Abstract
Phlegmonous gastritis was diagnosed in 2 yearling fillies that were presented with a 1-wk history of fever, lethargy, and hypoproteinemia, associated with a previous diagnosis of equine proliferative enteropathy based on clinical signs and PCR assay detection of Lawsonia intracellularis in fecal samples. Abdominal ultrasound revealed enlargement of the stomach and expansion of its submucosal layer with hypoechoic fluid, as well as thickened hypomotile small intestinal segments. Given the poor prognosis and poor response to treatment, both horses were euthanized, one on the day of presentation and the other after 3 wk of intensive medical management including a combination of antimicrobials, analgesics, and intravenous colloids. At autopsy, acute mural gastritis characterized by severe submucosal edema with suppurative inflammation (i.e., phlegmonous gastritis) and necroulcerative enteritis compatible with the necrotizing form of equine proliferative enteropathy were identified in both horses. The gastric inflammation was associated with thrombosis and mixed bacterial populations, including Clostridium perfringens, that were confined to the submucosa without evidence of mucosal involvement; toxin genes compatible with C. perfringens type C were identified in one case. Human phlegmonous gastritis is an uncommon, often-fatal pyogenic infection that is often associated with mucosal injury, bacteremia, or immunocompromise. Our finding of this unusual gastric lesion in 2 horses with similar signalment, clinical disease, and spectrum of postmortem lesions suggests a similar etiopathogenesis that possibly involves local, regional, or distant hematogenous origin, and should be considered a potential complication of gastrointestinal mucosal compromise in horses.
Phlegmonous gastritis (PG) is a rare disease in humans characterized by a severe pyogenic bacterial infection of the gastric wall; the overall mortality rate of 46% ranges from 20% for surgically treated cases to 50% for medically managed cases. 16 In humans, early reports of PG describe marked, regionally circumscribed to diffuse expansion of the gastric wall by edema and suppurative inflammation that primarily and most severely affects the submucosa, and affects the muscularis externa less severely; the mucosa remains mostly intact.3,17,21,24 The expansion of the gastric wall was discerned radiographically in these cases, and was distinguished from emphysematous gastritis (EG) by the lack of gas bubbles and extensive mucosal involvement.19,35 The condition can occur in previously healthy or debilitated patients; predisposing causes include primary gastric mucosal injury (e.g., traumatic or peptic ulcers, gastric neoplasia) in cases of primary PG, distant or systemic infection (e.g., cellulitis, endocarditis) in cases of secondary PG, and immunosuppression (e.g., patients on therapy for cancer or immune-mediated disease) in cases of idiopathic PG.6,12,14,15,29,30
In a retrospective review of 37 cases reported in humans from 1997–2003, the most common presenting symptoms were abdominal pain (95%), nausea and/or vomiting (68%), fever (57%), and hematemesis (14%). 16 The lesions were diffuse or focal and included marked expansion of the gastric submucosa, predominantly by neutrophilic inflammation, with necrosis, hemorrhage, and thrombosis. Microorganisms most frequently isolated were Streptococcus spp. (68%), Enterobacter spp. (18%), Escherichia coli (14%), and Proteus spp. (14%). One-third of infections were polymicrobial, and Clostridium perfringens and Clostridium septicum were isolated in 2 of 22 (9%) cases, with both patients having died within 3 and 14 d of presentation. The case in which C. perfringens was isolated had polymicrobial infection including 4 other bacteria (Staphylococcus spp., Streptococcus spp., Enterobacter spp., Proteus spp.). Although diagnosis of PG can be challenging and, in the past, was often made during exploratory laparotomy or autopsy, the advent of endoscopy, ultrasound, and computed tomography has greatly improved the ability to identify characteristic lesions for more expedient diagnosis and treatment. 30
In horses, primary gastritis is uncommon and most often associated with parasites such as Gasterophilus intestinalis, Trichostrongylus axei, or Draschia megastoma; idiopathic syndromes including eosinophilic enterocolitis or multi-systemic eosinophilic epitheliotropic disease, which are classified as immune-mediated diseases, with components of hypersensitivity-associated inflammation; or secondary to ulcerative processes such as nonsteroidal anti-inflammatory drug (NSAID) toxicosis, ischemia, or direct toxic or caustic insults (e.g., cantharidin, oleander).27,33,34 Reports of bacterial gastritis in horses are even less common and comprise 3 case studies of infections with C. perfringens or C. septicum manifesting as EG7,36 and hemorrhagic gastritis associated with renal failure and hemoglobinuria. 23
Herein, we report 2 equine cases of polymicrobial PG that was concurrent with necroulcerative enteritis associated with Lawsonia intracellularis. PG in both cases had distinct ultrasonographic, gross, and histologic features with intralesional bacteria, which may represent a unique manifestation of septicemia and indicates a poor prognosis.
Two yearling Thoroughbred fillies, identified as cases 1 and 2 according to their temporal sequence of hospitalization, were presented to emergency services of the University of Pennsylvania, School of Veterinary Medicine, New Bolton Center (Kennett Square, PA, USA; Table 1). The fillies arrived from unrelated farms and owners, and were referred because of persistent fever and lethargy (cases 1, 2), and with intermittent colic (case 2). Each filly had been diagnosed with and treated for equine proliferative enteropathy (EPE) by the referring veterinarian on the basis of clinical signs and the identification of L. intracellularis by PCR assays performed on fecal samples submitted to an AAVLD-accredited veterinary diagnostic laboratory service as a component of microbial tests for equine intestinal pathogens, including equine coronavirus, Clostridioides [Clostridium] difficile toxins A and B, Neorickettsia risticii, and Salmonella spp., which were all negative.
Case-by-case comparisons of physical exam findings, hematologic derangements, abdominal ultrasound findings, and postmortem lesions in 2 fillies with phlegmonous gastritis.

= present; – = absent; Cp = Clostridium perfringens; CpC = Clostridium perfringens type C; Gm(+/–) = gram-positive and -negative bacteria; Gm(–) = gram-negative bacteria; Kid = kidney; MS = musculoskeletal; ND = not determined; Pul = pulmonary.
Case 1 was referred with a 2-d history of fever and arrived at the hospital with a temperature of 39.3°C (102.8°F). The filly exhibited mild tachycardia (56 beats/min) and decreased borborygmi in all abdominal quadrants. Palpation per rectum was unremarkable, and the filly had a moderate-to-good appetite. Despite a PCV of 0.35 L/L (35%), the filly was hypoproteinemic with a serum total protein concentration of 25 g/L (2.5 g/dL), hypoalbuminemic with a serum albumin concentration of 11 g/L (1.1 g/dL), and hyperlactatemic with a serum lactate concentration of 4.6 mmol/L. Also noted were mild electrolyte derangements including mild hyponatremia and hypochloremia, as well as mild hyperfibrinogenemia and leukocytosis characterized by lymphocytosis, monocytosis, and neutrophilia. The filly had been treated on the farm with metronidazole, oxytetracycline, flunixin meglumine, probiotics, and electrolytes. Medical therapy initiated at the hospital included twice daily intravenous oxytetracycline infusions, oral rifampin, intravenous balanced electrolyte solution, intravenous non-steroidal and steroidal anti-inflammatories, analgesics, gastroprotectants, probiotics, and an intravenous infusion of equine plasma. The filly initially responded to therapy with transient improvement in serum total protein concentration (40 g/L) but remained persistently mildly to moderately hyperlactatemic and tachycardic, indicative of hypovolemia, and developed mild ventral and distal limb edema. Two and 3 d later, the filly received intravenous colloid fluid therapy and an additional infusion of equine plasma.
On day 5 post-admission, the filly had signs of colic and inappetence. Abdominal ultrasound revealed hypomotile small intestine (duodenum, jejunum, ileum) with moderately thickened walls, up to 7.7 mm. There was marked echoic mucosal thickening of the duodenum with a thickened hypoechoic submucosa and marked echoic submucosal thickening of the jejunum and ileum. Differential diagnoses for the intestinal lesions included proliferative enteropathy caused by L. intracellularis or infiltrative bowel disease, including inflammatory bowel disease and neoplasia. There was generalized enlargement and distension of the stomach that had severely thickened walls up to 4-cm thick, with the upper limit in normal horses reported as 0.75 cm, 26 and prominent rugal folds. The gastric submucosal layer was markedly thickened and hypoechoic (Fig. 1A). The stomach contained a moderate amount of hypoechoic fluid and slightly displaced the ventral lung fields dorsally. Differential diagnoses for the gastric lesions included mural edema as a result of severe prolonged hypoproteinemia, primary gastritis, or gastric ulceration, although an ulcer was not identified.
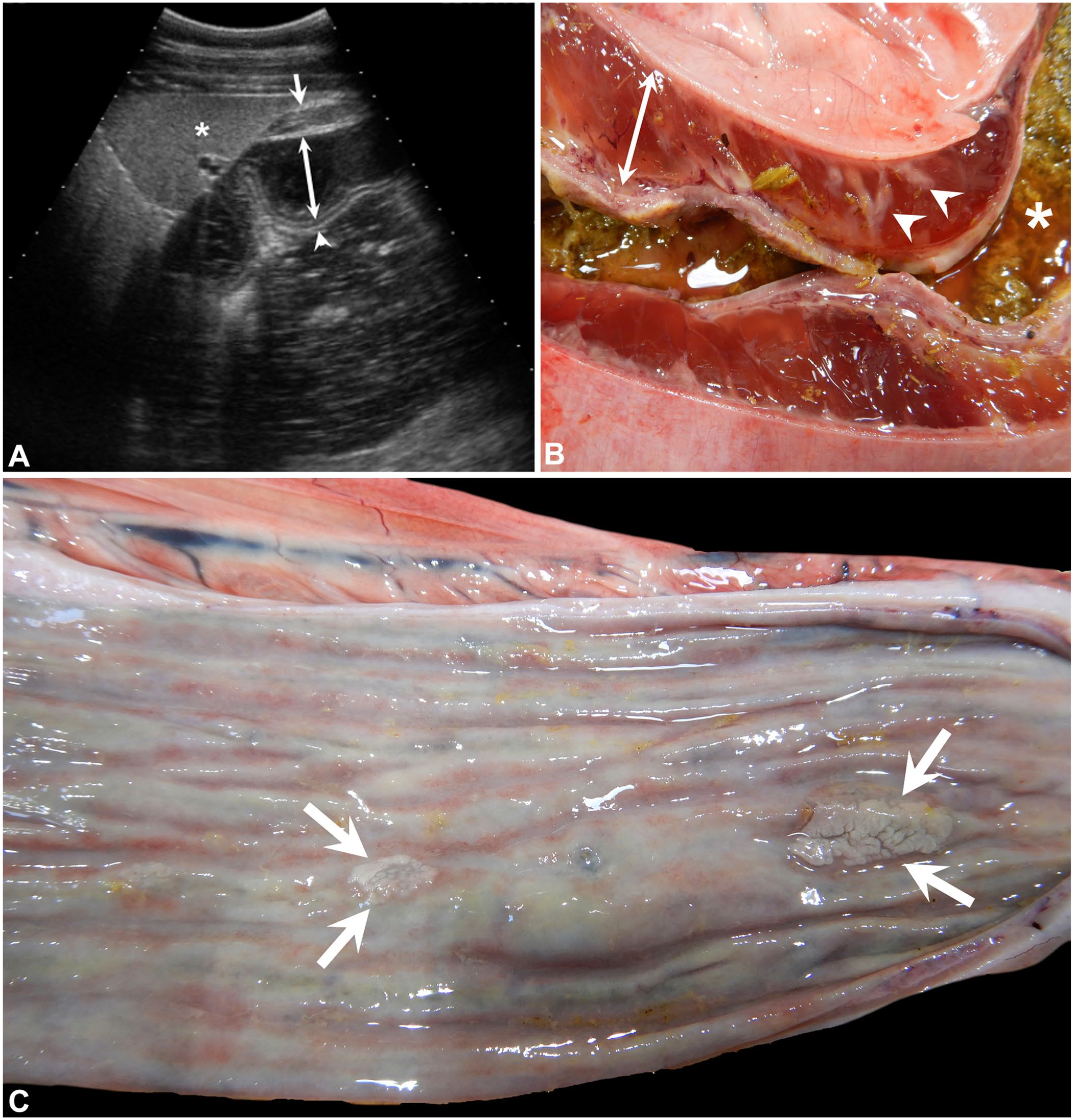
Horses with phlegmonous gastritis.
Given the poor response to therapy, oxytetracycline and rifampin were discontinued and replaced by oral chloramphenicol. Although the filly’s hyperlactatemia improved, her hypoproteinemia persisted, and a third intravenous dose of equine plasma was administered on day 13. On day 16, she developed severe tachycardia and concurrent acute non-weightbearing lameness in the right pelvic limb, with severe edema that was attributed to cellulitis given that pelvic limb radiographs were normal. The filly also developed intermittent rectal prolapse associated with straining to defecate, likely because of pain. Intensive analgesic therapy was instituted, including opiates, as well as nasogastric enteral fluid and nutritional support that produced favorable improvement in pain and appetite; however, hypoproteinemia, hypoalbuminemia, hyponatremia, and hypochloremia persisted, indicating continued enteric losses, and serum creatinine concentration increased to 213 μmol/L (1.9 mg/dL). On day 21, the filly was euthanized after she collapsed abruptly and was minimally responsive to stimuli.
Case 2 had a 1-wk history of persistent fever and lethargy, with intermittent, but progressively more severe, colic signs that were treated on the farm with intravenous oxytetracycline, oral chloramphenicol, and non-steroidal and steroidal anti-inflammatories (flunixin meglumine and dexamethasone). On arrival at emergency services, the filly had dull mentation with a fever of 40.3°C (104.6°F), severe tachycardia (88 beats/min), and mild tachypnea (20 breaths/min). Mucous membranes were dark-pink and injected. The abdomen was moderately distended with reduced borborygmi in all 4 quadrants. A stall-side ultrasound examination revealed findings similar to case 1, including generalized enlargement of the stomach that extended to the 14th/15th intercostal space and was filled with heteroechoic fluid. Small intestinal loops were hypomotile, distended and thickened (4–5-mm thick), and excess peritoneal fluid was identified. However, given the collapse of the patient related to colic, the ultrasonographic exam was truncated prior to assessment of the gastric wall thickness. No net reflux was obtained on nasogastric intubation, and abdominocentesis yielded yellow to pink-tinged cloudy fluid with a PCV of 0.05 L/L (5%), total protein concentration of 14 g/L, and lactate concentration of 12.9 mmol/L. A minimum database revealed a peripheral PCV of 0.66 L/L (66%) with a serum total protein concentration of 38 g/L, and lactate concentration of 4.7 mmol/L. Despite severe hemoconcentration, the serum sodium concentration was slightly low at 131 mmol/L and serum chloride concentration was in the low normal range at 97 mmol/L. Given the increasingly severe colic signs that persisted despite sedation, euthanasia was elected.
Autopsies were performed on both fillies, and similar gross lesions were noted (Table 1). Case 1 had diffusely contracted and thickened small intestinal segments with narrow lumina, scant contents, a thickened, brown granular-to-cobblestoned mucosal surface, and a few 0.5–1-cm diameter red-tan domed serosal plaques. The submucosa of the cecum and right ventral colon was expanded by edema. The squamous mucosa of the stomach had an adherent cluster of botfly larvae consistent with Gasterophilus intestinalis myiasis, and a single chronic oval ulcer with raised yellow-tan margins extending along a region of the margo plicatus. Although the gastric glandular mucosa was intact without evidence of necrosis or emphysema, the submucosa of the glandular portion was expanded up to 2.5-cm thick by coalescing foci of soft bulging translucent-to-opaque tan tissue with occasional dark-yellow foci. The esophageal mucosa had numerous thin linear raised yellow plaques. Additional postmortem lesions included marked edema of the subcutis and myofascial planes of the entire right pelvic limb, with enlarged, edematous inguinal lymph nodes, bilateral wedge-shaped tan foci in the cortices of both kidneys, with additional dry, orange-brown, wedge-shaped regions that extended to the renal crests, and cranioventral consolidation involving 5–10% of the left lung lobe. Given results from the previous microbiologic tests, including L. intracellularis detected in feces submitted for PCR assay, tissue sections were collected for routine histologic evaluation but samples for additional microbiology tests were not submitted.
In case 2, similar to case 1, the gastric wall was severely and diffusely expanded by gelatinous yellow-orange fluid interspersed with semi-opaque gray streaks that followed vascular segments without evidence of emphysema or mucosal necrosis (Fig. 1B). Two botfly larvae were attached to the squamous portion of the stomach; otherwise, the gastric mucosa was intact and grossly normal. Also similar to case 1, small intestinal segments were diffusely contracted with an ecchymotic, injected serosa and scant thick fluid and gas within a few segments. The mucosal surface from the duodenum to the ileum was diffusely gray-green with pale-pink linear streaks, scattered 1.5-cm long slightly raised gray oval foci, and loosely adherent tan-brown friable exudate (Fig. 1C). Within a segment of distal jejunum was a 1.5 cm raised umbilicated fistulous tract extending from the serosa into a 3-mm abscess involving the muscularis externa and serosa; the overlying mucosa was intact but in the process of re-epithelialization. Additional postmortem lesions included mild submucosal edema of the cecum and ventral colon, a moderate cloudy serosanguineous peritoneal effusion, and mild serous pleural effusion. Taking into consideration results from the previous tests, including a positive L. intracellularis fecal PCR assay, and given financial constraints, tissue sections were collected for routine histologic examination and limited microbiologic testing was performed, including a fecal swab submitted for Salmonella PCR assay, which was negative, and a swab of the gastric submucosa, which yielded moderate growth of Escherichia coli.
In both cases 1 and 2, histology of the stomach (Fig. 2A–C) showed severe expansion of the submucosa by edema infiltrated by sheets of neutrophils with multifocal fibrinoid vascular necrosis, thrombosis, and perivascular hemorrhage. The suppurative inflammation extended into and through the muscularis externa to the serosal surface, which was edematous and contained adherent plant material; however, emphysema was not a feature, and the overlying mucosa, which had mild glandular atrophy, remained intact. Sections of stomach that had been stained with Gram stain revealed a mixed bacterial population in case 1, including gram-positive cocci/diplococci with short and filamentous morphologies of gram-negative bacilli (Fig. 3A), and 1–3-µm long gram-negative bacilli in case 2 (Fig. 3B). In both cases, the bacteria were limited to the submucosa, and immunoreactivity for C. perfringens antigen was detected in intracellular and extracellular bacteria within necrotic and inflamed regions of the gastric submucosa (Fig. 3C, 3D); immunoreactivity for C. chauvoei, C. novyi, C. septicum, and C. sordellii antigens was not detected (not shown). PCR assay for C. perfringens genes of alpha (CPA), beta (CPB), epsilon (ETX), iota (ITX), enterotoxin (CPE), and necrotic B-like (NetB) toxins, performed on formalin-fixed, paraffin-embedded scrolls 10 of stomach tissue from case 2, were positive for CPA, CPB, and CPE toxins, compatible with C. perfringens type C. 28 C. perfringens toxin genes were not identified on scrolls of case 1.
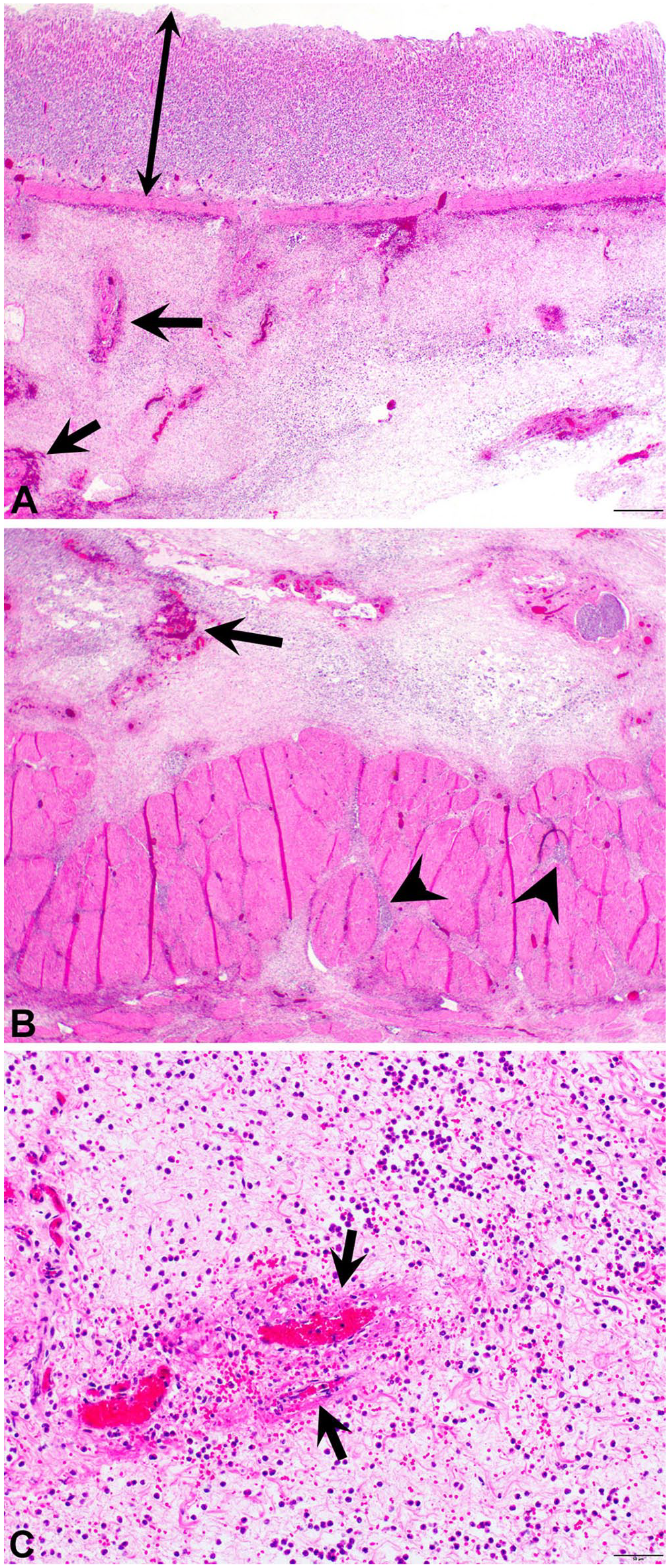
Histopathology of the stomach of a horse with phlegmonous gastritis (case 2).
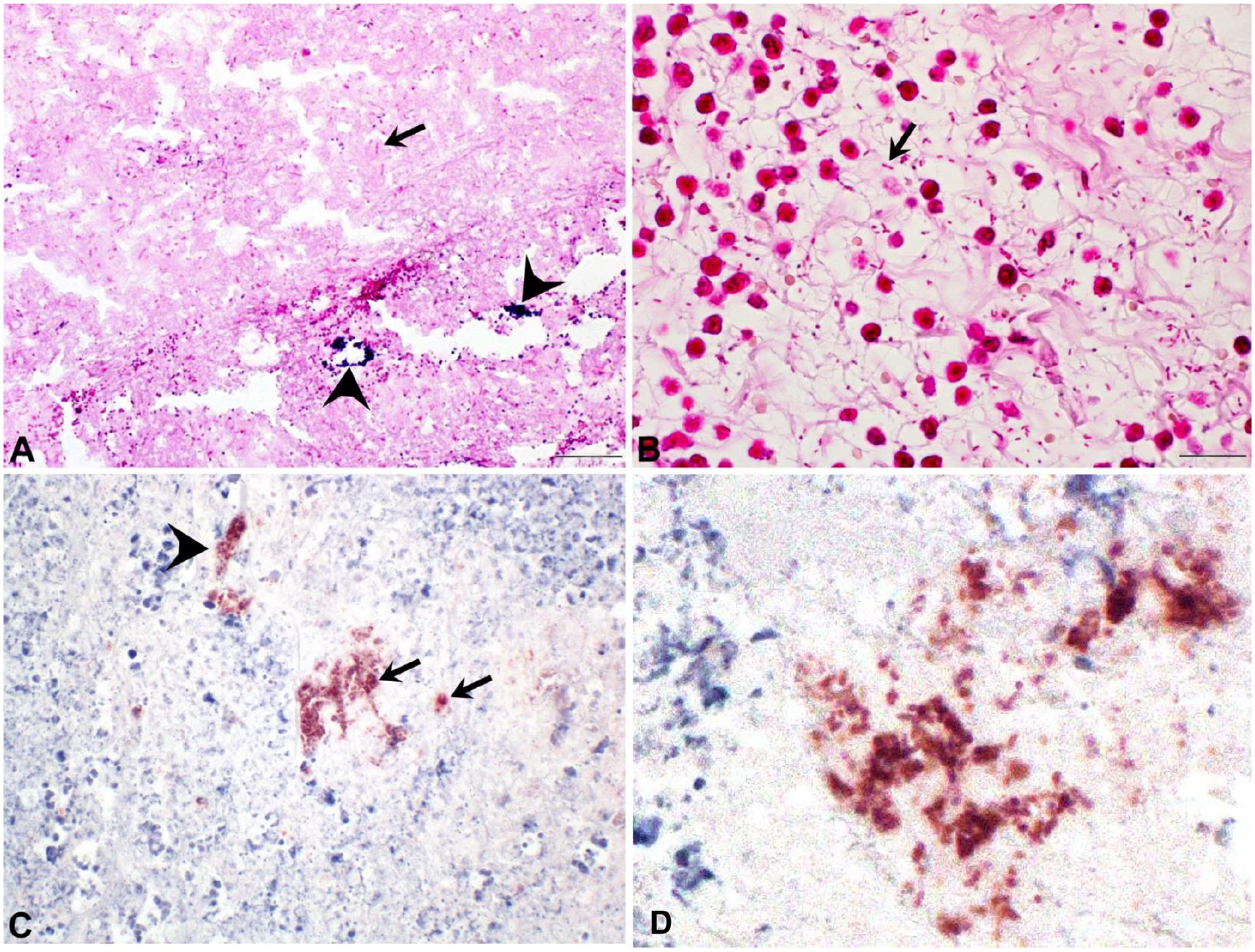
Special stains and immunohistochemistry of horses with phlegmonous gastritis.
Cases 1 and 2 had similar small intestinal histologic lesions (Fig. 4), including multi-segmental necroulcerative enteritis characterized by effacement of the mucosa by organizing granulation tissue with occasional thrombosis and diffuse diphtheritic membranes of fibrin, neutrophils, and cellular debris. Some intestinal segments had villus loss and blunting with epithelial attenuation, crypt necrosis, and dilation. In case 1, there were regions of mucosal hyperplasia characterized by an increased villus-to-crypt ratio (~8:1) with branching crypts and increased mitotic figures, as well as diffuse hypertrophy of the muscularis externa and remaining segments of muscularis mucosae, with expansion of the serosa by foci of maturing granulation tissue mixed with free erythrocytes and hematoidin- and hemosiderin-laden macrophages, akin to hemomelasma ilei. Warthin–Starry stains performed on sections of regenerating-to-hyperplastic intestinal mucosa did not reveal argyrophilic bacteria in case 1 or 2, and immunoreactivity for L. intracellularis antigen was not identified in case 1 or 2. Gram stains applied to intestinal segments revealed a mixed population of scattered gram-positive and gram-negative bacteria mixed with diphtheritic membranes, but immunoreactivity for C. perfringens, C. chauvoei, C. novyi, C. septicum, and C. sordellii antigens was not identified within intestinal samples from case 2, and the same PCR assays for C. perfringens toxins listed above were also negative in case 2 intestinal samples.
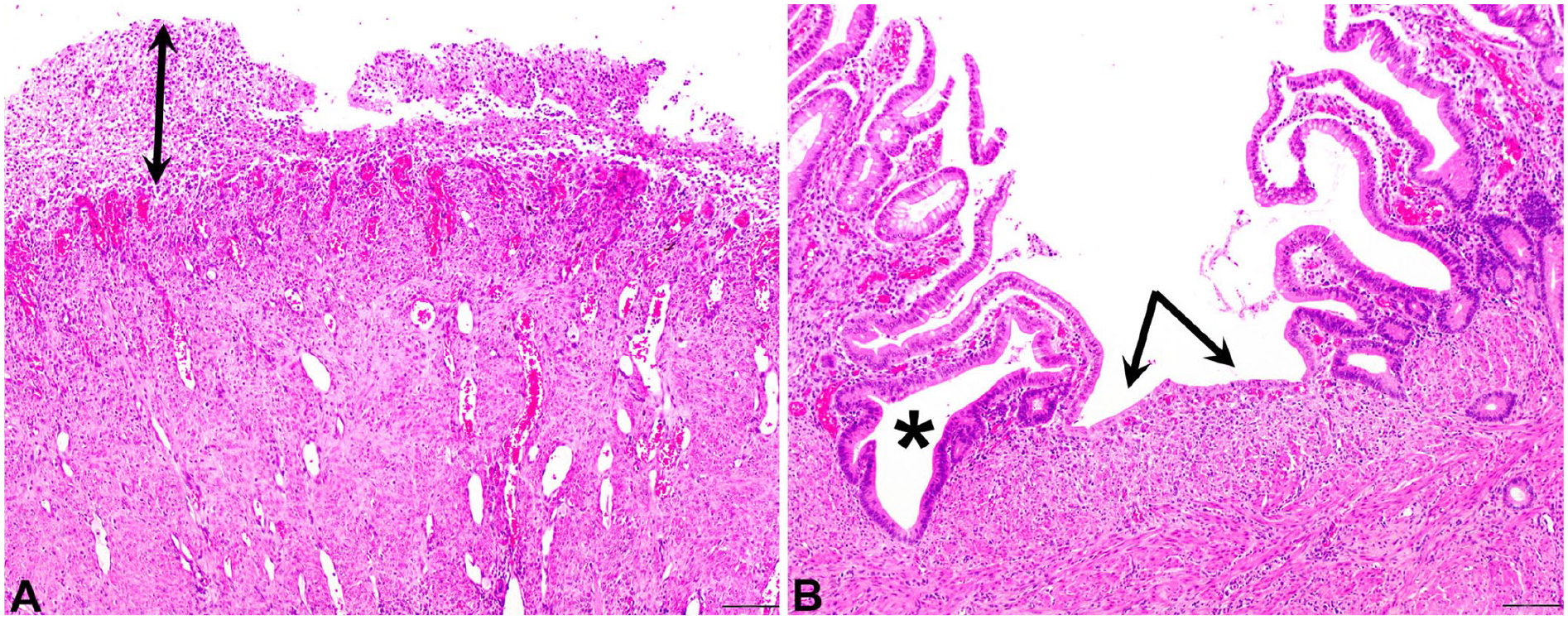
Histopathology of the intestine of a horse with phlegmonous gastritis (case 2).
The submucosal expansion of the cecum and colon in cases 1 and 2 was confirmed to be edema. Additional histologic findings in case 1 included severe suppurative cellulitis of the right pelvic limb with no bacteria identified by Gram stain; esophageal hyperkeratosis, with intracorneal microabscesses and yeast/hyphae compatible with candidiasis; bilateral renal cortical infarcts and renal crest necrosis; and focal suppurative bronchopneumonia with rare bronchiolar pigmented fungal/plant material (presumed aspiration pneumonia).
We have described here 2 cases of polymicrobial PG in which C. perfringens type C was identified by PCR assay (case 2) and C. perfringens (not typed) was detected by immunohistochemistry (cases 1, 2) along with other bacteria identified by Gram stain (cases 1, 2) and aerobic culture (case 2). The lesions of severe diffuse edema and suppurative inflammation with multifocal vascular necrosis were confined to the submucosa and muscularis externa without evidence of emphysema or mucosal involvement. PCR assay detected CPA, CPB, and CPE toxin genes, compatible with C. perfringens type C in the gastric wall of case 2. 28 Although our report presents an unusual lesion in the equine gastric wall that is ultrasonographically detectable in live horses, given the similar gross, histologic, and microbiologic features shared between PG and EG previously reported in horses and other species, it is possible PG and EG may represent morphologic variants of a similar process.
Although CPA is typically responsible for malignant edema and gas gangrene in humans and animals, 32 and its role in equine intestinal disease is controversial, CPA has been identified in a case of hemorrhagic gastritis associated with intravascular hemolysis in a horse. 23 In our cases, there was no hemolysis, and it is unlikely that CPA was involved. CPB produced by C. perfringens type C is a highly necrotizing toxin that causes necrotizing enteritis in newborn foals, whether mediated by direct mucosal or vascular damage. CPB also has lethal effects when circulated systemically and may have contributed to the acute clinical decline in case 2. It is possible that this toxin was at least in part responsible for the necrotizing enteritis observed, although toxin detection, the confirmatory test for C. perfringens type C enteric infection, was not performed in our cases. The localization of C. perfringens type C within the gastric wall of both cases is interesting and unusual. Although Gram stain did not reveal gram-positive bacilli, this staining method has reported variability that could reflect inherent characteristics of the cell wall of this particular population of bacteria, or changes occurring in the cell wall associated with different stages of cell growth and viability. 1 Although postmortem autolysis, which was more advanced in case 1, can reduce PCR assay sensitivity, it is more likely that prolonged formalin fixation, which can increase DNA fragmentation or cross-link cell membrane proteins within the cell wall that can limit exposure to assay reagents, may have impacted the PCR assay in samples from case 1. Our PCR assay did detect the CPE gene within the stomach of horse 2. The role of this toxin in enteric disease of horses has not been elucidated. However, CPE has been detected in feces from 20% of adult horses and 30% of foals with diarrhea, yet is not detected from feces in normal horses,20,32 suggesting that it may play a role in gastrointestinal disease of these animals.
Both of our cases had generalized gastric enlargement that included luminal distension and marked submucosal thickening. Antemortem ultrasonographic examination identified the submucosal thickening in case 1, which had a complete examination of the abdomen in contrast to case 2 in which, out of concern for patient safety, the exam was prematurely terminated. Given the postmortem findings, we suspect that a complete ultrasonographic examination in case 2 would have revealed gastric mural thickening similar to case 1. In both cases, ultrasonographic examination also confirmed concurrent small intestinal disease characterized by thickened, hypomotile small intestinal segments, and postmortem intestinal lesions were compatible with what has been described as the necrotizing form of EPE (N-EPE) associated with L. intracellularis infection,8,22 a syndrome that shares similarities with the spectrum of L. intracellularis intestinal disease in swine. 18 Although at the time of euthanasia, neither case had detectable immunoreactivity for L. intracellularis antigen, nor argyrophilic bacteria identified with Warthin–Starry stain, this is not unexpected considering that both horses had received systemic antimicrobials, including the tetracycline class of drugs, and studies have shown that fecal shedding of L. intracellularis bacteria is not detected by PCR assay after 4 d of antimicrobial initiation. 5 In swine, studies of L. intracellularis fecal PCR assay show consistently high specificity (i.e., 100%) but low sensitivity depending on stage of disease (i.e., 71% in early vs. 38% in later stages of disease), and reports of fecal PCR assays in horses show similar high sensitivity (100%) and lower sensitivity (i.e., 74–79%), especially with disease chronicity.9,25 Detection of L. intracellularis in postmortem samples is also reported to be inconsistent.2,22 Intestinal contents from case 2 were negative for Salmonella spp. by PCR assay after cultural enrichment, and C. perfringens antigen or toxins were not detected in the intestine of case 2 by immunohistochemistry or by PCR assay, respectively. Although characteristic antemortem data and autopsy findings are compatible with what is described for N-EPE caused by L. intracellularis, we cannot determine the potential contribution of a secondary process, such as intestinal dysbiosis associated with antimicrobial therapy, or secondary infection by opportunistic or pathogenic microbes (e.g., C. difficile) that were not discerned.
Case 1 had a longer duration of disease prior to euthanasia (21 d after presentation) compared to case 2 (euthanized the day of presentation) and other horses with clostridial gastritis (1–13 d) reported in the literature.7,23,36 This was despite both horses sharing similar physical examination findings (tachycardia, tachypnea, decreased borborygmi without reflux) and biochemical derangements (hypoproteinemia and hyperlactatemia with relatively low serum sodium and chloride concentrations, suggestive of enteric loss) and may be the result of the intense medical management implemented in case 1 versus case 2. Neither horse had gastric mucosal necrosis7,23,36 or emphysema7,36 as reported with C. perfringens type A and C. septicum gastritis in horses. Thus, the gross and histologic features in our 2 cases were more compatible with PG in humans.13,16,30 It is possible that the lack of mucosal necrosis or emphysema in our 2 cases may be an artifact of postmortem sampling and that other sections may have contained these features.
A retrospective cohort study of 23 calves with abomasitis shares clinical signs and biochemical derangements similar to the 2 horses in our report, including tachycardia, tachypnea, pyrexia, hyperlactatemia, hypoproteinemia, hypochloremia, and hyponatremia. 11 In 11 of 12 deceased calves that had postmortem examinations performed, 10 of 11 had lesions involving the submucosa and 7 of 8 had concurrent intestinal disease. Although a variety of gram-positive and gram-negative bacteria were identified within the abomasal wall in 7 of 11 cases, microbiology identified concurrent infection by E. coli and C. perfringens in 7 of 8 postmortem bacterial cultures. Although there are similarities between this report of calf abomasitis and our 2 cases, mainly the presence of concurrent intestinal disease and identification of mixed bacteria in the gastric wall including C. perfringens (case 1 and 2) and E. coli (case 2), differences in the calf study include mucosal involvement in 9 of 11 cases and emphysema in 6 of 11 cases. Similarly, polymicrobial gastritis with and without mucosal involvement is also reported in dogs 31 and cats,4,31 and although these canine and feline cases also have overlapping features with PG, including expansion and inflammation of the wall by edema and inflammation with polymicrobial infection including Staphylococcus spp., Enterococcus spp., E. coli, and C. perfringens type A, mucosal and mural emphysema was the defining gross feature of lesions in these canine and feline cases.
Therefore, although the gastric lesions presented here, including ultrasonographic, gross, and histologic features, as well as polymicrobial infection that included C. perfringens and E. coli, are most compatible with PG described in humans, there are some common overlapping features of Clostridium-associated gastritis reported in veterinary species, especially morphologic features reported in the case of equine C. perfringens–associated hemorrhagic gastritis, renal failure, and hemoglobinuria 23 that make it difficult to determine whether equine PG could be a pathologic process related to or disparate from EG. However, it important to recognize that despite ultrasonographic and gross appearances that suggested mural edema, the gastric lesions in these 2 horses were inflammatory and localized to the submucosa and outer muscularis with an infectious component including C. perfringens and other bacteria. Although the gastric lesions coincided with intestinal disease compatible with N-EPE associated with L. intracellularis infection, whether equine PG represents a nonspecific manifestation of regional or distant septicemia, as evidenced by the later development of hindlimb cellulitis and renal infarcts in case 1, or results from direct inoculation through an unidentified focus of compromised gastric mucosa, remains speculative. Moreover, the contribution of toxigenic C. perfringens bacteria to either N-EPE or PG has yet to be determined. Based on human reports, immunosuppression could also contribute to the pathogenesis of equine PG. A better understanding of their etiopathogenesis could be provided by further histologic and microbiologic investigation of similar lesions across veterinary species, including metagenomic and molecular phenotypic analyses to determine the mechanistic contribution of microbial populations within inflammatory gastric lesions.
PG should be considered a life-threatening condition in horses that can be identified ultrasonographically antemortem and has distinct gross and histologic features. Additional studies of PG and EG in horses are needed to determine whether these lesions represent different manifestations of the same condition, and whether aggressive medical management can be successful in resolving clinical signs and lesions.
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
