Abstract
Twenty-eight lactating dairy cattle in New York State were exposed to botulism toxin; 12 died and 16 recovered but never returned to full productivity. Pieces of a raccoon carcass were found in the total mixed ration on the first day of the outbreak. Clinical signs included anorexia, decreased milk production, decreased tongue tone, profound weakness, and recumbency.
On November 10, 2018, a tie-stall dairy herd in New York State (NYS) with 62 lactating and 12 nonlactating cows experienced a 50% drop in feed intake among the lactating cattle. By the next day, 1 lactating cow was recumbent. Two other cows were weak and exhibiting abdominal effort during respiration. On November 12, the 2 weak cattle progressed to recumbency, and herd milk production dropped by ~ 20%. Over the next 5 d, 28 cattle showed clinical signs of weakness starting in the hind legs and progressing to recumbency, anorexia, abdominal effort during respiration, dry manure, decreased rumen contractility, and a dramatic decrease in milk production. There was slow retraction of the tongues of affected cattle, and many would repeatedly attempt to rise on the hind legs until they were exhausted, and then remain in sternal recumbency. The cattle appeared alert, with normal ear carriage and normal tail tone. Total herd milk production was 50% lower than average, based on bulk tank weights from the milk hauler on day 5 of the outbreak. By November 17, 12 cattle had died, and the remaining 16 showed profound weakness. In some cases, disease progression was rapid; cattle developed clinical signs and died or were euthanized given prolonged recumbency for 36 h in a tie-stall barn. This outbreak had a 45% morbidity rate and a 19% mortality rate.
The day after the lactating cattle became anorexic, the veterinarian found a piece of raccoon carcass, identified by the attached tail, in the total mixed ration (TMR) in the manger in front of a mid-lactation cow. The farmer stated that the TMR batch containing the carcass had only been available for consumption for the past 24 h, given that one bale of hay was used to make a daily supply of feed for the lactating herd. Based on the severe state of decomposition, the raccoon carcass was presumably mowed in the first cutting of grass hay harvested earlier that year. The hay was stored in single round bales and covered with commercial plastic bags to promote fermentation. No spoilage was detected in the hay when opened. It is possible that other round bales from that field contained pieces of raccoon carcass, and previous feeding of the carcass to the cattle had occurred but was not noticed by the farmer. The farmer noted that there was more jimson weed in the hay fields that growing season compared to previous years. The TMR diet included grass haylage, corn silage, grain, cottonseed, and the ionophore monensin (Rumensin; Elanco) in a commercial pellet. After finding the raccoon carcass, the remaining TMR and first cutting haylage were discarded. Only lactating cows fed the TMR diet developed clinical signs. Nonlactating cows housed in the other end of the tie-stall barn, but on a different ration, remained asymptomatic. Young stock on the premises were also unaffected.
Based on the history, clinical signs, and number of animals affected simultaneously, a differential diagnoses list included botulism, ionophore toxicity, organophosphate poisoning, mycotoxin toxicity, nitrate poisoning, cyanide poisoning, vitamin E–selenium deficiency (white muscle disease or nutritional myodegeneration), toxic plant ingestion (coffee senna [
Four cattle were euthanized for postmortem examination and diagnostic testing. Cow 1 was autopsied on the farm, and ~ 500 mL of dark-red pericardial effusion was observed. Several mild changes were present in a chemistry panel performed on serum collected antemortem from cow 2: hypernatremia, hyperkalemia, hyperchloremia, elevated anion gap, hypocalcemia, hyperproteinemia, hyperglycemia, mildly elevated aspartate aminotransferase and creatine kinase activities, and hyperbilirubinemia (Table 1). Cow 2 was euthanized and sent with the carcasses of cows 3 and 4, and fresh and formalin-fixed tissues from cow 1, for autopsy and ancillary testing at Cornell University’s Animal Health Diagnostic Center (AHDC; Ithaca, NY). Upon examination, cow 3 also had ~ 500 mL of pericardial effusion, and cow 4 had ecchymotic and petechial hemorrhages affecting the pericardium, aortic adventitia, and tracheal mucosa.
Abnormal results in the serum chemistry panel for cow 2.
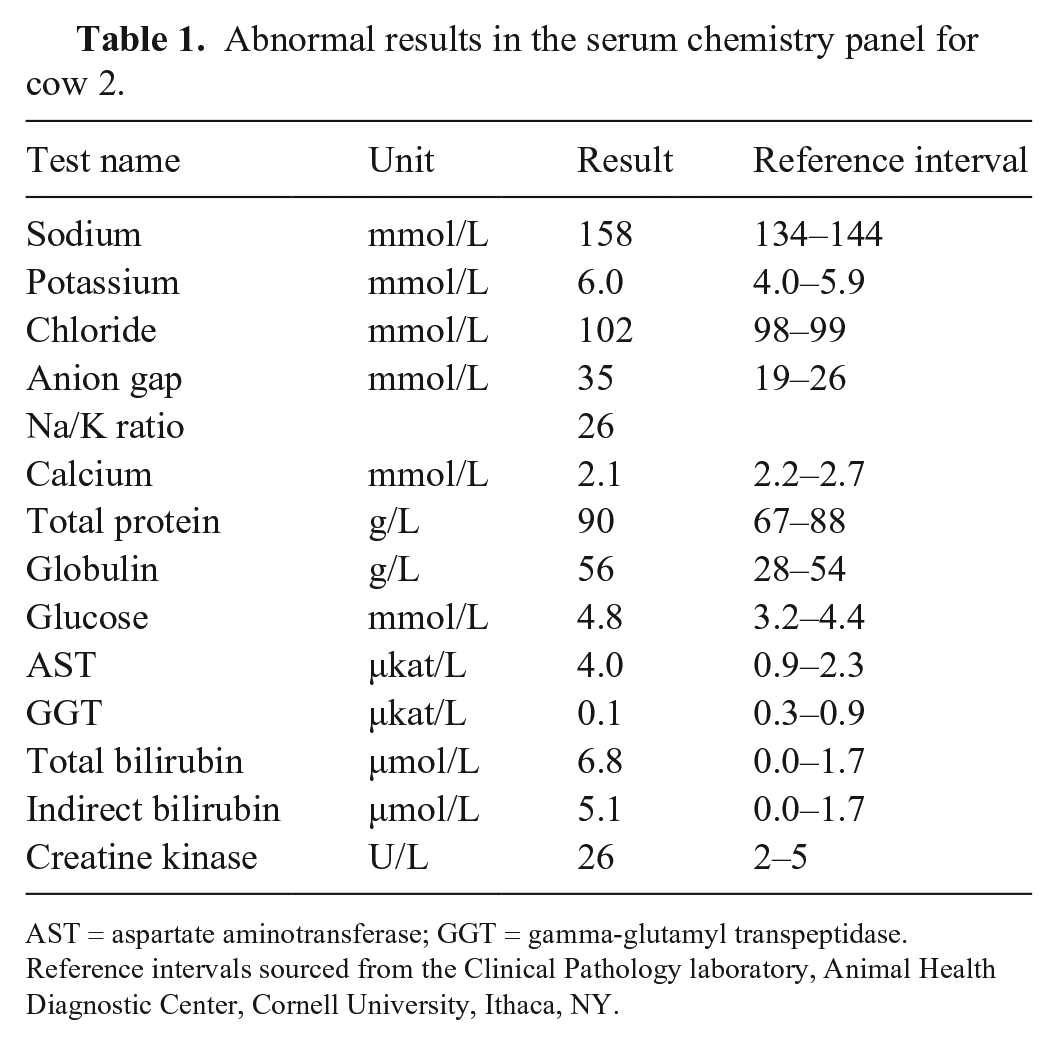
AST = aspartate aminotransferase; GGT = gamma-glutamyl transpeptidase. Reference intervals sourced from the Clinical Pathology laboratory, Animal Health Diagnostic Center, Cornell University, Ithaca, NY.
Although there were no observed gross lesions, histologic examination of the skeletal and cardiac muscle demonstrated acute mild-to-moderate multifocal monophasic degeneration of individual muscle fibers in the hindquarters of 4 of 4 cattle and hypereosinophilic cardiomyocytes in 2 of 4, raising concern for a toxic myopathy, particularly ionophore toxicity. 19 Commercial pellets (Rumensin) from the current supply on the farm were tested by the manufacturer (Elanco Animal Health Feed Assay Service) for monensin levels. Test results (65.2% g/ton as fed, wet weight) indicate 102% of label claim (63.9 g/ton), which is within acceptable limits (85–115%). TMR was not tested for monensin quantification, but the farmer and attending veterinarian confirmed that no mixing errors occurred. At the time of clinical signs or death from ionophore toxicity, the rumen contents may no longer contain representative as-fed ionophore levels, as a result of digestion and absorption. 6 This makes interpretation difficult when monensin is purposefully added to the diet; therefore rumen contents from deceased cattle were not tested for ionophore quantification in this outbreak. Feed was not tested for other ionophores, such as lasalocid or salinomycin, given that they were not part of the mixed ration. Monensin and other ionophores remain a potential cause of toxicity but their presence in the feed are unlikely based on the TMR diet formulation and clinical signs.
Ancillary testing included aqueous humor nitrate concentrations on all 4 cattle, liver selenium concentrations on all 3 cows tested (cows 1–3), and serum cholinesterase activity on cow 2. These tests were all within normal limits, ruling out nitrate poisoning, nutritional myopathy, and organophosphate poisoning. The TMR was tested for mycotoxins (Cumberland Valley Analytical Services, Waynesboro, PA), which were found to be within normal limits (Burbank KE, pers. comm., Nov 2018).
Samples of rumen contents from 3 autopsied cows were tested for
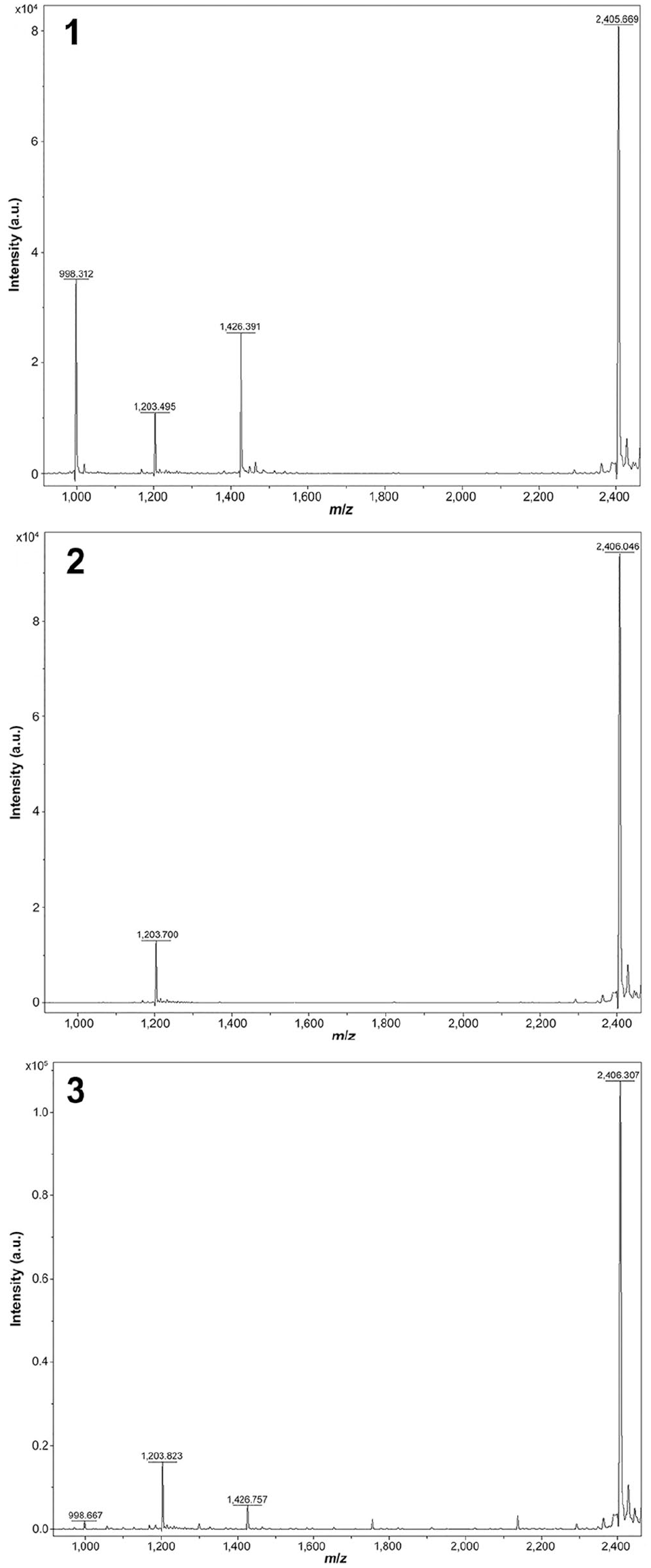
Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS)
The herd diagnosis was botulism caused by BoNT/A. The detection of preformed toxin A by MALDI-TOF MS in the rumen contents of cows 2 and 3 is considered diagnostic for exposure. The positive culture of
The gold standard for diagnosing botulism is detection of the toxin through the mouse bioassay (MBA),1,11 which is offered at only 2 veterinary laboratories in the United States. Although considered the most sensitive test, the LOD of the MBA may exceed the relatively low concentrations of preformed neurotoxin sufficient to intoxicate highly susceptible animals, such as horses, and less so cattle, both known to be more sensitive to the toxin than mice.11,13 Cattle are ~ 13 times more sensitive to BoNT/C than mice, 13 and this can lead to false-negative laboratory MBA results in cases of bovine botulism.
BoNTs are metalloproteinases, and in the neuromuscular junction, BoNTs cleave key vesicular transport proteins of the
BoNT/B–D have historically been found as the causative neurotoxins in bovine botulism.4,11,12 BoNT/C and /D are associated with pica and phosphorus deficiency and have been reported in cattle in South Africa, South America, and Australia.
11
Outbreaks of BoNT/C and /D in confined dairy cattle are typically linked to feed contaminated by dead carcasses or poultry litter, and have been reported in North America and Europe.3,5,14 BoNT/B has caused botulism in improperly ensiled forages such as rye silage,
4
plastic packed hay,
22
and round bale barley haylage fed to cattle.
10
The ensiling process creates an anaerobic environment, and if the pH does not drop below 4.5,
The presence of BoNT/A in rumen contents from 2 NYS dairy cows documents an outbreak of bovine botulism from toxin A in North America, detected by MALDI-TOF MS. The detection of BoNT/A in this herd may be related, in part, to the sensitivity of the MALDI-TOF MS. Toxin A was not detected in the third sample of rumen contents, possibly as a result of low toxin concentration. The source of both BoNT/A toxin and
In our outbreak, 2 autopsied cows had pericardial effusion, and all 4 had histologic findings of monophasic, multifocal degeneration variably affecting skeletal muscle and cardiac muscle consistent with toxic myopathy. Given that it targets the neuromuscular synapse, botulism is not associated with gross or histologic lesions, 12 although prolonged recumbency secondary to botulism can cause myodegeneration. Muscle toxins include ionophores, toxic plants, and plant-origin toxins that produce nonspecific segmental necrosis. In cattle, the clinical signs of acute monensin toxicity are nonspecific and include anorexia, diarrhea, dullness, weakness, and death.6,19 Monophasic, multifocal degeneration is usually present by 48 h after exposure. 19 In this herd, elevated ionophore levels were not detected in the commercial pellets (Rumensin), and other toxins were considered unlikely based on exposure history. Ultimately, the cause of these lesions is unknown.
Marked pericardial effusion consistent with cardiac failure was observed in 2 of 4 autopsied cows; pericardial effusion with fibrin strands is reported as a relatively consistent feature of shaker foal syndrome (toxicoinfectious botulism), as are pulmonary edema and congestion. 21 Petechial and ecchymotic hemorrhages have been reported sporadically with botulism toxicity 2 ; however, these findings are also nonspecific agonal changes.
There are no established meat and milk withdrawals for cattle affected by botulism. Botulinum neurotoxin has not been detected in milk from Holstein cattle injected with various doses of BoNT/C.
13
Cooking and pasteurization will denature botulinum toxin. Although the risk for milk and meat contamination appears low, in previous reports of bovine botulism, carcasses have been incinerated and milk was not shipped for human consumption.5,8,10 In our outbreak, the farmer was instructed to not drink raw milk and not feed raw milk to calves. Bovine veterinarians, diagnostic laboratories, and federal and state health officials should be aware of
Footnotes
Acknowledgements
We thank Amy Rourke, Melissa D’Amico, Dominick Centurioni, and Maureen Conlon from the Wadsworth Center Biodefense Laboratory, New York State Department of Health, for providing laboratory technical support; Dr. Kelly A. Johnson, Cornell University Outreach & Information Resources Librarian for reference organization and manuscript review; Dr. Jen Nightingale from Countryside Veterinary Clinic for her sample submission; and Dr. Francisco Uzal, California Animal Health and Food Safety Laboratory System for manuscript review.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
