Abstract
Blackleg is an infectious disease of cattle and rarely other ruminants, produced by Clostridium chauvoei and characterized by necrotizing myositis. In most cases of blackleg, the large muscles of the pectoral and pelvic girdles are affected, with other skeletal muscles and the heart involved less frequently. We studied 29 blackleg cases selected from the archives of the California Animal Health and Food Safety Laboratory, 1991–2015. Immunohistochemistry was also evaluated to detect C. chauvoei in formalin-fixed, paraffin-embedded (FFPE) tissues of cattle. Nineteen animals had gross and/or microscopic lesions in both skeletal muscle and heart, 9 had lesions in the skeletal musculature alone, and 1 in the heart alone. Gross lesions in the skeletal musculature involved the following muscle groups: hindquarters (n = 8), forequarters (n = 5), neck (n = 5), lumbar area (n = 3), brisket (n = 2), diaphragm (n = 2), abdominal wall (n = 1), thoracic wall (n = 1), and tongue (n = 1). Of the 20 animals that had lesions in the heart, 11 had pericarditis and myocarditis; 7 had pericarditis, myocarditis, and endocarditis; and 1 each had pericarditis and myocarditis. Immunohistochemistry was 100% sensitive to detect C. chauvoei in FFPE skeletal muscle and/or heart of cattle with blackleg. Simultaneous lesions in skeletal musculature and heart were relatively common in blackleg cases in California; the most affected skeletal muscles were those of the hindlimbs.
Introduction
Blackleg is an infectious disease of cattle and rarely other ruminants, produced by Clostridium chauvoei and characterized mainly by necrotizing myositis. C. chauvoei is an anaerobic, gram-positive, spore-forming rod, which is found in the soil and, less frequently, in the digestive tract of many animals,1,2,4,17 mainly in the form of spores that are highly resistant to environmental factors and disinfectants.15,18 The spores of this microorganism have been found within the cytoplasm of macrophages in the skeletal muscle of healthy cattle. 15 C. chauvoei spores or vegetative forms present in the feces of healthy or sick animals or in carcasses can contaminate the soil 17 where the spores can survive for many years. 21 Because of this, blackleg most commonly affects pastured animals. However, the disease can also occur in housed animals when feed is contaminated with soil that contains spores. 11 Most cases of blackleg occur during the rainy season or periods of high rainfall,1,21 and a positive correlation exists between annual rainfall and outbreaks of blackleg. 21 It is thought that high rainfall may assist in the dissemination of spores; additionally, water-saturated soils are more prone to conditions with minimal or no oxygen, which favor the germination of C. chauvoei spores and multiplication of the microorganism.1,21
The pathogenesis of blackleg is not fully understood but it is believed to involve ingestion of spores, followed by absorption through the intestinal mucosa.1,2 The spores are then distributed via the bloodstream to multiple tissues, including skeletal and cardiac muscle, where the spores are phagocytized by tissue macrophages.1,2,13,15 Once in the cytoplasm of these macrophages, the organisms can survive for months to years without deleterious effects to the host. However, when the oxygen tension drops in areas of muscle in which spores are present, usually as a consequence of blunt trauma and associated tissue hemorrhage, degeneration, and necrosis,1,2,13,15 the spores germinate, proliferate, and produce toxins that are responsible for most clinical signs and lesions of blackleg. Because of this proposed pathogenesis, blackleg is usually referred to as an endogenous infection. C. chauvoei toxin A is considered to be the main virulence factor of this microorganism,8,9,16 although the so-called molecular Koch postulates have not yet been fulfilled and it is therefore possible that other toxins may be involved in the pathogenesis of the disease.
It is believed that in most cases of blackleg, the large muscles of the pectoral and pelvic girdles are affected; other skeletal muscles are involved less frequently. 2 However, to our knowledge, there is only one article published on the distribution of lesions in cattle with C. chauvoei infection. 14 Also, although lesions in the heart were traditionally considered unusual,17,23 anecdotal evidence suggests that they are rather common.
We therefore performed a retrospective study of the gross and microscopic changes in 29 cases of blackleg in cattle in California. In addition, we evaluated the performance of immunohistochemistry (IHC) to detect C. chauvoei in formalin-fixed, paraffin-embedded (FFPE) tissues of these animals.
Materials and methods
Case selection and clinical history
Cases of blackleg occurring between 1991 and 2015 (n = 29) were selected from the archives of the San Bernardino, Davis, and Tulare laboratories of the California Animal Health and Food Safety Laboratory System (CAHFS). The cases were selected from a larger pool of cases on the basis that they fulfilled the following 2 conditions: 1) bovine carcasses received for autopsy with gross and microscopic findings consistent with blackleg, and 2) positive culture and/or fluorescent antibody test (FAT) for C. chauvoei in affected muscle. The information available on signalment, clinical history (Table 1), gross and microscopic pathology (Table 2), and ancillary tests (Table 3) was reviewed. Unless otherwise specified, all laboratory tests done on samples from these animals were performed following standard operating procedures (SOPs) of CAHFS.
Signalment and clinical information of 29 animals with blackleg.

Some animals had more than one clinical sign.
Number of animals with different microscopic lesions in skeletal muscle and heart in 17 and 15 cattle with blackleg, respectively.
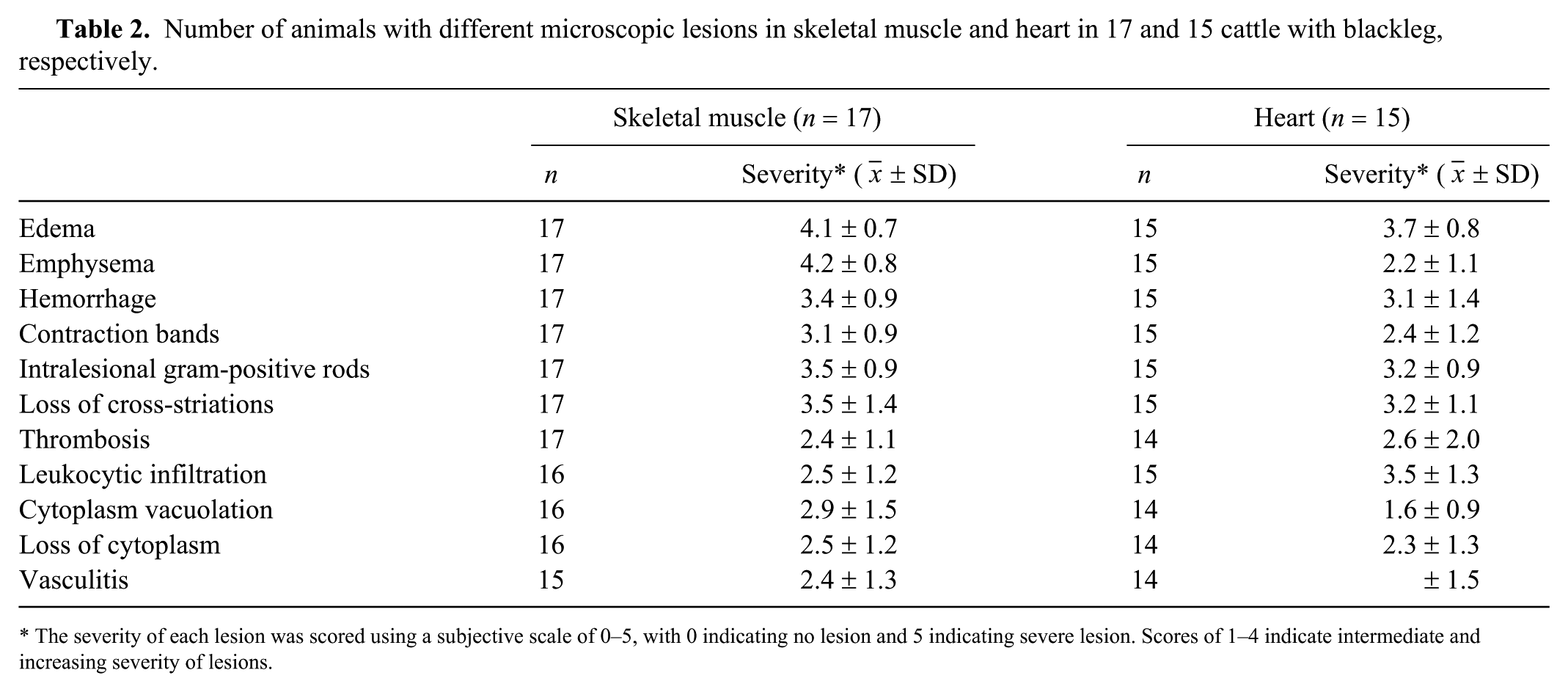
The severity of each lesion was scored using a subjective scale of 0–5, with 0 indicating no lesion and 5 indicating severe lesion. Scores of 1–4 indicate intermediate and increasing severity of lesions.
Results of anaerobic culture, fluorescent antibody test, and immunohistochemistry for Clostridium chauvoei in cattle with blackleg.
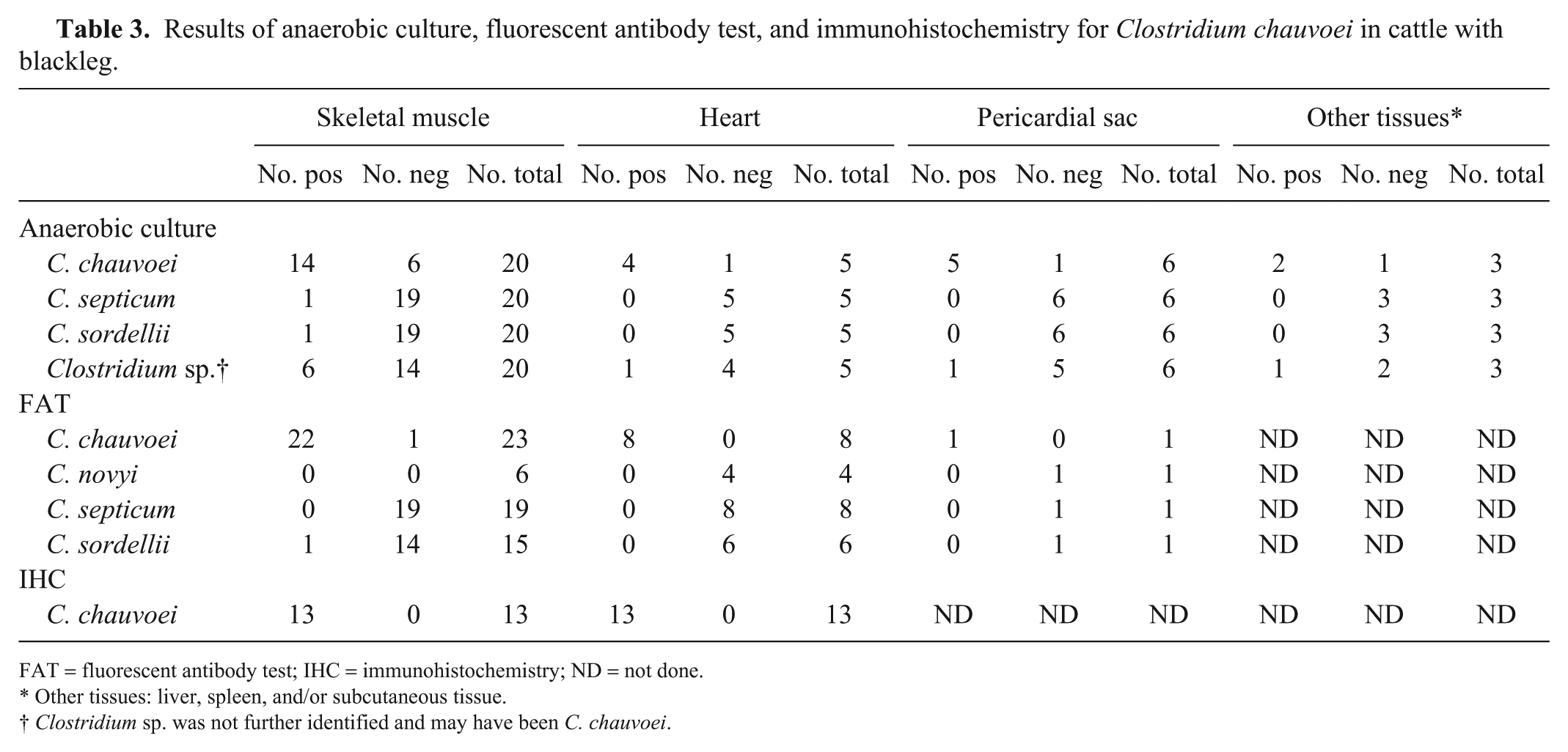
FAT = fluorescent antibody test; IHC = immunohistochemistry; ND = not done.
Other tissues: liver, spleen, and/or subcutaneous tissue.
Clostridium sp. was not further identified and may have been C. chauvoei.
Gross and microscopic pathology
Autopsy had been performed on all animals by a total of 11 different pathologists of CAHFS (Davis, Tulare, and San Bernardino laboratories). We reviewed the gross information from each individual autopsy report. Samples of skeletal muscle and heart, and of a variety of other tissues including subcutaneous tissue, skin, lung, trachea, liver, kidney, adrenal gland, brain, spleen, thymus, various lymph nodes, esophagus, rumen, reticulum, abomasum, small intestine, cecum, colon, peri-vaginal tissue, mammary gland, and/or placenta, had been collected in most cases and fixed in 10% buffered formalin (pH 7.2) for 24–72 h. All tissues had been processed by routine histologic techniques to produce 4-µm thick sections, which were stained with hematoxylin and eosin (H&E). Selected sections of skeletal muscle and heart were also stained with Gram stain. Histologic sections that were still available were re-examined by one of the authors (CC Abreu). Special emphasis was put on sections of skeletal muscle (17 cases) and heart (15 cases), which were analyzed for the presence or absence, and, if present, severity of the following changes: edema, emphysema, hemorrhage, contraction bands, loss of cross-striations, cytoplasmic vacuolation, loss of cytoplasm, leukocytic infiltration, thrombosis, vasculitis, and intralesional gram-positive rods. The severity of each of these lesions was scored using a subjective scale of 0–5, with 0 indicating no lesion and 5 indicating severe lesion. Scores of 1–4 indicated intermediate and increasing severity of lesions.
Immunohistochemistry
Paraffin blocks of skeletal muscle and heart were available from 13 animals. Sections from these blocks were processed by an indirect immunoperoxidase technique for C. chauvoei (R.T.U. VECTASTAIN Elite ABC system, Vector Laboratories, Burlingame, CA), according to the manufacturer’s instructions and SOP of CAHFS. Briefly, endogenous peroxidase was blocked with a 3% solution of hydrogen peroxide. Antigen retrieval was achieved with pepsin. The sections were treated (Background Punisher, Biocare Medical, Concord, CA) to block nonspecific binding, before being incubated with a goat anti–C. chauvoei polyclonal antibody (VMRD, Pullman, WA) for 60 min at 37°C. Vector NovaRED (Vector Laboratories) was used as chromogen for visualization. Skeletal muscle and heart sections from a calf from which C. chauvoei had been isolated and that was also positive by FAT for C. chauvoei were used as positive controls. Skeletal muscle and heart tissues from a clinically healthy steer that was negative for anaerobic culture and C. chauvoei FAT were used as negative controls. Additional negative controls included sections from each case that were treated with normal goat IgG (Vector Laboratories) instead of the primary antibody. The primary C. chauvoei antibody had been shown to be specific for this microorganism; muscle sections from animals infected with Clostridium septicum, Clostridium novyi, Clostridium sordellii, or Clostridium perfringens did not stain with this antibody.
Bacterial cultures and PCR
Samples of skeletal muscle, heart, and/or subcutaneous and/or interstitial edema of 25 animals had been collected aseptically and subjected to anaerobic culture. Briefly, the samples had been inoculated onto pre-reduced, anaerobically sterilized Brucella agar plates (Hardy Diagnostics, Santa Maria, CA) and cooked meat medium (Hardy Diagnostics), and incubated anaerobically at 37°C for up to 5 d. Samples of subcutaneous tissue, skeletal muscle, pericardium, heart, pleura, lung, liver, kidney, spleen, thymus, and/or mesenteric lymph node, from 25 animals, had also been inoculated onto 5% sheep blood Columbia agar plates (Hardy Diagnostics) and MacConkey agar plates (Hardy Diagnostics) and incubated aerobically at 37°C for 48 h.
Fluorescent antibody test
Impression smears of skeletal muscle and heart from 23 and 8 animals, respectively, were used to perform FAT for C. chauvoei. Smears from at least 1 of these 2 locations were processed by C. chauvoei FAT in all animals included in our study. In addition, smears of the same tissues from a variable number of animals had been processed for FAT for C. novyi, C. septicum, and/or C. sordellii (Table 3). The smears were fixed in anhydrous acetone for 10 min at 4–8°C. They were then incubated with the corresponding polyclonal antiserum conjugated to fluorescein isothiocyanate (VMRD), mixed with 1% Evans blue for 30 min at 37°C in a humidified chamber. After this, the slides were rinsed with phosphate-buffered saline, and FAT rinsing buffer for 10 min. The smears were finally mounted in buffered glycerol (pH 9.0), and observed with a fluorescence microscope.
Results
Signalment and clinical history
The animals were 2–19 mo old, with most of the cases occurring in animals 6–12 mo old (Table 1). Most of the animals died in winter (rainy season in California), although several cases also occurred in spring, summer, and autumn. Most animals were female (n = 20), were in good nutritional condition, and were on pasture. In those cases in which clinical signs were observed, the signs consisted of leg weakness, painful swelling of musculature (both of which were most frequently seen in rear quarters), difficulty in standing, and dehydration. Death occurred within 24 h of onset of clinical disease in all cases in which this information was reported. Most animals for which vaccination history was available were not vaccinated against C. chauvoei. Breeds represented in our study were Holstein (n = 12), crossbreed (n = 6), Angus (n = 3), Charolais (n = 3), Limousin (n = 2), Red Angus (n = 1), and Angus cross (n = 1). The breed was not reported in 1 case.
Gross pathology
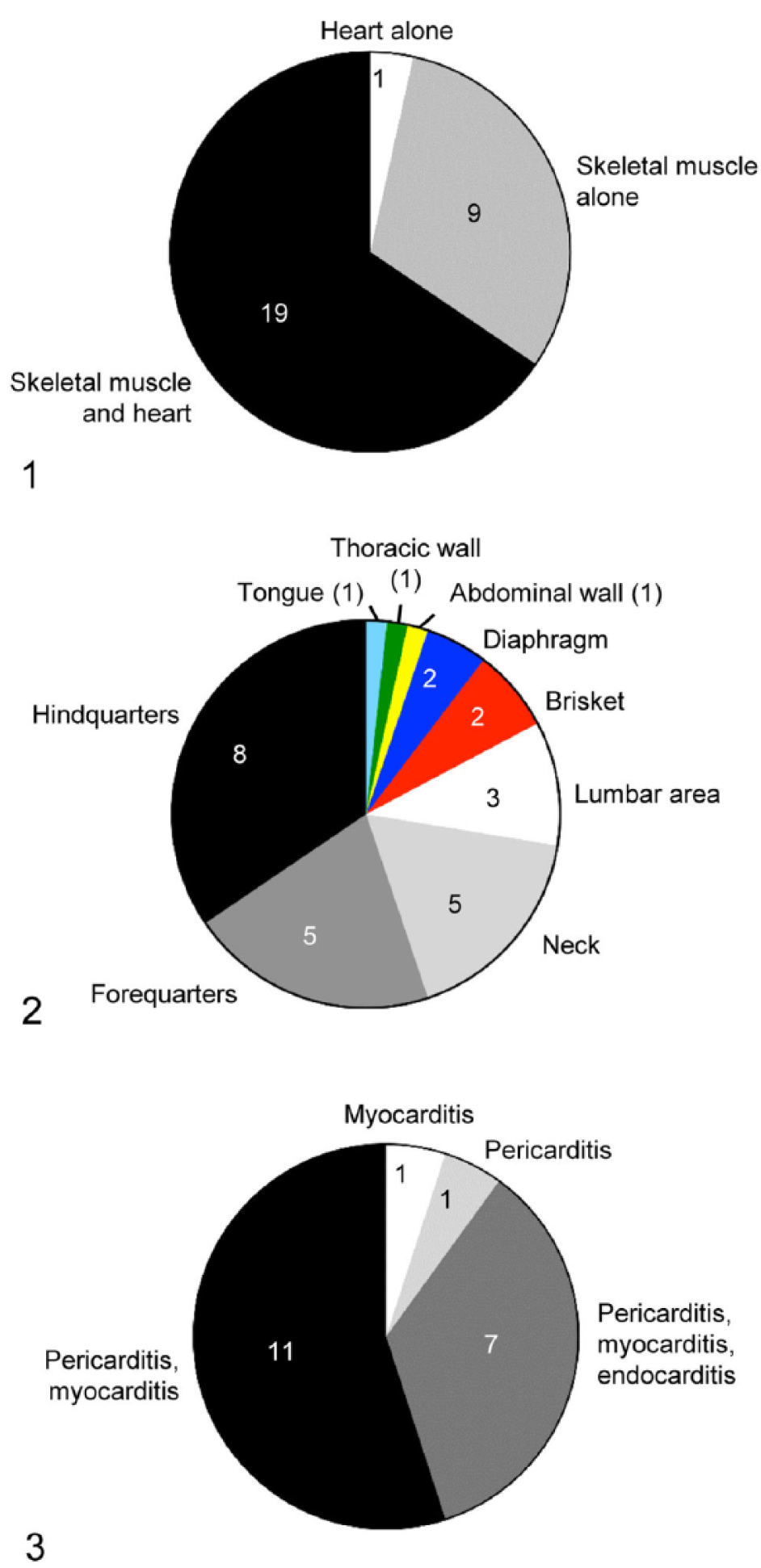
Most of the animals (n = 19) had lesions in both skeletal and heart musculature, and a few animals (n = 9) had lesions in skeletal musculature alone. Only one animal had lesions only in the heart (Fig. 1). The hindquarters were the skeletal muscle groups most frequently affected, followed by muscles of the forequarters, neck, lumbar area, brisket, diaphragm, abdominal wall, thoracic wall, and tongue (Fig. 2). Of the 28 animals that had lesions in skeletal muscle, 12 had lesions affecting 2 muscle groups, 9 showed lesions in 1 muscle group, 4 in 4 muscle groups, and 1 in 5 muscle groups.
Lesions in cattle with blackleg.
Of the 20 animals that had gross anatomic lesions in the heart, 19 had myocardial lesions. Although the specific location of the myocardial lesions was not recorded in some cases, in those in which this information was available, gross lesions were seen equally distributed in the walls of both ventricles and both atria. Most of these animals (n = 19) also had pericarditis and/or endocarditis, but one animal had only myocarditis and another one had only pericarditis (Fig. 3).
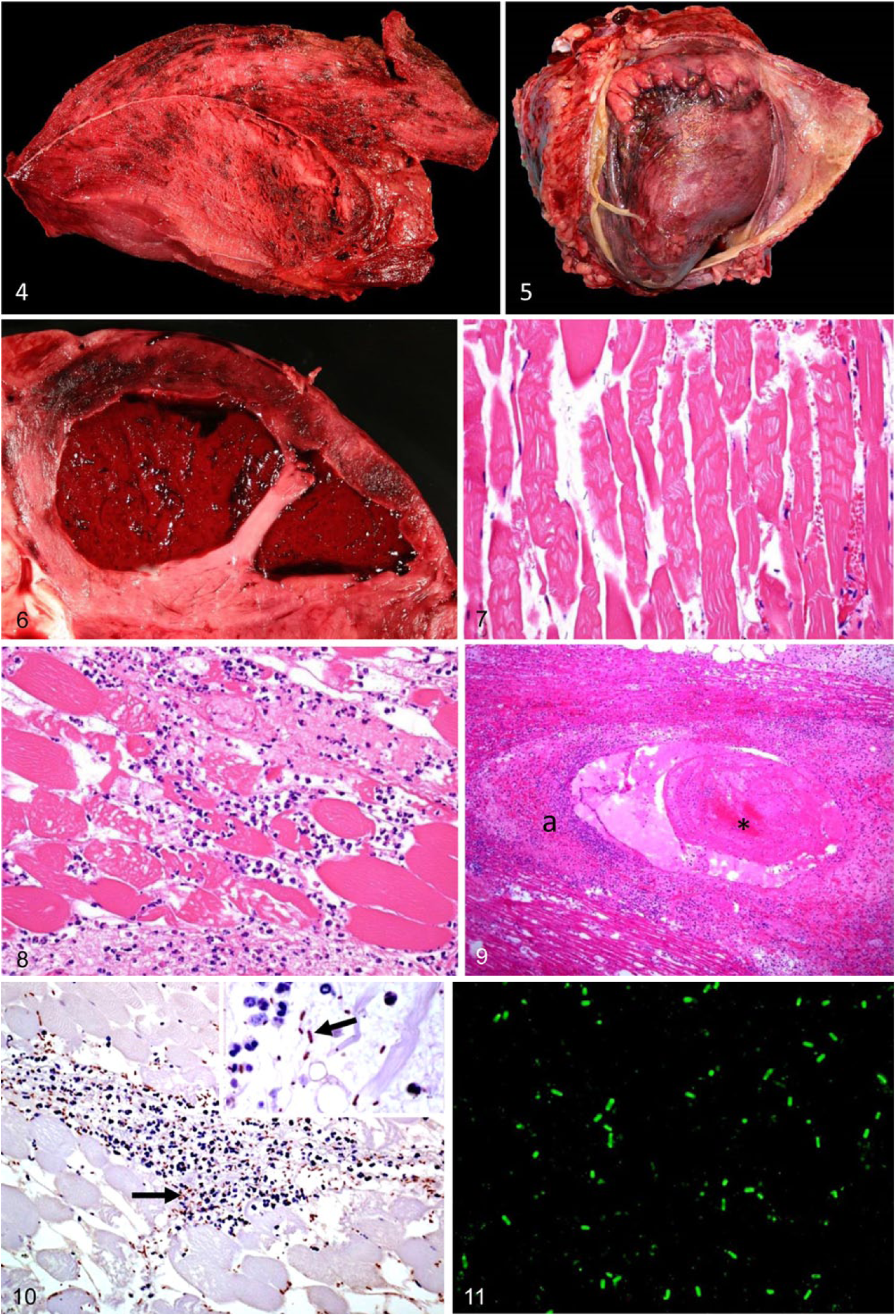
Gross anatomic lesions in cases in which superficial skeletal muscles were affected were seen externally as swelling, with the overlying skin being taut and usually dark. Crepitation was noticed in the subcutaneous tissue of affected areas by palpation. The subcutaneous tissues and fascia around the lesions were expanded by serosanguineous fluid in which gas bubbles were apparent. Affected muscles were dry, friable, dark-red to black, and had multiple small cavities because of gas bubbles (Fig. 4). Affected muscles were edematous in the periphery of the lesions. The odor that emanated from the affected tissues was sweet and reminiscent of rancid butter.
Lesions in cattle with blackleg.
In those cases in which pericarditis and/or epicarditis were present, the pericardial sac was attached to the epicardial surface by fibrinous inflammatory exudate (Fig. 5), and the pericardial sac contained serosanguineous fluid. Myocardial lesions consisted of focally extensive dark areas of hemorrhage, necrosis (Fig. 6), and occasional emphysema.
Other common lesions included fibrinous pleuritis affecting the pleura adjacent to the heart (n = 7), hepatic (n = 1) and pulmonary congestion (n = 15), hydrothorax (n = 1), and ascites (1).
Microscopic pathology
The microscopic lesions were similar in all animals and, with a few exceptions, were similar in cardiac and skeletal muscles (Table 2). Microscopic changes consisted of interstitial hemorrhage and proteinaceous effusion, muscle fiber swelling, hypereosinophilia, loss of cross-striations, contraction bands (Fig. 7), vacuolation, and fragmentation (Figs. 7, 8). Some of the fragmented myofibers were infiltrated by small to occasionally large numbers of neutrophils (Fig. 8). Within perimysial and epimysial stroma of skeletal muscles, there was multifocal infiltration of viable and degenerating leukocytes, with a predominance of neutrophils and fewer lymphocytes, plasma cells, and macrophages. There was multifocal interstitial accumulation of gram-positive rods with rounded ends, some of them with a central or sub-terminal spore that deformed the cell; these rods were observed singly or in clusters. Multifocally, a few areas had less interstitial hemorrhage and proteinaceous edema, but more pronounced disruption of interstitium and muscle bundles by large gas bubbles. Many interstitial vessels contained thrombi. In addition, arterioles and arteries were frequently affected by fibrinoid degeneration and necrosis, and were infiltrated by neutrophils (Fig. 9); this change was more prominent in the myocardium than in the skeletal muscle. Large numbers of sporulated and non-sporulated gram-positive rods surrounded some of these affected blood vessels. The epicardial and endocardial stroma had multifocal hemorrhages, fibrinous effusion, and leukocytic, mostly neutrophilic, infiltration. Arterial thrombosis in the lung was seen in one case.
C. chauvoei IHC
IHC for C. chauvoei was performed on skeletal muscle and heart of 13 animals that were positive by FAT and culture. IHC was positive in all cases, and positively stained bacteria were observed in a similar location to that observed in the sections stained with H&E and Gram (Fig. 10).
Bacteriology and FAT
C. chauvoei was isolated from skeletal muscle, heart, pericardial sac, subcutaneous tissue, liver, and/or spleen of most animals in which anaerobic culture was performed (Table 3). Clostridium sp., C. septicum, and/or C. sordellii were also isolated from a few animals (Table 3). Escherichia coli, other coliform bacteria, Streptococcus gallolyticus, Bacillus sp., Proteus sp., or mixed flora were isolated from several specimens of a few animals. FAT was positive for C. chauvoei on smears of skeletal muscle and heart of the great majority of animals subjected to this test (Table 3, Fig. 11). FAT for other clostridia investigated gave variable results (Table 3).
Discussion
Our aim in this retrospective study was to document the gross and microscopic changes in a series of cases of blackleg. Additionally, we evaluated the ability of IHC to detect C. chauvoei in archival FFPE tissues of cattle with blackleg. In our case series, simultaneous lesions in several skeletal muscle groups and heart were very common; 19 cases had lesions in both locations. Only one article 14 has been published describing the distribution of lesions in cattle with C. chauvoei infection. That article, however, indicates that skeletal myositis-only lesions account for half of blackleg cases as compared to the approximately one-third of the cases (n = 9) described herein. Our results also differ from previous ones by finding that cardiac lesions are more common than thought previously. Surprisingly, the paper referred to above 14 describes pericarditis in 6 of 29 cases, but not myocarditis. Most references indicate that heart lesions are relatively uncommon.6,23
A possible explanation for the discrepancy between our results and those published previously is that it is plausible that, in most of the reported cases of blackleg, once the easier-to-find skeletal muscle lesions were detected, the carcasses were not investigated further and possible myocardial lesions went unnoticed. This would have been most likely the case in field postmortem examinations but less probable for autopsies performed in a postmortem room where most carcasses are examined systematically. The high rate of concurrent heart lesions is important when field postmortem examinations are performed because if blackleg is not suspected and the animal dies lying on the affected side, only a heart lesion may be noticed and submitted to the laboratory for testing. Although a diagnosis of blackleg can be established by testing heart lesions, this might reduce the chances of establishing a final diagnosis. Finally, the number of cases included in our study is limited, as is the geographic area, which could have led to skewed results.
The skeletal muscles affected most frequently in our study were those of the rear quarters, which is consistent with previous descriptions of the disease.1,2,11,20 However, in most animals of our study, the lesions in skeletal musculature were observed in more than one muscle group. Given that this was a retrospective study based on autopsies performed by several different pathologists, we cannot be sure that in every case an extensive search of the carcass for lesions was performed once one affected area was found. The tongue and some non-superficial groups of skeletal musculature, such as the diaphragm and sublumbar muscles, are not always inspected and they may have been missed, at least in some cases.
Most of the animals in our study were on pasture, which agrees with current knowledge of the pathogenesis of blackleg. According to this generally accepted mechanism, ingestion of C. chauvoei spores from pastures is the most common form of exposure. This is believed to be the reason why most cases of blackleg occur in pastured animals.5,19,21,23 Exercise-associated hypoxia in pastured animals may also play a role, although this is speculative. The detailed pathogenesis of blackleg, in particular the spreading of C. chauvoei from the digestive tract to muscles, is largely unknown. 9 It is likely that various virulence factors, including toxins and a hypoxic intramuscular environment, are required for blackleg to occur. 9
The predisposing factors for the cardiac lesions of blackleg have not been definitively determined, but it has been suggested that pre-existing myocardial injuries, or stress associated with yarding and management procedures that might result in increased levels of plasma cortisol and catecholamines, may lead to this form of the disease.7,10,23 Cardiac toxicants such as ionophores or gossypol, and nutritional deficiencies of selenium and/or vitamin E, may result in myocardial injury that is favorable for spore germination. These substances were not investigated in our series of cases. However, cottonseed and ionophores are common in corralled dairy cattle, not in pastured animals, and our results suggest that blackleg is not common in yarded dairy cattle, perhaps because of more appropriate vaccination practices than in pastured animals.
Although these predisposing factors have been postulated in previous studies, their role has not been demonstrated.10,23 In cases in which both cardiac and skeletal musculature are affected, the myocardial lesions could be secondary to skeletal muscle lesions followed by septicemia. The reduced oxygen tension associated with the systemic effects of C. chauvoei could act as a predisposing factor for the germination of spores in the heart.
The same principle may apply for other groups of muscles that are unlikely to be affected by external blunt trauma, such as the diaphragm and the sublumbar muscles. The reason muscle tissue seems to be primarily affected in cases of blackleg is unknown. In a previous study, 14 pericarditis was considered to be the result of hematogenous infection arising from clostridial myositis or, in the few cases in which lesions were seen only in the pericardium, this organ was considered to be the primary focus of infection. In our study, pericarditis without involvement of the heart was only observed in 1 animal. Although it is possible that in this animal the pericardium was the primary focus of infection, this is unlikely as no inflammation of other serous membranes was observed. It is plausible that in this case there was a myocardial lesion that was overlooked by the pathologist performing the autopsy.
Traditionally, blackleg has been reported to occur in animals 6–24 mo old,11,19,23 with a few cases occurring outside this age range.1,5,20 The results of our study are consistent with previous studies; all cases occurred in animals 2–19 mo old, with most cases affecting animals 6–12 mo old.1,2 No information has been published that might help explain the age susceptibility for blackleg. It is possible that younger animals are more at risk because of lack of full development of their immune system. This is, however, purely speculative.
Blackleg typically affects animals in good nutritional condition, and often selectively causes death in the best-grown or best-fattened animals of a group.5,19,23 This was also the case in our study, with at least 17 cases occurring in cattle that were in good nutritional condition. Information about nutritional condition was not available in the remaining 12 animals, but it is possible that it was also good or very good.
Vaccination against C. chauvoei is a routine practice in most cattle operations around the world, and it is widely assumed by veterinarians and farmers alike that this procedure prevents disease and death produced by this microorganism. Most studies published show that vaccination against C. chauvoei is close to 100% efficacious in preventing blackleg after natural exposure. 22 However, the efficacy of vaccination to prevent blackleg after experimental challenge was reported to be 50–100%, depending on the dose of the inoculum.1,22 This could be the explanation for the small number of cases (n = 4) that occurred in confirmed vaccinated animals in our study. This information, however, needs to be interpreted cautiously, as information on vaccination status was not available in almost half of the animals included in our study.
Clinical signs in affected animals were often not observed in our study, with most of the animals having a history of being found dead without clinical signs being observed previously. These data are consistent with the literature.5,11,12,19,20,23 This may be because most animals dying of blackleg are on pasture, and cattle on pasture are often not observed daily. Most animals died in winter which is when most of the rainfall occurs in California; this is also consistent with previously reported cases in the literature, and there is a positive correlation between annual rainfall and incidence of blackleg. It has been postulated that dissemination of spores is aided by water saturation of the soil, which would also favor reduced oxygen concentration, a requirement for germination and multiplication of C. chauvoei spores.1,2,21,23 Rainfall seems to have been a predisposing factor for the blackleg cases in our study.
The standard method for detection of C. chauvoei is anaerobic culture and/or FAT on skeletal and/or heart samples. However, contaminant flora, particularly other fast-growing and swarming anaerobes such as C. septicum, may hamper cultural identification. FAT is a useful alternative because if smears are prepared soon after death, it provides a snapshot of the bacterial population at the time they are prepared, which avoids the problems generated with some anaerobic species growing faster than others. In our study, although C. septicum and other clostridia were isolated from one animal, these microorganisms were not detected by FAT, suggesting that only a small number of individuals from these species, or perhaps only spores, were present in the samples collected.
IHC is a simple and quick method that has shown in our study to specifically identify C. chauvoei in FFPE tissues. This technique may complement the bacteriologic procedures used to confirm the diagnosis of blackleg, especially where laboratory facilities for anaerobic culture are not readily available, or when specimens for laboratory testing are to be sent from areas far from diagnostic centers. IHC also allows retrospective studies using FFPE tissues. 3 IHC was useful to demonstrate C. chauvoei in sections and to correlate the presence of the microorganisms with lesions.
Footnotes
Acknowledgements
We thank all of the CAHFS pathologists that provided cases for this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We acknowledge the Brazilian fostering agency, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – CAPES, for Dr. CC Abreu’s PhD scholarship.
