Abstract
Mesotheliomas are uncommon neoplasms that arise from mesothelial cells in either the abdominal or thoracic cavities and are rarely diagnosed in cats. A 10-y-old spayed female domestic shorthair cat was presented to the Louisiana State University oncology service for evaluation of a large amount of abdominal effusion. Abdominal ultrasound identified a large mesenteric mass with numerous ill-defined nodules. An abdominocentesis was performed with cytologic and immunocytochemical findings consistent with a neoplastic effusion, with large clusters of epithelioid cells that exhibited strong cytoplasmic expression of pancytokeratin, vimentin, and Wilms tumor 1 antigens. Further testing was declined, and meloxicam was prescribed until the cat died 23 d after initial presentation. Upon postmortem examination, the omentum was contracted into a firm mass adhered to multiple organs and accompanied by numerous small white nodules throughout the abdominal cavity. On histopathology and immunohistochemistry, neoplastic cells were found throughout the abdominal cavity; 60–95% exhibited moderate-to-strong cytoplasmic immunoreactivity for cytokeratin, vimentin, and Wilms tumor 1 protein. The final diagnosis was an epithelioid mesothelioma. Our case illustrates the utility of cytology, immunocytochemistry, and its relation to histology and immunohistochemistry. We also reviewed the reported cases of feline mesothelioma.
Mesotheliomas are neoplasms of mesothelial cells, which line the abdominal and thoracic cavities and cover the organs within these cavities. Up to 90% of human mesothelioma cases have been associated with asbestos exposure, which is supported by experimental animal models. 19 Although some canine mesothelioma cases have also been associated with asbestos exposure, the majority of cases in domestic animals are considered to be spontaneous; ferruginous bodies (asbestos fibers coated with ferroproteins) on histopathology have never been reported with mesothelioma in cats, to our knowledge. 11 Mesotheliomas can be challenging to diagnose antemortem given the inherent difficulty in differentiating a neoplastic population from reactive mesothelial hyperplasia on cytomorphology alone. Additionally, mesothelioma cells frequently express multiple cytokeratins and vimentin, mimicking the characteristics of normal and reactive mesothelial cells. 11 Thus, the diagnosis often relies on the presence of disseminated nodules and histologic evidence of tissue invasion. We highlight a case of feline mesothelioma with a focus on comparison of the cytologic, histologic, and immunochemical staining of the tumor, and review the limited literature on mesotheliomas in cats.
A 10-y-old spayed female domestic shorthair cat was presented to the Oncology service at the Louisiana State University (LSU) Veterinary Teaching Hospital (Baton Rouge, LA, USA) for evaluation of a large volume of abdominal effusion. Weight gain was noted by the owner 2 wk prior to original presentation to her primary veterinarian. She was maintaining a normal appetite and thirst but had increasing abdominal distension. Her primary veterinarian noted a tight abdomen and performed a CBC and serum biochemistry, neither of which revealed any clinically significant findings. Abdominal radiographs showed loss of serosal detail and a large amount of peritoneal effusion with nephroliths in the left kidney. Approximately 600 mL of effusion was removed from the abdomen, and she was referred to LSU for further testing.
On presentation 3 d later, a palpable abdominal fluid wave and a mid-abdominal mass were found. Abdominal ultrasound was performed, which revealed a moderate-to-large amount of echogenic free fluid and a hyperechoic and clumped mesentery with multiple, small, ill-defined, hypoechoic nodules. Hydronephrosis of the right kidney was present, along with nephroliths in the left kidney.
Peritoneal fluid analysis indicated an automated nucleated cell concentration of 4.7 × 109/L (4,700/µL) and a protein concentration of 39 g/L (3.9 g/dL). On both direct and cytocentrifuged preparations, the nucleated cells were ~60% nondegenerate neutrophils with ~30% round-to-polygonal epithelioid cells, ~10% macrophages, and rare small lymphocytes. The epithelioid cells were arranged individually and in clusters, the latter of which were somewhat concentrated along the feathered edge of the direct smears. Their clustering likely led to underrepresentation of the nucleated cell concentration. The epithelioid cells displayed moderate anisokaryosis and anisocytosis and occasional binucleation. The nuclei were round, central-to-paracentral, 10–17 μm diameter, and contained coarsely to finely stippled chromatin with 1–3 variably sized round nucleoli. Cells had moderate-to-abundant amounts of cytoplasm with deep basophilia, dispersed fine clear vacuolation, occasional blebbing, and rare neutrophilic emperipolesis (Fig. 1). Neoplastic effusion, either carcinomatosis or mesothelioma, was suspected, with mild neutrophilic inflammation. However, reactive mesothelial hyperplasia could not be excluded entirely. Immunocytochemistry was performed, as described previously, 5 and ~95% of the epithelioid cells exhibited strong cytoplasmic labeling with pancytokeratin (AE1/AE3, 1:50; Dako), vimentin (V9, 1:50; Dako), and Wilms tumor 1 protein (WT1; 6F-H2, 1:1; Cell Marque) indicating mesothelial origin (Fig. 1, inset A–C).
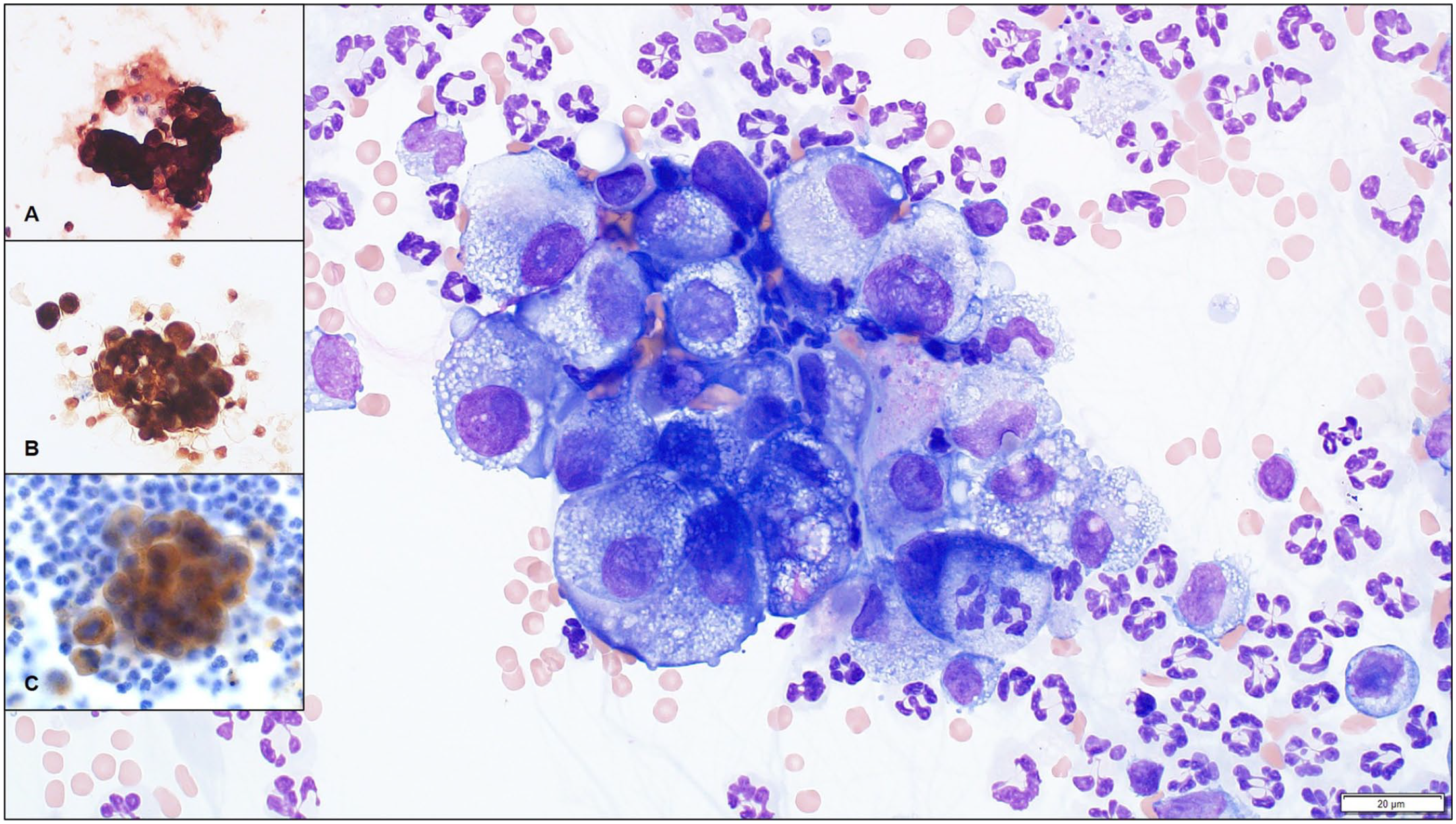
Cytocentrifuged preparation of peritoneal fluid from a 10-y-old domestic shorthair cat with mesothelioma. Several neutrophils and a cluster of atypical epithelioid cells exhibit strong cytoplasmic expression of cytokeratin, vimentin, and Wilms tumor 1 protein on immunocytochemistry, suggesting mesothelial lineage. Wright–Giemsa; 60× objective. Bar = 20 μm. Insets:
Further testing was declined, and meloxicam treatment (0.05 mg/kg PO q24h for 7 d, then 0.025 mg/kg PO q24h; Metacam; Boehringer Ingelheim) was initiated. A therapeutic abdominocentesis was performed by the primary veterinarian approximately once per week until the cat died at home 23 d after initial presentation to her primary veterinarian.
A postmortem examination was performed. The abdominal cavity was filled with a large amount of serosanguineous fluid, and the omentum was contracted into a 10 × 5 × 3 cm firm, white, multinodular mass adhered to the spleen, stomach, liver, and right kidney (Fig. 2). This kidney had a white, irregular surface and adhered to the liver and the dorsal peritoneal wall. On cut surface, the renal architecture was completely lost and replaced by pale-yellow to white coalescing nodules with small cavitations containing scant cloudy white fluid. The left kidney, which retained the normal architecture, contained multiple nephroliths. The liver had a smooth light-tan to red capsular surface with an accentuated lobular pattern.
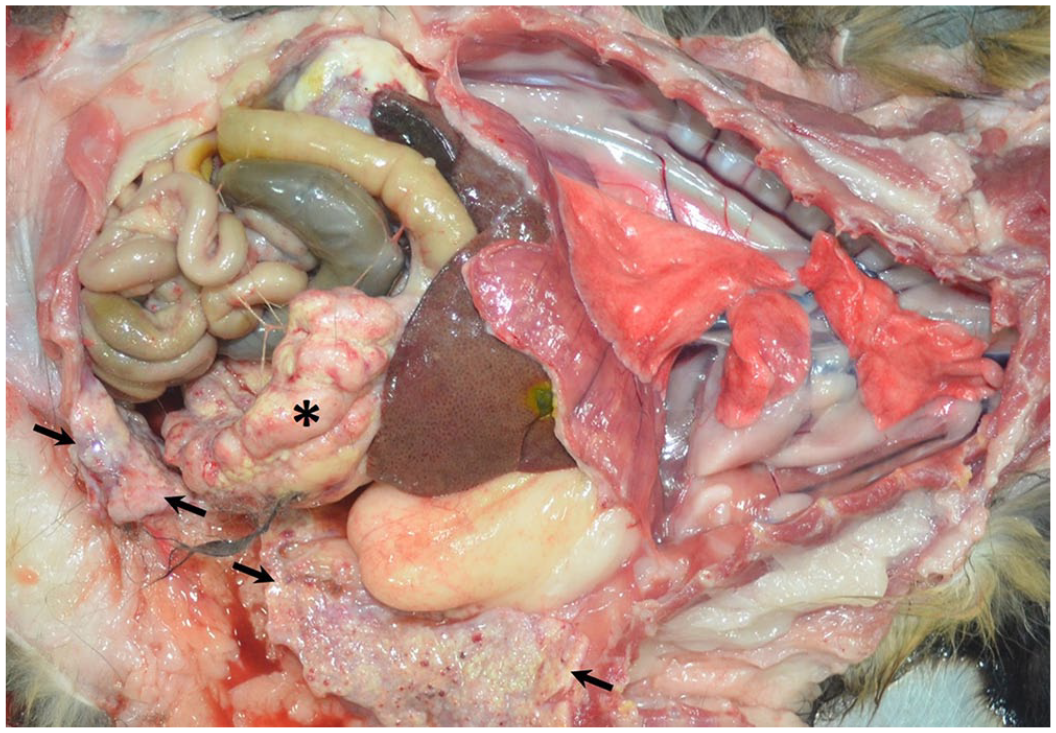
Peritoneal mesothelioma in a cat. The omentum is contracted into a firm, white, multinodular mass (asterisk). The parietal peritoneal surface is extensively coated with multifocal-to-coalescing firm, white, slightly raised nodules (arrows).
Histologically, the omentum and the serosal surfaces of the diaphragm, abdominal wall, small intestine, and urinary bladder were variably effaced by irregular, nonencapsulated, poorly delimited neoplastic nodules, with extensive central necrosis. The neoplasm was composed of predominantly polygonal cells arranged in nests and tubular and micropapillary structures sustained by variable (often abundant) fibrous stroma (Fig. 3A). Neoplastic cells had moderate-to-abundant eosinophilic cytoplasm and round or oval nuclei with finely stippled-to-open chromatin and often a large nucleolus. Anisocytosis and anisokaryosis were moderate to marked; occasional karyomegalic cells were present. The mitotic count was 9 per 2.37 mm2 (0–1 per 400× field). Many blood and lymphatic vessels were infiltrated by neoplastic cells. Lymphocytes infiltrated the neoplastic stroma multifocally, often around peripheral blood vessels. Neoplastic cells also effaced the parenchyma of the right kidney. In the left kidney, there was an extensive region of renal papillary necrosis, with multifocal tubular mineralization and interstitial fibrosis extending into the renal cortex. In addition, there were intravascular neoplastic emboli in the liver and lungs. Immunohistochemistry for pancytokeratin and vimentin showed strong dual cytoplasmic reactivity among the majority (~95% and 80%, respectively) of the neoplastic cells (Fig. 3B, 3C). Approximately 60% of the neoplastic cells also exhibited moderate cytoplasmic expression of WT1 (Fig. 3D). The final diagnosis was peritoneal epithelioid mesothelioma with renal, hepatic, and pulmonary metastases.
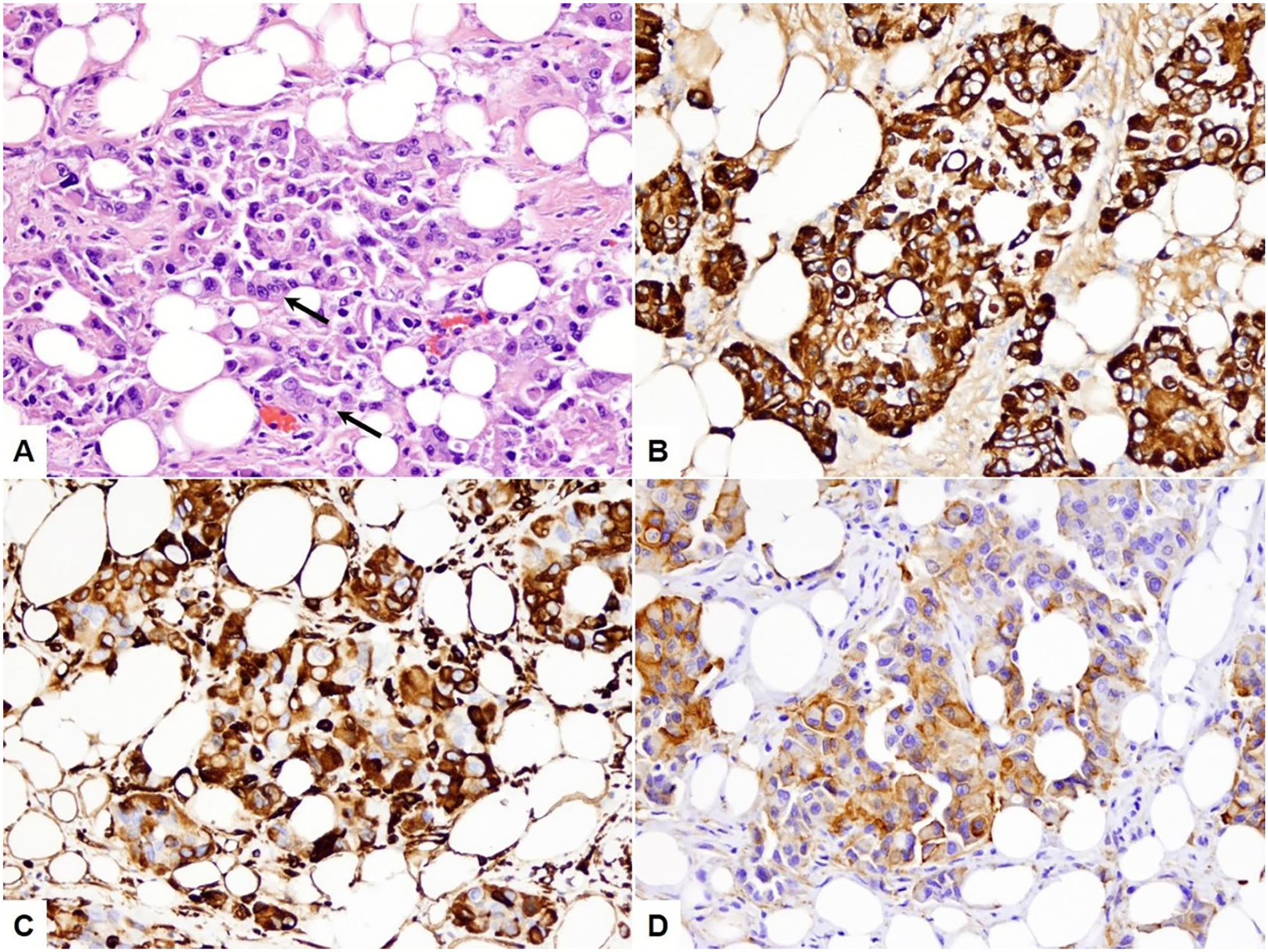
Peritoneal mesothelioma in a cat.
Mesotheliomas are rare neoplasms in cats, with the largest pathology study consisting of 10 cases gathered over 20 y. 2 We performed a literature review to summarize the patient data and lesions in feline mesothelioma cases, confirmed histologically by invasion or metastasis (Suppl. Table 1). Cases were omitted if there was no histopathology performed, if there was only a brief mention of the case, or if the source was not peer-reviewed. The 30 known cases that fit the criteria included 11 males and 18 females; one case did not specify sex. The affected cats were 11 mo to 17 y old, with a median age of 10 y with one case of unknown age. Neoplasms were recorded in the pleura (n = 18), peritoneum (n = 10), and pericardium (n = 4).The breeds affected most commonly were domestic shorthair (n = 22) and Siamese (n = 4), followed by mixed breed (n = 2), Persian (n = 1), and domestic medium hair (n = 1). The types of mesothelioma reported are epithelioid (n = 8), sarcomatoid (n = 6), and biphasic (n = 8); the subclassification was unspecified in 8 cases. When combining the 17 cases in which immunohistochemistry was performed, all had dual reactivity of both pancytokeratin and vimentin. HBME1 and calretinin, 2 commonly used markers for human mesothelioma, were expressed in 7 of 11 and 0 of 10 feline mesotheliomas, respectively.1,2,6,8,9,14,15,20 Reactive feline mesothelial cells have been found to label with WT1 protein, 10 prompting investigation of expression of this protein in our case. Most (~60–95%) mesothelioma cells in our case exhibited strong cytoplasmic labeling with WT1. Interestingly, despite being a DNA-binding transcription factor with nuclear distribution, several neoplasms, including mesotheliomas, exhibit cytoplasmic labeling of WT1.12,17 Diagnosticians should be aware of this staining pattern to avoid assumption of nonspecific labeling. Although validation studies and expression profiles for WT1 in the cat are still needed, this protein may be helpful in identifying mesothelial cell lineage in this species.
Less than half of the reports of feline mesotheliomas have cytology results available to compare to the histopathology, and none of the papers reviewed utilized immunocytochemistry to aid in the diagnosis of mesothelioma.3,4,7,8,16,18 Although distinguishing reactive mesothelial hyperplasia from mesothelioma or carcinomatosis will remain a limitation of cytology, in our experience, hyperplastic mesothelial cells rarely exhibit significant nuclear atypia in cats as can be seen in dogs. Thus, the observation of mesothelial cells with nuclear atypia in an effusion of a cat favors the diagnosis of neoplastic effusion but should be verified with histologic documentation of invasion or metastasis. Along with cytomorphology, dual cytokeratin and vimentin expression will likely remain the basis of confirming mesothelial cell lineage. However, as other neoplastic cells may co-express cytokeratin and vimentin, notably feline bronchogenic adenocarcinoma, 13 additional immunomarkers such as WT1 may further support mesothelial cell lineage. Our case report illustrates the comparable cytopathologic, histopathologic, and immunolabeling characteristics in a case of feline mesothelioma, as well as the utility of using immunocytochemistry to identify mesothelial lineage when neoplastic effusion is suspected.
Supplemental Material
sj-pdf-1-jvd-10.1177_10406387211017489 – Supplemental material for Feline mesothelioma: case report and review of cytologic, immunocytochemical, histopathologic, and immunohistochemical findings
Supplemental material, sj-pdf-1-jvd-10.1177_10406387211017489 for Feline mesothelioma: case report and review of cytologic, immunocytochemical, histopathologic, and immunohistochemical findings by Andrew H. Schlueter, Shannon D. Dehghanpir, Bonnie Boudreaux, Christopher Robinson, Jose Cesar Menk P. Lima and Ingeborg M. Langohr in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the Histology Laboratory of the Louisiana Animal Disease Diagnostic Laboratory and the University of Georgia Veterinary Diagnostic Laboratory for tissue processing and immunolabeling.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
