Abstract
Exposure of wildlife and domestic animals to anticoagulant rodenticides (ARs) is a worldwide concern, but few methods exist to determine residue levels in live animals. Traditional liver detection methods preclude determining exposure in live wildlife. To determine the value of assessing AR exposure by fecal analysis, we compared fecal and liver residues of ARs in the same animals. We collected liver and fecal samples from 40 apparently healthy red foxes (Vulpes vulpes) potentially exposed to ARs, and quantified brodifacoum, bromadiolone, coumatetralyl, difenacoum, difethialone, and flocoumafen residues by liquid chromatography–tandem mass spectrometry. Residues of ARs were detected in 53% of the fecal samples and 83% of the liver samples. We found good concordance between AR residues in feces and liver for coumatetralyl, difenacoum, and difethialone. Bromadiolone occurred in significantly greater frequency in livers compared to feces, but no significant difference in concentration between feces and liver in individual foxes could be detected. Brodifacoum displayed a significant difference in concentration and occurrence of positive samples between liver and feces. Our findings demonstrate that fecal analysis of ARs provides a feasible and valuable non-lethal means of determine AR exposure in live wildlife.
Anticoagulant rodenticides (ARs) have been used worldwide in pest control since the 1950s. ARs include first-generation ARs (FGARs), such as warfarin, diphacinone, coumatetralyl, and chlorophacinone, and second-generation ARs (SGARs), such as brodifacoum, bromadiolone, difenacoum, difethialone, and flocoumafen.
Secondary exposure (ingestion of poisoned prey) in wildlife is a worldwide problem, and AR residues have been verified in 84–99% of livers from predators such as the red fox (Vulpes vulpes), stone marten (syn. beach marten; Martes foina), and European polecat (Mustela putorius).4,21 Subtoxic levels of ARs may induce behavioral changes and reduced body condition in predators, impairing hunting ability and predisposing them to accidents and injury. 2 The threat of secondary poisoning in the critically endangered arctic fox (Vulpes lagopus) is of particular concern. The red fox may act as a sentinel for this species because of its widespread distribution and similar feeding resources.
ARs accumulate in the liver, and the major route of elimination is through feces. 5 Exposure in wildlife is normally assessed by residue analyses in liver, restricting examination to potentially biased opportunistically sampled dead animals. ARs have been analyzed in plasma or assessed by coagulation test to verify AR exposure in animals,1,12 but this is inadequate in verifying sublethal exposure because residues can be detected in feces even when ARs are no longer detectable in plasma of either foxes or dogs.17,18 During chemical immobilization and radio-tagging procedures of endangered species, there is an opportunity to use noninvasive techniques to sample feces from sedated animals. However, to be able to interpret such results, studies are needed to compare concentrations of ARs in feces with corresponding liver concentrations. We measured concentrations of ARs in liver and fecal samples collected from the same animal to evaluate the value of assessing AR exposure by analyzing AR concentrations in feces from live wildlife.
The 40 wild red foxes included in our study were apparently healthy animals shot in Norway by experienced hunters during the winter and spring of 2016. Feces and a piece of the liver were removed immediately after death and submitted to the Norwegian Veterinary Institute (NVI) within 2 d. The submitted samples were frozen at −80°C and kept frozen at this temperature for 3 d, before being stored at −20°C until preparation and analysis. In our study, we analyzed the ARs used most commonly in Norway: brodifacoum, bromadiolone, coumatetralyl, difenacoum, difethialone, and flocoumafen.
Fecal samples were lyophilized to dryness and analyzed at the laboratory of the Department of Forensic Sciences, Oslo University Hospital. Procedures for fecal extraction and analysis of ARs have been validated in our laboratory and applied in our previous study in foxes.18,19 Briefly, fecal samples were homogenized and aliquots of 100 mg removed. ARs were extracted with acetonitrile and dichloromethane from the aliquots and separated (Acquity ultra performance liquid chromatography BEH C18 column; Waters) with a mobile phase consisting of ammonium formate buffer and methanol. Positive electrospray ionization (ESI) tandem mass spectrometry (MS/MS) detection was performed on a triple quadrupole mass spectrometer (Waters), using 2 multiple reaction monitoring transitions. Signal-to-noise ratios were >10; precision and accuracy were within ±20%. In feces, limits of quantification (LOQs) were 1.5 ng/g for coumatetralyl, 2.2 ng/g for difenacoum, 2.6 ng/g for brodifacoum and bromadiolone, and 2.7 ng/g for difethialone and flocoumafen.
Liver samples were analyzed at NVI by a previously validated method. 9 Liver extracts (0.5 ± 0.1 g) were homogenized twice with acetone, before evaporating the liquid fraction to dryness. Residues were re-dissolved in acetonitrile and washed twice with hexane (Fluka Chemika; Millipore Sigma). ARs were separated (1200 series high performance liquid chromatography, Agilent Technologies; Xbridge C18 column, Waters). The column was equilibrated with ammonium acetate (Fluka Chemika) in water and acetonitrile at a ratio of 20:80 (v/v). The ARs were detected (negative ESI; G6470A triple quadrupole LC-MS; Agilent Technologies). Fragment ion spectra were recorded using 2 multiple reaction monitoring transitions. The recovery rates of ARs from liver tissue were 87–95%. Wet liver tissue LOQs were 0.5 ng/g for coumatetralyl, 0.8 ng/g for difenacoum, 1.8 ng/g for brodifacoum and bromadiolone, and 0.3 ng/g for difethialone and flocoumafen.
Comparisons between frequencies of AR occurrence between compounds in feces and liver were assessed by the Fisher exact test, and statistical comparisons were conducted using statistical software (Epi Info v7.2.3.1; Center for Disease Control and Prevention, Division of Health Informatics & Surveillance, Atlanta, GA). Statistical computations of AR concentrations between feces and liver were assessed by Wilcoxon signed rank test and conducted by JMP Pro (v14.2.0; SAS Institute). Nonparametric tests were used when data were not normally distributed; p ≤ 0.05 was considered statistically significant.
Of the 40 wild red foxes examined, 35 of 40 (88%) contained detectable residues of 1 or more ARs. Residues of ARs were detected in 21 of 40 (53%) fecal samples and 33 of 40 (83%) liver samples. The number of detected ARs differed between feces and liver, but brodifacoum was most prevalent in both (Table 1). Given the low number of samples positive for flocoumafen, we excluded this substance from further statistical comparisons.
Number of red foxes from which anticoagulant rodenticides were detected.
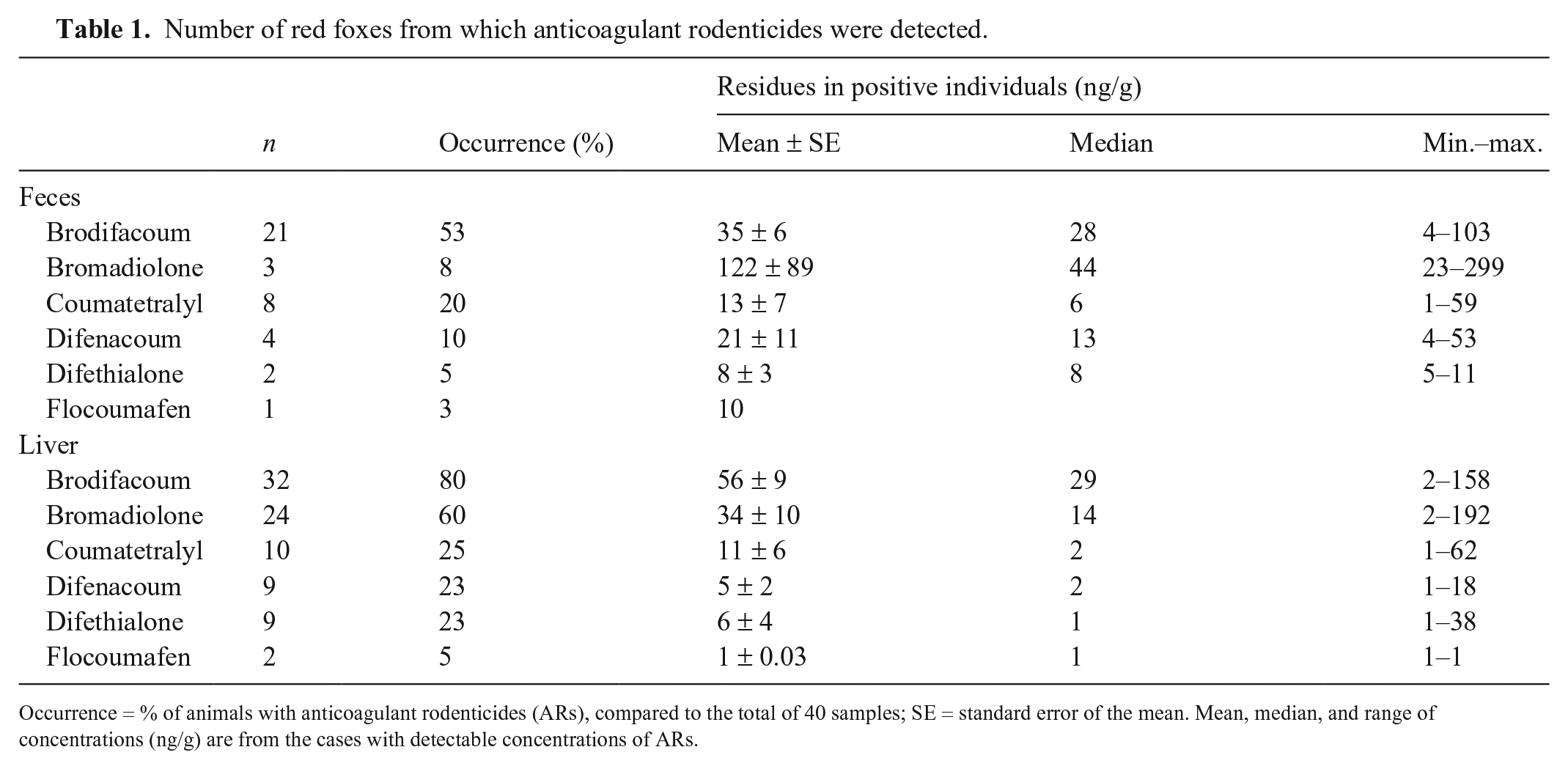
Occurrence = % of animals with anticoagulant rodenticides (ARs), compared to the total of 40 samples; SE = standard error of the mean. Mean, median, and range of concentrations (ng/g) are from the cases with detectable concentrations of ARs.
Comparing summed number of positive samples for each substance between feces and liver, there was a statistically significant difference between specimens for brodifacoum (p = 0.018) and bromadiolone (p < 0.0001; Fig. 1). No significant differences were found for coumatetralyl (p = 0.790), difenacoum (p = 0.225), and difethialone (p = 0.051).
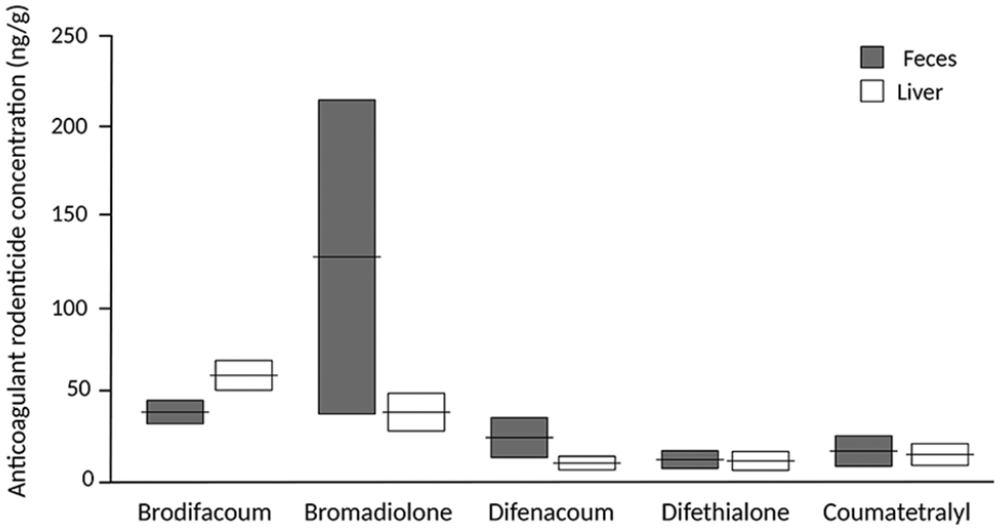
Fecal and hepatic concentrations of anticoagulant rodenticides from 40 wild red foxes. The results are given as means ± SE. For n, see Table 1.
Comparisons of AR concentrations between feces and liver demonstrated no statistically significant difference for bromadiolone, coumatetralyl, difenacoum, or difethialone. Brodifacoum, however, was detected in significant higher concentration in liver than feces (p = 0.003).
In 11 of 40 (28%) foxes, the hepatic AR concentrations were >100 ng/g (mean: 178 ng/g). In 4 of these animals, the concentrations were >200 ng/g (202–354 ng/g). Concentrations >100 ng/g were also detected in 2 of the fecal samples (113 and 362 ng/g).
Two or more ARs were detected in 11 of 40 (28%) fecal samples, with a mean of 1.9 ARs in the positive foxes. In the liver samples, 2 or more ARs were found in 27 of 40 (68%), with a mean of 2.6 in the positive foxes (Fig. 2). There was a significant difference between number of substances in liver compared to feces (p = 0.001).
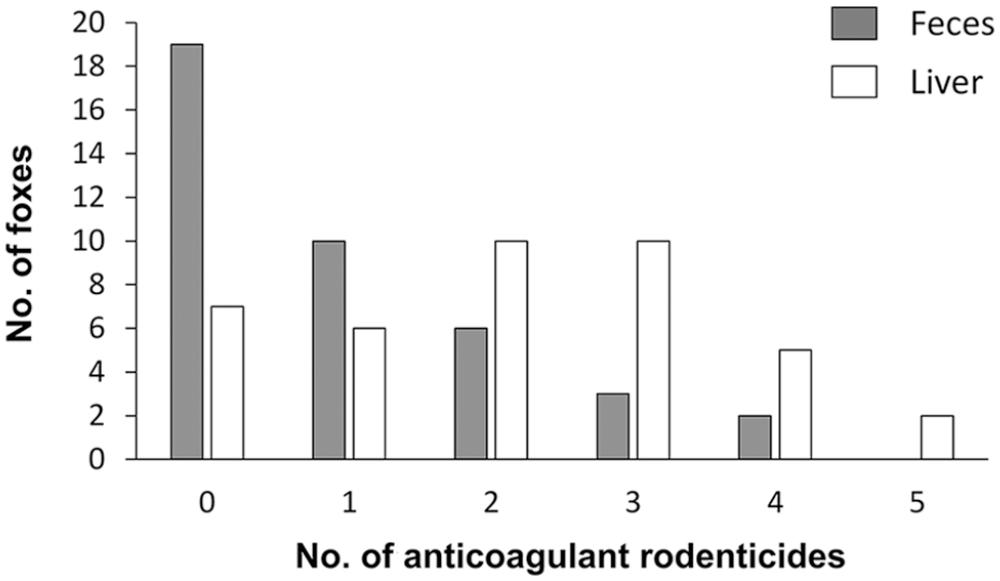
Number of anticoagulant rodenticides detected in samples of feces and liver collected from 40 wild red foxes. The samples were analyzed for brodifacoum, bromadiolone, coumatetralyl, difenacoum, difethialone, and flocoumafen.
Overall, our results revealed good concordance between residues in feces and liver for coumatetralyl, the only FGAR analyzed. We detected the compound in 20% of the fecal and 25% of the liver samples; this is a high number considering previous suggestions of more rapid elimination of FGAR than SGAR. 16 Earlier studies have estimated the half-life of coumatetralyl of 15.8 d in mice and 55 d in rats.16,23 The prevalence detected in our study suggests that coumatetralyl has a longer half-life in red foxes than previously estimated in rodents, which is in accordance with previous findings of estimated terminal half-life of at least 81 d after a single ingestion in a dog. 18
We found good concordance of difenacoum and difethialone residues between feces and liver, both in concentration and frequency of positive foxes. The consistency between similar concentrations of difethialone found in liver and feces is probably a result of its exclusive fecal elimination as unchanged parent material. 8 Difenacoum displays similar elimination in feces with <2% excretion in urine. 22 On the other hand, 5% of bromadiolone is eliminated through urine, and similar excretion is seen with brodifacoum. 6 Although this difference in urinary elimination is small, a contribution to the difference in the concentrations between liver and feces of bromadiolone compared to difethialone and difenacoum is possible.
Bromadiolone was identified in a significantly higher number of livers compared to fecal samples. However, no significant difference in concentration of bromadiolone between feces and liver in the individual foxes was detected. This discrepancy is probably a result of the low number of positive fecal samples compared to liver. We detected bromadiolone in feces in only 3 animals, but in high concentrations. In one of these foxes, fecal concentration was 299 ng/g, with corresponding liver concentration of 35 ng/g. The high fecal concentration could indicate recent ingestion of either bait or rodent containing a high amount of bromadiolone. Another reason for the discordance in results may be low sensitivity in detection of bromadiolone in feces. A comparatively low detection in feces was identified in a previous experiment in 4 foxes, with a mean of only 1.1% bromadiolone in feces compared to liver 26 d after exposure. 17 On the other hand, given the low number of foxes in that experiment, direct comparison to our results is specious.
We detected brodifacoum significantly more often and in higher concentration in liver than in feces. We examined whether the significance in our results was influenced by the different LOQs in feces (2.60 ng/g) and liver (1.80 ng/g), but no such effect was found. One reason for this discrepancy in test results could be the result of variation in metabolism. Rats resistant to bromadiolone are suggested to have different metabolism of the compound compared to susceptible rat breeds or strains. 14 Whether this is valid for other ARs or affects the animals’ metabolism after secondary exposure is not known. Furthermore, the discordance could in part be the result of a longer liver elimination half-life of brodifacoum (350 d detected in rats). 5 Bromadiolone has an equivalent half-life of 318 d. 6 In comparison, difenacoum and difethialone have an estimated liver elimination half-life of 118 and 126 d, respectively.7,8 On the other hand, as bromadiolone was detected in only 3 fecal samples, extended comparisons are inconclusive. Furthermore, feces from foxes contain plant material and hair influencing extraction recovery and AR concentration, which is likely to contribute to the lower detection in feces compared to liver. 19
Thresholds of toxicity for liver residues of ARs have not been established. In barn owls (Tyto alba), hepatic concentrations >200 ng/g SGAR were previously determined as potentially lethal 15 ; a later study indicated a significant risk of acute intoxication with levels <100 ng/g. 20 However, one study demonstrated no signs of ill health in barn owls with liver residues up to 690 ng/g brodifacoum, 140 ng/g difenacoum, and 520 ng/g flocoumafen. 11 This discrepancy could be the result of large variation in individual susceptibility to ARs within species. Furthermore, tolerance to ARs is highly variable between species. Liver concentrations of 39 ng/g and 160 ng/g bromadiolone were lethal in poisoned dogs. 3 In contrast, liver residues of up to 2,060 ng/g bromadiolone were detected in randomly shot wild red foxes. 10 This difference could be because of a large variation in metabolism and vitamin K epoxide reductase activity between species. 24 In our study of presumed healthy foxes, 28% of the hepatic samples of SGAR were >100 ng/g, the previously stated threshold of acute toxicity. In 4 of these foxes, the residues were >200 ng/g, previously indicated as potentially lethal concentrations. This confirms that residue levels can verify exposure, but AR concentrations alone cannot be used to determine effect on animal health or serve as an indicator of toxicosis.
We collected feces directly from the rectum after death. Other studies have suggested analyzing ARs in scats sampled from the ground, but DNA analyses have detected 18–25% misclassification of presumed fox feces in these studies.10,13 In addition, repeated fecal samples from one individual could skew the results. We therefore suggest collecting feces directly from the animals, also avoiding natural degradation of scats in the environment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded in part by The Norwegian Environment Agency (19S45D8A).
