Abstract
Five Bulldog pups, 4 weeks of age or younger, were presented over a 2-day period for postmortem examination and diagnostic evaluation. The pups originated from 2 different litters but had been cared for at a common facility since their birth. All 5 pups died after exhibiting symptoms consisting of lethargy, dyspnea, nasal discharge, anorexia, vomiting, diarrhea, and abdominal pain. Necropsy examination revealed locally extensive to diffusely red, firm, consolidated lungs in all pups. Histopathologically, the lungs were variably effaced by multifocal areas of necrosis. The alveolar lumens contained fibrin, edema fluid, macrophages, and neutrophils. Many of the bronchioles contained cellular debris and neutrophils admixed with sloughed bronchiolar epithelium, which often contained large intranuclear amphophilic inclusion bodies that peripherally displaced chromatin. Fluorescent antibody testing was positive for Canine adenovirus. An adenovirus isolated via cell culture was positive on direct fluorescent antibody test and was identified as Canine adenovirus serotype 2 via polymerase chain reaction. Electron microscopy revealed typical viral inclusions within bronchiolar epithelial cells. Hemolytic Escherichia coli was also isolated from the lungs in 3 of the 5 pups. The current case demonstrates a natural and rare fatal infection with a viral agent that is typically associated with immunosuppression in both animals and humans.
Two distinct adenoviruses have been identified in dogs. Canine adenovirus serotype 1, the more well known of the 2 canine adenoviruses, is the cause of infectious canine hepatitis, which is typically seen in young or unvaccinated dogs and may result in a rapid course of clinical disease leading to death. Canine adenovirus serotype 2 (CAdV-2) is most commonly associated with upper respiratory disease in dogs and is often considered a component of infectious tracheobronchitis, or kennel cough. Pneumonia rarely results from infection with CAdV-2, but when it does occur, the animal is typically immunosuppressed. 3
Five Bulldog pups were presented over a period of 2 days to the Kansas State Veterinary Diagnostic Laboratory (Manhattan, Kansas) for necropsy examination and diagnostic evaluation. All pups were raised at the same facility with a common history but were from 2 separate litters. Litter A consisted of 8 pups that were 13 days of age at the time of submission. Litter B was a litter of 3 pups that were 4 weeks old at the time of death. Four of the 5 examined pups were from litter A, and 1 pup was from litter B. The dam of litter B originated from a different location than that of litter A, but after delivery by cesarean section at a neutral site, all pups were returned to the original residence of the dam of litter A. All pups were normal at the time of birth and received serum transfusions from the appropriate dam. They were then separated from the dams and hand raised, during which time litters A and B were cohabitated. Neither dam exhibited any signs of illness during gestation or after delivery of the pups, and both dams were reportedly current on all vaccinations. Reported clinical signs were similar for all pups and consisted of lethargy, dyspnea, nasal discharge, anorexia, vomiting, diarrhea, and abdominal pain. They were treated with supportive care, but all 11 pups from the 2 litters eventually expired. Six pups were not submitted for diagnostic evaluation.
Gross postmortem examination revealed similar findings among all pups: nasal and/or ocular discharge, a small amount of serous pleural effusion, and cranioventral to diffuse, dark red, consolidated lungs that failed to collapse after removal from the thoracic cavity, with bilateral rib impressions on the pleural surfaces (Fig. 1). The total volume of affected lung ranged from 60% to 90% between the pups. Two of the pups also had moderately enlarged submandibular lymph nodes. All other organs including the brains were grossly normal.
Samples of the affected lung from all of the pups along with all other organs were removed and placed in 10% buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Additional samples of lung from 1 pup were fixed in Karnovsky cacodylate fixative (a 50/50 solution of 2.0% paraformaldehyde and 2.5% glutaraldehyde in 0.1 M cacodylate buffer), processed routinely for transmission electron microscopy, and embedded in resin. a Ultrathin 90-nm-thick sections were stained with uranyl acetate and lead citrate and viewed with an electron microscope. b
Histologically, the affected lungs in all 5 examined pups contained multifocal to occasionally coalescing areas of necrosis characterized by obliteration of normal pulmonary architecture (Fig. 2) and replacement by celular and eosinophilic and basophilic karyorrhectic debris admixed with alveolar macrophages and sloughed epithelial cells. Within these areas of necrosis, large numbers of bronchiolar epithelial cells and rare alveolar macrophages contained intranuclear inclusion bodies, which peripherally displaced nuclear chromatin or completely obscured the nucleus (Fig. 3). The inclusion bodies were 5–7 μm in diameter and amphophilic with a homogenous, glassy appearance. Diffusely throughout the remainder of the lung, alveoli contained large numbers of alveolar macrophages and moderate amounts of edema fluid along with scattered infiltrates of nondegenerate neutrophils and fibrin. Multifocally, small numbers of Gram-negative coccobacilli were evident with Gram stain, consistent with the Escherichia coli cultured, and were also present within alveoli.
Liver and splenic lesions were also present in 1 pup. Within the liver, there was individual hepatocyte necrosis and vacuolar degeneration; hepatocytes often contained intranuclear inclusion bodies similar to those described within the bronchiolar epithelium of the lung. There was moderate lymphocyte depletion within the periarteriolar lymphoid sheaths of the spleen and occasional macrophages contained similar intranuclear inclusion bodies.
There were no histologic lesions present in the gastrointestinal tracts of any of the examined pups to explain the cause of the observed clinical signs of abdominal pain, anorexia, vomiting, and diarrhea. Generalized systemic illness resulting from the infection with CAdV-2 is the most likely explanation.
Fluorescent antibody test (FAT) was performed for Canine distemper virus, CAdV, Canid herpesvirus 1, and Alphacoronavirus 1 on tissues from the 4 pups submitted together on the second day. Fresh tissues were cut at 4–5-μm sections using a cryostat and, after drying, were fixed in cold acetone for 15 min. After fixation and drying, conjugate c was added and was incubated for 30 min in the dark at 37°C. The sections were then placed in phosphate buffered saline (PBS) wash for 10 min and mounted on a slide with a 50/50 solution of glycerin and PBS. Lung from all 4 pups and liver from 1 pup were positive for CAdV and displayed diffuse nuclear staining of the respiratory epithelium. All other FATs were negative. Samples from the single pup submitted the previous day were also positive for CAdV and negative for Canid herpesvirus 1 via FAT. Bacterial cultures yielded growth of hemolytic E. coli from 3 of the 5 pups and Klebsiella pneumoniae from 1 pup.
Virus isolation was performed on pooled tissue samples from each of the 5 pups. To make 10% homogenates, 2 g of spleen and lung taken from each animal was minced with Eagle minimal essential medium (EMEM). d The homogenate was further processed into slurries and clarified by centrifugation. One milliliter of each supernatant was then inoculated onto 25-cm 2 cell culture flasks seeded with Madin–Darby canine kidney cells maintained in EMEM with 7% fetal calf serum and antibiotics. The cultures were incubated at 37°C with 5% CO2 until viral cytopathic effect with frequent intranuclear inclusion bodies was observed in 75–80% of the cells in the monolayer. Cultures were harvested, and cell debris was removed by centrifugation. The supernatant was assayed for the presence of CAdV using a direct fluorescent antibody test (DFAT) with a polyclonal conjugate. e Once the presence of CAdV was verified, DNA was extracted from the supernatant using a commercial kit. f Polymerase chain reaction (PCR) was performed using HotStar Taq master mix g with primers designed to the E3 and flanking regions of CAdV-1 and CAdV-2 6 : HA1 (5′GCGCTGAACATTACTACCTTG TC-3′) and HA2 (5′-CCTAGAGCACTTCGTGTCCGC TT-3′). These primers amplify a 508-bp region for CAdV-1 and a 1,030-bp region for CAdV-2. The PCR products were run on agarose gels and stained with ethidium bromide.
Adenovirus was grown from all 4 pups via cell culture (Fig. 4), which was confirmed with positive DFAT (Fig. 5), and further characterized as CAdV-2 by PCR (Fig. 6). Electron microscopy revealed the presence of a crystalline array of adenoviral particles in the nucleated cells of the lung (Fig. 7). These viral particles were nonenveloped, displayed icosahedral symmetry, and averaged between 80 and 110 nm in diameter. Because of the degree of postmortem autolysis present in the tissue, the exact cell type could not be determined.
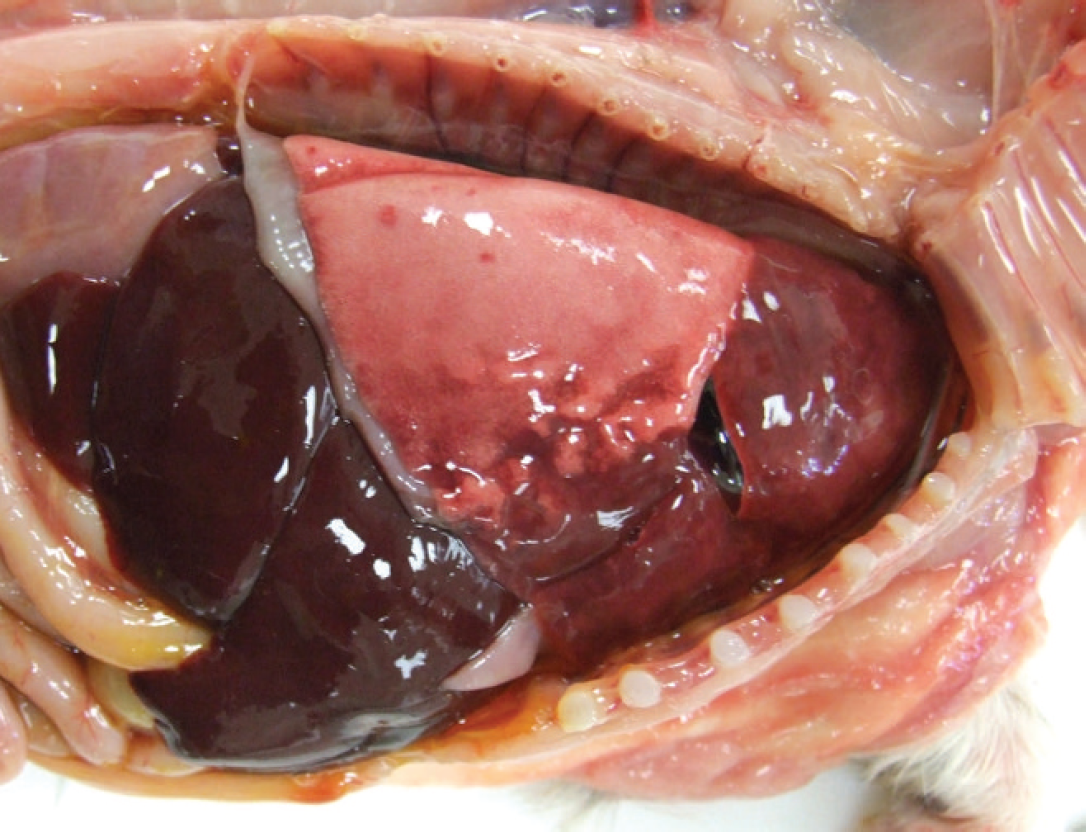
Bulldog puppy; gross necropsy image of Bulldog pup with cranioventral pulmonary consolidation and rib impressions.
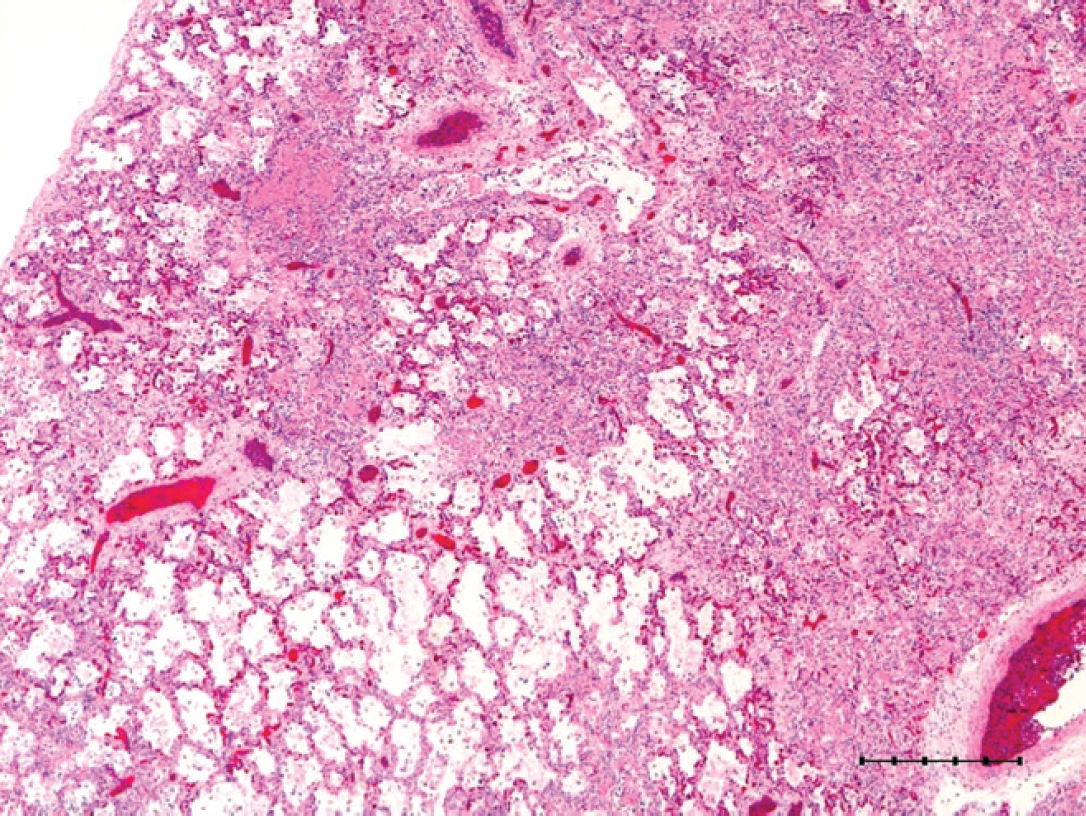
Lung; multifocal to coalescing necrosis with loss of normal architecture with large amounts of cellular and karyorrhectic debris. Hematoxylin and eosin. Bar = 500 μm.
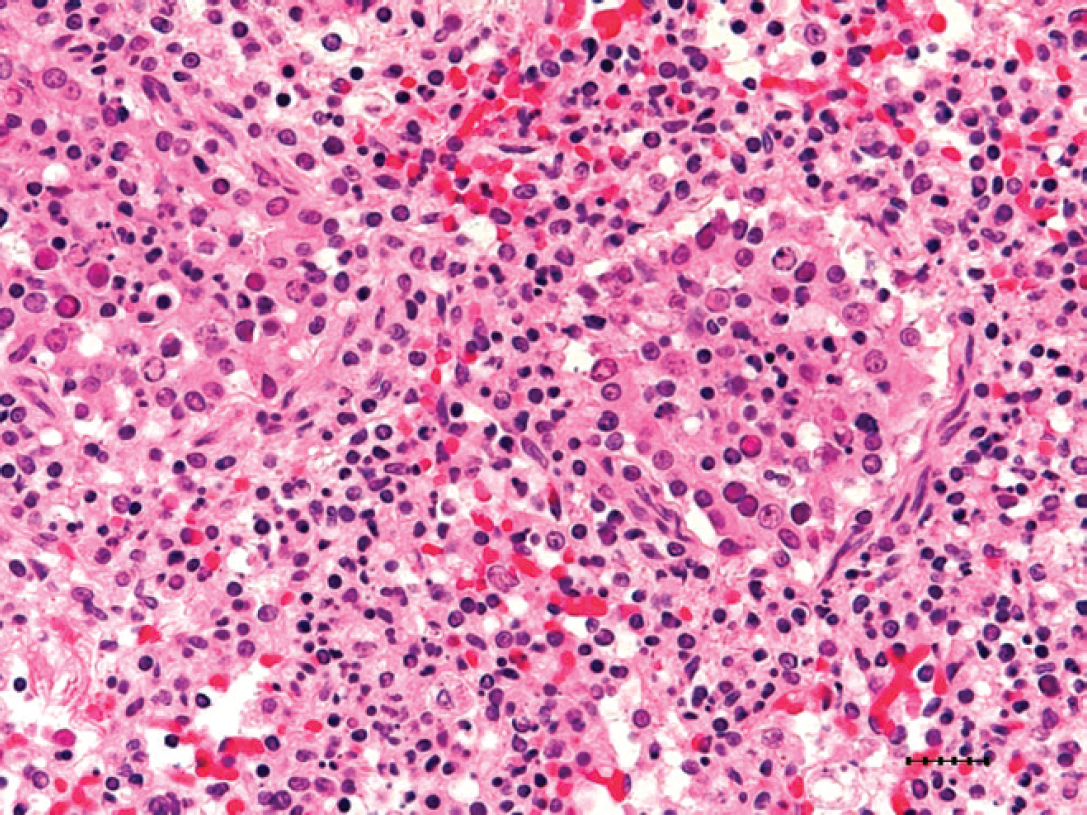
Lung; necrosis of bronchiolar epithelium with epithelial and histiocytic intranuclear amphophilic inclusion bodies, which peripherally displace nuclear chromatin and measure approximately 5–7 mm in diameter. Hematoxylin and eosin. Bar = 25 μm.
Canine adenovirus serotype 2 is a common viral infection in dogs that is typically transient and causes asymptomatic or mild upper respiratory disease. It is easily aerosolized from one canine to another, and after inhalation, the virus replicates in pneumocytes. Characteristic pulmonary lesions consist of epithelial cell necrosis, type II pneumocyte hyperplasia, a mild inflammatory infiltrate, and distinctive large intranuclear inclusion bodies within epithelial cells or alveolar macrophages. 5 Recently reported cases of infection by CAdV-2 in conjunction with other canine pathogens have resulted in fatal pneumonia 4,7 and neurologic signs. 1 Experimental infections with CAdV-2 have been documented to cause severe necrotizing and proliferative bronchitis and bronchiolitis. 2 In general, CAdV-2 is not highly regarded as a virulent primary pathogen. In many domestic animal species and in human beings, adenoviruses are typically classified as a disease of the immunosuppressed population.
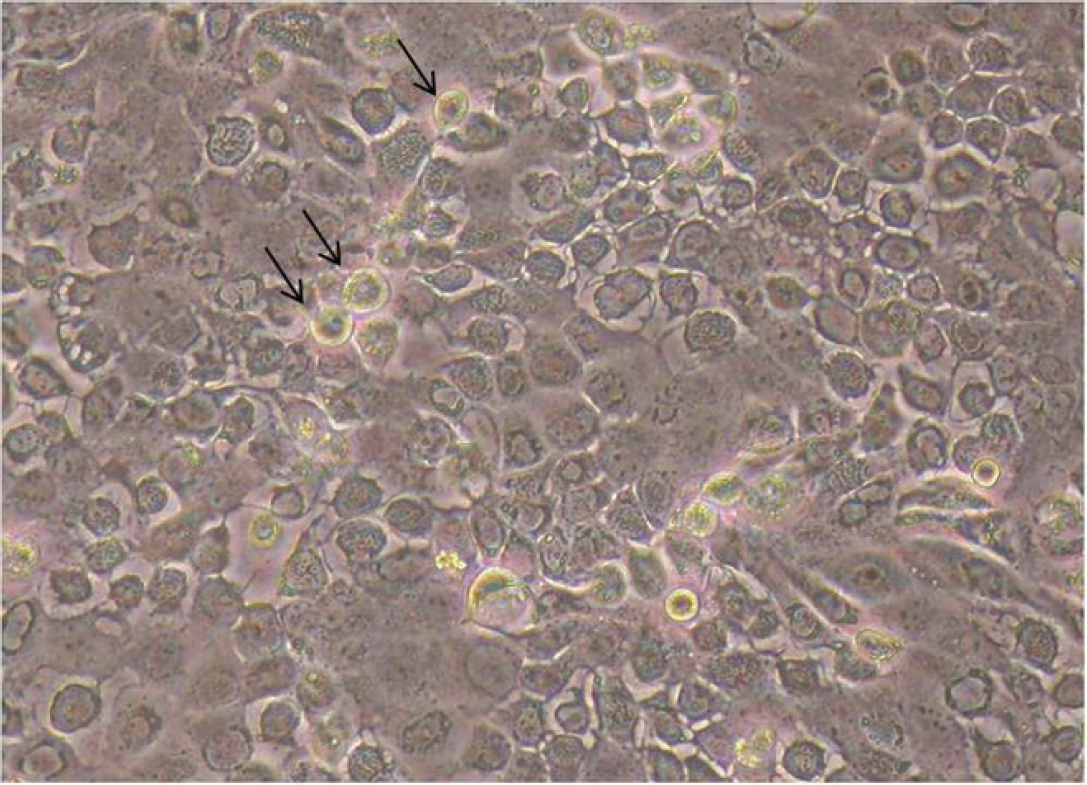
Virus isolation; cell culture monolayer with cytopathic effects and intranuclear inclusion bodies (arrows) present in 75–80% of cells. 200× magnification.
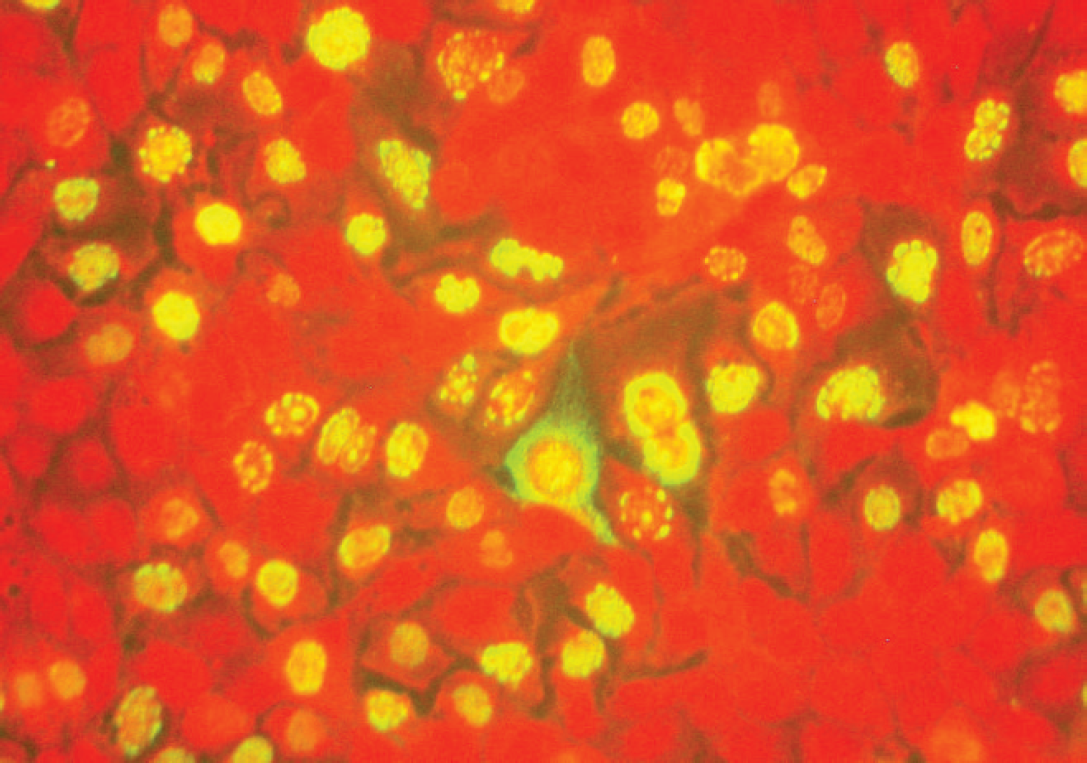
Direct fluorescent antibody staining of Madin–Darby canine kidney cells approximately 24 hr postinoculation with passage 1 of Canine adenovirus–positive cells extracted from primary cell culture. Notice the partial to diffuse, intense, nuclear fluorescence that is also associated with intranuclear inclusions (center). Antiadenovirus fluorescent antibody staining with Evan's blue counterstain. 200× magnification.
The current case represents a unique manifestation of this infection. The pups in the present case were not known to be immunosuppressed, and the dams were reported to be adequately immunized, including vaccination against CAdV-2. Serological titers of the dams were not obtained to confirm adequate vaccination status; thus, failure of the vaccine is possible. Likewise, function tests of the immune system of the pups were not done. However, given the age of the pups, it is feasible that lack of a mature immune system, specifically a poor cell-mediated immunological response, could have played a role in the infection of these pups. Viral disease control is performed via T-cell interaction with major histocompatibility complex (MHC) class I presentation of viral particles. The MHC class I type of these pups is not know; however, given that the all pups presented were closely related, in-breeding may have predisposed the pups to a similar and potentially abnormal immunological response to a normally low pathogenicity virus. Likewise, the anatomical brachycephalic conformation of the pups may have allowed for a higher dose of the virus to breach the nasal clearance mechanisms present within the normal canine nose, thus allowing deeper penetration of the viral particles and subsequent infection of the lower respiratory tract. The source of the virus is not known, but the introduction of litter B, the pups from the secondary location that were relocated to the origin of litter A after birth, is suspected.
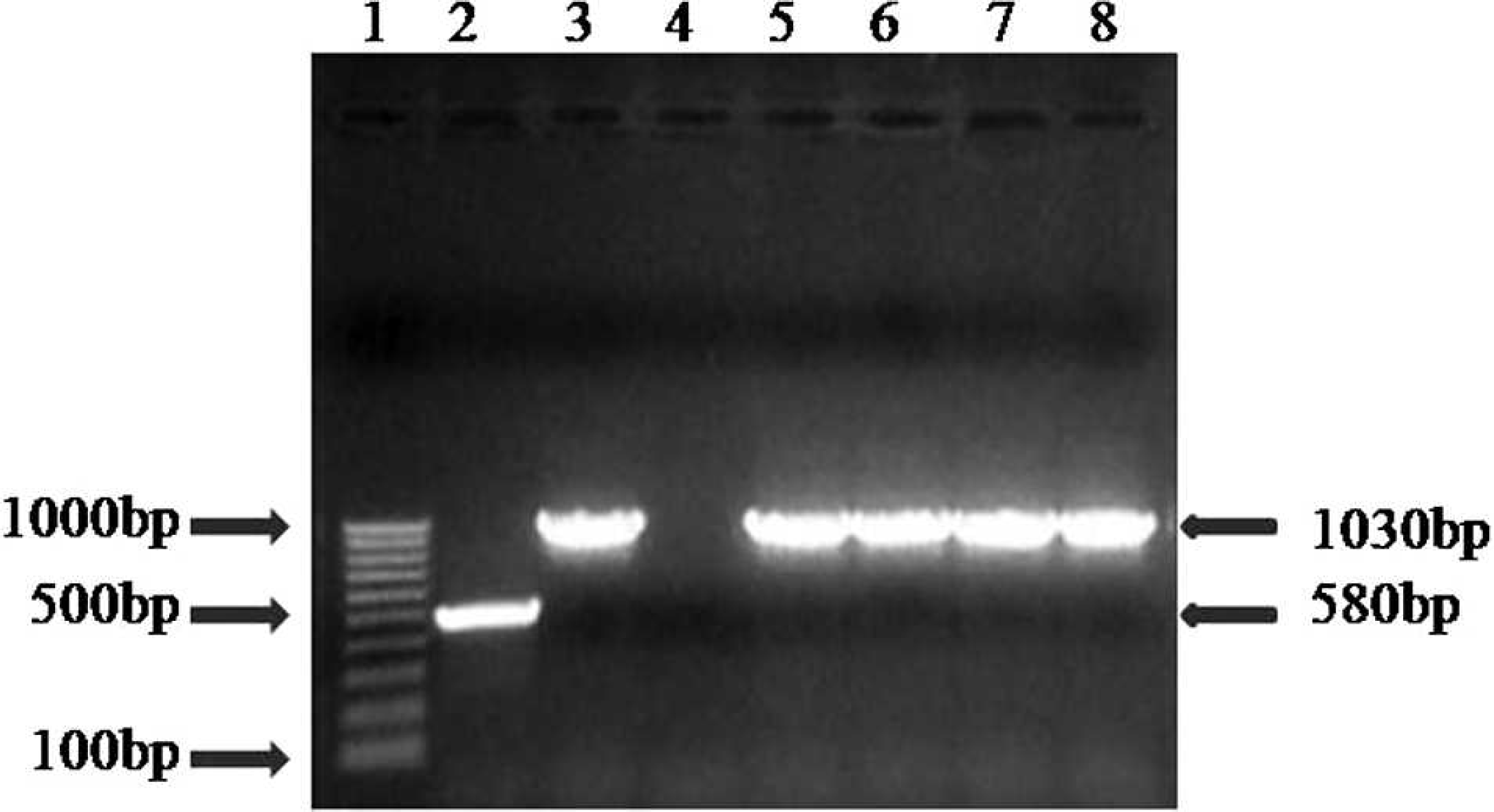
Polymerase chain reaction gel. Lane 1: marker; lane 2: ICH (Canine adenovirus serotype 1 [CAdV-1]) positive control, 508 bp; lane 3: ICH (CAdV-2) positive control, 1,030 bp; lane 4: negative control; lane 5: lung, puppy no. 1 (cell culture isolate), 1,030 bp (CAdV-2); lane 6: spleen and lung, puppy no. 2, 1,030 bp (CAdV-2); lane 7: spleen and lung, puppy no. 3, 1,030 bp (CAdV-2); lane 8: spleen and lung, puppy no. 4, 1,030 bp (CAdV-2).
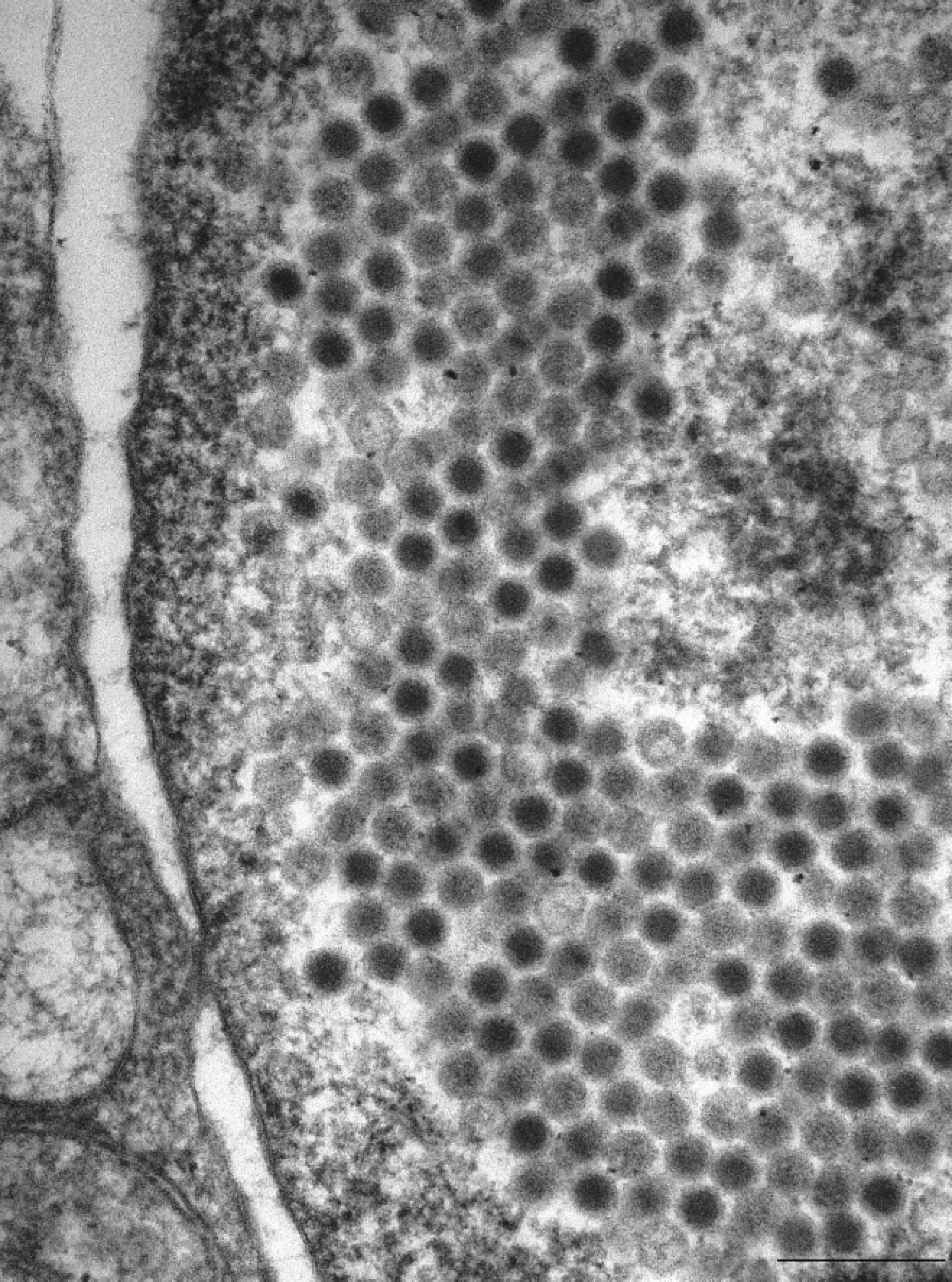
Electron microscopy. Crystalline array of adenovirus particles in nucleated pulmonary cells. Bar = 250 nm.
This is believed to be the first case report of naturally occurring fatal infection with CAdV-2 accompanied by a complete diagnostic workup including virus isolation, FAT, PCR, electron microscopy, and DFAT. During the writing of the present article, 3 more Bulldog pups from the same owner were submitted for gross necropsy and histopathologic evaluation at the Kansas State Veterinary Diagnostic Laboratory. Gross necropsy revealed pneumonia with a similar appearance and pattern to that described in the current study. Histologic abnormalities were restricted to the lungs and consisted of multifocal to coalescing necrosis with loss of normal architecture and frequent intranuclear inclusion bodies consistent with CAdV-2. Diagnostic testing was limited to histopathology.
Acknowledgements
The authors wish to thank Ms. Stephanie Heersink for her technical support with fluorescent antibody testing and Mr. Lloyd Willard for his assistance with electron microscopy.
Footnotes
a.
Epon LX112, Ladd Research, Burlington, VT.
b.
Hitachi H-300, Hitachi Ltd., Japan.
c.
Canine Adenovirus (CAV) FA FITC Conjugate, VMRD Inc., Pullman, WA.
d.
Minimum Essential Media Eagle, BioWhittaker Inc., Walkersville, MD.
e.
Canine Adenovirus Polyclonal Conjugate, VMRD Inc., Pullman, WA.
f.
DNA Blood Min-kit, Qiagen Inc., Valencia, CA.
g.
Hotstar Taq Master Mix, Qiagen Inc., Valencia, CA.
