Abstract
Lens-free technology (LFT) is a user-friendly, simple imaging technique, based on in-line holography and numerical processing. LFT gives access to a reconstructed optical field revealing phase and absorption contrasts of the samples studied. It can generate high-contrast images of biological objects on a large field-of-view with a micrometer scale resolution using a basic optical setup without optical magnification.2-4 Briefly, a sample is placed on a high-resolution optical sensor and illuminated from above by a point-like source of light. The sensor records interference patterns created by the light diffracted by the objects and the transmitted undiffracted light. The raw data are then processed with a dedicated reconstruction algorithm.2-4 The wide field-of-view and the high resolution then lead to statistical counting (> 10,000 cells) in a single-shot acquisition consistent with performance required in hematology.
LFT has already proved useful for numerous biological applications, such as sperm motility assessment,
Our aims were: 1) to confirm that LFT can create a hologram of microfilariae in whole blood that can be visually detected (visual LFT detection), 2) to design an algorithm to detect microfilariae in the LFT acquisitions, and 3) to determine if LFT with a specific algorithm combined with automated specimen preparation is sensitive and specific for detection of microfilariae in canine whole blood (algorithmic LFT detection). Our study was approved by the Science et Santé animale ethics committee (SSA_2017_007) of the École nationale vétérinaire de Toulouse. After informed and signed owner consent, canine blood specimens were collected at the Aristotle University of Thessaloniki and at the École nationale vétérinaire de Toulouse.
We examined 3 groups of 10 canine EDTA blood specimens. Group A was composed of microfilariae-positive specimens, confirmed by direct microscopic counting of microfilariae on 2 May-Grünwald/Giemsa–stained blood smears. Group B was the negative control group composed of specimens without any hematologic abnormality. Group C was composed of blood samples from dogs with diverse hematologic abnormalities except microfilaremia. All EDTA blood specimens were: 1) examined by a European Board of Veterinary Specialisation–certified specialist in veterinary clinical pathology for identification and counting of microfilariae in stained smears, and 2) directly sampled by the LFT prototype for 3 cycles of image acquisition at low (1/100) and high (1/900) dilutions.
The LFT prototype is a small benchtop hematology analyzer, developed by Horiba ABX. For each cycle, the analyzer rinsed the photographic chamber with saline, filled it with a new 0.03-µL aliquot of 1:100 or 1:900 whole blood diluted in saline, and took 5 pictures; this sequence was repeated up to 75 times, using a total volume of 2.2 µL of whole blood. We recorded 8-bit uncompressed gray-scale raw images, and all pictures were then analyzed by visual LFT and algorithmic LFT (Suppl. material) to identify and count microfilariae. Briefly, after a numerical reconstruction step to obtain an in-focus image, 2-step sorting of regions of interest was performed on each image (Fig. 1). The first step uses morphologic criteria of the detected objects, and the second takes into account their gray-level intensity to refine the detection. 1 In a final step, the algorithm compared the regions of interest detected in the 5 images of each series. A series was classified as positive (i.e., microfilariae detected) if the same region of interest was detected in ≥1, ≥2, or ≥3 images of the series, depending on the chosen cutoff. The number of detections observed for each specimen was considered to indicate the intensity of the parasitemia.
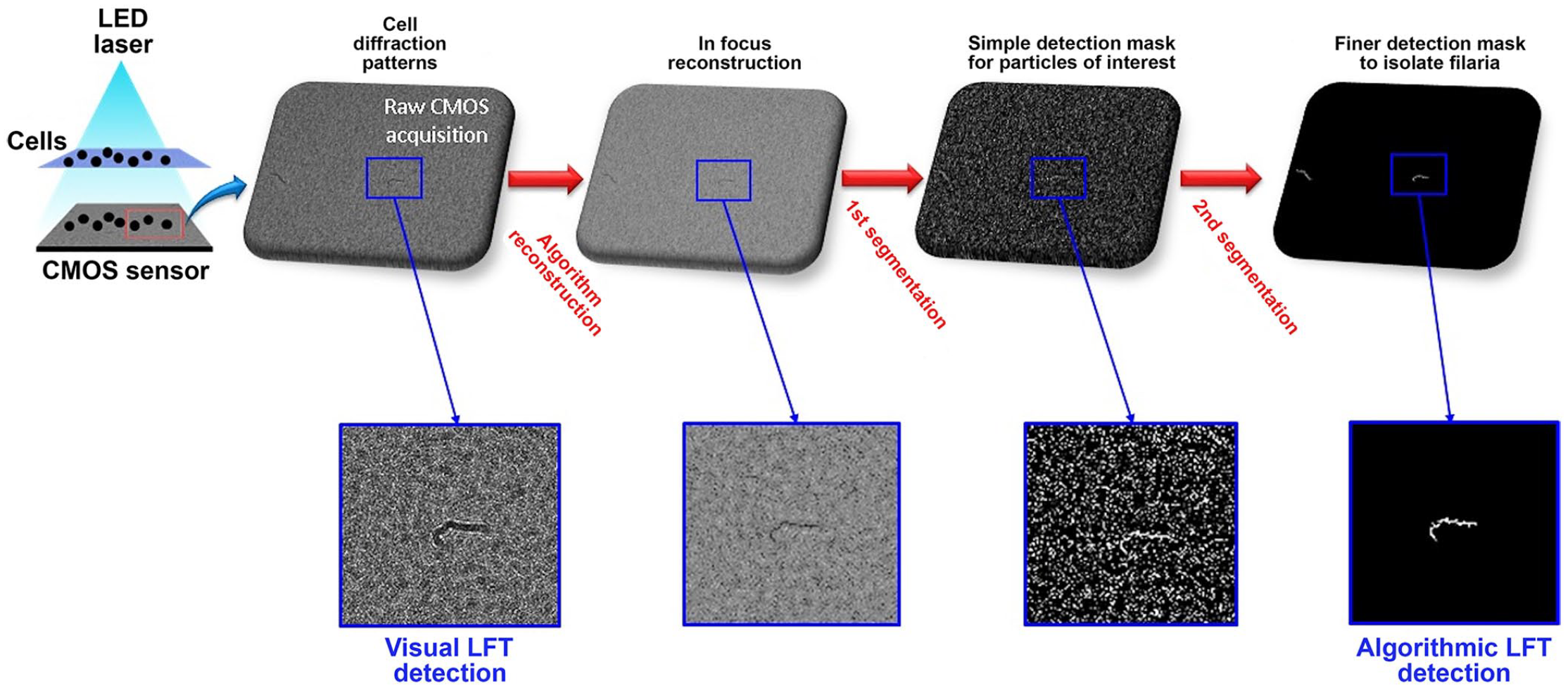
The steps of the algorithm for image reconstruction and detection of regions of interest, based on eccentricity and solidity characteristics.
Group A was composed of 1 spayed and 3 intact females, 1 castrated and 5 intact males; ages were 3–9 y. Concurrent diseases were leishmaniasis in 3 dogs, 1 case each of megaesophagus and bronchopneumonia secondary to regurgitation, immune-mediated hemolytic anemia (IMHA), and idiopathic epilepsy. One of the 3 dogs with leishmaniasis also had a very mild infection by
The total number of images obtained for each case was variable, given the variable number of series of acquisitions (min. and max. number of series: 20 and 77, respectively) and the fact that series were incomplete in 6 of 30 cases as a result of technical issues. At high dilution, no microfilariae could be detected by visual LFT or algorithmic LFT on any image, even in specimens with a heavy parasite load. At low dilution, no microfilariae were identified by visual LFT on images of groups B and C. Microfilariae were identified by visual LFT in images of 9 of 10 cases of group A.
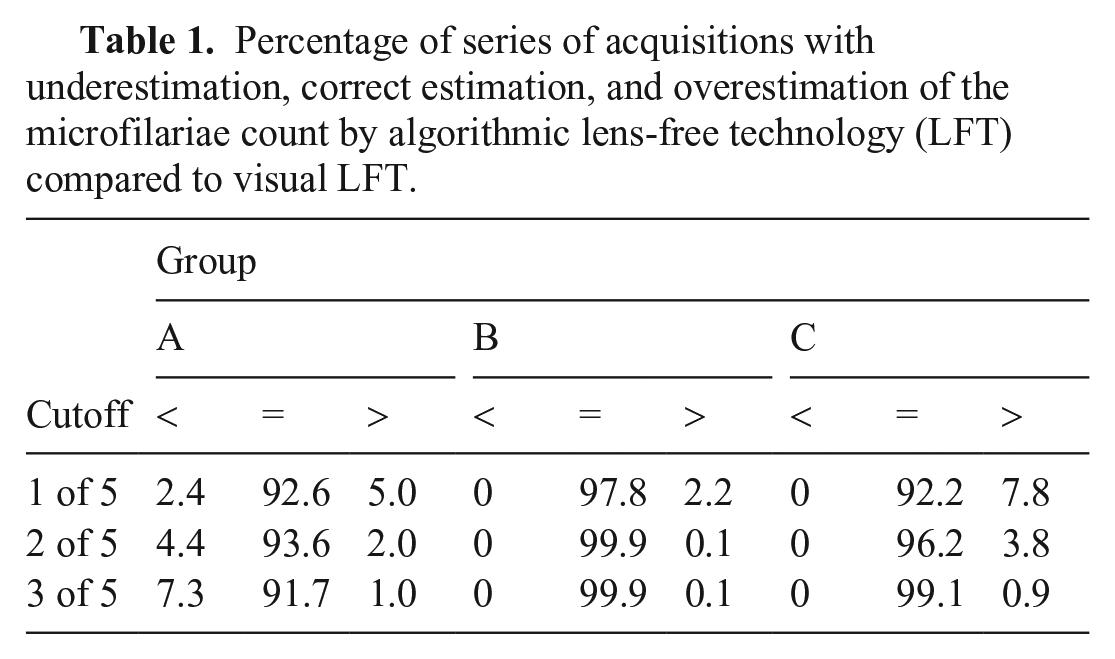
In images of group A, the percentage of series in which microfilariae were identified by the algorithmic LFT decreased moderately when the cutoff was increased from 1 to 3, whereas in groups B and C, the percentage of series in which microfilariae were falsely identified decreased (Table 1). In group A, the number of series in which microfilariae were observed by visual LFT or by algorithmic LFT was positively correlated with blood smear counting (Spearman r = 0.83,
Percentage of series of acquisitions with underestimation, correct estimation, and overestimation of the microfilariae count by algorithmic lens-free technology (LFT) compared to visual LFT.
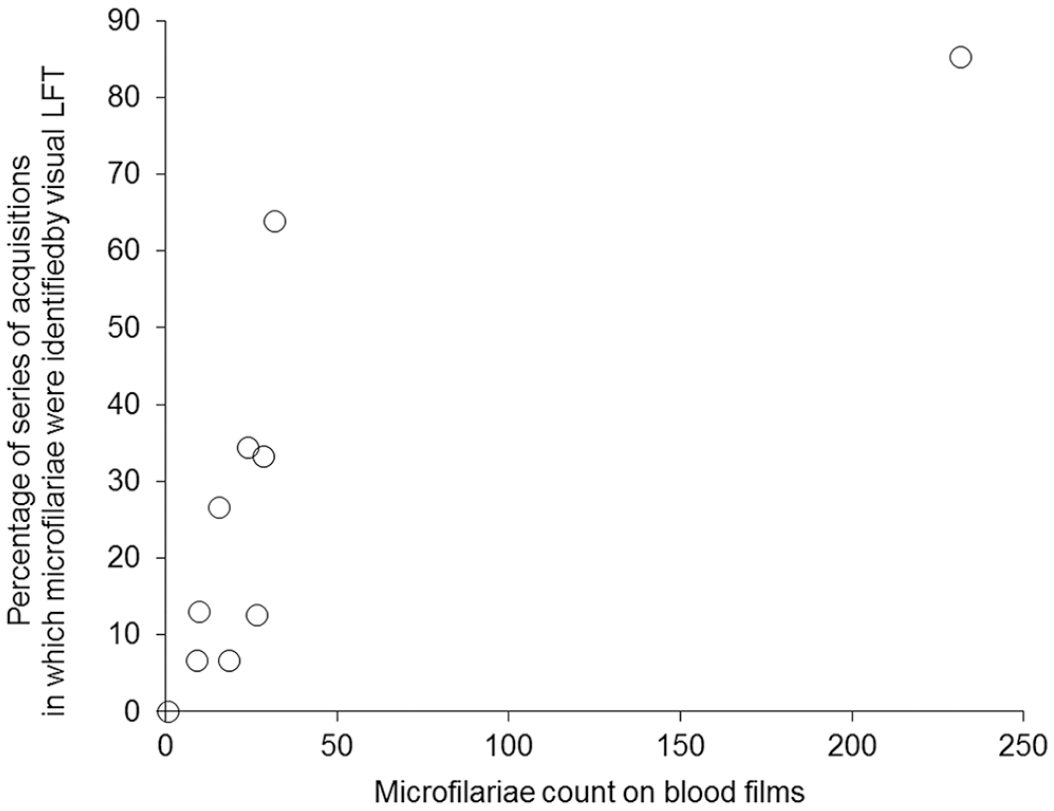
Scatterplot of the percentage of series with visual lens-free technology identification of microfilariae versus microfilariae count on blood films.
Microfilariae were correctly identified in 9 of 10 cases on images either by visual LFT or algorithmic LFT (Table 2). The false-negative was the case in which only one parasite had been identified by microscopic examination of blood films. In groups B and C, microfilariae were falsely identified by algorithmic LFT in at least 1 series of images of 3 of 20 cases at the 3 of 5 cutoff. In the latter, visual observations of images were poor RBC spreading, agglutinating RBCs, or foreign objects.
Number of cases identified as diseased versus non-diseased by algorithmic lens-free technology (LFT); sensitivity (Se) and specificity (Sp) of microfilaria detection by algorithmic LFT technology.
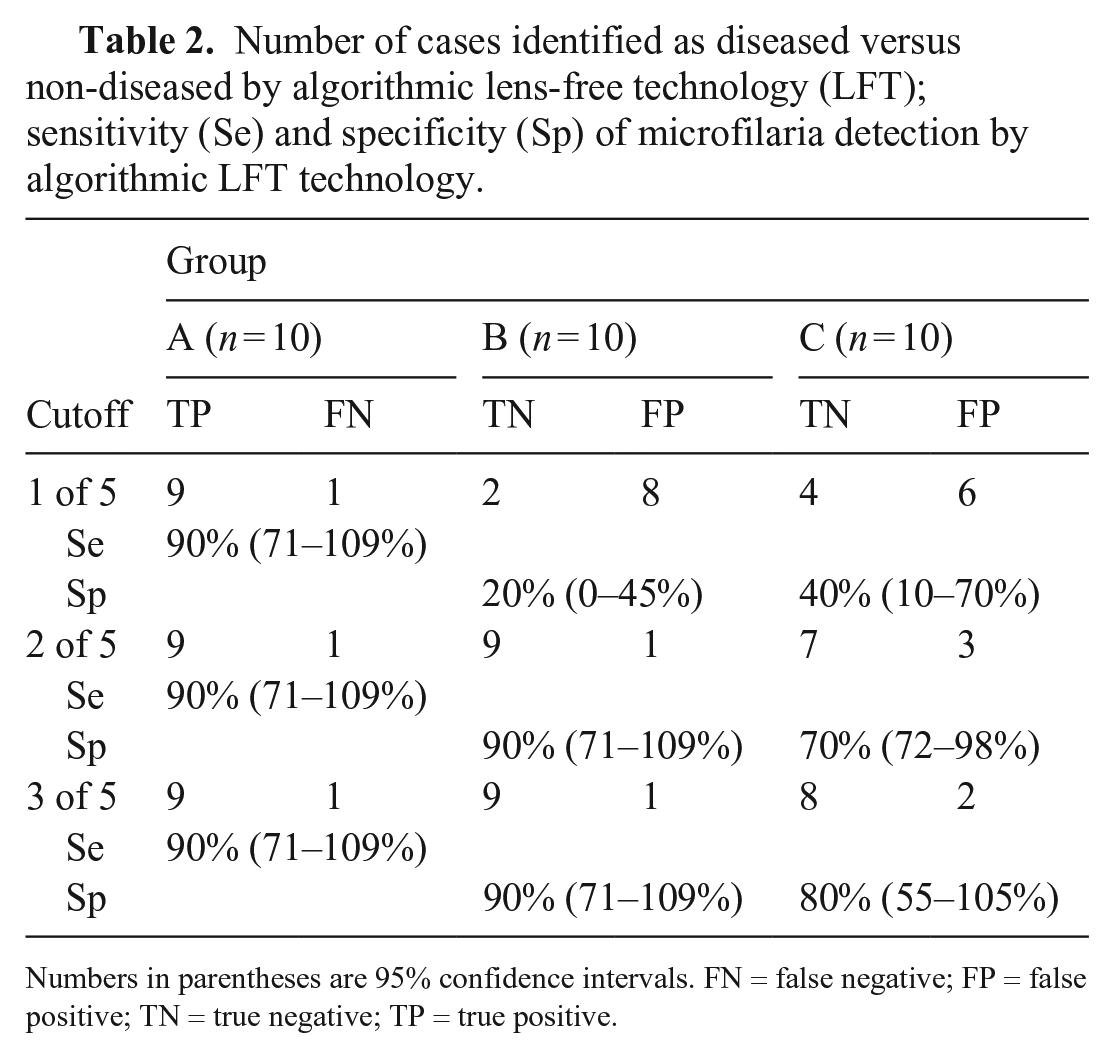
Numbers in parentheses are 95% confidence intervals.
FN = false negative; FP = false positive; TN = true negative; TP = true positive.
Human hemoparasite recognition using LFT with a detection method based on parasite motility has been reported, 5 but, to our knowledge, confirming that LFT can create holograms of circulating microfilariae in diluted whole blood (without lysis) that are identifiable to the human eye and an algorithm have not been reported. Given that the device is cost-effective because its components are widely available and not expensive, and does not require advanced technical skill, it seems adequate for the detection of microfilariae in a large number of individuals (i.e., for screening at-risk populations in endemic areas).
The high- and low-dilution cycles had different performances. Using high dilution, the wide spread of cells simplified object discrimination and recognition, and the algorithm had 100% specificity (i.e., found no false-positives). However, sensitivity was null given that no microfilariae could be identified on the LFT images, even in specimens with a heavy parasite load.
Using low dilution, sensitivity was high (90%). Given the higher volume of analyzed blood and the high number of processed images, false-negative images led to few false-negative series, and no false-negative cases when compared to visual LFT counting, given that the algorithm always recognized at least one of the microfilariae photographed in each case in group A (Fig. 3). When evaluating the whole method (LFT acquisitions coupled to the algorithmic image recognition), a false-negative result occurred in only one case, because no microfilaria was photographed. Importantly, this case had a very low parasite load (only one microfilaria in 2 blood smears), and the number of series of LFT acquisitions was lower than for other cases (only 20 compared to 30–77). Another cause of false-negatives was microfilariae located on the edges of the images. A movement-based detection method, as reported previously, 5 was not chosen because some microfilariae were dead in our specimens. Supplementary information regarding motion and location might decrease the false-negative rate in the case of microfilariae close to the edges.

Lens-free technology image (hologram) of a microfilaria surrounded by red blood cells in a canine whole blood specimen.
With low dilution, a few false-positives were found because of RBC overlapping or agglutination (Fig. 4) or contaminants (dust). At low dilution, RBCs overlap each other in the acquisition chamber, resulting in regions with morphometric properties (solidity characteristics) that could be mistaken by the algorithm for microfilariae. Between 2 consecutive images, micro-movements of RBC in the acquisition chamber from residual flow caused by the chamber filling would generally separate RBCs and break the equivocal pattern, whereas a microfilaria would still create a region of interest even if it moved during acquisition, giving a positive result on nearly all images of the same series. This explains why the algorithm specificity improved from the 1 of 5 to the 3 of 5 image cutoff. In the one case of IMHA, agglutinated RBCs created regions of interest with increased solidity that would not disappear between 2 consecutive images. Therefore, this case was the main source of error in group C. If the IMHA case was excluded, the rate of false detections with the 2 of 5 and 3 of 5 cutoffs was very low and was caused by dust or textile fibers. Moreover, visual control can easily eliminate these false detections.
One limitation of our study was the absence of a standardized method for the detection and counting of microfilariae in blood smears (uncalibrated volume of the blood drop for the blood smear); however, examinations were performed in European College of Veterinary Clinical Pathology–certified laboratories by specialists in veterinary clinical pathology according to the procedures of the laboratory. The LFT method was not standardized because these were only preliminary investigations. The main limitation was the limited number of cases, which prevented us from calculating relevant statistics as shown by the very wide 95% confidence intervals of the values estimated for sensitivity and specificity (Table 2). However, given the high number of images that were analyzed for each specimen, we believe that these preliminary results can be trusted.
There are several ways to improve algorithmic LFT detection of microfilariae, the first being increasing the number of series of acquisitions. Supplementary criteria could also be defined to detect the microfilariae missed by the algorithm. However, building an overly complex algorithm based on a limited number of cases leads to the risk of over-fitting, meaning that the algorithm would perform extremely well in the dataset used for the algorithm writing, but poorly when used in other cases. A deep-learning method could also be used, but would require a very high number of cases, which was not possible in our study. A second improvement would be lysing RBC, thus creating a protocol combining the advantages of the low-dilution protocol (larger analyzed blood volume) and the high-dilution one (better visualization of other-than-RBC elements). Enriching the blood after the lysis step, for instance by filtration, would increase sensitivity by increasing the concentration of microfilariae. Such a protocol would still be simple and cost-effective and meet the requirements for a point-of-care test. Because dead microfilariae are easier to detect than live ones, adding a step in the process to kill the microfilariae before running the specimen would facilitate the analysis.
Our preliminary results need to be confirmed in a larger study, in order to test technical improvements and increase the number of cases to obtain a statistically valid evaluation of sensitivity and specificity.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211001092 – Supplemental material for Detection of circulating microfilariae in canine EDTA blood using lens-free technology: preliminary results
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211001092 for Detection of circulating microfilariae in canine EDTA blood using lens-free technology: preliminary results by Typhaine Lavabre, Zoe S. Polizopoulou, Damien Isèbe, Olivier Cioni, Véronique Rebuffel, Pierre Blandin, Nathalie Bourgès-Abella and Catherine Trumel in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
Damien Isèbe is employed by Horiba ABX.
Funding
The authors received material support from Horiba ABX who provided the LFT analyzer, and funded the algorithm development by the Univ. Grenoble Alpes, CEA, LETI team.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
