Abstract
Two separate late-term abortion outbreaks in Jersey heifers in July 2020 and December 2020 were investigated by the Iowa State University Veterinary Diagnostic Laboratory. We evaluated 3 whole fetuses and 11 sets of fresh and formalin-fixed fetal tissues during the course of the outbreaks. The late-term abortions were first identified at a heifer development site and subsequently observed at the dairy farm. Aborted fetuses had moderate-to-marked postmortem autolysis with no gross lesions identified. Observed clinical signs in cows at the dairy farm ranged from intermittent loose stools to acute post-abortion pyrexia and reduced feed intake. Routine histopathology and reproductive bacterial culture revealed acute, suppurative placentitis with moderate-to-heavy growth of Salmonella spp. group B from stomach contents, liver, placenta, and heifer fecal contents. Serotyping identified Salmonella enterica subsp. enterica serovar Brandenburg in all 14 fresh tissue cases, as well as individual and pooled heifer feces. Whole-genome sequencing analysis revealed that all isolates belonged to ST type 873 and possessed typhoid toxin genes, several fimbrial gene clusters, type III secretion system genes, and several pathogenicity islands. Abortions caused by Salmonella Brandenburg have not been reported previously in dairy cattle in the United States, to our knowledge.
Salmonella spp. cause a wide range of clinical insults in most avian, reptilian, and mammalian species, including humans, resulting in systemic, enteric, and reproductive disease. 22 Salmonella spp. are highly diverse, and include ~2,600 serovars (serotypes). 10 Salmonella enterica is comprised of >1,500 serovars, with 99% capable of causing infection in animals and humans. 6
The most common Salmonella serogroups isolated from cattle with enteric disease include B, C1, C2, and D, with serovars Typhimurium, Montevideo, Newport, and Dublin represented commonly.2,17 Salmonella enterica subsp. enterica serovar Dublin is considered host-adapted to cattle, primarily dairy cattle, and causes severe enteric disease and death. 11 In a 2006–2015 study, Salmonella Dublin was the most common serotype isolated in dairy cattle, with and without detected disease (23%), of almost 5,000 isolates evaluated. 24 Disease manifestation and clinical signs depend on the distribution and dissemination of the pathogen, with enterocolitis, septicemia, and abortions observed most commonly in dairy cattle. Microscopic lesions typical of systemic disease are detected most frequently in lung, liver, spleen, and lymph nodes. 15
Although exposure to Salmonella spp. in cattle operations is inevitable, control of salmonellosis within herds is attempted through management practices by optimizing nutritional and hygienic practices, thereby limiting the infectious dose. 11 Autogenous and commercial vaccines, along with optimized management practices, are all part of the control and prevention measures available to limit disease within the herd.11,12,20,21
We diagnosed and investigated 2 primary outbreaks of Salmonella Brandenburg in a Jersey heifer development feedyard and local dairy farm A, in late July and early August of 2020 and again in December and January of 2020–2021. A third outbreak occurred on dairy farm B in February 2021, in a similar geographic area but with no relationship to feedyard and dairy farm A. The case definition for recurrent outbreaks of Salmonella Brandenburg–associated fetal abortions within these 2 dairy systems consisted of late-term abortions at 220–260 d of gestation, with culture results and serotyping of Salmonella Brandenburg from fetal tissues and fecal contents, coupled with typical microscopic changes within fetal tissues.
The initial outbreak in the feedyard was confined to a single pen, beginning in late July 2020 and extending for 3 wks, with ~40 heifers aborting in the last trimester. Heifers from this pen were moved to a different pen for isolation; abortions continued until the heifers were returned to the dairy farm because they were due to calve. During the initial outbreak, no significant clinical signs were detected in aborted or pregnant heifers within the feedyard.
The second outbreak occurred in mid-December 2020 in heifers from dairy farm A, with 22 of ~150 heifers aborting in a feedyard pen adjacent to the pen with the initial outbreak in July. Again, no clinical signs were observed within the feedyard heifers prior to or after abortion. Upon being returned to dairy farm A, ~50 additional abortions were observed, for a total of 70 abortions within the group. Cows that aborted at the dairy farm were observed to have loose stools and post-abortion pyrexia with reduced feed intake. A third outbreak occurred in a separate dairy farm (B), with a matching case definition and unknown number of abortions in February 2021.
Samples submitted to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, IA, USA) included fresh and formalin-fixed tissues (placenta, brain, heart, liver, lung, spleen, adrenal gland, thymus) and stomach contents from 11 cases received July 2020 to February 2021. Two whole fetuses in late gestation (~220 d) were evaluated during the initial outbreak in July 2020, and another late-term abortus (~260 d) was examined in January 2021 during a site visit, for a total of 14 aborted fetal tissue submissions. Additional samples evaluated during the outbreak included serum, feed, feces, environmental water, and vaginal swabs. All fetuses and tissues examined had advanced postmortem autolysis with abundant hemoglobin imbibition and no other significant gross tissue alterations.
The most frequent microscopic changes observed were suppurative placentitis with embolized bacteria (Fig. 1A, 1B), meconium aspiration, occasional neutrophils in alveoli, and neutrophilic vasculitis within the placenta and umbilicus (Fig. 1C; Table 1). In the 10 cases with placentitis, neutrophilic exudate adhered to the chorioallantoic villi; the epithelial barrier was attenuated or denuded with infiltration of neutrophils into the subjacent interstitium. Gram staining revealed the abundance of uniformly short, gram-negative bacilli within vascular structures, further supportive of bacterial-associated abortion (Fig. 1D). On occasion, neutrophils extended into the amniotic membrane, surrounded vascular structures within the umbilicus, and expanded into the surrounding mesenchyme (Fig. 1C). Two cases had hepatitis, characterized by random foci with a loss of tinctorial properties, hepatocyte swelling, or individualization with mineralization; in 3 animals, bile canaliculi were engorged. Systemic bacterial colonization was observed histologically as colonies filling vessels within distant organs in 3 of 7 cases (Table 1). Because of the advanced postmortem autolysis in all cases, the presence of bacteria in blood vessels could be the result of postmortem invasion given that lesions such as vasculitis were not distinctly visible.
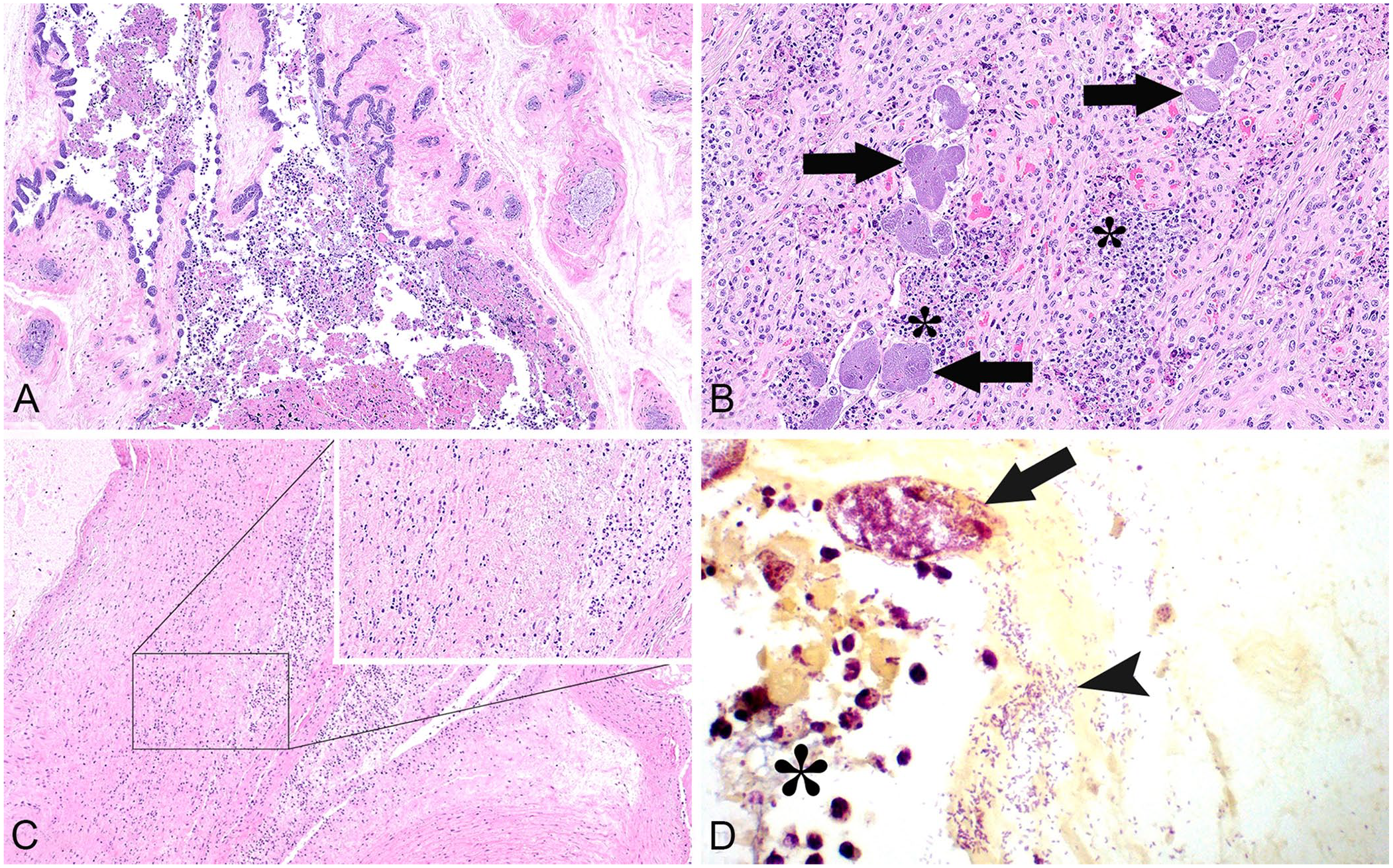
Salmonella enterica subsp. enterica serovar Brandenburg abortions in dairy cattle.
Microscopic lesions observed in fetal tissues with Salmonella Brandenburg–associated abortion.

− = negative; + = positive; Br = brain; Hrt = heart; Li = liver; NA = not applicable; NR = not received.
Whole fetus examined.
In all 14 cases, there was moderate-to-heavy growth of Salmonella spp. from direct culture on Hektoen enteric agar as identified by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS). Stomach contents and liver samples yielded pure growth most frequently, with mixed growth observed frequently in placental and umbilical tissues (Table 2). Other bacteria isolated and identified by MALDI-TOF MS included Bacillus cereus, B. licheniformis, Klebsiella variicola, and Enterococcus hirae, among many other contaminants that were observed; however, these organisms were not pursued further because they were not isolated consistently, showed comparatively low growth, and were not considered to be common pathogenic organisms in the bovine reproductive tract. Serogrouping at the ISU-VDL indicated that all Salmonella spp. isolates belonged to serogroup B. The organism was isolated most commonly from placenta (9 of 9 cases), followed by liver (7 of 7 cases), stomach contents (6 of 6 cases), and lung (2 of 3 cases; Table 2). Classic auto-agglutination and genomic-based serotyping performed at the National Veterinary Services Laboratories confirmed Salmonella Brandenburg in all fetal Salmonella sp. group B isolates submitted.
Salmonella Brandenburg–associated abortion culture results.
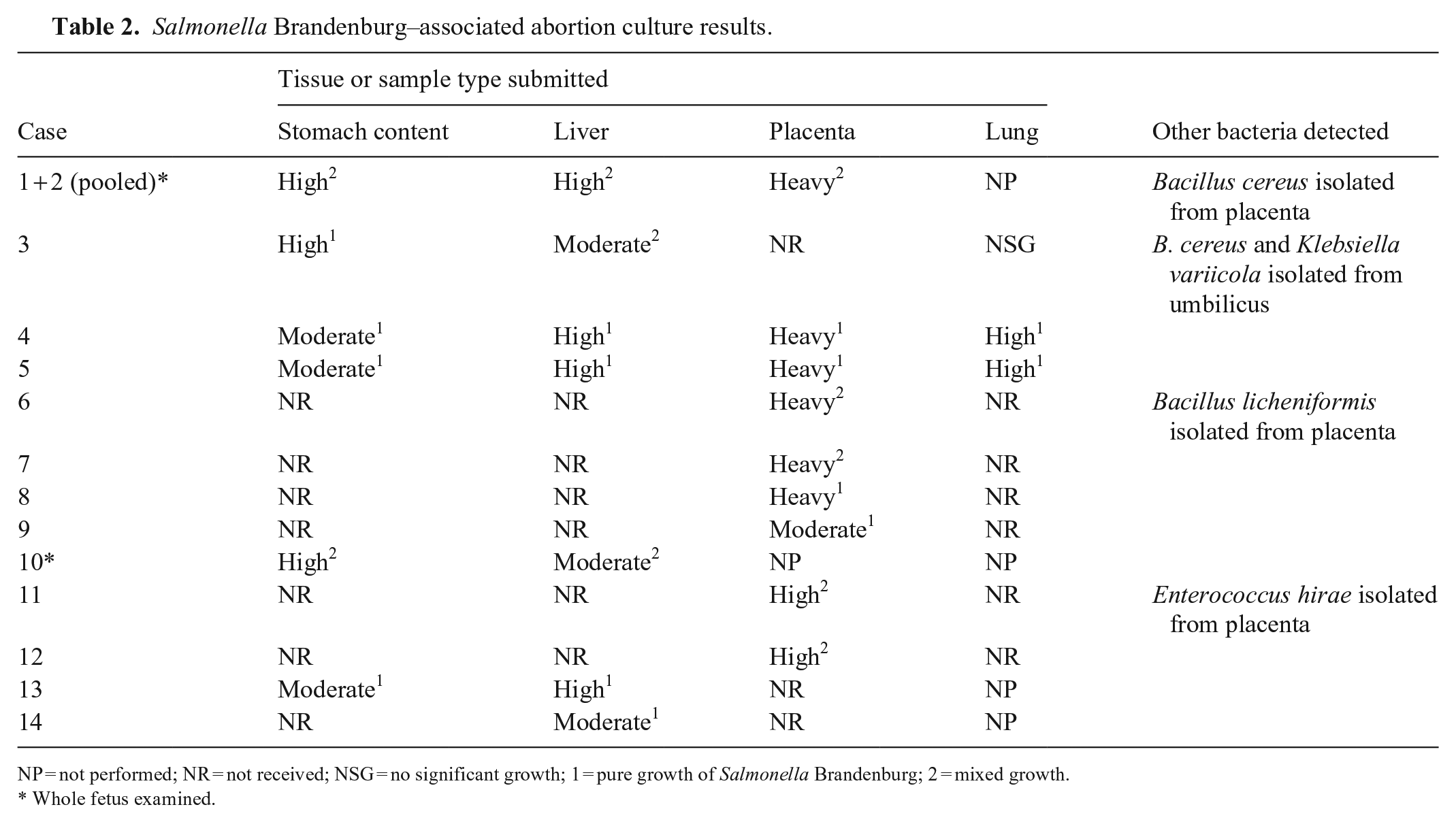
NP = not performed; NR = not received; NSG = no significant growth; 1 = pure growth of Salmonella Brandenburg; 2 = mixed growth.
Whole fetus examined.
Fetal tissues from abortions tested negative for bovine herpesvirus 1 (BoHV1; Bovine alphaherpesvirus 1), bovine viral diarrhea virus (BVDV; Pestivirus), ovine herpesvirus 2 (Ovine gammaherpesvirus 2), Toxoplasma gondii, Coxiella burnetii, and Leptospira spp. by in-house, real-time PCR (all PCR tests validated per ISU-VDL quality program). Stomach contents from all submissions were negative for Campylobacter spp. by culture. Serum from affected heifers had negative titers for BoHV1, BVDV, Neospora caninum, and Leptospira spp. by ELISA and virus neutralization. No significant concentrations of mycotoxins were detected in feed or silage by liquid chromatography–tandem MS.
Additionally, bacterial isolates from fetal liver, placenta, feces, and stomach contents were obtained from the development feedyard, dairy A, and dairy B, and subjected to whole-genome sequencing analysis via next-generation sequencing (NGS; MiSeq, Illumina). All isolates were grouped in the multi-locus sequencing type ST-873, and typhoid toxin genes pltA, pltB, and cdtB. Five Salmonella pathogenicity islands (SPI-1–5), which are often found in many other Salmonella strains, were also detected in each abortion-associated isolate. In addition, all abortion-associated isolates encoded fimbrial operons staABCDEFG and pegABCD (Suppl. Table 1). Both fimbrial operons have been identified much less frequently in other Salmonella spp. strains (GenBank CP024164, CP022494, CP040701, CP034703, CP026327, CP026379, CP061118, CP003278, CP038507, CP032449, CP028151, CP022963, CP006631, CP031359, CP040686, CP007523, CP051368, CP045761, CP029991, CP022139, CP034074, CP022015, CP007222).
Isolates from fetal liver and placenta from the development feedyard were subjected to antimicrobial susceptibility testing by a broth microdilution testing panel (BOPO7F, Sensititre; Thermo Fisher). Both isolates were found to be susceptible to only fluoroquinolone antibiotics (e.g., danofloxacin, enrofloxacin). Salmonella Brandenburg NGS analysis detected antimicrobial resistance genes for aminoglycosides, beta-lactamases, sulfonamides, tetracyclines, trimethoprim, and florfenicol (Suppl. Table 2), which correlated with the in vitro broth microdilution susceptibility testing.
In addition to submitted tissues, fecal samples were obtained from 18 heifers from the affected feedyard pen in late December 2020 to screen for fecal shedding of Salmonella spp. Samples were pooled by 3, 2, 2, 5, and 5 (samples [1–3], [4, 5], [6, 7], [8–12], [13, 14, 16–18]) for a total of 5 pools, with 1 sample (15) submitted individually from an animal from a different dairy farm. Pooled and individual testing were based on animal ear tag numbers per request of the submitting veterinarian. All feces were cultured with an individual sterile swab. Two of 5 pools ([8–12], [13, 14, 16–18]), and the individual swab (15) were positive for Salmonella spp. via routine culture without enrichment in low-to-moderate growth; all were serogroup B. Corresponding pooled samples were also submitted for Salmonella spp. PCR, with 3 pooled samples ([6, 7], [8–12], [13, 14, 16–18]) testing positive for Salmonella spp. Sample 15 was negative for Salmonella spp. by PCR. Salmonella spp. serotyping was not performed on these isolates given the recent detection of Salmonella Brandenburg within the pen; however, fecal shedding was likely, based on observed clinical signs with corresponding abortions and increased non-pregnant cows received at the dairy farm.
In early January 2021, Salmonella spp. were isolated from 7 of 9 fecal samples from dairy farm A submitted for culture without enrichment (Table 3). Two of the isolates were serotyped and confirmed to be Salmonella Brandenburg, confirming fecal shedding. Extensive fecal bacterial cultures were performed in the pre-breeding pens at the feedyard as well, with 1 of 100 fecal samples positive by routine culture with tetrathionate enrichment and confirmed as Salmonella Brandenburg by serotyping. One pool of avian environmental samples from the feedyard was also cultured, with enrichment, and found to have moderate growth of Salmonella Brandenburg. Fecal samples from 2 cows with enteric signs but that had not aborted from dairy farm B were also positive with enrichment in late January. As expected, culture results often had mixed growth with other colonies or organisms considered fecal contaminants.
Salmonella spp. detection in feces by culture.
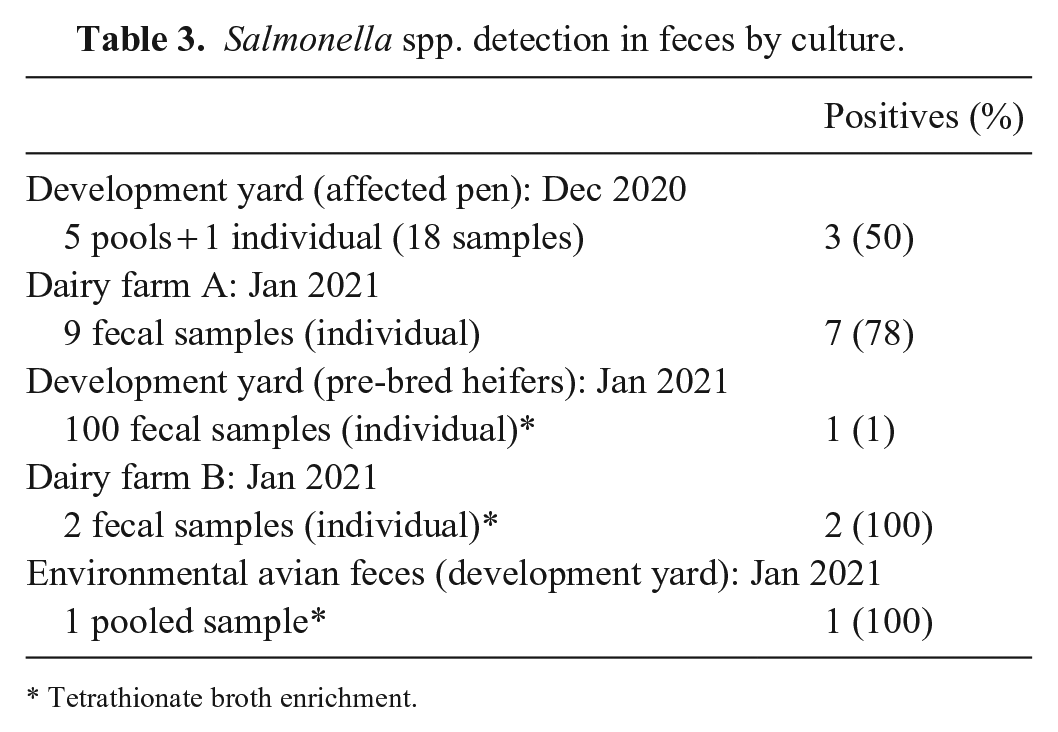
Tetrathionate broth enrichment.
During routine milk surveillance testing, Salmonella Brandenburg was isolated from a solitary milk sample in late August 2020 from dairy farm A, suggesting environmental presence. Overall, 14 cases received by the ISU-VDL had features (histopathology and culture) diagnostic for Salmonella Brandenburg abortion, with suppurative placentitis observed in 10 of 11 cases in which placental tissue was received. Salmonella Brandenburg was isolated most commonly from placental (9 cases) and liver (7 cases) tissues from 14 cases. In all cases in which Salmonella Brandenburg was isolated, stomach content, placenta, and liver were considered the preferred postmortem samples for culture of this organism.
Salmonella Brandenburg abortions were first described in a sheep flock in the South Island of New Zealand in 1996, spreading to other farms within the region until 2003. Most of the affected flocks had high stocking rates and were considered capable of spreading the disease for at least 12 mo. 1 In 1999, cattle within the same area were diagnosed with abortions and enteric disease in calves caused by Salmonella Brandenburg. 1 Most cattle identified were heifers aborting in the last trimester. 3 Additionally, a sudden increase in human Salmonella Brandenburg cases was observed; this organism has also been linked to illness in people having contact with live poultry.1,16
In cattle, Salmonella spp. and particularly host-adapted strains, such as Salmonella Dublin, are often associated with enterocolitis, hematogenous dissemination, and abortion. Transmission is generally fecal-oral via consumption of contaminated feed or water sources; in utero, and to a lesser degree, rodent and avian transmission are also considered routes.7,9,19,23 Infection requires sufficient numbers of Salmonella spp. to colonize and invade enterocytes. The infectious dose is often strain dependent; however, one study showed that 106 cfu of Salmonella Dublin per os was capable of causing clinical disease and shedding of bacteria in healthy calves. 25 In clinical cases, an infectious dose is dependent on many factors such as passive transfer, acquired immunity, host age, and physiologic state. 14 M cells may aid in transportation and colonization within local Peyer patches in the intestine; dissemination of the organism relies on survival and replication within histiocytes that spread to distant organs.5,13,18 To cause abortion, the organism colonizes the fetus or uterus via the circulatory system or ascends through the reproductive tract, resulting in placentitis, metritis, and fetal loss. Salmonella spp.–associated abortions are commonly associated with stress in the host, such as changes in diet or spoiled feed. 18
Overall, the pathogenesis of Salmonella Brandenburg in our outbreaks was not fully elucidated; however, a pathogenesis similar to other Salmonella spp. is highly likely. We suggest that fecal-oral transmission with bacterial dissemination within the heifer development pens and dairy farm to be the most likely transmission route, similar to that described previously with other Salmonella spp. in cattle.4,8 The heifer development feedyard was densely populated with mixed dairy and beef cattle, which, coupled with substandard hygienic and biosecurity practices, are thought to have contributed significantly to ongoing disease. Future studies should focus on the pathogenesis of Salmonella Brandenburg in pregnant cattle and careful observation of clinical signs post-infection. Additionally, the development of serologic and molecular assays would be important for understanding herd status and rapid detection and classification of the organism.
The increases in abortions and non-pregnant cows being received at the dairy farm waned following the initial investigation, likely as a result of the implementation of hygienic practices on the dairy farm, optimization of heifer nutrition, and increased administration of Salmonella Newport subunit, Salmonella Dublin modified-live, and autogenous Salmonella Brandenburg vaccines. To our knowledge, Salmonella Brandenburg abortions in dairy cattle have not been reported previously within the United States and should be considered a differential diagnosis in cases associated with late-term abortions, particularly in dairy cattle.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221105890 – Supplemental material for Salmonella enterica serovar Brandenburg abortions in dairy cattle
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221105890 for Salmonella enterica serovar Brandenburg abortions in dairy cattle by Christopher L. Siepker, Kent J. Schwartz, Tyler J. Feldhacker, Drew R. Magstadt, Orhan Sahin, Marcelo Almeida, Ganwu Li, Kristin P. Hayman and Patrick J. Gorden in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
