Abstract
Hepatic trematodes, such as Fasciola hepatica and Fascioloides magna, have variable distribution throughout the United States. F. magna is endemic in the upper midwestern United States, and F. magna infections are diagnosed frequently in weaned calves and adult beef cattle at the North Dakota State University Veterinary Diagnostic Laboratory (NDSU-VDL). Rarely, liver fluke infestation has also been observed in much younger calves, including aborted fetuses. We describe here, in 2 fetal and 7 neonatal beef calves submitted to the NDSU-VDL between 2011 and 2020, parasitic migration tracts in livers, consisting of regionally extensive, random, linear tracts of fibrosis admixed with black porphyrin pigment, along with foci of necrosis and hemorrhage, and mixed inflammatory cells, which were caused presumptively by F. magna infection. Samples were not available from our 9 cases for PCR assay and sequencing, but we did confirm F. magna within liver samples collected from regional cattle in 2020 and 2021. Fetal and neonatal trematodosis was often concurrent with other common causes of fetal abortion and neonatal calf loss in our cases; however, based on the prepatent period of F. magna, fetal and neonatal beef calf trematode infestations occurred in utero.
Hepatic trematodosis, or liver fluke infection, is a worldwide disease of both animals and humans. 1 Two liver flukes commonly described in U.S. cattle are Fasciola hepatica and Fascioloides magna. The geographic distribution of F. hepatica in the United States extends along the Gulf coast, West coast, and Rocky Mountain region; F. magna is limited to the Pacific northwest, Rocky Mountains, Great Lakes, and southeastern and southcentral United States.7,9,11 To date, there are no confirmed reports in the literature of F. hepatica in native cattle of North Dakota or Minnesota.
Both trematode species have an elaborate life cycle wherein non-embryonated eggs are shed in feces of an infected definitive host before they become embryonated in water, followed by hatching of miracidia from the eggs. Miracidia subsequently seek out and penetrate their intermediate host—the snail—where they develop within tissues until released as cercariae into the environment. Free-swimming cercariae encyst on plants to form metacercariae that are ingested by a human or animal host.1,9,11 F. hepatica metacercariae migrate through the duodenal wall and into the peritoneal cavity to reach the liver. Immature F. hepatica flukes migrate through the liver parenchyma to bile ducts where they mature to adulthood. 2 In contrast, within definitive hosts such as white-tailed deer (Odocoileus virginianus) and caribou (Rangifer tarandus), immature F. magna flukes migrate randomly within the liver parenchyma, leading to severe tissue destruction along the way, before they mature to adults encased in fibrous tissue.9,11
F. magna can also infect animals considered dead-end hosts given the inability of the organism to fully mature into adulthood and the subsequent lack of eggs able to reach the small intestine for fecal shedding and continued cycling. 11 Cattle are one such dead-end host. Between 2012 and 2021, 132 cases of hepatic trematodosis in beef and dairy cattle from North Dakota and Minnesota were submitted to the North Dakota State University Veterinary Diagnostic Laboratory (NDSU-VDL; Fargo, ND, USA) as carcasses for postmortem examination or as tissues from field autopsies. In these 132 cases, liver hemorrhage, necrosis, abscesses, and/or infarcts were observed, and 45 of 132 (34%) had evidence of concurrent bacillary hemoglobinuria. The liver flukes historically have been assumed to be F. magna based on size (typically >3 cm long; F. hepatica adults can be up to 3 cm long 11 ); location of the lesions with the liver parenchyma (instead of bile ducts); association with necrosis, hemorrhage, and tissue destruction; and the known geographic distribution of the trematode species in the Great Lakes region, near the location of the NDSU-VDL. Upon review of each case report, there were <5 cases of suspected F. hepatica; however, the flukes were described to be 3 cm long, which could represent immature F. magna. Nevertheless, definitive identification of the fluke species in these cases was not confirmed by PCR.
Given that the low-ends of the prepatent periods of F. hepatica and F. magna are 2.5 and 3 mo, respectively,1,8 it is surprising that 9 of the 132 trematodosis cases were from neonatal or aborted calves that could not have ingested cercariae from the environment within that timeframe. We focus here on these fetal and neonatal calves, namely 7 neonatal calves 1-h to 1.5-mo-old and 2 late third-trimester gestation or full-term fetuses. Fetuses and neonates were Angus, Simmental, or beef crossbreds from both North Dakota and Minnesota.
Grossly, 8 of 9 calf livers were streaked with random, black, variably sized, firm and/or gritty, linear tracts (Fig. 1). The only case with no known liver lesions was from a field autopsy; black linear tracts were noted within the lung parenchyma both grossly by the referring veterinarian and histologically by the pathologist, suggesting that the liver sample submitted might not have been representative. Interestingly, extrahepatic migration was noted in 2 other cases. In a 2-wk-old calf that died with suspected neurologic signs, a large fluke was present within the calvaria (Fig. 2), along with hemorrhage extending from the olfactory lobes to the brainstem and covering the meninges. Microscopically, the leptomeninges were markedly expanded by accumulations of neutrophils, eosinophils, and fewer lymphocytes, plasma cells, and hemosiderophages. In some sections, there were extensive areas of malacia; moderate numbers of lymphocytes, plasma cells, and macrophages surrounded vessels in the adjacent neuropil. In the second case, a full-term aborted fetus, a fluke was found within the peritoneal cavity, as well as marked subcapsular hepatic hemorrhage.
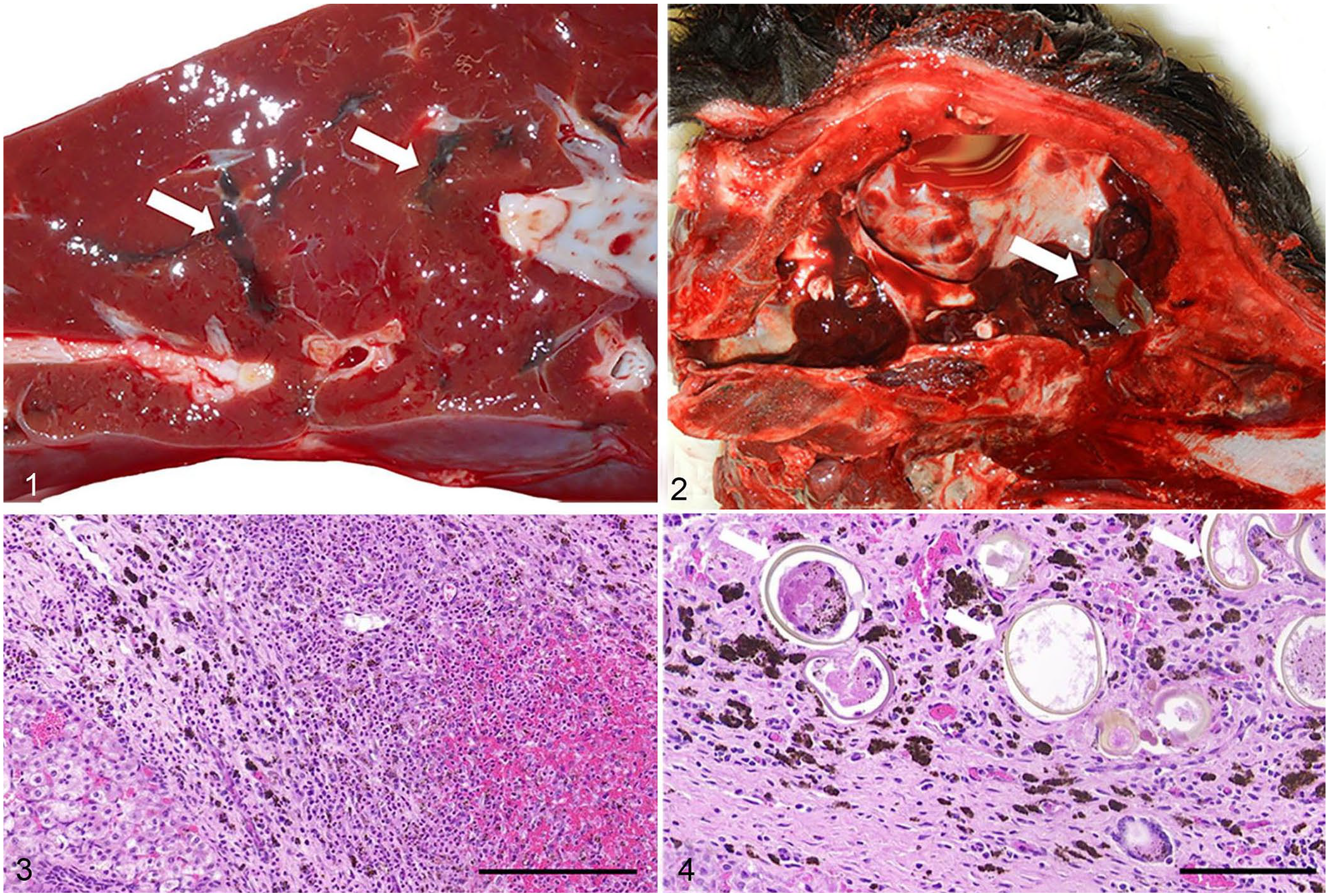
Hepatic and aberrant extrahepatic fluke migration in fetal and neonatal calves.
In all cases, parasitic migration tracts observed grossly in livers corresponded histologically to regionally extensive, random, linear tracts of fibrosis admixed with black porphyrin pigment, along with foci of necrosis and hemorrhage, and mixed inflammatory cells (Fig. 3). Within the liver, there was variable biliary hyperplasia and, in 6 of the 9 cases, golden-brown to yellow-walled eggs were present, with an occasional operculum denoting fluke ova (Fig. 4). The 2 cases with a fluke present on gross examination also had intrahepatic ova.
Six of the neonatal calves had concurrent diseases that are often observed in neonates without liver flukes. These included septicemia confirmed histologically and by positive Escherichia coli culture, fibrinosuppurative and necrotizing bronchopneumonia and omphalitis confirmed both grossly and histologically and by positive Mannheimia haemolytica cultures, monensin-associated myocardial necrosis confirmed histologically and by ionophore screen, and liver mineral abnormalities (high copper, selenium, and manganese concentrations) confirmed by inductively coupled plasma–optical emission spectroscopy. Only the 2 aborted fetuses and the calf with meningeal trematodosis were thought to have fluke migration as the primary cause of abortion and death.
There were challenges in characterizing molecularly the flukes and ova in the fetal and neonatal trematodosis cases. For the 2 neonates in which flukes were observed grossly, the flukes and tissues were no longer available for testing. Based on our experience, attempts to recover fluke DNA from formalin-fixed, paraffin-embedded tissues from other fetal and neonatal cases were not pursued because they would have resulted in a lower yield of fluke DNA than from fresh tissues. Therefore, to determine the fluke species present within North Dakota and Minnesota, fresh liver and flukes were collected in 2020 and 2021 from all cattle of any age that exhibited hepatic parasitic migration tracts on gross examination. PCR was performed on those samples as described previously. 6 Briefly, 20–50-mg sections of frozen liver from 1 fetal case and frozen liver or flukes from 10 adults with hepatic trematodosis in 2019–2021 were placed in a tube with 2 mL of 1 × PBS. Samples were homogenized (200 homogenizer; VWR) and centrifuged for 10 min at 2,780 × g. A total of 50 µL of the supernatant was transferred for extraction (MagMAX pathogen RNA/DNA kit; Applied Biosystems). Each 20-µL reaction included 4 µL (1×) of PCR buffer, 5.3 µL of nuclease-free water, 1.2 µL (1.5 mM) of MgCl2, 0.4 µL (0.2 mM) of dNTPs, 2 µL (1 µM) of each primer, 0.1 µL (0.5 U) of Taq (GoTaq Flexi polymerase; Promega), and 5 µL of DNA template. Reaction conditions consisted of an initial denaturation of 94°C for 3 min; followed by 35 cycles of 94°C for 30 s, 54°C for 30 s, 72°C for 30 s; and a final elongation of 72°C for 5 min. Product was visualized on ~3% agarose gel under UV illumination.
PCR assays targeting a portion of the ribosomal DNA (ITS2) region with universal primers for F. magna and F. hepatica had variable results. Of the 11 flukes or tissues used for the PCR analysis, ITS2 regions were amplified from 3 flukes and 2 liver sections. Unfortunately, these amplicons were from yearling or adult animals and not from the fetus. Amplified ITS2 regions from the 3 flukes were then purified and sequenced (3730xl DNA analyzer; Applied Biosystems). Synthetic IDT gBlock gene fragments were designed and used as positive controls. The consensus of accessions EF534992, EF534993, EF534995, DQ683545, EF612487, EF051080, and EF534994 was used for F. magna, and DQ683546, AJ272053, and AB207148 for F. hepatica. Two fluke samples were successfully amplified and sequenced (GenBank OL890681, OL890682). A BLAST search (https://blast.ncbi.nlm.nih.gov/Blast.cgi) resulted in 99.7% identity to F. magna and <90% identity to F. hepatica. Thus, given the historical presence of F. magna within the region, large size, and morphology of rounded anterior end on the fluke found in the calvaria of case 3 (Fig. 2), molecular confirmation of F. magna from 2 recent liver fluke cases submitted to the NDSU VDL, and no reports of F. hepatica in the upper midwestern United States, we attributed the fetal and neonatal trematode infections to F. magna.
All aborted fetal and neonatal calves were presented during late-winter to mid-spring, which coincides with the regional calving season. There is also an increased incidence of liver fluke infestation in older cattle at that time compared to the summer months (58 cases in January through April 2012–2020 vs. 27 cases in June through September 2012–2020). This temporal pattern corresponds well to the 3–7 mo prepatent period of F. magna and indicates that cattle typically become infected during the late summer and early fall months when the snails harboring the trematode larvae are likely to be more abundant because of higher precipitation. It is also important to note, however, that the period of highest liver fluke diagnoses also corresponds to a general seasonal increase of submissions of bovine abortion, scours, and pneumonia cases to the NDSU-VDL. Thus, the temporal pattern may be part of this seasonal increase.
Hepatic trematodosis can cause economic damage in addition to the fetal and neonatal calf losses presented here. Although bovine liver fluke infestation is often asymptomatic or considered incidental at time of slaughter, a subset of cattle has adverse outcomes associated with F. hepatica infection, including lower body weights, decreased fertility, and offal condemnation.3,8,13,14 Additionally, several clostridial species sporulate during liver fluke migration, resulting in significant disease and mortality. Bacillary hemoglobinuria, caused by Clostridium haemolyticum, and infectious hepatitis caused by C. novyi type B, are 2 such diseases.10,15
Although, to our knowledge, the gross and histologic lesions of F. magna infection in fetal and neonatal calves in the United States have not been reported previously, bovine neonatal hepatic trematodosis caused by F. hepatica has been reported in other countries, albeit rarely and not for many decades. A 1959 study from Germany described a 7.9% infection rate in 661 calves up to 12-wk-old. 4 A study from Australia in 1975 found a lower incidence (0.5% in >16,000 1–3-wk-old calves) 12 ; however, the authors suspected the prevalence may have been higher given accounts from practitioners of disease in the field.
Our case study does not attempt to determine infection rate of fetal, neonatal, or older cattle given the mixed nature of whole carcass and limited tissue submissions to the NDSU-VDL, but neonatal hepatic trematodosis may be an area for future study considering the economic loss caused by bacillary hemoglobinuria in our region. Furthermore, the route of transmission of liver flukes to these neonatal calves has yet to be determined. Given the prepatent period of 3–7 mo,5,8 infection in these calves must have occurred in utero. One route of transmission may be hematogenous spread from the dam to the fetus. Another possible, and more likely, scenario is direct penetration of the uterus and fetus from aberrant migration within the abdominal cavity of the dam given that aberrant migration to lung tissue has been described in cattle. 5 In our fetal and neonatal cases, liver infection was the lesion present most commonly, suggesting similar tissue tropism of F. magna in both adult and young cattle. Thus, determining homing mechanisms of F. magna to fetal liver tissues would be useful for understanding liver fluke migration in older ruminants as well as other animals and humans.
Footnotes
Acknowledgements
In 2011, Dr. Teresa Newell was the first pathologist at the NDSU-VDL to observe fetal and neonatal trematodosis. We thank the necropsy, histology, bacteriology, virology, and molecular staff of the NDSU-VDL for technical assistance, and Mary Finseth and Kenfrey Mbuba for help in examining records.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was supported in part by the USDA, National Institute of Food and Agriculture, Hatch project ND04405.
