Abstract
Three calves were submitted to the Iowa State University Veterinary Diagnostic Laboratory for diagnostic evaluation following an abrupt increase in morbidity and mortality in a calf herd associated with epistaxis and widespread hemorrhage. Each of the submitted calves had moderate-to-severe hemorrhage within various tissues and body cavities, including the thymus, subcutaneous region of the neck, mediastinum, lungs, pericardial sac, heart, spleen, perirenal fat, urinary bladder, and skeletal muscle, including the diaphragm. An anticoagulant rodenticide screen was performed on the livers of each calf. Significant concentrations of chlorophacinone were detected at 4.2, 3.6, and 2.9 ppm in liver. Multiple piles and an open pail of white powdery material were present within the facility in which the calves were housed and were identified as the sources of chlorophacinone. Acute hemorrhage and death occurred in fourteen 1.5-mo-old, crossbred calves following ingestion of the vitamin K antagonist chlorophacinone.
We describe here the etiology, clinical presentation, lesions, and testing associated with chlorophacinone poisoning in 14 calves. Originally derived from dicoumarol, which is found in moldy sweet clover (Melilotus officinalis and Melilotus alba), anticoagulant rodenticides (ARs) are the most commonly used rodenticide compounds.6,8,13 Given the stable nature and high potential for relay toxicosis in non-target species, use of these compounds has been scrutinized and, in some regions, has undergone increased regulation and restriction. 11 Although a common occurrence in small animals, poisonings associated with AR compounds in livestock are observed only rarely. 5 Dicoumarol is most commonly associated with hemorrhage in cattle that have consumed moldy sweet clover. However, dicoumarol-associated hemorrhage has also been reported in cattle consuming sweet vernal grass (Anthoxanthum odoratum).2,12 To our knowledge, there is only one prior report of chlorophacinone intoxication in cattle. 4
In February 2021, sudden death was reported over 5 d in fourteen 1.5-mo-old, crossbred calves. Preceding death, several calves were reported to exhibit ataxia and mild respiratory distress. Seven days prior to the onset of complications, ~60 cow–calf pairs were placed in the vicinity of a slatted hog confinement barn. The facility had not contained any livestock for ~20 y. Although access by the cows was restricted, calves could access the interior of the building. The facility was used to provide calves reprieve from a bout of cold weather, with temperatures declining to −35°C. The manure pit of the building was 2.4 m deep; manure had been removed from the pit 20 y prior, and the pit had been packed with dirt. Cornstalks harvested from the previous year were blown into the building to serve as bedding for the calves several days before placing the calves in the confinement building.
Three fresh deceased calves were submitted to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, IA, USA) for postmortem examination and toxicologic evaluation. All 3 calves were in good body condition and had minimal-to-no autolysis. Abomasal and rumen contents were examined for noxious plants, including those associated with dicoumarol, and other suspicious substances. The rumen content of each calf consisted primarily of cornstalks and leaves in addition to strands of bale netting. Abomasa contained moderate amounts of milk clot. In all 3 calves, petechial-to-ecchymotic hemorrhage was present throughout the thymus, endocardium, spleen, and various skeletal muscle groups, including the diaphragm. Each calf had various degrees of hemothorax (Fig. 1). If devoid of hemorrhage, the lungs, spleen, kidneys, abdominal viscera, and skeletal muscle were pale. The oral cavity, other mucous membranes, and skin lacked erosive or ulcerative lesions. Blood was not present in the perianal region. Hemorrhage was more widespread and pronounced in calves 2 and 3; severe subcutaneous hemorrhage and edema was observed in the cervical region extending from the mandible to the thoracic inlet in both calves (Figs. 2, 3). Calf 2 also had significant hemorrhage within the perirenal fat and the wall of the urinary bladder; blood was not present in the urine. Petechial hemorrhages were present throughout the lungs. Blood was present in the abomasum (~1 L), small intestine, and colon of calf 2. Calf 3 had an ~12 cm-diameter hematoma within the mediastinum ventral to the esophagus and extending from the thoracic inlet to the diaphragm. The joints were not swollen in any of the calves and were not examined further. Prior to submission of calves to the ISU-VDL, a postmortem examination was performed on only one calf at the site given concern for a potential zoonotic agent. Clinical signs of respiratory distress and ataxia, as well as epistaxis, were similar in calves that did not undergo postmortem examination as well as in calves in which a postmortem examination was performed.
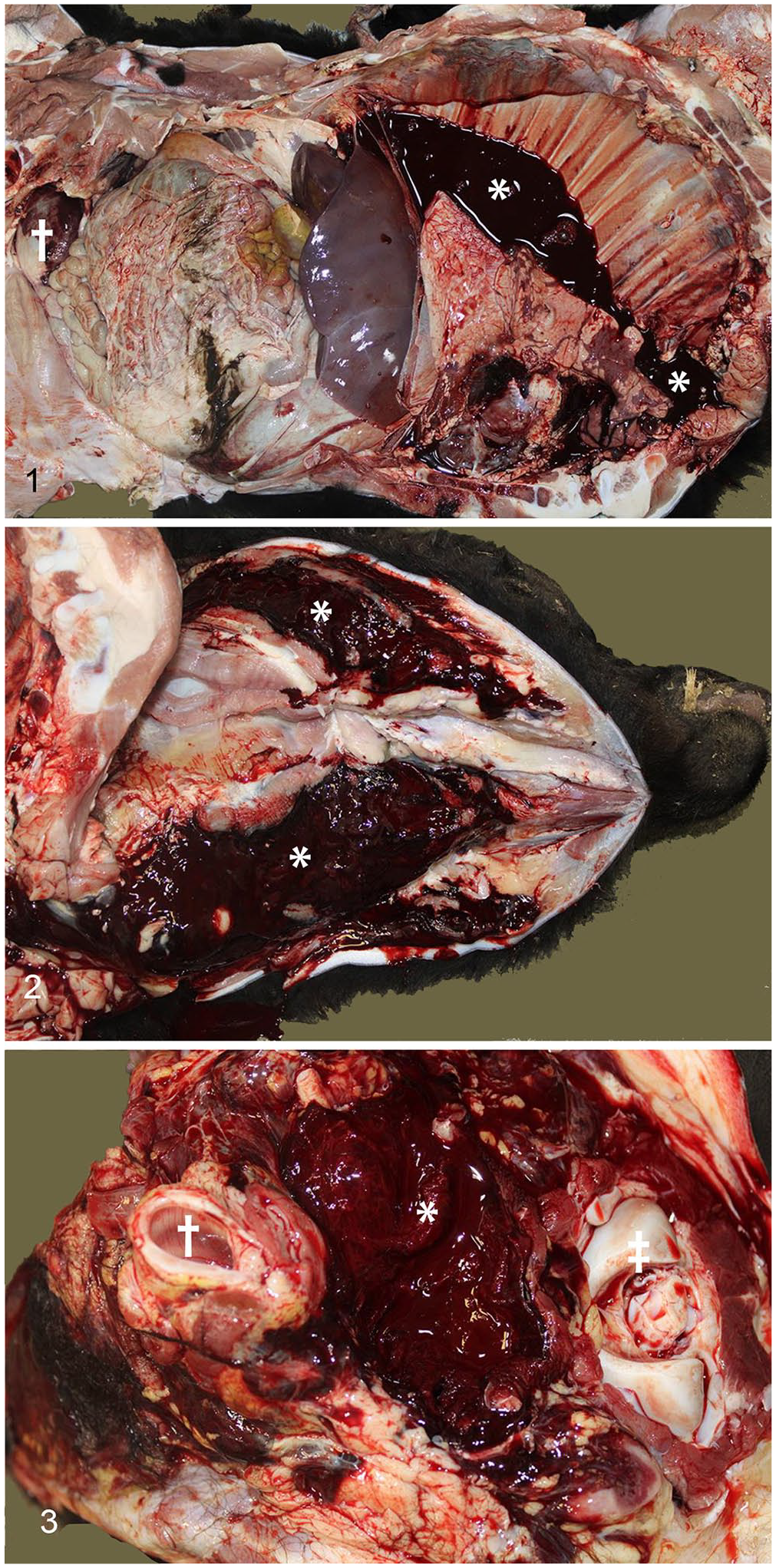
Hemorrhagic lesions in calf 2. Calf in left lateral recumbency, cranial is to the right in all images.
Given the concern for zoonotic potential of a possible infectious agent, 5 mL of whole blood was collected from each calf and submitted to the University of Iowa State Hygienic Laboratory (Coralville, IA, USA) for PCR evaluation for Bacillus anthracis. No bacilli were observed in blood smears. Results of the PCR assay for B. anthracis were negative.
Tissues for histologic evaluation were placed in 10% neutral-buffered formalin for ~16 h, processed routinely, and sections were stained with H&E. Acute thymic and splenic petechial and ecchymotic hemorrhage was noted in each calf. Hemorrhage was noted in the lymph nodes of calf 1, and in the lungs of calf 2. Clotted blood intermixed with feed material was noted in the lumen of the small intestine of calf 2. Skeletal muscle was collected for histologic evaluation from areas of pallor and non-hemorrhagic regions to evaluate for myopathies, particularly those associated with ionophore intoxication given that calves had access to the cow feed that contained monensin; no histologic abnormalities were noted. No microscopic lesions indicative of inflammation or suggestive of infectious agents were observed in any tissue of any of the 3 calves, hence, bacterial culture was not performed.
Based on clinical signs, owner concerns, and observed lesions, differentials included dicoumarol, ARs, stachybotryotoxins, B. anthracis, and Salmonella spp. Liver, ocular fluid, abomasal content, and rumen content were collected for toxicologic evaluation. A nitrate dipstick (Merck) was used to screen ocular fluid for nitrate and nitrite; nitrate at a concentration >10 ppm or nitrite were not detected. A toxic element screen, including essential macro- and micro-minerals, as well as antimony, arsenic, barium, boron, cadmium, lead, mercury, and thallium, was performed on the liver of calf 2 by inductively coupled plasma–optical emission spectrometry. Aside from the below-normal concentration of manganese, no other abnormal concentrations of other elements were observed. Although nitrate and metals were not on the differential list for our case nor are they typically associated with the observed lesions, we routinely perform these analyses in ruminant cases with neurologic signs and sudden death.
Contents of the abomasum and rumen, as well as the corn stalks used for bedding, were evaluated by gas chromatography–mass spectrometry. Cornstalks were also tested for mycotoxins, including stachybotryotoxins by liquid chromatography–tandem mass spectrometry (LC-MS/MS) at the North Dakota State University Veterinary Diagnostic Laboratory (Fargo, ND, USA). No toxic compounds associated with the reported clinical signs or of diagnostic significance were detected by either analysis.
Livers were evaluated by LC-MS/MS for the anticoagulant compounds brodifacoum, bromadiolone, chlorophacinone, coumachlor, dicoumarol, difenacoum, difethialone, diphacinone, and warfarin, using a validated method. 4 Chlorophacinone was detected above the assay quantification limit of 1.5 ppm. Further quantification of chlorophacinone in the livers was conducted by high-performance liquid chromatography with a UV/Vis detector. Chlorophacinone was quantified at 4.2, 3.6, and 2.9 ppm in calves 1–3, respectively. A white powdered rodenticide was reported to have been utilized in the building at the time swine were housed 20 y prior. Upon further inspection of the facility, a bucket of powdered material was found, with chlorophacinone being identified as the active ingredient on the label. Piles of the powder were located throughout the facility. No powdered material from either the bucket or any of the piles was submitted for analysis. Evidence of calf activity was present near the observed piles. Once the piles were identified, it was determined that all deceased calves had been able to access these piles prior to death. Although a postmortem examination was performed on only 4 of 14 calves, and chlorophacinone was confirmed in 3 of 14, exposure to the compound, clinical signs, and observed hemorrhage were consistent in all 14 calves, strongly suggesting that morbidity and mortality in the remaining 11 calves was caused by chlorophacinone. Detection of chlorophacinone, a vitamin K antagonist, in the liver, the accessibility of the powder to calves, and widespread hemorrhage provided clear evidence of AR poisoning.
Chlorophacinone (2-alpha-4-chlorophenyl-a-phenylacetyl-1,3-indandione) is an indandione first-generation anticoagulant. Chlorophacinone is found in oral baits (0.05%) and tracking dusts (0.2%). 10 In our case, the chlorophacinone was a tracking dust formulation that had been stagnant in the environment for 21 y. The powder was not submitted and hence the concentration of chlorophacinone within the powder could not be determined. Decay of most of the label precluded determination of the original concentration. Hemorrhage following ingestion of ARs is the result of impairment of the clotting cascade. Vitamin K antagonist rodenticides act through inhibition of the vitamin K epoxide reductase enzyme, which is essential for the recycling of vitamin K through the reduction of inactive vitamin K epoxide to the active form. 6 Vitamin K is necessary in the production of clotting factors II, VII, IX, and X. Factors VII and IX are associated with the extrinsic and intrinsic pathways, respectively; factors II and X are associated with both pathways. Hemorrhage is the hallmark manifestation of AR poisoning. Clinical signs following AR ingestion may be delayed for 24 h to several days, during which time the clotting factors present within the body become depleted. 8
Reported ingestions of AR compounds are rare in livestock species. 5 Chlorophacinone has been reported once in cattle and twice in lambs.4,7 In the reported bovine case, 7 cows died, with subcutaneous hemorrhage, unclotted blood, and pale livers. 4 The hemorrhage and lesions in that case, although less severe, were similar to those in the calves in our case. The concentration of chlorophacinone in the liver in the cows was not reported. 4 The clinically affected lambs 7 and the calves in our case had extensive hemorrhage within the neck region, thymus, and the musculature leading to the thoracic cavity. The lack of hepatic lesions in our case differed from the report in lambs, which had centrilobular necrosis. 7 The concentrations of chlorophacinone in the calf livers in our case were 5–7 times greater than those reported in the lambs. 7 Similarities and degree of hemorrhage between lambs in the previous cases and calves in our case may be associated with age and ruminal development.3,7,8 Although the chlorophacinone concentration in the powder was not determined in our case, consumption of either the 0.05% or 0.2% formulation could result in the same lesions and mortality. Consumption of smaller amounts of the 0.2% formulation than of the 0.05% formulation would be expected to cause clinical signs and mortality. Lack of hepatocellular necrosis may have been the result of the acute-to-peracute nature of death, which did not allow time for the development of lesions.
Chlorophacinone was accessible to calves for several days, but it is unknown how much of the compound was ingested by the calves. The toxicity of AR varies by species. 10 No toxic dose of chlorophacinone has been determined for cattle; the toxic tissue level has not been reported for chlorophacinone or other ARs in livestock or other domesticated species. Diagnosis of AR toxicity is dependent on a history of exposure, clinical signs, and detection of an anticoagulant compound in the tested tissue or fluid.
Treatment of AR poisoning focuses on the restoration of clotting factors. 6 In dogs, supplementation of vitamin K1 (phytonadione) at 2.5 mg/kg PO q12h to q8h has been recommended. Administration of vitamin K1 at 1.1–3.3 mg/kg IM has been successful in the treatment of animals consuming dicoumarol. Treatment of dicoumarol toxicity with menadione sodium bisulfate (vitamin K3) at up to 44 mg/kg PO and 22 mg/kg IM has been attempted with little success. 1 The duration of treatment can be up to 4 wk.8,13 Blood transfusions offer an immediate supply of clotting factors. 10 Given the cost and limited availability of transfusions, treating multiple large animals is not feasible. 1 Decontamination through the use of activated charcoal and cathartics is of little use unless initiated early or the affected animals have been observed consuming such compounds.6,10,13 The futility of decontamination methods following observation of clinical signs is the result of the delayed onset following ingestion. 8 In our case, following the initial deaths, calves were immediately removed from the barn. Although chlorophacinone has been reported to be excreted in milk, there was minimal concern of further exposure of surviving calves through nursing because cows did not have access to the interior of the barn. 9
Although poisonings associated with synthetic ARs by livestock species are rare, as indicated by our case being only the fourth report of chlorophacinone intoxication in livestock species since 1992, occasional exposures do occur.4,5 If marked hemorrhage is found in livestock species, postmortem evaluation of liver for vitamin K antagonists should be considered. To limit and prevent exposure, any environment in which animals are placed should be evaluated for potential toxic agents. Such agents should be removed from the environment regardless of the time elapsed given the retention of toxic properties. 8 Our case exemplifies the stability of chlorophacinone over a 21-y period. Although the facilities had been abandoned for >20 y, chlorophacinone was still able to inflict substantial morbidity and death in calves.
Footnotes
Acknowledgements
We thank the University of Iowa State Hygienic Laboratory and the North Dakota State Veterinary Diagnostic Laboratory for their assistance and analytical efforts. We also thank the ISU-VDL staff for timely processing of the collected samples.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or this publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
