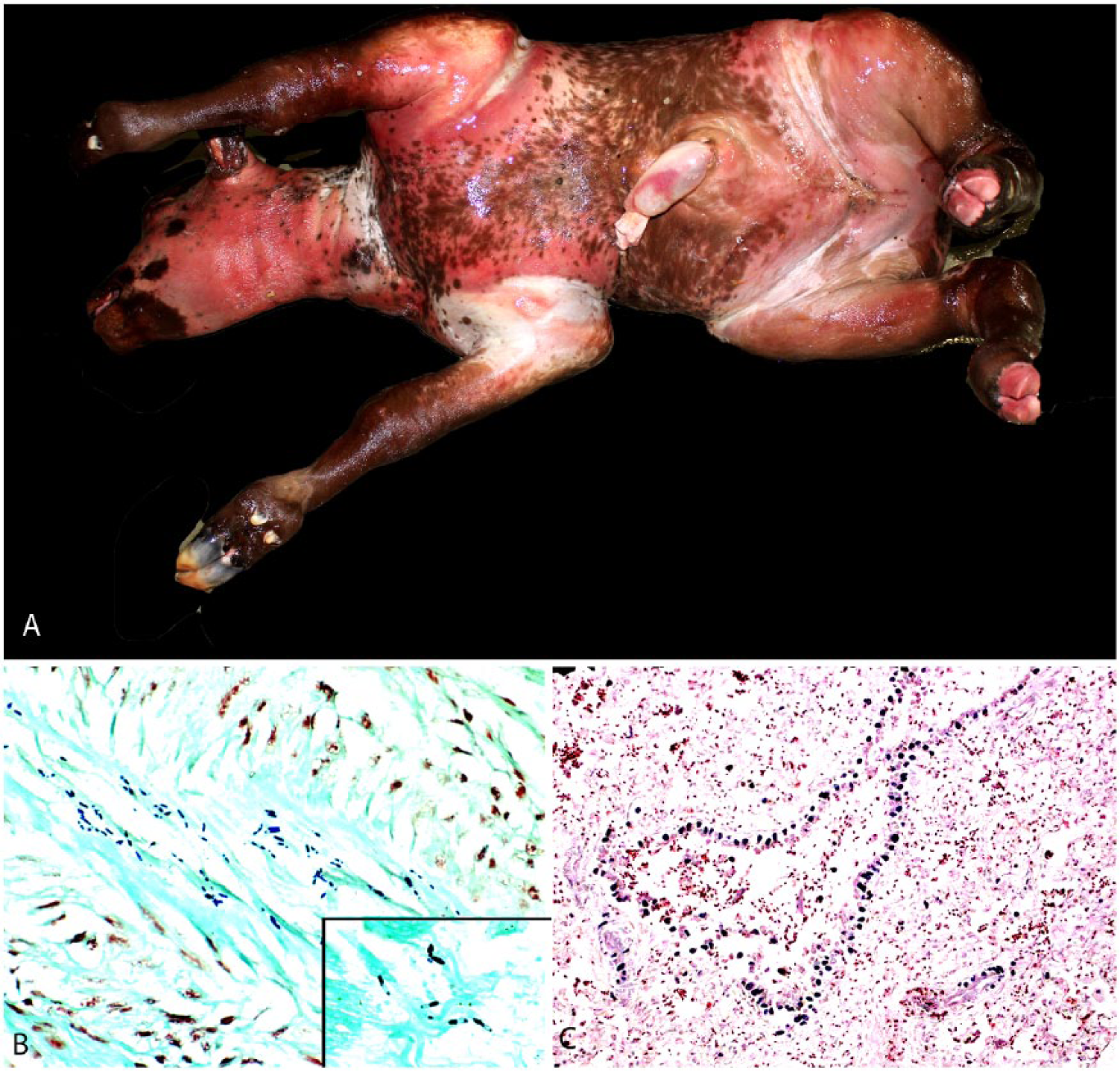
Clostridium chauvoei infection in a ~217-d bovine fetus.
Samples of skeletal muscle from the dam and the fetus were collected aseptically and processed for aerobic and anaerobic culture. Smears from these tissues were prepared, air-dried, and processed for a fluorescent antibody test (FAT) using conjugates against Clostridium novyi, Clostridium septicum, and Clostridium sordellii (VMRD, Pullman, WA). Briefly, samples for bacterial culture were inoculated into cooked meat medium and on 5% sheep agar blood, and incubated anaerobically for 48 h at 37°C. Additional blood agar plates were also incubated aerobically under the same conditions. Isolated colonies were identified by conventional biochemical techniques. Impression smears were fixed in acetone prior to conjugate application, after which the slides were incubated at 37°C for 30 min before being rinsed and soaked for 10 min in FAT rinse buffer, mounted, and observed in a fluorescence microscope.
Multiple tissue specimens, including hindlimb skeletal muscle, heart, tongue, liver, kidney, placenta, spleen, and lung from the dam, and fetal skeletal muscle, heart, and lung, were fixed in 10% buffered formalin for 24 h, embedded in paraffin, and routinely processed to make 4 µm thick hematoxylin and eosin–stained histologic sections. Selected sections of hindlimb skeletal muscle, heart, and liver from the dam, and fetal heart and lung were stained with Hucker–Twort Gram stain. Immunohistochemistry (IHC) for C. chauvoei and C. septicum were performed at the California Animal Health and Food Safety Laboratory System on formalin-fixed, paraffin-embedded tissues, including hindlimb skeletal muscle, heart, liver, and placenta from the dam, and on fetal lung tissue using the RTU VECTASTAIN Elite ABC system (Vector Laboratories, Burlingame, CA) according to the manufacturer’s instructions. A 3% solution of hydrogen peroxide was used to block endogenous peroxidase, followed by antigen retrieval with pepsin. Nonspecific binding was blocked by treating the samples (Background Punisher, Biocare Medical, Concord, CA) before incubation with goat anti–C. chauvoei or C. septicum polyclonal antibodies (VMRD) for 60 min at 37°C. NovaRED (Vector Laboratories) was used as chromogen for visualization. Tissues incubated with normal goat serum instead of primary antibodies served as negative controls. Skeletal muscle from 2 steers with blackleg or gas gangrene, and from which C. chauvoei and C. septicum, respectively, had been isolated, served as positive controls.
The most significant microscopic findings in the dam were necrohemorrhagic and emphysematous skeletal myositis, necrohemorrhagic myocarditis, and fibrinosuppurative epicarditis. These lesions were primarily characterized by large areas of coagulative necrosis. Necrotic myocytes were hypereosinophilic, fragmented, and had loss of cross-striations, cytoplasmic vacuolation, and pyknotic nuclei. Degenerate myocytes were swollen with frequent sarcoplasmic vacuolation. Occasionally, hypercontraction bands spanned myocytes. Skeletal muscle tissue architecture was further disrupted by large, clear, emphysematous cavities, with only a few similar cavities seen in the myocardium. Necrotic areas contained extravasated erythrocytes, strands of fibrin, and a small number of neutrophils, both viable and degenerate. Inflammation was more severe in the myocardium than in skeletal muscle. The epicardium was covered by a thick layer of fibrin and degenerate neutrophils with scattered extravasated erythrocytes. Multiple gram-positive, often paired, 3–8 μm long by 1 μm thick rods with sub-terminal spores were scattered throughout the skeletal and cardiac muscle fibers and separating fascia.
Multifocally, the fetal subepicardium was expanded by hemorrhage, and occasional focal hemorrhages were seen within the myocardium. Skeletal myofibers and cardiomyocytes were often notably vacuolated. Eosinophilic fibrillar material was present throughout the fetal heart, infiltrating between and separating cardiomyocytes, and in the lung, filling alveoli and expanding interstitial septa. This material did not stain with Fraser–Lendrum stain, and its identity could not be confirmed. The fetal lungs were significantly emphysematous. Alveoli were inflated and contained a moderate amount of extravasated erythrocytes, frequent neutrophils, and squamous epithelial cells (squames). Gram-positive rods, similar to those seen in the skeletal muscle and myocardium of the dam, were distributed widely throughout the fetal lung and heart, and in higher numbers than in the dam (Fig. 2).
C. chauvoei and Escherichia coli were cultured from the skeletal muscle of the cow; a pure culture of C. chauvoei was isolated from the fetal skeletal muscle. C. chauvoei was identified by FAT in skeletal muscle and myocardium of both animals; FAT for the other clostridial species tested was negative in all tissues. Abundant C. chauvoei was present in all dam and fetal tissues tested by IHC (Fig. 2). Occasionally, a few C. septicum IHC-positive rods were also present in all tissues of both dam and fetus.
A diagnosis of blackleg was established for the dam and fetus based on gross and histologic findings, coupled with results of anaerobic culture, FAT, and IHC. Small numbers of C. septicum were detected by IHC on tissues of the dam and fetus. This is a fast-growing and common postmortem invader that is present in the intestine of many normal cows and invades tissues very soon after, or sometimes before, death. This was the most likely explanation for the presence of C. septicum in tissues of the cow and fetus. This conclusion is supported by the fact that both culture and FAT were negative for this microorganism, whereas C. chauvoei was detected in tissues of both the dam and fetus by all of the techniques used.
Blackleg has not been described in pregnant cattle, to our knowledge. Fetal emphysema from which C. chauvoei, then called C. feseri, was isolated, was described in fetuses from 2 ewes following shearing.5,39 Because of the association with shearing wounds, however, that disease was likely gas gangrene and not blackleg.
Neonatal C. chauvoei infection was reported in a 3-d-old bull calf with emphysematous myositis in the hindlimbs. 42 Infection was presumed to be postpartum rather than in utero, because the dam did not die and because C. chauvoei was isolated from both the skeletal muscle and the forestomachs, suggesting ingestion of the spores. Neonatal skeletal muscle trauma from parturition presumably activated the infection.
Blackleg is a common and relatively well-described disease. Thus, the lack of reports in pregnant cattle is interesting. One possible explanation is that the disease typically affects young cattle (usually 6 m to 2 y) before the ideal breeding age. The cow in our report was bred early and at 2 y of age carried a near-term fetus. In many cases of blackleg, a postmortem examination is not performed, lowering the likelihood of finding fetal infections. The lack of other cases leaves it unclear whether the fetus is always infected, or whether there may be some level of fetal protection through the placental barrier. Given the rapid disease progression and eventual death of the dam by toxemia, fetal death is inevitable regardless of whether the fetus is exposed to C. chauvoei. In our case, we hypothesize that, following activation of spores, both bacteria and toxins were hematogenously spread to the fetus, given the large number of bacteria seen histologically and immunohistochemically, and isolation of C. chauvoei from fetal tissues. Toxemia presumably caused the ultimate demise of the fetus.
Literature review
Etiology
C. chauvoei was discovered in 1887 and was later named after a French veterinarian, JBA Chauveau. 40 C. chauvoei is a gram-positive, anaerobic, and spore-forming rod found in the soil, feces, and the digestive tract of many animals,3,7,38 mainly in the form of spores that are highly resistant to environmental factors and many disinfectants. 40
C. chauvoei can occur in pairs and rarely in short chains, but it is most frequently found as a single organism. Although traditionally considered a gram-positive rod, the staining characteristics are variable and, particularly in old cultures, C. chauvoei may appear gram-negative. Most strains are motile, a feature that is associated with the presence of peritrichous flagella. This microorganism sporulates readily, producing ovoid, and usually sub-terminal, spores. Central and occasionally terminal spores also may occur. Iodine, chlorine, and other oxidizing disinfectants readily destroy vegetative and sporulated forms of C. chauvoei. Spores, however, tolerate the action of quaternary ammonium and phenolic disinfectants, and boiling.1,40
On blood agar, most colonies of C. chauvoei are circular and surrounded by a thin ring of hemolysis. This characteristic somewhat depends upon the source of the red blood cells in the media and the strain of the isolate.1,40 For instance, hemolysis is seen on media containing red blood cells of sheep, cattle, rabbits, pigs, or dogs, but not on media containing erythrocytes of humans, horses, guinea pigs, or chickens. The colonies of C. chauvoei are slightly raised, white-gray, and glossy, becoming increasingly opaque as incubation progresses. When spores are formed, usually after 24 h of incubation, the colonies are completely opaque. They usually do not merge, but older cultures may show some degree of confluence. Although C. chauvoei ferments several carbohydrates, this microorganism can also grow on media without carbohydrates, obtaining energy from amino acid fermentation. 40
The pathogenicity of C. chauvoei is linked to several toxins and virulence factors (Table 1). Among these, several hemolysins result in cytolysis and hemolysis, causing the characteristic lesions of blackleg.8,17,29,38 The main virulence factor of C. chauvoei is toxin A (CctA), a β-barrel, pore-forming hemolysin.11,28,29 CctA is a soluble monomer that oligomerizes on the cell surface to form a polymer pore that perforates the cell membrane, resulting in disruption of membrane permeability and cell lysis. Novel experimental vaccines containing CctA demonstrate protective immunity in immunized guinea pigs. 11 Another β-barrel, pore-forming hemolysin produced by C. chauvoei is chauveolysin. This is a cholesterol-dependent cytolysin that targets cholesterol on the cell surface and forms a larger pore composed of 40–50 monomers.28,29 C. chauvoei also produces other virulence factors whose roles in clostridial disease pathogenesis are less well described, including DNase, hyaluronidase, and neuraminidase (a sialidase).8,12,29,45,46 C. chauvoei DNase, a beta toxin, is responsible for DNA degradation through cleavage of phosphodiester bonds. Hyaluronidase, a gamma toxin, enzymatically cleaves hyaluron, an important component of the extracellular matrix, thereby loosening tissues and presumably favoring the spread of C. chauvoei. 12 Neuraminidase enzymatically cleaves sialic acids on host cell surfaces, resulting in decreased rigidity of the cell membrane and degradation of tight junctions. 50 Similar to hyaluronidase, these effects are thought to enhance the spread of C. chauvoei through tissues. Flagella are known to be important virulence factors that contribute to the infectious process by providing C. chauvoei mobility and facilitating the spread of the organism.6,22,29,43,44 Flagellar antigens have been considered as potential candidates for blackleg vaccines, 22 and antibodies to flagellar proteins have been shown to be protective.6,44
Major toxins and virulence factors produced by Clostridium chauvoei.

Epidemiology
Blackleg affects mainly nonvaccinated cattle between 6 mo and 2 y of age,7,15,41,48 with occasional cases occurring in animals outside this age range.4,7 The disease affects mostly animals in good nutritional condition, often on pasture.3,4,7,41,47,48
Despite the frequently repeated claim that both spores and vegetative forms of C. chauvoei are found in feces of healthy and sick animals, little information is available in this regard in the scientific literature. 38 The current, but unproven, dogma is that regardless of the origin, the spores of C. chauvoei can contaminate the soil, survive in the environment for decades, and can infect animals grazing on contaminated pastures.9,38,47 Less commonly, the disease can occur in housed animals, when the silage, forage, hay, or other feed is contaminated with spore-containing soil.3,14 Ingestion of C. chauvoei spores is probably the most common form of exposure, and infected ruminants do not directly transmit the disease to other animals.
A correlation between the incidence of blackleg and rainfall rate seems to exist, with most cases of the disease occurring in wet seasons, and cases of blackleg being frequently reported in cattle that were moved to a new pasture following rainfall.18,48 It has been suggested that this correlation is based on 1) the rain facilitating the dissemination of spores, and 2) water saturation of the soil favoring anaerobiosis and thus multiplication of C. chauvoei. 47
A few cases of a disease thought to be blackleg have been described in sheep.13,14 However, because in many of those cases the site of entry was a cutaneous or mucosal wound rather than ingestion, it is likely that most, if not all, of those cases were C. chauvoei–associated gas gangrene rather than blackleg. Infections by C. chauvoei of mostly unknown pathogenesis have been reported in goats, deer, oryx, elephants, horses, pigs, mink, fresh-water fish, whales, frogs, and hens.16,21,25,30,32,40 Gas gangrene 26 and enterocolitis 51 associated with C. chauvoei have been reported, albeit very rarely, in humans.
Pathogenesis
Surprisingly, there is little scientific evidence to support the classical model for this disease (Fig. 3). According to this model, spores of C. chauvoei present in contaminated pastures are ingested and undergo one or more replication cycles in the intestine before being absorbed through the intestinal mucosa to the bloodstream and/or being excreted in feces.7,12,31 Once absorbed, the spores are distributed to multiple tissues, including skeletal and cardiac muscle. Transport is assumed to be mostly hematogenous, although lymphatics may also play a role in dissemination of spores.7,23,31,41,52 Once the spores of C. chauvoei reach the muscle, they are phagocytized by resident macrophages, and can survive in the cytoplasm of those cells for long periods of time without affecting the host. 27 But when anaerobic conditions are created in areas where the spores are present, most frequently related to trauma and associated hemorrhage and necrosis, those latent spores germinate, proliferate, and release toxins that produce the clinical manifestations and lesions of blackleg.1,7,19,23,31,41,45
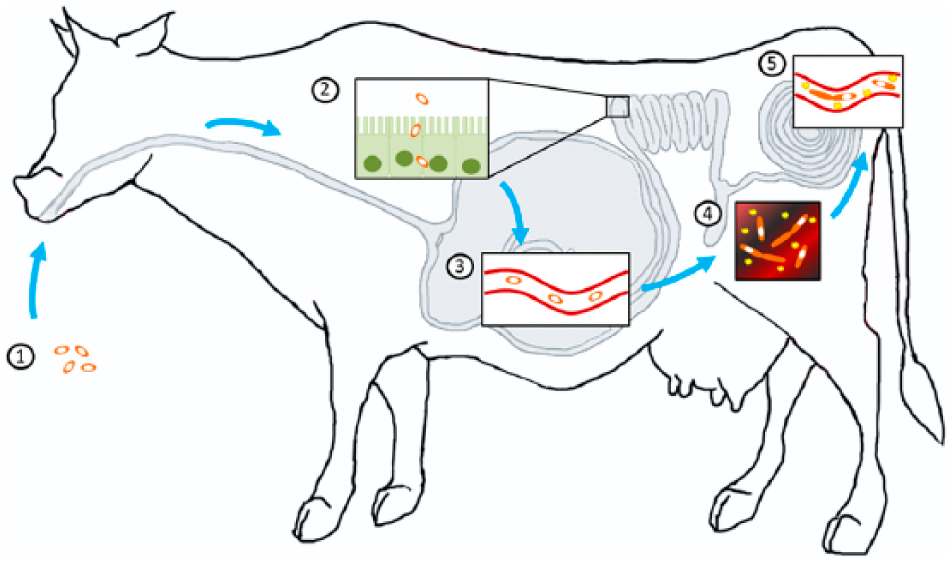
Proposed pathogenesis of blackleg. Clostridium chauvoei spores are initially ingested from contaminated soil (1) and are absorbed across the intestinal mucosa into the bloodstream (2). Spores are then spread to multiple tissues, primarily skeletal muscle, where they become dormant (3). Later, a low oxygen environment is created in the skeletal muscle, often from an acute injury, favoring spore germination. The bacteria produce potent exotoxins that cause local necrosis and inflammation (4). Finally, proliferating bacteria and toxins enter the bloodstream causing exotoxemia and ultimately death (5).
It has been suggested that, in addition to blunt trauma, hypoxia associated with excessive exercise or other factors may also predispose to spore germination in sheep. 14 This mechanism would help explain the few cases in which lesions have been seen in the heart but not in skeletal muscles in cattle. 48 Based on this model of pathogenesis, blackleg is referred to as an “endogenous” infection. It is important to distinguish this pathogenesis from the so-called exogenous mechanism of gas gangrene, a disease produced when spores or vegetative forms of one or more clostridial species, including C. chauvoei, enter subcutaneous and/or muscular tissues via skin or mucosal wounds. 7 The complex pathogenicity of C. chauvoei requires various virulence factors including potent toxins, but also enzymes and organelles enabling the bacterium to reach its destination, the muscle tissue (Table 1). 12
Although this pathogenesis model of blackleg has been accepted by veterinarians for many years, several inconsistencies remain unexplained. For example, cases or outbreaks of blackleg that occur: 1) recurrently, usually every year, in the same geographic region, 2) soon after movement of animals to new pastures, and 3) in animals fed feedstuff believed to be heavily contaminated with C. chauvoei. These special circumstances do not contradict the classic model of blackleg; rather, they suggest that other pathogenic mechanisms may also play a role. A possible explanation for the 3 circumstances mentioned above is that septicemia with active infection occurring in target organs without the need for trauma occurs when animals are exposed to unusually large loads of C. chauvoei. If this is the case, the mechanism generating anaerobiosis in those tissues remains unknown. The reason for the preference of this microorganism to selectively colonize muscle is unknown. 1
In most cases of blackleg, heart and skeletal muscle lesions occur in the same animals. It has been postulated that the latter develops first and that metastatic colonization of the heart and/or deeper skeletal muscles such as those of the sub-lumbar area and diaphragm occurs secondarily. If this is the case, toxemia and/or bacteremia-associated hypoxia would act as a predisposing factor for the cardiac and/or deep muscles lesions. Pericarditis, another lesion usually observed associated with cardiac lesions, is thought to be a consequence of local extension; however, pericarditis as a direct consequence of septicemia cannot be ruled out. 1
The pathogenesis of cardiac lesions, particularly in those rare cases in which cardiac but no skeletal muscle lesions are observed, has not been satisfactorily explained. Although it is possible that at least in some cases, once cardiac lesions are found at autopsy, skeletal muscle lesions are overlooked, there seems to be enough evidence that, albeit rarely, cardiac lesions may occur in the absence of skeletal muscle lesions. Several predisposing factors were suggested for an outbreak of blackleg in sheep in which myocardial, but no skeletal muscle lesions, were found. These include increased cortisol 10 and catecholamine levels associated with stress, several toxins such as ionophores and gossypol, and vitamin E and selenium deficiency. 14 It is possible that at least some of those factors are implicated in cases of cardiac lesions in cases of blackleg of cattle in which no skeletal muscle lesions are found.
Clinical signs
Blackleg is typically an acute or subacute disease, with many animals dying suddenly or rarely surviving more than 36 h after the onset of clinical disease. Occasionally chronic cases may occur.15,41,42 In cases in which clinical signs are observed, they include one or more of the following: lethargy, anorexia, reluctance to move, lameness, and recumbency.4,15,41,42,48 When superficial muscles are affected, swelling and crepitus are evident.4,15 Cases with cardiac lesions have similar clinical signs, in addition to increased diffuse lung sounds and dyspnea. Signs of congestive heart failure such as jugular vein distension and brisket edema have rarely been reported. 41
Pathology
Animals that succumb to blackleg are usually in good nutritional condition. 41 When superficial muscles are affected, the overlying skin is stretched by underlying inflammation and is dark. Emphysema of the subcutaneous tissue causes palpable crepitus. Classical necrohemorrhagic and emphysematous myositis is present in the skeletal muscles, mainly in the hindlimbs, giving those muscles a black appearance, from which the name of the disease derived (Fig. 4). The large muscles of the rear quarters are most frequently affected; other muscular groups are less frequently involved (Abreu CC, et al. Pathology of blackleg in cattle. Proc Ann Conf Am Assoc Vet Lab Diagn; 2015; Providence, RI).15,42 Additional, often overlooked, skeletal muscles that should be examined for lesions include the tongue and diaphragm (Fig. 4).
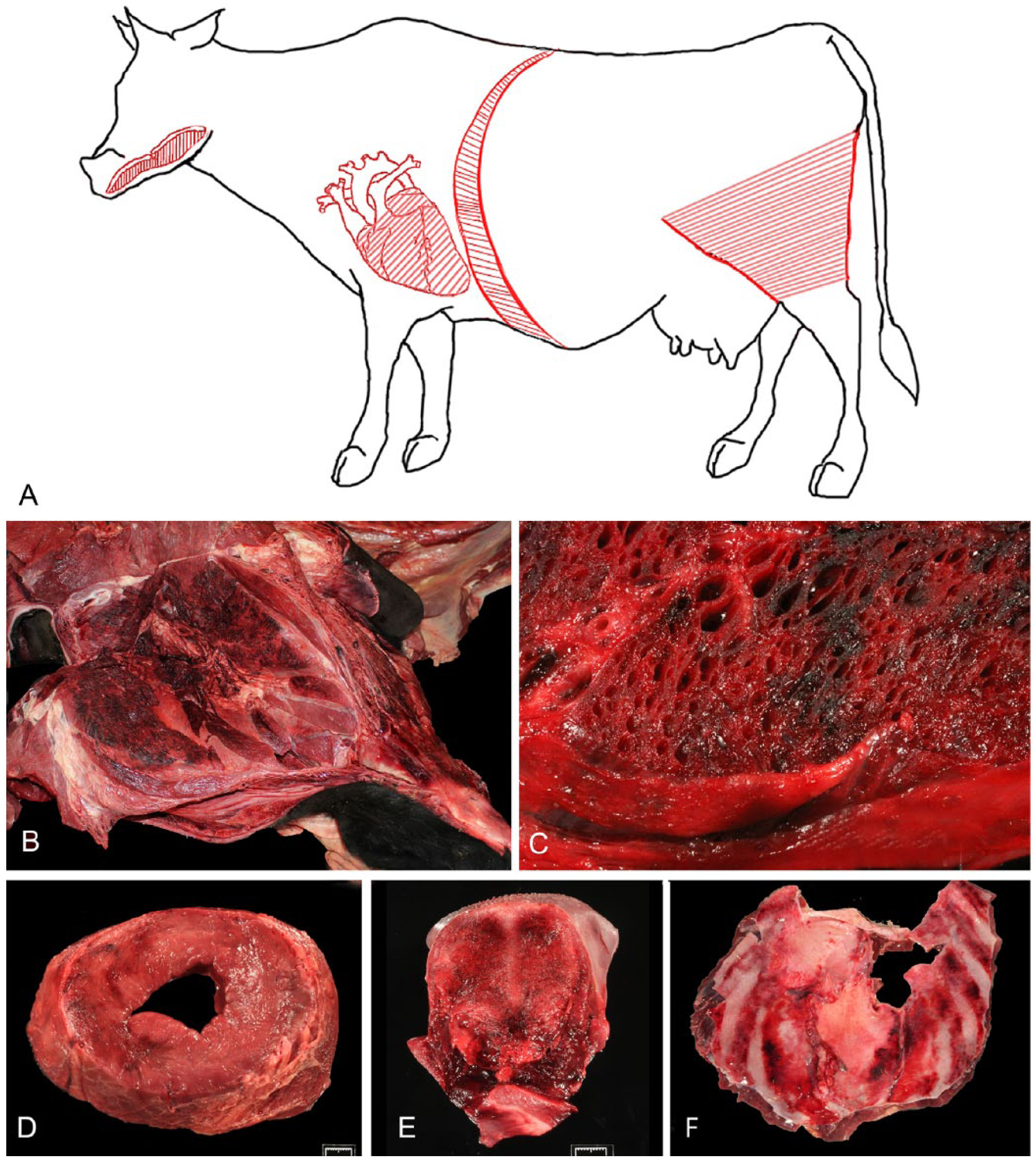
Gross lesions of blackleg in cattle.
Serosanguineous fluid and emphysema expand the subcutaneous tissues and fasciae of affected areas. The affected muscles in these areas show variably sized, dark red to black, dry, friable areas with many small cavities produced by gas accumulation (emphysema; Fig. 4). The peripheral areas of affected muscles are edematous. Typically, a scent described as reminiscent of rancid butter emanates from those lesions.1,7,15,42 When present, lesions in the tongue and diaphragm (Fig. 4) are similar to those seen in peripheral skeletal muscles although less severe. Emphysema has not, however, been described in the tongue.
A recent study from California (author’s unpublished) suggested that, contrary to previous beliefs,41,48 lesions in the heart are seen in the great majority of cases of blackleg. Cardiac lesions are mainly characterized by necrohemorrhagic myocarditis (Fig. 4), usually in association with skeletal muscle lesions, although, a few cases have been described with cardiac, but no skeletal muscle, lesions.14,20,33,48 The myocardial lesions can affect the walls of any of the chambers of the heart, and they may be focal or multifocal. Cardiac lesions generally lack the emphysema that typically accompanies the skeletal myositis, or have only a small amount of gas. Pericarditis is a characteristic gross finding, the pericardium being diffusely congested and hemorrhagic, and the 2 layers of the pericardial sac often have multiple adhesions by variable amounts of fibrin (Abreu CC, et al. Pathology of blackleg in cattle). Occasionally, fibrinous endocarditis may be observed associated with myocardial lesions. Pulmonary congestion, edema, and hemorrhage are almost always present, and sometimes are accompanied by adjacent fibrinous pleuritis in areas in contact with the heart.41,48
The histologic lesions of blackleg are similar in cardiac and skeletal muscle (Fig. 5). At low magnification, large empty vacuoles, representing emphysematous spaces created by gas bubbles, usually are evident in the interstitium of skeletal muscle but, rarely in the myocardium. Myocytes show degenerative and/or necrotic changes consisting of hypereosinophilic, swollen, vacuolated, and fragmented myofibers that may display hypercontraction bands and loss of cross-striations. Leukocytic infiltration is not a prominent feature in most blackleg cases. This is believed to be a consequence of the acute nature of the disease and also the leukocytoclastic effect of some of the exotoxins produced by C. chauvoei. 29 Occasionally, however, neutrophils, and later on in the process, macrophages, may infiltrate some of the affected myofibers. The interstitium is usually expanded by hemorrhage and proteinaceous edema.
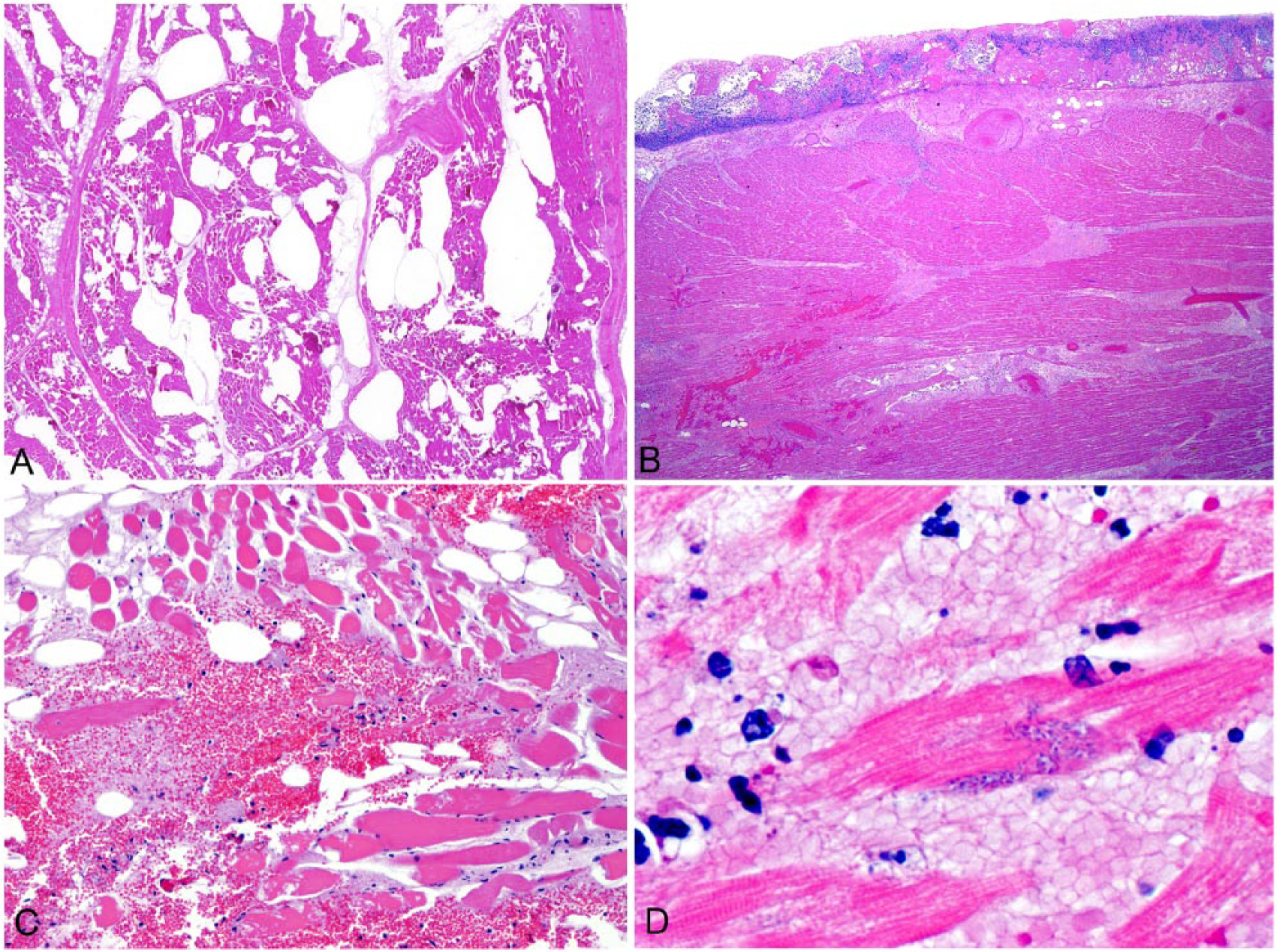
Histologic lesions of blackleg in cattle.
Vascular fibrinoid degeneration and necrosis, with many blood vessels infiltrated by neutrophils and containing fibrin thrombi, is a prominent feature of blackleg. Vascular changes are more prevalent in cardiac lesions. Large numbers of gram-positive rods, which may contain sub-terminal, central, or, rarely, terminal spores, are present in the interstitium (usually surrounding affected blood vessels) or within myofibers. Fibrinosuppurative and hemorrhagic pericarditis and pleuritis, with occasional vascular changes similar to those described in muscle, are also observed.1,7,41,42,48
Diagnosis
Although a presumptive diagnosis of blackleg is usually based on clinical history, signs, and gross and histologic changes, a final diagnosis requires detection of C. chauvoei in affected tissues. This can be achieved by culture, PCR,24,34–37 and/or immunodetection methods, including FAT and IHC. 1 Decisions on which tests to use are often based on availability at local diagnostic laboratories.
The primary differential diagnosis for blackleg is gas gangrene, also known as malignant edema, a clostridial cellulitis and sometimes also myositis, associated with wound contamination. This disease may be produced by one or more of several clostridial species, including C. septicum, C. chauvoei, C. perfringens, C. sordellii, and C. novyi.2,7,19,31 Gross microscopic lesions of blackleg and malignant edema can be difficult to differentiate when the disease is restricted to skeletal muscle, especially in cases in which wounds are not readily evident. Additional tests are often needed for distinction.2,7,19,23,31
Prevention and treatment
Blackleg can be prevented by vaccination, a procedure that is an important component of the health management of many cattle-producing operations. 9 Conventional blackleg vaccines are bacterins, prepared from formalin-treated cultures of C. chauvoei, that are generally available in polyvalent formulations together with other clostridial components. The evidence for the efficacy of these vaccines is mostly anecdotal or based on measurement of antibody titers in vaccinated animals. However, the literature on clinical trials of these vaccines in cattle is surprisingly scant. 49 Nevertheless, the limited information available shows that these vaccines are nearly 100% effective in preventing blackleg after natural exposure, and 50–100% effective against experimental challenge with C. chauvoei. 49
Because of the age distribution of the disease, initial vaccination against blackleg is usually recommended at 2 mo of age, followed by a booster 4–6 wk later. Annual or bi-annual boosters are recommended after that, until 2 y of age. 1
Guinea pigs vaccinated with recombinant CctA were protected against challenge with a virulent strain of C. chauvoei, making this toxin a promising candidate for novel vaccines against blackleg. 11 Another likely candidate for vaccine production is the neuraminidase of C. chauvoei. It is expected that a combination of this antigen with one or more polypeptides derived from the toxins produced by C. chauvoei will have a potentiated protective effect against blackleg. 50 Additionally, antibodies against C. chauvoei flagella have been shown to induce a protective immune response.6,22,44 Reducing stress and trauma, especially in younger cattle, has been suggested to reduce the incidence of blackleg. 1
Conclusions
Blackleg is a preventable histotoxic clostridial disease that mainly affects young cattle raised on pasture. The clinical course of disease is rapid and often results in death within 36 h. At autopsy, there is classically necrohemorrhagic and emphysematous myositis in the skeletal muscles of the hindlimbs, giving the leg a black appearance that inspired the disease name, and often similar findings in the heart occasionally with fibrinous pericarditis. Clinical, gross, and histologic findings can lead to a presumptive diagnosis of blackleg. Diagnostic confirmation requires the detection of C. chauvoei by culture, PCR, or immunostaining methods. Blackleg is preventable by vaccination.
The currently accepted pathogenesis of blackleg involves ingestion of spores, with absorption through the intestinal mucosa into the blood circulation. In skeletal and cardiac muscle, the spores are phagocytized by macrophages, and when anaerobiosis develops, usually associated with blunt trauma, the spores germinate and produce highly toxic virulence factors. Although the currently accepted pathogenesis of blackleg seems plausible, most evidence to support the proposed chain of events in the pathogenesis is circumstantial with no definitive supporting proof available. In addition, anecdotal evidence suggests that cases of blackleg may also occur in animals not subjected to trauma, for which a mechanistic explanation is not currently available.
Footnotes
Acknowledgements
We thank Dr. Gwen Levine for her cytology expertise and Dr. Sonia Lingsweiler for her microbiology assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
