Abstract
Pentobarbital is used commonly to euthanize animals. Occasionally during a death investigation, it is necessary to determine whether a cat or dog was euthanized via pentobarbital overdose. Screening for the detection of barbiturates including pentobarbital can be performed using commercial immunochromatographic tests. We used a commercial immunochromatographic test for barbiturates in humans to screen for barbiturates in urine collected postmortem from 20 dogs and 20 cats to determine whether they had been euthanized with pentobarbital. Additionally, we analyzed the urine for pentobarbital using liquid chromatography–mass spectrometry as a confirmatory test. Screening and confirmation testing revealed 100% agreement between the tests and with the euthanasia status of each animal. Our results support the use of the immunochromatographic test for the screening of urine collected postmortem to assess for the presence of barbiturates, specifically pentobarbital, used for euthanasia.
Pentobarbital is a drug in the barbiturate class of sedative-hypnotic drugs and is a U.S. Drug Enforcement Agency class II–controlled substance. Other drugs in the barbiturate class include secobarbital and phenobarbital. The primary action of pentobarbital is as a depressant in the central nervous system. 7 In veterinary medicine, pentobarbital, either alone or in combination with phenytoin, is used commonly to euthanize animals. 6 In human medicine, pentobarbital has been used as an anesthetic, for the treatment for seizures, and for the treatment of insomnia.4,5,8,10 In addition to the medical uses for pentobarbital, secobarbital, and phenobarbital, these are also drugs of abuse and are referred to on the street as “yellow jackets,” “red devils,” and “goof balls,” respectively. 15
When performing an autopsy on an animal, whether for diagnostic or forensic purposes, it may be important to confirm whether the animal died of natural causes or by chemical euthanasia such as by a pentobarbital overdose. For example, if several comatose and deceased birds were found around a dog carcass in a landfill, a likely cause would be pentobarbital relay toxicosis; however, there is no validated rapid test to be used on the deceased dog. Another example occasionally encountered in media reports involves cases of pets being euthanized accidentally by a veterinarian or, in other instances, pet owners blaming the veterinarian for euthanizing their pet without permission.2,11,12 These incidents lead distraught pet owners to ask questions that often guide legal actions that the owners wish to pursue against the attending veterinarian. Detection of barbiturates in these animals is paramount to determining if the animal was euthanized. Additionally, in several cases in our laboratory, for reasons unknown to us, we have explicitly been told by pet owners that their dog had died naturally at home, and only after performing expensive toxicology analysis did we determine that the animals were actually euthanized via barbiturate overdose.
Mass spectrometry (MS) is the current gold standard in forensic analysis of substances and, combined with a separation technique such as gas chromatography (GC-MS) or liquid chromatography (LC-MS), it can be used to detect and quantify a variety of drugs. 3 These procedures are commonly used for the detection of drugs in the barbiturate class in biological samples from humans. Additionally, commercial rapid tests that can be used to screen for barbiturates, including pentobarbital, are available for use in humans using urine as the biological matrix. Urine screening of humans for intoxication by barbiturates typically involves use of an immunochromatographic test. The available rapid tests for barbiturates have a limit of detection (LOD) of 300 ng/mL; screening tests will be positive if drug concentrations are above this level. 1
In veterinary medicine, blood samples or liver samples collected postmortem can be submitted for GC-MS or LC-MS to test for drugs in the barbiturate class. However, it takes several days at minimum to receive results given that samples must be shipped to specialized laboratories. Once in the laboratory, the time-consuming process involves both sample processing and extraction, as well as analysis time and data interpretation from the instrumentation. Additionally, the cost for analysis is US$100–250, depending on the laboratory. This high cost and time needed for testing are major issues for clients and veterinarians. In the emergency setting, the use of on-site, urine multidrug tests that screen for barbiturates as well as other drugs have been shown to be useful for clinical practice use in dogs. 13 However, there is currently no validated rapid and cost-effective test that can be used to screen dogs and cats postmortem for barbiturates when the situation arises.
We evaluated the usefulness of a commercial human immunochromatographic test to screen for barbiturates in the urine of dogs and cats postmortem to determine whether or not the animal had been euthanized with pentobarbital.
Materials and methods
Specimens
Urine was collected by cystocentesis from 20 dogs and 20 cats at autopsy that were submitted to the Veterinary Forensic Pathology Service (VFPS) at the University of Florida College of Veterinary Medicine (Gainesville, FL, USA). Animals were a combination of stray and client-owned. All samples were collected by a board-certified veterinary pathologist. We collected 1–25 mL of urine in each case; 0.5 mL of urine was required to perform our study. Urine was transferred to a red-top tube and refrigerated (4°C) until analysis. VFPS submission forms submitted with each animal indicated whether the animal was euthanized or died naturally. Sample collection was approved by the University of Florida Institutional Animal Care and Use Committee. All of the animals enrolled in our study had been euthanized for purposes unrelated to our study.
Immunochromatographic test
We used a barbiturate detection strip test (Alere one step barbiturates single drug test; Abbott), which is a competitive-binding immunoassay. The drug test is reported to detect 10 different barbiturates, including pentobarbital, secobarbital, amobarbital, alphenal, aprobarbital, butabarbital, butalbital, butethal, cyclopentobarbital, and phenobarbital. When a barbiturate is present in the specimen, it competes with the respective drug conjugate for binding sites for its specific antibody. If a drug is present in the specimen and it is above the LOD, it will saturate all of the binding sites of the antibody and will not generate a colored line on the test strip. The LOD for pentobarbital is 300 ng/mL.
The canine and feline urine samples were tested within 4 h of collection, and the remaining urine sample was frozen at −80°C. The samples were analyzed according to the manufacturer’s instructions. 1 Briefly, the test strip was immersed in a urine specimen for 10–15 s and then the test strip was placed on a flat surface. The test result was read 5 min after immersion in the urine. Two lines appear for a negative result, and one line appears for a positive result. The control line must appear for the test to be valid.
Determination of drug concentration
The analytical reference standards for pentobarbital and amobarbital were obtained from Cerilliant. The analytical reference standard for pentobarbital-d5 was obtained from Cayman Chemical. High-performance liquid-chromatography–grade or better solvents were purchased from Thermo Fisher.
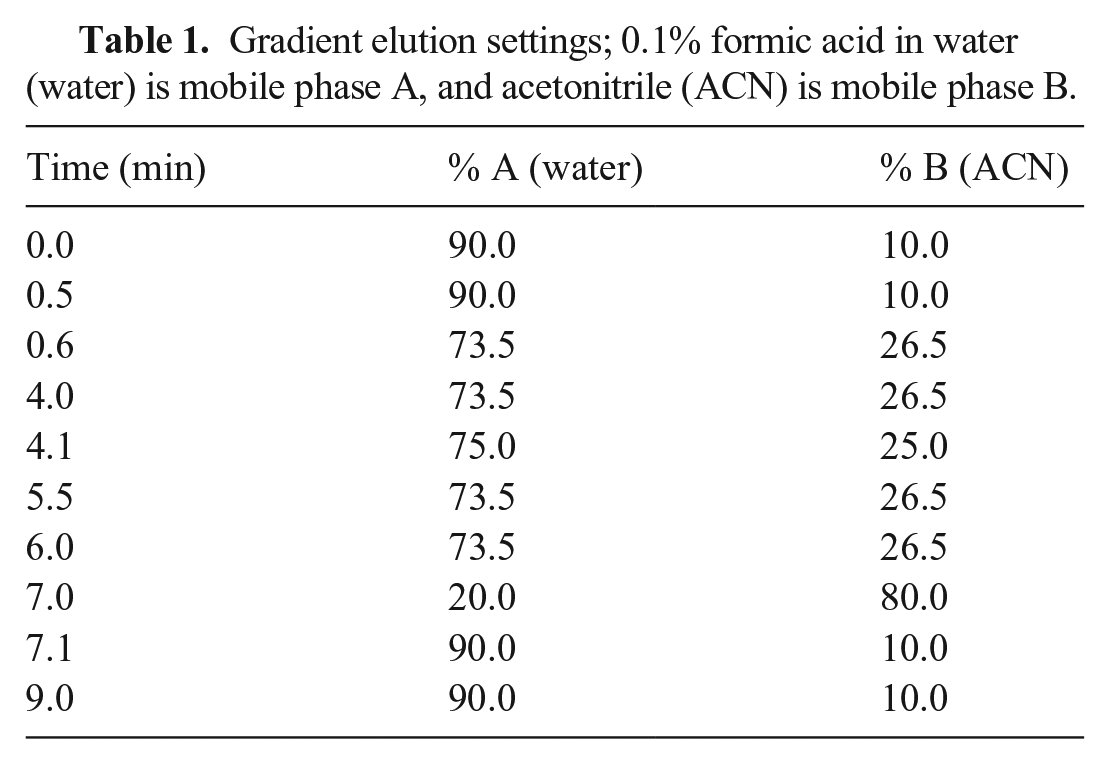
The analyte concentrations were determined in canine and feline urine samples using a liquid chromatography–tandem MS (LC-MS/MS) system equipped with a Quantum Ultra triple quadrupole mass spectrometer (Thermo Fisher) and an Accela liquid-chromatography system (Thermo Fisher) with a HCT pal autosampler (CTC Analytics). The operating parameters for the mass spectrometer included a spray voltage of 450 V, a vaporizer temperature of 325°C, and sheath and auxiliary gases of 60 and 15 arbitrary units, respectively. The product masses and collision energies of the analyte were optimized by infusing the standards into the TSQ Quantum Ultra MS. Chromatography employed a C18 column (50 mm × 2.1 mm, 1.9-μm column, Hypersil Gold; Thermo Fisher). Deionized water (solvent A) and acetonitrile with 0.1% formic acid (solvent B) were used as the mobile phases, at a flow rate of 0.3 mL/min (Table 1).
Gradient elution settings; 0.1% formic acid in water (water) is mobile phase A, and acetonitrile (ACN) is mobile phase B.
Urine sample analysis
Urine calibrators were prepared at concentrations of 300–20,000 ng/mL by supplementing drug-free canine or feline urine with working standard solutions. Drug-free urine was obtained from animals that were known to not have been euthanized and were not being administered any medications. Quality control samples were also prepared in the same fashion as the calibrators, with pentobarbital at a concentration of 2,000 ng/mL, and were included with each sample set. Because pentobarbital and amobarbital have identical molecular weights and identical product ions, they could only be distinguished by the special gradient outlined above that resulted in different retention times compared to the internal standard (i.e., the relative retention time). Pentobarbital yielded a relative retention time of 1.01; amobarbital eluted slightly later and yielded a relative retention time of 1.04. To verify within each run that the separation remained consistent, a separate quality control sample fortified with amobarbital only at a final concentration of 2,000 ng/mL was included with each sample set. Prior to analysis, 0.1 mL of each urine sample was diluted with 0.5 mL of saline and supplemented with the internal standard pentobarbital-d5 equivalent to 2,000 ng/mL. Supported liquid extraction was performed (SLE+ columns; Biotage). Samples were added to the SLE+ columns, allowed to sit for ~10 min, and were then eluted with 2 volumes (2.5 mL each) of methyl-tert-butyl-ether (MTBE). The samples were dried under nitrogen at 40°C and reconstituted in 1 mL of 10% acetonitrile in water; 20 μL was injected into the LC-MS/MS system. Software (GraphPad) was used to generate the calibration curves, and Excel (Microsoft) was used to determine the concentration of pentobarbital in all samples using linear regression analysis, with a weighting factor of 1/X used for the calibration curve.
Results
Using the barbiturate immunochromatographic test, urine samples from 13 cats were positive for barbiturates and negative for 7 cats (Fig. 1; Table 2). Barbiturate immunochromatographic test results were concordant with the submission form histories; the 13 positive cats had been euthanized and the 7 negative cats had died naturally. The LC-MS/MS results for all cats agreed with the results of the immunochromatographic test, and, when detected, barbiturate concentrations were 609 to >20,000 ng/mL, which exceeded the upper limit of quantification (LOQ).
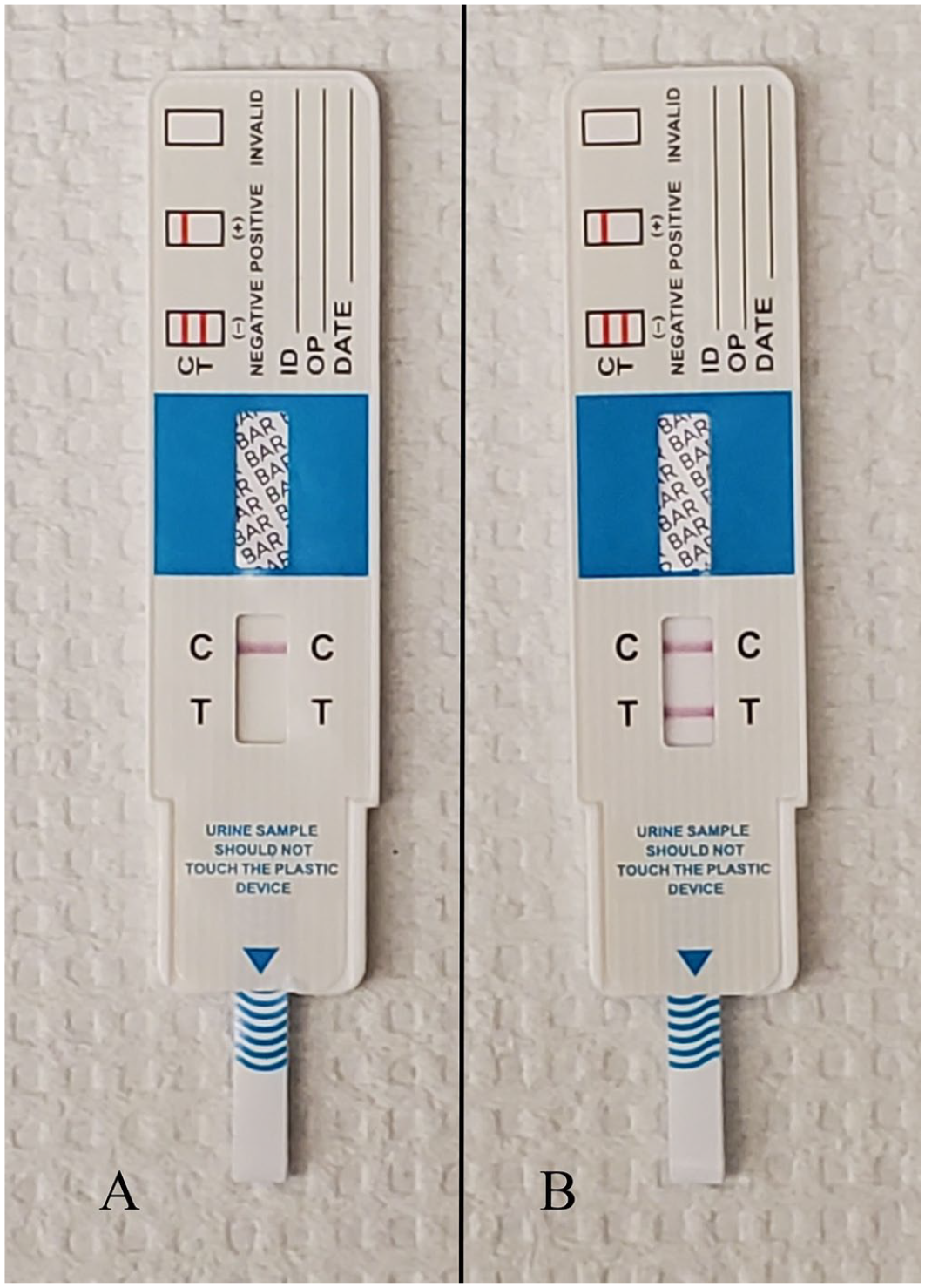
Barbiturate detection strip test.
Results of the immunochromatographic screening test for barbiturates and liquid chromatography–mass spectrometry (LC-MS) for pentobarbital from the urine of cats.
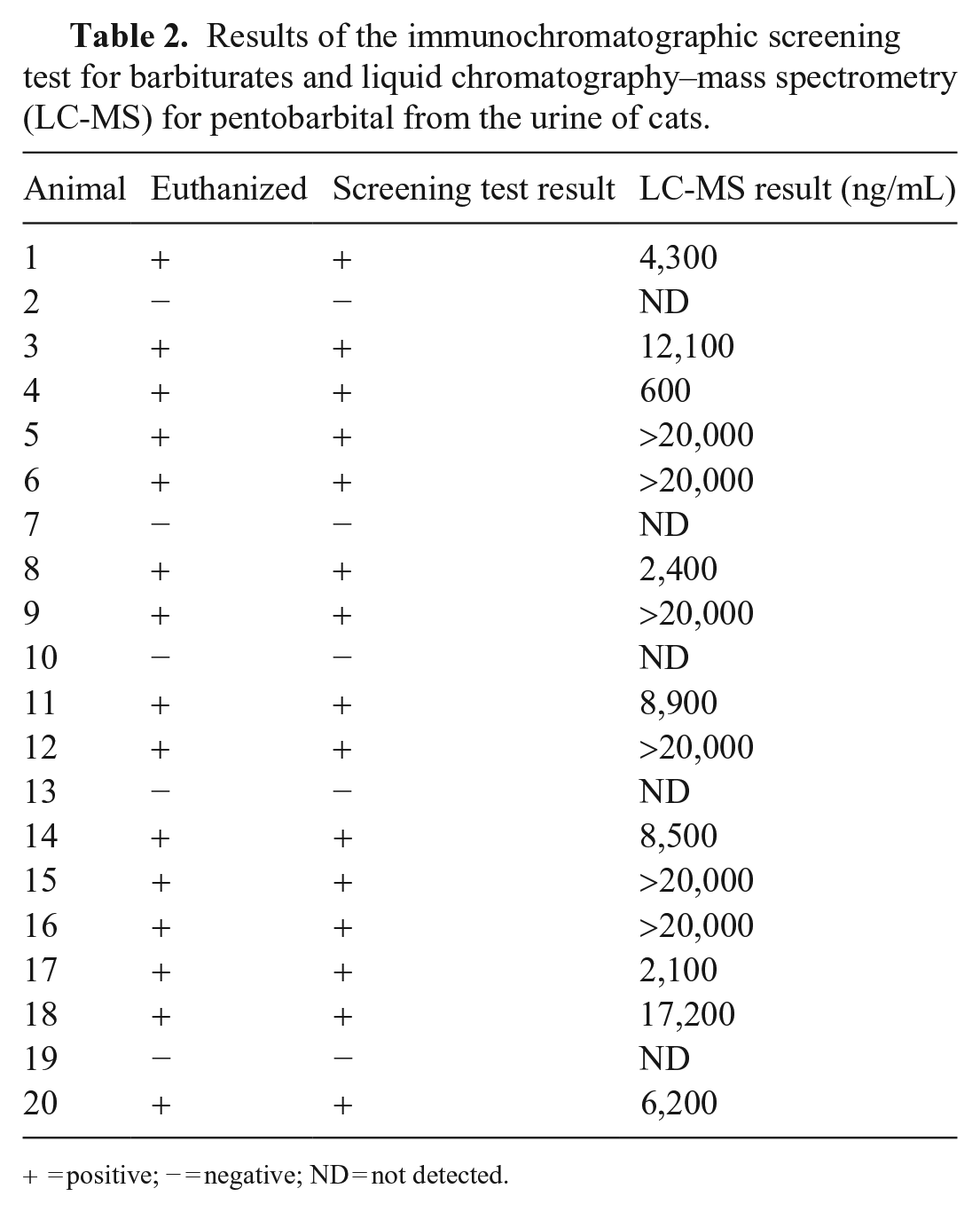
+ = positive; − = negative; ND = not detected.
Urine samples from 15 dogs were positive for barbiturates and negative for 5 dogs (Table 3). Barbiturate immunochromatographic test results were concordant with the submission form histories; the 15 positive dogs had been euthanized and the 5 negative dogs had died naturally. The LC-MS/MS results for all dogs agreed with the results of the immunochromatographic test, and, when detected, barbiturate concentrations were 305 to >20,000 ng/mL, which exceeded the upper LOQ.
Results of the immunochromatographic screening test for barbiturates and liquid chromatography–mass spectrometry (LC-MS) for pentobarbital from the urine of dogs.
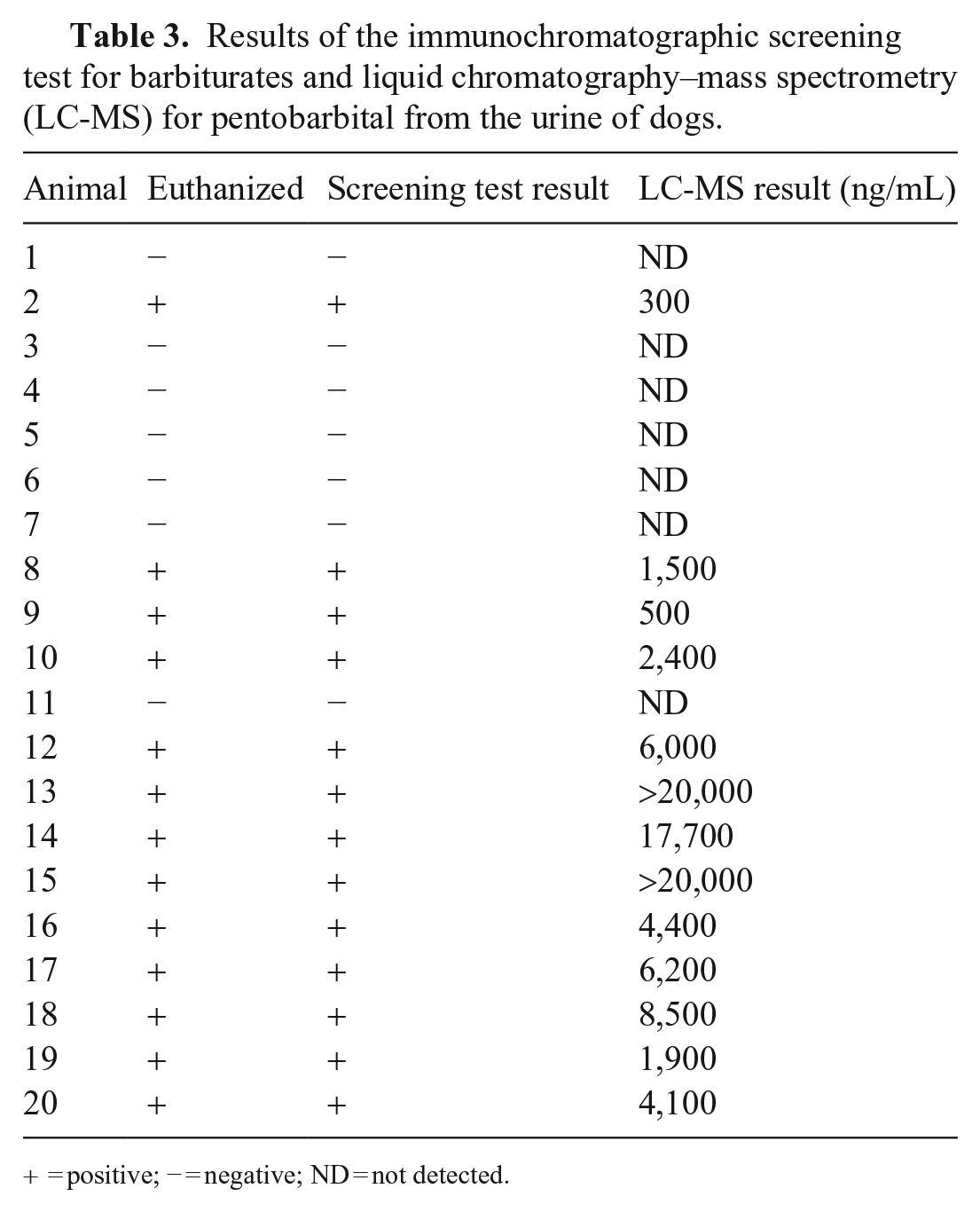
+ = positive; − = negative; ND = not detected.
In urine samples from both species, pentobarbital was distinguished from amobarbital by differences in the retention time of the 2 molecules. There was no indication of amobarbital in any of the samples tested.
Discussion
When the question of whether an animal was euthanized via pentobarbital overdose is asked, the conventional confirmatory test is GC-MS or LC-MS. We have demonstrated that a commercial immunochromatographic test can be used to screen urine samples from cats and dogs postmortem for the presence of the barbiturate class of drugs, which includes pentobarbital. There was 100% agreement observed in both the feline and canine urine samples analyzed. Based on screening test results, we were able to rule in the possibility of pentobarbital overdose with a positive test result and rule out the possibility of a pentobarbital overdose with a negative test result.
To confirm the presence or absence of pentobarbital in the urine samples, LC-MS was performed on all samples in a blind manner. There was 100% agreement between the immunochromatographic test results and the LC-MS results for all feline and canine samples tested. Our results confirmed that the immunochromatographic tests were detecting pentobarbital within the urine samples. The concentrations of pentobarbital in all canine and feline urine samples were above the LOD of 300 ng/mL for the immunochromatographic test, which supports the use of urine for testing for barbiturates with this rapid test. The LOQ and LOD for the LC-MS/MS method were 300 ng/mL and 231 ng/mL, respectively.
Screening of feline and canine urine for barbiturates with an immunochromatographic test when questions regarding the use of chemical euthanasia arise could result in the presumptive identification of barbiturates in urine samples, at which point confirmatory testing for pentobarbital should be performed. The identification of barbiturates in a carcass that was being scavenged by birds that were comatose and dying would allow for the presumptive diagnosis of pentobarbital relay toxicosis and ultimately the rapid treatment of the surviving birds could be instituted. For the pet owner who thought that their dog was euthanized by a veterinarian without their consent, a negative screening test might provide some comfort that the veterinarian did not euthanize their pet and ultimately avoid a costly legal confrontation.
Confirmatory testing using LC-MS or GC-MS can be performed after a rapid screening test is completed. 9 Use of a screening test is appropriate given that the average veterinary hospital or laboratory would not have access to the equipment required to conduct confirmatory analysis and would require the samples to be sent out for testing. In medical examiner’s offices, it is common for the pathologist to keep rapid tests on hand to screen for drugs of abuse during the autopsy. 14 The results from this initial screening step help the pathologist determine if there is further toxicologic examination required for the case. The same can be said for veterinary pathologists screening for drugs. The Alere immunochromatographic test is inexpensive and rapid and can provide valuable information when screening for barbiturates at autopsy of a dog or cat.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
