Abstract
A 9-y-old, spayed female rabbit was presented for evaluation of hypoglycemia and lateral recumbency. The patient was hypothermic and had diffuse muscle wasting; weight loss since a previous visit was also noted. Hematologic abnormalities included progressive nonregenerative anemia and severe heteropenia. Evaluation of a bone marrow aspirate sample revealed active hematopoiesis with abundant pink matrix. The matrix material stained positively with periodic acid–Schiff and alcian blue, and a diagnosis of gelatinous transformation of the bone marrow (GTBM, serous atrophy of fat) was made. Although its precise prevalence remains to be determined, GTBM should be suspected in rabbits with persistent cytopenias following prolonged starvation or gastrointestinal disease.
Gelatinous transformation of the bone marrow (GTBM), also termed starvation marrow or serous atrophy of fat, is the replacement of normal hematopoietic tissue with glycosaminoglycans (e.g., hyaluronic acid).1,15 GTBM has been reported in cattle, 22 cats,12,20 minipigs, 4 a miniature horse, 2 and reindeer. 11 In a study conducted in the 1970s, the bone marrow of 3 rabbits subjected to starvation had similar abnormalities to those previously described in humans with GTBM and in our case. 19
The pathogenesis of GTBM is unknown, but it appears to be related to activation of bioregulatory responses in severe illness involving fat mobilization.3,16,18 Disorders most commonly reported with GTBM in people are anorexia nervosa, malnutrition and malabsorption, and other diseases resulting in marked weight loss and anorexia (e.g., malignant neoplasia, diabetes mellitus). 17 The subsequent deposition of glycosaminoglycans in bone marrow interferes with the normal hematopoietic microenvironment, resulting in decreased production and peripheral cytopenias.5,13
Previous studies conducted with humans and rabbits indicate that, although prognosis is guarded, GTBM is reversible if the underlying etiology is eliminated.9,10,14,18,19 Nutritional management is the main therapeutic goal, but red blood cell transfusion, hormone replacement, and therapy with colony-stimulating factors have also been reported as beneficial.5,14,17 We detail here the diagnosis and successful treatment of GTBM in a domestic rabbit.
A 9-y-old, spayed female Holland Lop rabbit (Oryctolagus cuniculus) was referred to the William R. Pritchard Veterinary Medical Teaching Hospital (VMTH) of the University of California–Davis for further evaluation following a severe hypoglycemic episode. The rabbit had been presented initially to the referring veterinarian because of acute onset of lateral recumbency following a 1.5-wk history of decreased fecal production with soft feces. Severe hypoglycemia, bradycardia, hypothermia, and anemia were observed at the referral visit. The rabbit was provided with supportive care (intravenous fluids with dextrose, heat) and transferred to the VMTH within 36 h.
The patient’s medical history included severe right-sided otitis externa and media with associated facial nerve deficits, treated with partial ear canal ablation and bulla osteotomy 3 y earlier. Intermittent corneal ulceration of the right eye persisted following surgery, presumably secondary to facial nerve paralysis. Right-sided head tilt and ataxia, with possible seizure-like episodes, developed one year following surgery. A computed tomography (CT) scan showed no significant changes to the middle ear. Additional history included bilateral pelvic limb pododermatitis, mild chronic acquired dental disease, and mild progressive rhinitis. The patient was not receiving any medications at the time of the current visit.
Upon presentation to the VMTH, the patient was quiet, alert, responsive, and ambulatory, but hypothermic (36.7°C), moderately dehydrated, and its body condition was emaciated (body condition score 2 of 9, with 5 being ideal). The weight had decreased by 23% (0.28 kg) since the last VMTH visit 3 y prior (1.20 kg then, vs. 0.92 kg upon presentation). The gastrointestinal (GI) tract contents were decreased on palpation, and borborygmi were reduced. Grade I/VI pododermatitis was present on both hind feet.
Neurologic examination revealed a postural slope toward the right when the patient was sitting, and right-sided facial nerve paralysis. Other neurologic parameters were within normal limits.
Blood samples obtained by lateral saphenous venipuncture were placed into EDTA and lithium heparin tubes (Microtainer; Becton Dickinson) for complete blood count (CBC) and plasma chemistry analysis, respectively. An Advia 120 hematology analyzer (Siemens) was used for automated cell counts. A blood smear was stained with Wright–Giemsa (Millipore Sigma) for a 200-cell differential count and morphology assessment. Total protein concentration was determined via refractometry (REC-300ATC; Tekcoplus). Chemistry analyses were performed on an automated analyzer (Cobas 6000 C501; Roche).
CBC results at presentation included mild normocytic, normochromic anemia, mild heteropenia, and moderate lymphopenia (Table 1). Polychromasia was absent, and a reticulocyte count on day 5 confirmed nonregenerative anemia. Clinical chemistry abnormalities included marked hypoglycemia (3.4 mmol/L, reference interval [RI]: 6.0–10.9 mmol/L), moderate hyponatremia (130 mmol/L, RI: 140–148 mmol/L), mild hypochloremia (91 mmol/L, RI: 96–109 mmol/L), mild hypercholesterolemia (2.54 mmol/L, RI: 0.36–2.15 mmol/L), moderate hypotriglyceridemia (0.18 mmol/L, RI: 0.45–1.10 mmol/L), decreased creatinine concentration (38 µmol/L, RI: 53–114 µmol/L), and increased creatine kinase (CK) activity (118 U/L, RI: 0–16 U/L) activity. Total protein concentration was at the lower reference limit (59 g/L, RI: 59–83 g/L); albumin was not measured because of an invalid protocol assay for the species and, thus, a value for globulins was also not obtained. Results for potassium, bicarbonate, phosphorus, calcium, urea, bilirubin, and magnesium concentrations, and alanine transaminase, aspartate aminotransferase, alkaline phosphatase, and gamma-glutamyl transferase activities were within RIs.
Complete blood count results for a 9-y-old Holland Lop rabbit from presentation (day 1) to 2 mo after discharge on day 13.
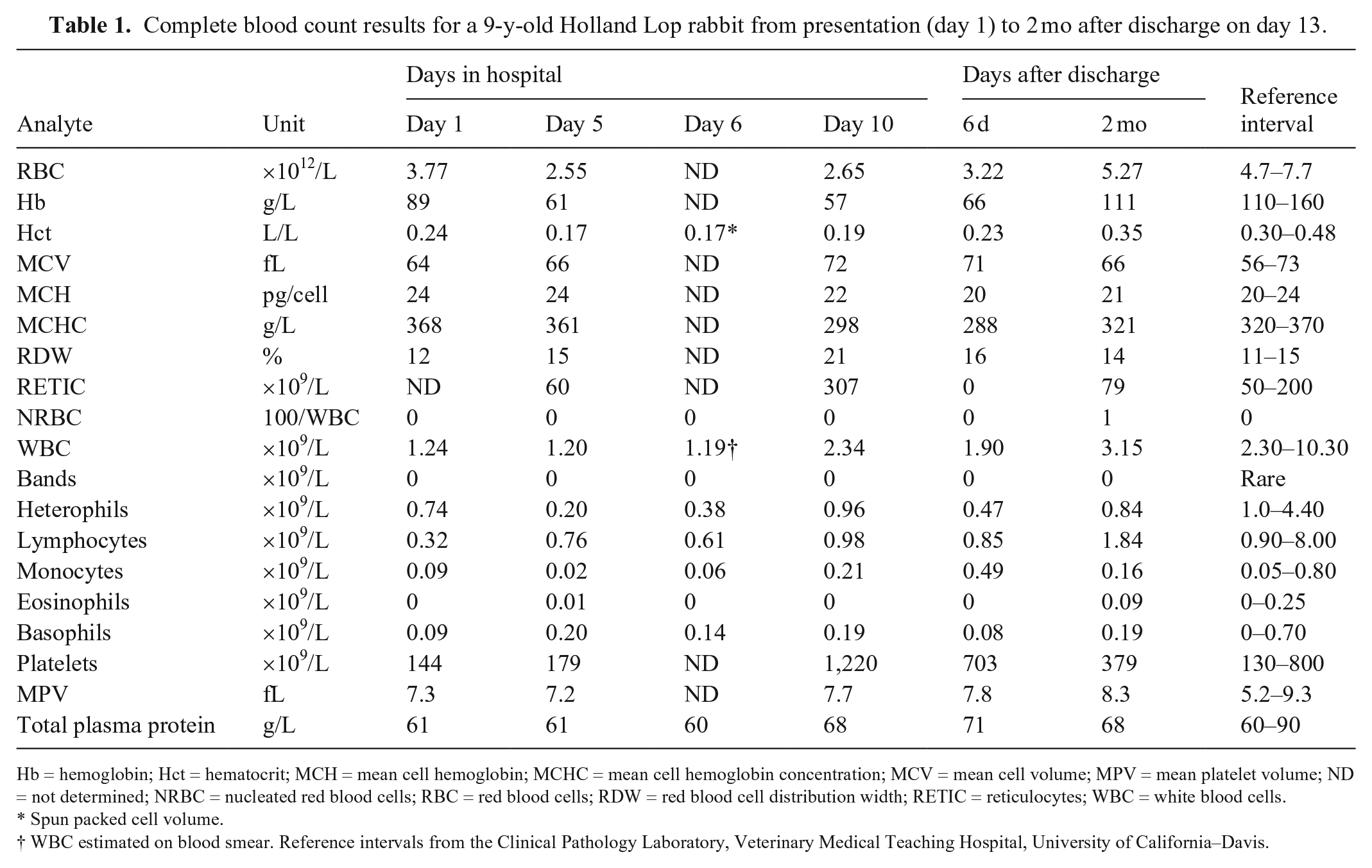
Hb = hemoglobin; Hct = hematocrit; MCH = mean cell hemoglobin; MCHC = mean cell hemoglobin concentration; MCV = mean cell volume; MPV = mean platelet volume; ND = not determined; NRBC = nucleated red blood cells; RBC = red blood cells; RDW = red blood cell distribution width; RETIC = reticulocytes; WBC = white blood cells.
Spun packed cell volume.
WBC estimated on blood smear. Reference intervals from the Clinical Pathology Laboratory, Veterinary Medical Teaching Hospital, University of California–Davis.
Abdominal ultrasonography revealed diffuse nonspecific hepatopathy, cholecystic debris in the gall bladder, and focal intrahepatic biliary mineralization. Abdominal radiographs showed a gas-filled cecum. Skull radiographic findings were consistent with the historical asymmetric thickening of the right tympanic bulla and cheek tooth malocclusion. Thoracic radiographs showed mild generalized rounding and enlargement of the cardiac silhouette, and a mild bronchial pattern suggestive of airway inflammation. Echocardiography revealed generalized cardiac enlargement, hyperechoic left ventricular endomyocardium consistent with ischemia or infiltrative disease, and a thickened aortic valve. Imaging modalities also showed mild peritoneal, pleural, pericardial, and retroperitoneal effusions that were attributed to fluid overload; the effusions resolved 2 d later. Results of a CT scan of the skull showed static abnormalities of the right tympanic bulla and small points on the premolars and molars.
Fecal flotation and direct fecal examination were negative for endoparasites, but a fecal occult blood test was positive, confirming GI bleeding. Given that the rabbit was not fed anything meat-derived, results were interpreted as true positive. Preliminary differentials for the recumbency and hypoglycemia included starvation, GI malabsorptive disease, and sepsis; the skull CT scan showed no signs of osteomyelitis of the bullae. GI ulceration with translocation or aortic valve endocarditis were considered possible niduses of infection.
Following admission, the patient was treated with supplemental oxygen and intravenous crystalloid fluid (70 mL/kg/d) with 2.5% dextrose. Following diagnoses of cardiomegaly (day 2) and tri-cavitary effusion (day 4), fluids were decreased to 27 mL/kg/d and then 13 mL/kg/d, respectively, for dextrose administration only, which was discontinued on day 5. Intravenous potassium phosphate supplementation was added on day 5 to correct hypophosphatemia. Over the subsequent 3 d of hospitalization, parenteral antimicrobials (enrofloxacin, penicillin), parenteral vitamins (phytonadione, cyanocobalamin, B complex), gastroprotectants (oral sucralfate, parenteral pantoprazole), and oral tramadol were initiated.
The patient was hospitalized for 13 d. On day 5, the anemia worsened (hematocrit [Hct] 0.17 L/L) with no reticulocyte response (Table 1), and heterophils decreased to 0.2 × 109 cells/L; the rabbit was given a whole blood transfusion. On day 10, the Hct had increased to 0.19 L/L with reticulocytosis (307 × 109 cells/L), and heterophils had increased to 0.96 × 109 cells/L. Marked hypophosphatemia (0.16 mmol/L, RI: 0.55–1.61 mmol/L) was observed on day 4 but returned to within the RI (0.65 mmol/L) the day after initiation of supplemental phosphate.
On day 11, bone marrow was aspirated from the proximal right humerus using a 25G × 2.5 cm needle and a 20-mL syringe coated with 1.8 mg/mL of EDTA. The marrow specimen was placed in a petri dish and particles collected using a microhematocrit tube for preparation of direct smears, which were stained with Wright–Giemsa. On microscopic examination, numerous normocellular unit particles were observed (estimated at 30–40% cellularity, RI: 30–50% cellularity; Fig. 1A). A large amount of amorphous, smooth-to-fibrillar, bright-pink matrix was found between spicules and among the cells (Fig. 1A, 1B). Megakaryocytes appeared adequate and were primarily mature. The estimated granulocytic:erythroid ratio was 1.0 (RI: 0.4–1.4); both the granulocytic and erythroid series were complete and normally distributed. Numerous small adipocytes with signet-ring morphology were observed (Fig. 1B), as well as a few plasma cells and several hemosiderin-laden macrophages. The cytologic diagnosis was active hematopoiesis with abundant pink matrix. Primary differentials for the matrix were mucin, other glycoproteins, and collagen.
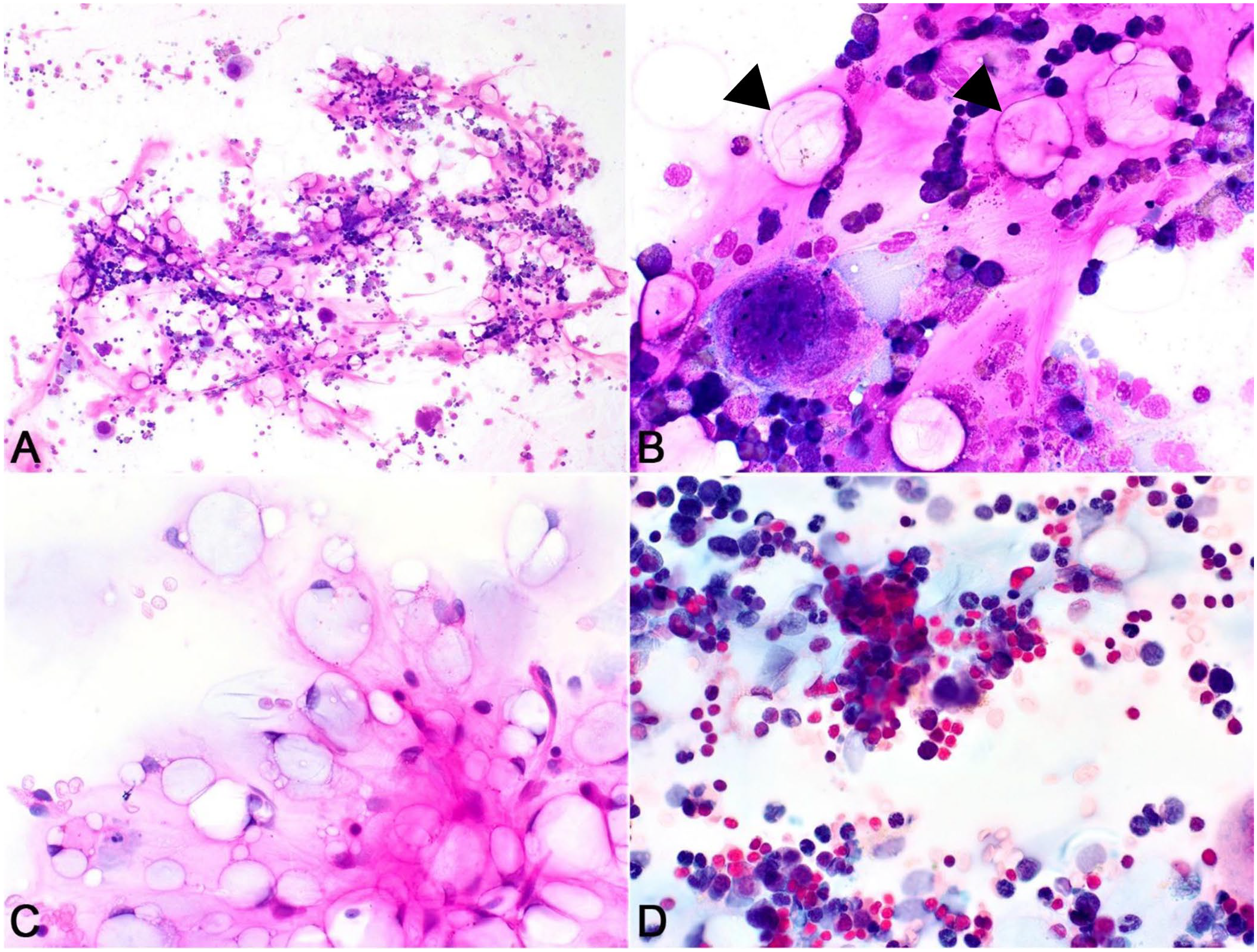
Bone marrow aspirate from a 9-y-old rabbit with gelatinous transformation of the bone marrow. Wright–Giemsa stain.
Cytochemical stains (alcian blue, periodic acid–Schiff [PAS]) were applied to unstained bone marrow aspirate smears using standard methods. 1 The matrix stained strongly positive with PAS (Fig. 1C) and weakly positive with alcian blue (Fig. 1D), confirming a glycoprotein composition. A diagnosis of GTBM was made.
The patient was discharged on day 13 weighing 1.1 kg (19% increase from presentation). At home, the patient received timothy and alfalfa hay, leafy greens and carrots, and apples as treats, as well as an assist feeding formula (Critical Care, 45 mL/kg/d; Oxbow Animal Health). Antimicrobials, sucralfate, tramadol, and vitamin supplementation were maintained as in hospital; pantoprazole was transitioned to oral omeprazole.
The patient was rechecked 6 d after discharge. The heterophil count had decreased to 0.47 × 109 cells/L, and the Hct and platelet count had increased to 0.23 L/L and 703 × 109 cells/L, respectively. The patient was normothermic (38.2°C), and no other laboratory or clinical abnormalities were noted except for residual poor body condition.
Upon recheck 2 mo later, the owner reported that the rabbit had improved energy and had gained weight. On physical examination, the body condition score was still slightly below normal (3 of 9), and facial paralysis and mild pododermatitis persisted. The body weight was 1.4 kg (27% gain compared to discharge, and similar to the elective visit 3 y prior of 1.3 kg). Persistent mild heteropenia (0.84 × 109 cells/L) was the only laboratory abnormality.
Diagnosis of GTBM is based on hematologic findings and medical history and is confirmed by microscopic evaluation of a bone marrow aspirate or biopsy.15,19 Hematologic abnormalities usually include nonregenerative anemia, with or without other cytopenias, and normal-to-hypocellular marrow, with hypoplasia of one or more cell lineages. 10 Abundant amorphous pink-to-purple mucinous material associated with marrow spicules that reacts positively with alcian blue or PAS confirms the presence of glycosaminoglycans (e.g., hyaluronic acid).3,6 Signet-ring cells, reported to be atrophic adipocytes, are also typically observed. 8 These expected findings are consistent with those observed in our case. Although the marrow in this rabbit was normocellular, it was obtained 11 d after presentation and after normalization of blood glucose levels. Despite normocellularity, cytopenias were observed, and the subsequent reticulocytosis and increasing heterophil count indicated a regenerative response, likely in response to an earlier decrease in marrow production. The mild heteropenia of 0.84 × 109 cells/L at 2 mo after discharge may have been normal for this rabbit, given that a heterophil count of 0.82 × 109 cells/L was reported during an elective visit 3 y earlier.
Differential diagnoses for GTBM include marrow necrosis, marrow edema, amyloidosis, and aplastic anemia. We did not observe karyorrhectic debris, as would be expected in necrosis, and hematopoietic activity ruled out aplastic anemia. We did not completely rule out amyloid, which also stains positively with alcian blue and PAS; however, amyloid is usually more homogeneous, associated with blood vessels and numerous plasma cells, and unlikely to be limited to the bone marrow (other findings did not support systemic amyloidosis). Stains for collagen were not done, but the relatively rapid clinical and hematologic resolution did not support myelofibrosis. A limitation of our case is the lack of a bone marrow core biopsy and additional special stains. However, the literature suggests that a final diagnosis of GTBM can be reached based on cytologic findings and a positive reaction for PAS with or without alcian blue.3,17
The GTBM in our case was likely the result of the rabbit’s poor body condition, malnutrition, and history of anorexia, with resultant hypoglycemia. Even though the owner initially reported a normal appetite, the rabbit had lost 23% of its body weight compared to its last visit and was producing less feces; thus, we suspect the owner did not realize the animal was not eating. Further, rapid development of marked hypophosphatemia after therapy with dextrose is the hallmark of refeeding syndrome. It is unclear whether another underlying condition may have led to anorexia. Dental disease has been reported as a predisposing factor for GTBM among ruminants 21 and may have been a factor in the rabbit in this case. The facial paralysis, if affecting deeper tissues, could have compromised mastication or deglutition. Diarrhea and GI bleeding also raised the possibility of malabsorptive disease, which could cause weight loss in the face of a normal appetite.
Even though the erythrocytic indices were not consistent with iron deficiency (i.e., microcytic hypochromic), and total protein was not low enough to support acute hemorrhage, the history of GI bleeding and eventual marrow regeneration could suggest that the anemia was multifactorial. If that were the case, spontaneous hemorrhage could have been a consequence of the GTBM, through a primary hemostatic disorder caused by hypoplastic thrombocytopenia. Although platelet numbers were never below the RI we used, thrombocytopenia may have occurred prior to presentation. It is also possible that the patient had a concurrent functional thrombopathy; however, further coagulation analyses were not pursued.
Low creatinine concentration was consistent with a lower muscle mass, and low triglycerides and protein were likely secondary to decreased feed intake. Increased CK was consistent with multiple venipuncture and capillary sticks to monitor glycemic levels but, in cats, has also been attributed to skeletal muscle breakdown to provide amino acids for energy metabolism following hypoglycemia. 7
Pododermatitis did not appear to be directly linked to GTBM in this rabbit. Facial paralysis could have been secondary to otitis. An autopsy would be necessary to fully characterize the full range of disease entities in this rabbit. However, at the time of writing, the patient was alive and doing well.
Footnotes
Acknowledgements
We thank Dr. Michael Kent for his assistance with bone marrow collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
