Abstract
We assessed the health status and hematologic and blood chemistry values of 48 juvenile green turtles (Chelonia mydas) on the southeastern coast of Brazil (25°S 48°W). We investigated the impact of size and weight, nutritional condition, and presence of epibionts and fibropapilloma tumors. Nutritional status was good for 39 animals; these animals had higher serum albumin, cholesterol, and phosphorus concentrations than 9 animals with a fair nutritional score. Reference values for hematology and biochemistry were calculated for 39 individuals without fibropapillomas (FPs). Turtles with epibionts (n = 33) had lower hemoglobin (Hb) than turtles without epibionts (n = 15; t = −2.09, p = 0.04), and the area occupied by epibionts was positively correlated with the white blood cell count (r = 0.37, p = 0.03). FP turtles had significantly lower hematocrit (Hct), Hb, and red blood cell (RBC) counts than non-FP turtles; serum albumin and cholesterol were higher in non-FP than FP turtles. A negative correlation between curved carapace length (CCL) and Hct (r = −0.51, p = 0.0002) and RBC (r = −0.47, p = 0.0007), and between serum cholesterol, sodium, and uric acid and CCL (r = −0.53, p = 0.0001; r = −0.38, p = 0.007; r = −0.35, p = 0.014, respectively) were identified. The health of turtles appears to deteriorate as they get larger, which manifests in more FPs, decreased body condition, and systemic physiologic changes consistent with chronic disease including lower Hct, RBC counts, serum cholesterol, sodium, and uric acid.
Introduction
The southwest coast of Brazil encompasses an important feeding ground for juvenile green turtles (Chelonia mydas). The area is formed by a complex of rivers and estuaries, wherein turtles from different Central American localities feed on algae, invertebrates, seagrass, monocotyledons, and black mangrove leaves. 26 The area is surrounded by several conservation units and is part of the Atlantic Forest biodiversity hotspot. 25 Considering the relatively low anthropogenic disturbance experienced by these animals (i.e., low boating and fishing activities) in protected areas, most animals are expected to be healthy. Hematologic values and serum biochemistry analysis have been used to provide information on the health status of different species, including green sea turtles. 28 Other external body features, such as the presence or absence of fibropapilloma (FPs), epibiotic load, and body condition also provide information about the general health of individuals.11,19,37
Diseases in green turtles, including fibropapillomatosis, have been connected to heavily polluted areas, agricultural runoff, and areas with high human density. 4 FPs are associated with infection by chelonid herpesvirus 5 (species Chelonid alphaherpesvirus 5).23,24 However, the virus alone does not necessarily cause FPs, and disease expression appears to be a multifactorial interplay among the virus, the host, and environmental factors. 20 Turtles from low-disturbance habitats tend to have a lower incidence of FP and better body condition than turtles who feed in low-quality environments.14,35 Hematologic values, which are an important health indicator, can deteriorate proportionally with the number of FPs. 19 Red blood cell (RBC) indices are commonly lower and outside the reference interval (RI) in green turtles that are affected by FP.1,44
Epibionts are commensal organisms that attach to the carapace and plastron of sea turtles; most of these species do not cause harm, although their presence may serve as an indicator of sea turtle health. 37 Chelonibia testudinaria barnacle epibiotic occurrence was higher in unhealthy than healthy green turtles, especially among small immature individuals from the east coast of Australia. 13
Annually, ~530 green turtles are stranded alive and 2,200 are stranded dead on the coast of São Paulo State (https://segurogis.petrobras.com.br/simba/web/sistema/pmp/1/individualfaunaoccurrence/). Living stranded turtles are transported to rehabilitation centers in different locations of São Paulo State for subsequent release. Hematologic parameters are crucial for determining the appropriate treatment for rescued turtles and the rehabilitation of debilitated individuals. Although natural or expected variation in hematologic values may occur within a species given geographic location, sex, body length, season, and diet,8,21,41,43 turtles that inhabit the same area tend to have similar blood values. Hematologic data from turtles in habitats with low anthropogenic areas can provide baseline values for turtles undergoing rehabilitation in centers along the Brazilian coast. Our aim was to develop RIs for blood values from non-FP juvenile green turtles inhabiting the coast of São Paulo State, and measure trends related to variations in body weight and length, nutritional status, presence of epibionts, and the extent of FPs.
Material and methods
Our study was conducted with permission of the Federal Government Chico Mendes Institute for Biodiversity Conservation–Biodiversity Authorization and Information System (ICMBio-SISBIO) permit 59901-1. Every effort was made to ensure the safety and health of both animals and personnel throughout the capture, examination, and blood sampling procedure. Anesthesia, euthanasia, and surgical procedures were not used in our study. The turtles were constantly shaded and kept wet while behavioral and respiratory patterns were monitored closely.
Study area and turtle handling
The study area encompassed the Lagamar estuary lagoon complex, which extends ~110 km along the southern coast of São Paulo State, Brazil (25°S 48°W). The area is formed by several islands isolated from the mainland by interconnected channels. 40 Turtles inhabiting this area voluntarily enter the artisanal traps used by local people for fishing. 7 Forty-eight turtles from this study were collected directly from 4 of these traps using a dip net. Individuals were brought to a camp field station by boat where all procedures related to data collection were performed on 8 different occasions from October 2017 to April 2018. Turtles were handled individually, for an average of 14 min, while the remaining animals were placed in a shaded area in ventral recumbency and kept moist for periods that varied from 15 to 180 min. Following manipulation, both of each turtle’s flippers were tagged with a disinfected metal tag provided by ICMBio-SISBIO under permit 59901-1 and then released into the estuarine channel.
Nutritional status
Turtle nutritional score was determined by visual observation of the plastron while the turtle was briefly held in the air by one person with both hands. When needed, a hand was passed along the underside of the turtle to assess plastron curvature. The plastron curvature classified the animal’s score as poor = concave plastron; fair = flat plastron; and good = convex plastron. 39
Body measurements and weight
Conventional body measurements were performed using a flexible tape measure (Wyneken J. The anatomy of sea turtles. Miami, FL: U.S. Department of Commerce, 2001. NOAA Technical Memorandum NMFS-SEFSC-470. Available at: http://ibimm.org.br/wp-content/uploads/2017/05/Wyneken-2001-The-anatomy-of-sea-turtles.pdf); the curved carapace length (CCL) was calculated from the cranial-most part of the carapace to the caudal-most tip of the carapace along the dorsal midline. The curved carapace width (CCW) was the widest part of the carapace; the curved plastron length (CPL) was the distance from the caudal-most part of the plastron hard structure to its cranial-most hard structure along the ventral midline; and the curved plastron width (CPW) was the widest part of the plastron without including the ventral surface of the costal scutes. Turtles were weighed using a calibrated hanging scale (kg) after being secured properly.
Fibropapillomatosis
Turtles were examined visually and assessed for the presence of FPs by the same researcher (DMD Mello). The absence or presence of FPs was recorded and classified as absent, mild, moderate, or severe, which accounted for both the tumor number and size.19,44 The area occupied by FPs relative to the carapace area (PAPI) was calculated (cm2) by analysis of photographs using image analysis software (ImageJ 1.51K software; National Institutes of Health, Bethesda, MD). Photos were taken from 90 cm above the turtle parallel to the long axis of the turtle’s body with a 0.1-cm scale. Each tumor was outlined, and the total area was calculated. Additionally, the extent of FPs was analyzed using the fibropapillomatosis index (FPI) to score the FP prevalence based on the number and size of tumors. 32 The FPI calculation was conducted by measuring and categorizing the FPs into 4 size groups: < 1 cm, 1–4 cm, > 4–10 cm, and > 10 cm. Based on FPI, individuals were then grouped into another FPI index—Southwest Atlantic Fibropapillomatosis Score (FPSSWA): as mild (FPI < 40), moderate (40 ≤ FPI < 120), or severe (FPI ≥ 120). 32
Epibionts
Epibionts were quantified using a photogrammetric technique in which photographs of the dorsal and ventral surface were taken with a 0.1-cm scale using the same method used to calculate the FPI. Images were analyzed with ImageJ 1.51K software to calculate the total carapace and plastron area in cm2. Each epibiont was outlined, and the total area occupied by all epibionts of each turtle was added and determined as a percentage of the total area of the carapace and plastron.
Blood collection
Approximately 5 mL of blood was drawn from the dorsal cervical sinus 29 using a 5-mL syringe and 22 G needles. The final volume of collected blood and the needle size varied slightly based on the size of the animal, but the value did not exceed 3 mL/kg. 42 Approximately 2 mL of blood was deposited into a 5-mL heparinized tube (Vacutainer: BD, Curitiba, Paraná, Brazil) for a complete blood cell count. Smears of whole blood were made immediately after blood collection. The remaining blood was placed in a 5-mL tube with clot activator and gel for serum separation (Vacutainer SST II Advance; BD) and placed in a cooler with ice packs before being taken to the laboratory for centrifugation and serum separation.
Laboratory procedures
Hematology
The blood cell analysis and differential were performed at the Instituto de Pesquisas Cananéia (IPeC; Cananéia, SP, Brazil), following conventional techniques for cell counting in reptiles,33,34 within 4 h of blood collection. The Hct was determined by filling a microcapillary tube with heparinized blood, centrifuging the sample at 11,360 × g for 5 min, and reading on a microhematocrit card (EZ reader; LW Scientific, Lawrenceville, GA). The Hb concentration was measured using a spectrophotometer at a wave length of 540 nm after placing 20 µL of whole blood into 5 mL of hemoglobin kit solution (Labtest Diagnóstica, Lagoa Santa, MG, Brazil) and centrifuging the sample for 5 min at 4,400 × g. RBCs, white blood cells (WBCs), and thrombocytes were counted using a Neubauer chamber with a light microscope at a magnification of 400×. 6 The WBC differential count was performed on one Wright-stained slide per animal. One hundred WBCs were counted under 1,000× total magnification in a back-and-forth serpentine pattern, and the percentage of each cell type was recorded for calculation of total cell counts.
Serum biochemistry
The clot activator and gel-containing tubes were centrifuged for 10 min at 4,400 × g for serum separation within 5 h of collection. None of the serum was hemolyzed or lipemic after centrifugation; serum was placed in a −20°C freezer until biochemical analysis. The quantification of serum analytes was conducted using semiautomatic equipment (Mindray BA-88A; Shenzhen Bio-Medical Electronics, Shenzhen, China) within 1 mo of blood collection; biochemical assays were validated in-house (Supplementary Table 1). Changes in aspartate aminotransferase (AST) and gamma-glutamyl transferase (GGT) activities, glucose, and uric acid have been reported for serum of marine turtles refrigerated up to 96 h, 12 but not for samples frozen for up to 4 wk. 13 Considering that sera in our study were frozen within 5 h of collection, significant alterations were not expected. Commercial kits were used to measure albumin, alkaline phosphatase, alanine aminotransferase (ALT), AST, bilirubin, calcium, cholesterol, creatine kinase (CK), creatinine, GGT, globulin, glucose, lactate dehydrogenase (LDH), magnesium, phosphorus, sodium, total protein, triglycerides, urea, and uric acid.
Reference intervals and statistical analysis
RIs were determined from apparently healthy individuals after careful inspection for the presence of FPs, physical injuries or illness, alertness, responsiveness, and observation of body shape. Turtles with FP were considered unhealthy, and their results were not included in RI calculations. RIs were determined by the calculation of the lower and upper 90% confidence intervals according to guidelines from the American Society for Veterinary Clinical Pathology. 17 Additionally, the minimum and maximum values and the means for both non-FP turtles and FP turtles were calculated.
Before statistical analysis, data were evaluated for distribution (Gaussian or non-Gaussian) and outliers using the Shapiro–Wilk test and histograms, respectively. Levene homoscedastic test (p > 0.05) was used when comparing 2 or more groups to determine the use of parametric or nonparametric tests. The difference between 2 groups was tested with the Student t-test when data were parametric; the Mann–Whitney test was used when data were nonparametric. The Pearson test was used to analyze the correlations between blood values and CCL, body weight, total FP-affected area, and epibiont area. Correlations between the FP prevalence, epibiota prevalence, body weight, and CCL were also analyzed. All statistical analyses (p ≤ 0.05) were performed using Statistica v.12 (StatSoft, Tulsa, OK).
Results
Hematologic and biochemical values from 48 free-range green turtles were collected. Body weight range was 1.58–11.26 kg, and the CCL was 23.9–53.1 cm (Table 1). CCL and body weight were highly correlated (r = 0.96, p < 0.001), consequently the correlation of both parameters with blood values was similar. For this reason, only correlation between CCL and blood values is presented (Figs. 1, 2).
Body measurements, weight, nutritional status, and prevalence of epibionts and fibropapillomas in 48 juvenile green turtles sampled in São Paulo State, Brazil.
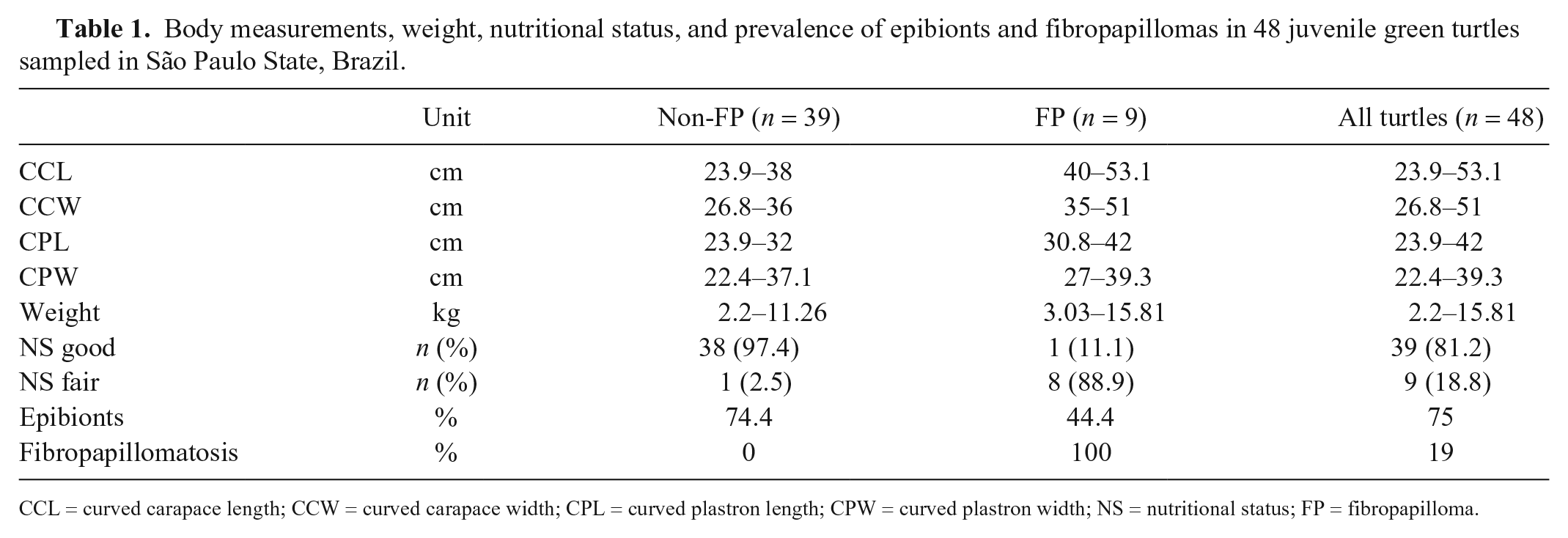
CCL = curved carapace length; CCW = curved carapace width; CPL = curved plastron length; CPW = curved plastron width; NS = nutritional status; FP = fibropapilloma.
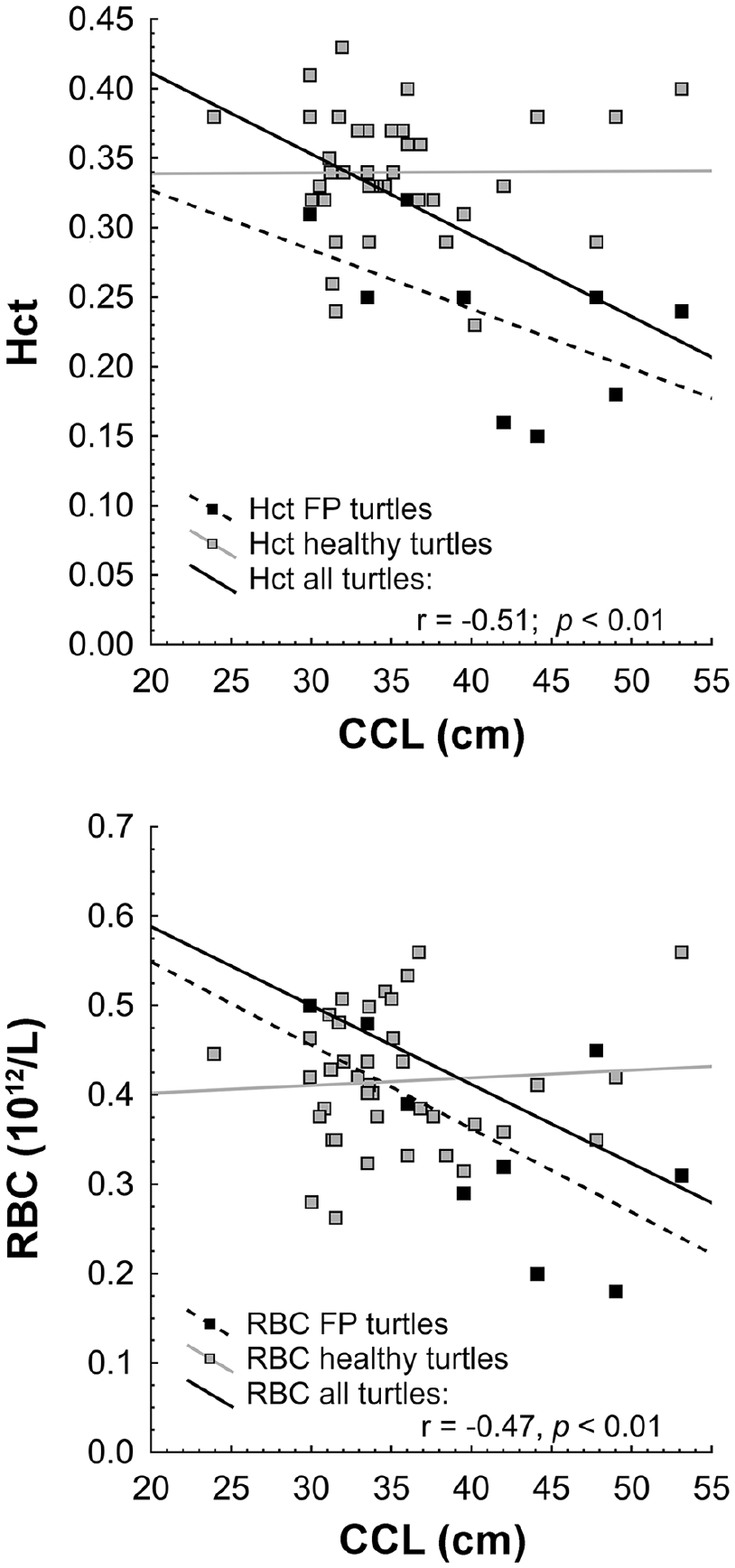
Hematocrit (Hct) and red blood cell (RBC) counts of juvenile green turtles from southern São Paulo State, Brazil, decreased as turtles grew larger. CCL = curved carapace length; FP = fibropapilloma.
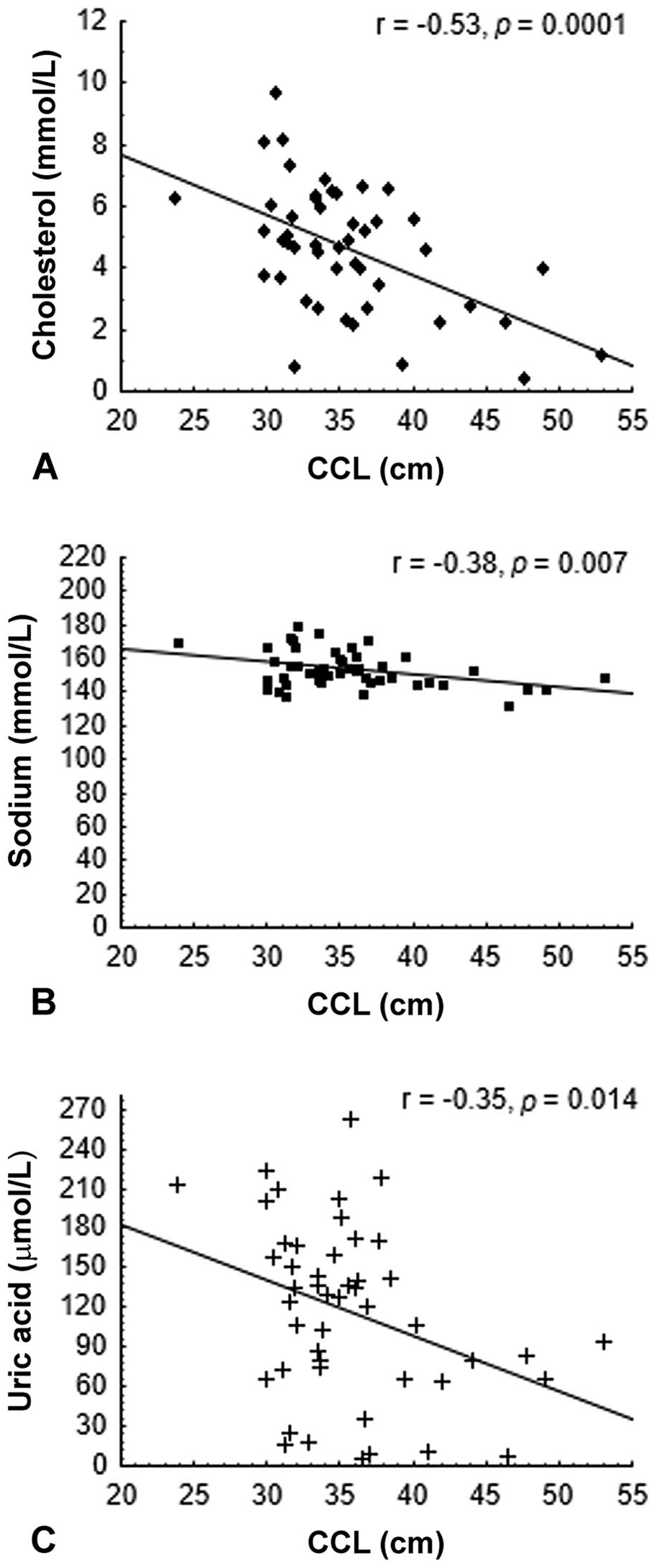
Serum cholesterol (
Epibiont prevalence, fibropapillomatosis, and other external body features
Among the 9 turtles with fair nutritional condition, one had no FP tumors, whereas the remaining 8 had clinical signs of fibropapillomatosis; conversely, only 1 of the 39 turtles with good nutritional condition had FPs (Table 1). In total, 33 of 48 (69%) sampled turtles had epibionts. Five of the turtles with fair nutritional condition had epibionts, whereas 4 had no epibionts; 28 of those in good nutritional status had epibionts, whereas 11 did not have any epibionts. Of the 33 affected turtles, 4 had epibionts located exclusively on the carapace, 4 had epibionts only in the plastron, and most (25 individuals) had epibionts on both plastron and carapace. A few epibionts were observed on the front and hind flippers, and the head and neck, but these were not included in the epibiont percentage calculation.
The CCL was positively correlated with the presence of FPs (r = 0.54, p = 0.0001). A correlation was not found between the epibiont area and FP tumor area (cm2; r = 0.61, p = 0.27) or between the epibiont area and FPI (r = 0.63, p = 0.25).
Most turtles did not have FPs (n = 39), whereas 8 were considered to have mild fibropapillomatosis, and one animal had moderate fibropapillomatosis according to FPSSWA. None of the turtles had severe fibropapillomatosis. Most of the tumors (90.8%) occurred on the ventral aspect of the body. Tumor diameter was 0.25–3.76 cm on the ventral aspect and 1.31–3.19 cm on the dorsal aspect, whereas the tumor estimated total area was 1.01–3.54 cm2 (dorsal) and 0.05–6.56 cm2 (ventral).
Reference intervals
RIs were determined from the apparently healthy non-FP turtles (n = 39; Tables 2 and 3, respectively). Nine of the captured turtles with FPs had indicators of poor health, such as fair nutritional score; lower Hct, Hb, and RBC values; and lower serum albumin or cholesterol values (Tables 2 and 3, respectively).
Hematologic values for 39 non-fibropapilloma (FP) and 9 FP juvenile green turtles captured in a feeding area along the southern coast of São Paulo State, Brazil.
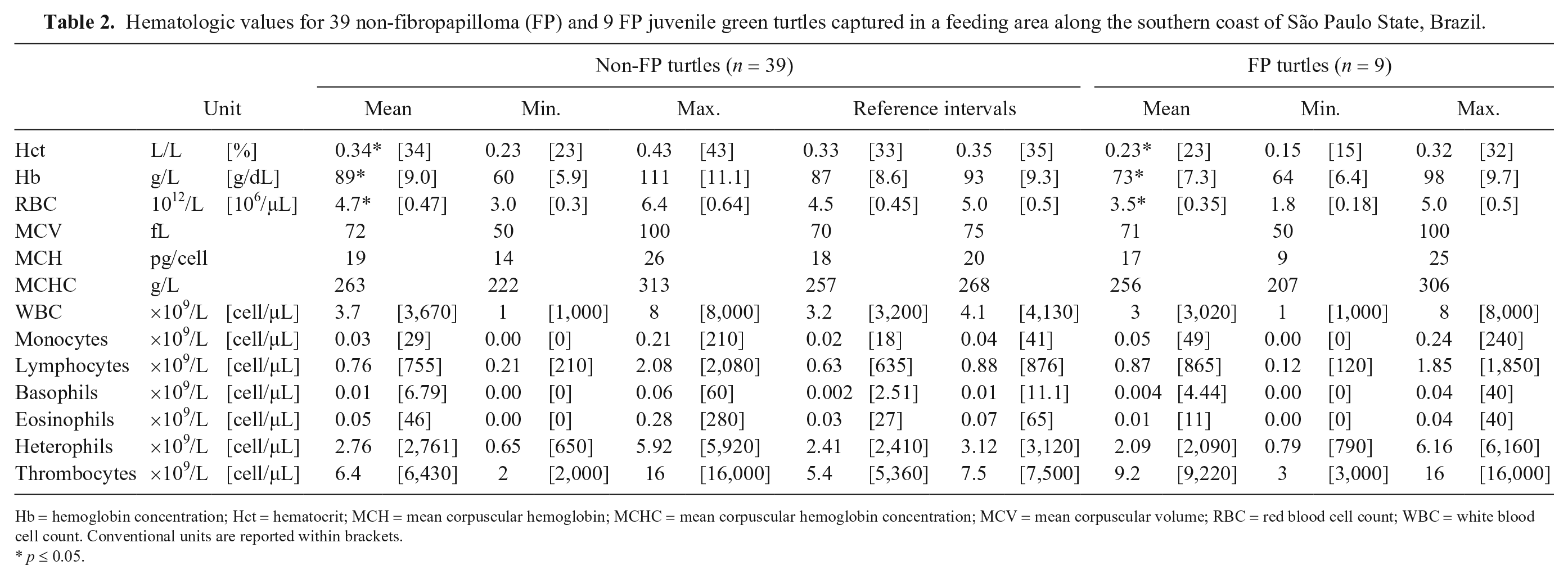
Hb = hemoglobin concentration; Hct = hematocrit; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; MCV = mean corpuscular volume; RBC = red blood cell count; WBC = white blood cell count. Conventional units are reported within brackets.
p ≤ 0.05.
Blood chemistry values for 39 non-fibropapilloma (FP) and 9 FP juvenile green turtles capture in a feeding area along the southern coast of São Paulo State, Brazil.*
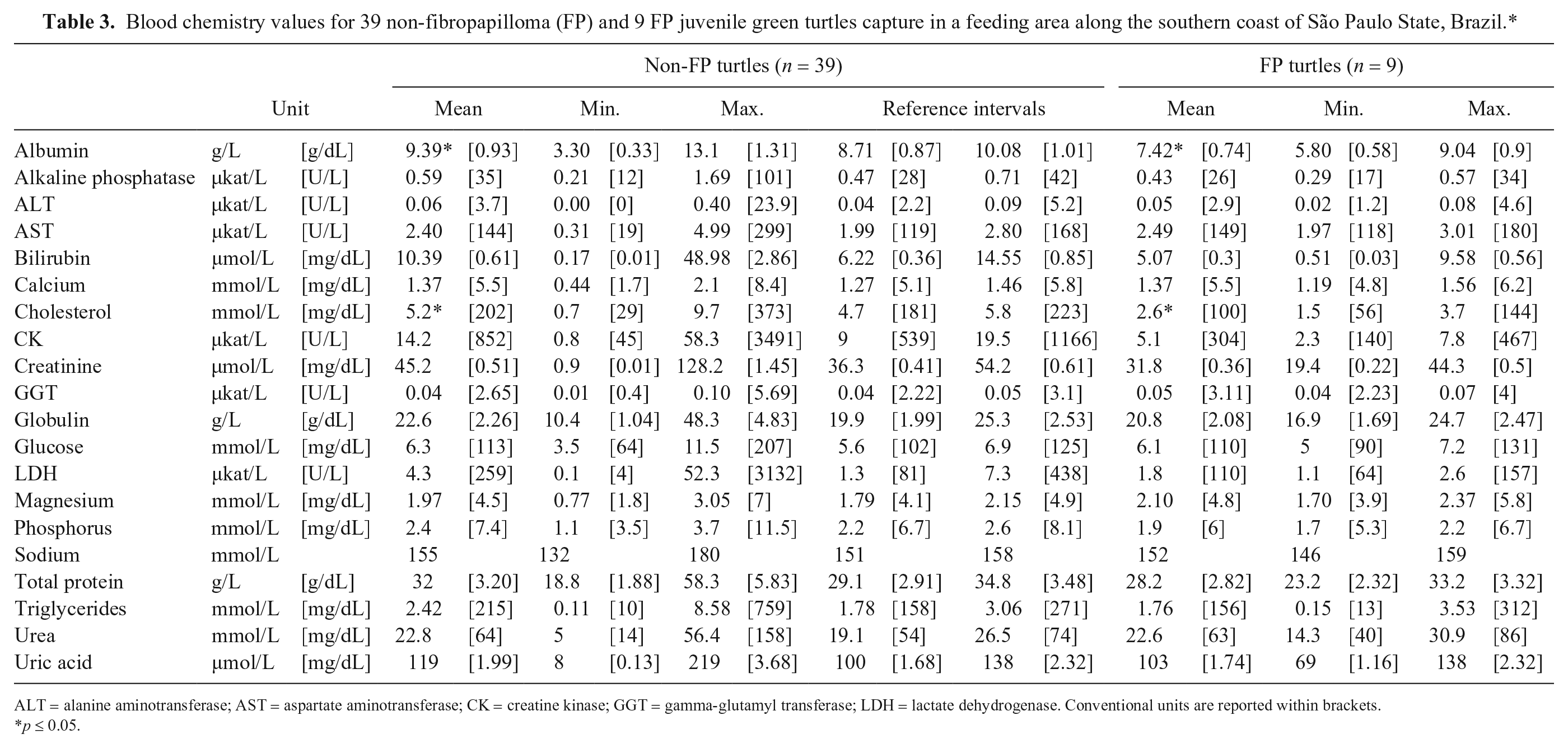
ALT = alanine aminotransferase; AST = aspartate aminotransferase; CK = creatine kinase; GGT = gamma-glutamyl transferase; LDH = lactate dehydrogenase. Conventional units are reported within brackets.
p ≤ 0.05.
Blood values and nutritional status
The nutritional score determined by visual inspection of plastron curvature was good for most animals (n = 39), whereas 9 animals were scored as fair, and none were classified as poor. Animals with fair body condition exhibited significantly lower levels of Hct (mean = 0.24), Hb (mean = 61 g/L), and RBC (0.34 × 1012/L) than those in good condition: Hct (mean = 0.34), Hb (mean = 89 g/L), and RBC (0.48 × 1012/L; t = 5.0, p < 0.001; t = 2.67, p = 0.01; t = 4.08, p < 0.001, respectively; Supplementary Table 2); additionally, turtles with good body condition had higher levels of serum albumin (mean = 9.8 g/L), cholesterol (mean = 5.02 mmol/L), and phosphorus (mean = 2.4 mmol/L) than animals with fair body condition: albumin (mean = 7.2 g/L), cholesterol (mean = 2.79 mmol/L), and phosphorus (mean = 1.9 mmol/L; t = 2.94, p = 0.005;t = 3.27, p = 0.002; t = 2.02, p = 0.04, respectively; Supplementary Table 3).
Blood values and external features
The Hct and RBC values decreased as the CCL increased (r = −0.51, p = 0.01; r = −0.47, p > 0.01, respectively; Fig. 1). Serum cholesterol, sodium, and uric acid were negatively correlated with CCL (r = −0.53, p = 0.0001; r = −0.38, p = 0.007; r = −0.35, p = 0.014; Fig. 2).
Blood values and epibiont prevalence
Turtles with epibionts had significantly lower Hb (mean = 86 g/L) than turtles without epibionts (mean = 94 g/L; t = −2.09, p = 0.04). The WBC was not different between turtles with (mean = 3.6 × 109/L) and without (mean = 3.3 × 109/L) epibionts (t = 0.69, p = 0.49). However, the area occupied by epibionts was positively correlated with the WBC count (r = 0.37, p = 0.03).
Turtles with epibionts had higher levels of cholesterol (mean = 4.9 mmol/L), sodium (mean = 153 mmol/L), and uric acid (mean = 128 µmol/L) than turtles without epibionts: cholesterol (mean = 3.6 mmol/L), sodium (mean = 149 mmol/L), and uric acid (mean = 82 µmol/L; t = 2.09, p = 0.04; t = 2.17, p = 0.03; t = 2.21, p = 0.03, respectively).
Blood values and fibropapilloma prevalence
Turtles with FPs had significantly lower Hct, Hb, and RBC counts than did turtles without the disease (t = 5.99, p = 0.0001; t = 3.66, p = 0.0006; t = 3.80, p = 0.0004, respectively; Table 2). The mean serum albumin and cholesterol levels were higher in non-FP turtles than those with FPs (t = 2.58, p = 0.01; t = 3.73, p < 0.001, respectively; Table 3).
Eight FP turtles were classified as “mild” according to the FPISWA index. The FPI and PAPI (cm2) were negatively correlated with serum total protein (r = −0.70, p = 0.04; r = −0.73, p = 0.02) and albumin (r = −0.77, p = 0.01; r = −0.79, p = 0.01), and positively correlated with triglycerides (r = 0.89, p = 0.001; r = 0.90, p = 0.0008, respectively; Fig. 3). FPI index and the surface area calculation of papilloma had similar correlations to albumin, total protein, and triglycerides levels (Fig. 3).
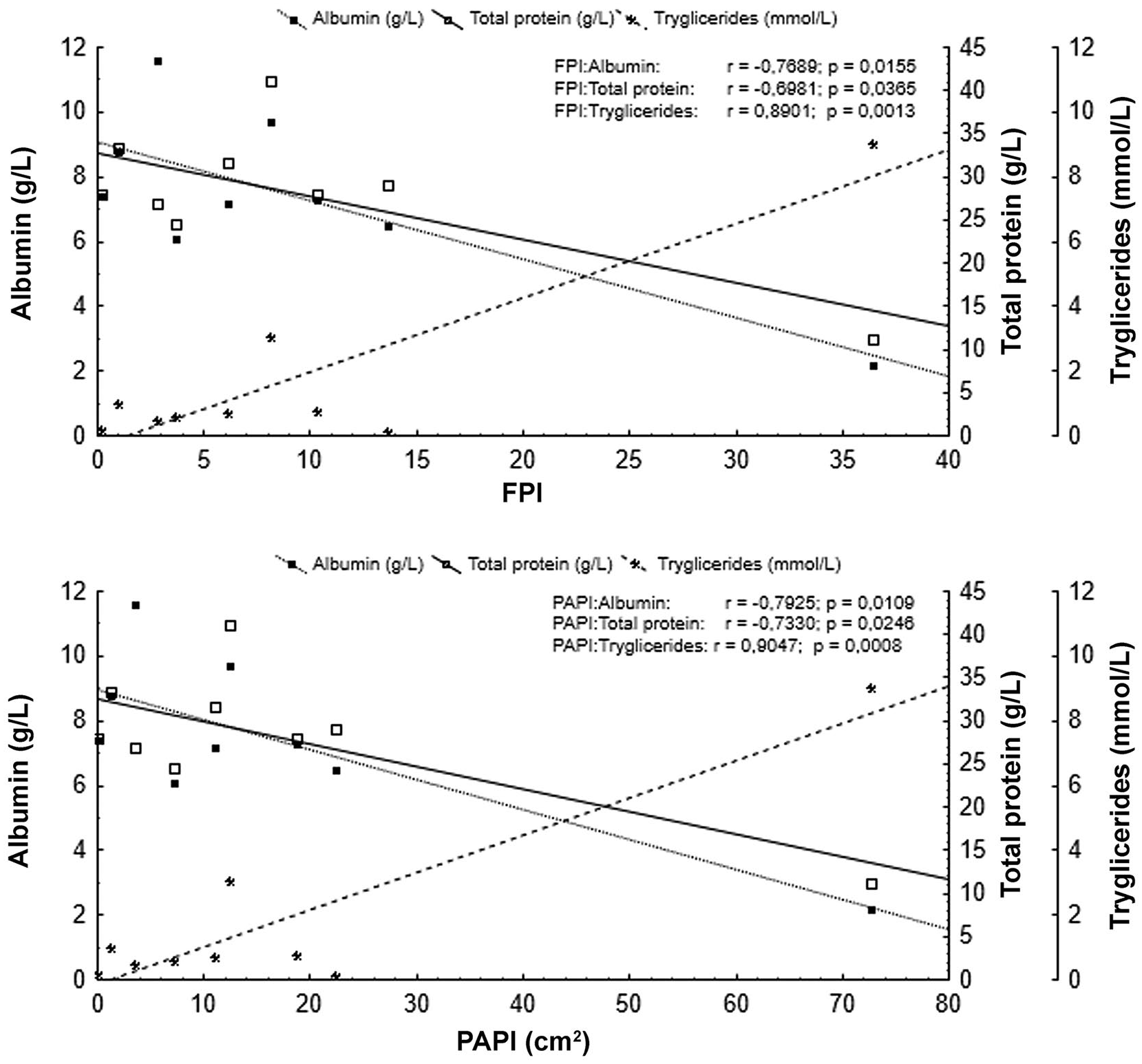
Serum albumin and total protein of juvenile green turtles from southern São Paulo State, Brazil, were negatively correlated with the occurrence and severity of fibropapillomatosis (FPI), whereas serum triglycerides were positively correlated. PAPI = papilloma area.
Discussion
Most of the turtles sampled at the Lagamar estuarine lagoon complex were apparently healthy based on analysis of blood values, nutritional score, and absence of FPs or other disease. In general, the blood RIs of non-FP turtles are within those reported for clinically healthy wild juvenile green turtles from the United Arab Emirates, Hawaii, and Mexico,2,21,34 with the exception of the WBC count, which was lower in turtles from Brazil (mean = 3.7 ± 1.8 × 109/L) than in Hawaii (mean = 9.3 ± 4.4 × 109/L). When comparing the blood cell values from turtles of our study with juvenile green turtles sampled in other studies on the northern coast of São Paulo, Brazil, the non-FP individuals from our study had higher values of Hct, Hb, and RBC, and lower WBC counts.31,47 All nutrients but magnesium, which was lower in our study, were within the RIs for healthy juvenile green turtles sampled at Espírito Santo State, Brazil. 11 The 9 turtles that were considered unhealthy had lower Hct, Hb, and RBC values than non-FP turtles, which was also observed in FP turtles from Hawaii, 2 Florida, 19 and Indonesia. 1
Food intake deficiencies may occur when wild turtles are injured or ill, which could be the result of reduced body movements caused by fishing gear entanglements, limb amputation, presence of FP on the eyes or limbs, and plastic ingestion, among others, that leads to inappropriate food intake and poor nutritional status. 27 Turtles with good nutritional score from our study had higher levels of serum albumin, cholesterol, and phosphorus than did turtles with fair nutritional score. Animals with fair nutritional score also had lower mean values of Hct and Hb and were also more likely to have grossly evident FPs, similarly to C. mydas in Hawaii and southeastern Brazil.11,46 Why some individuals had poorer nutritional scores is not clear. The occurrence of FPs appears to be directly related to a poorer nutritional score, although anthropogenic disruptors such as fishing interaction and marine debris ingestion should be considered. Debris ingestion followed by death is the most common reason for green turtle admission to the IPeC rehabilitation center (Barreto A. Gerenciamento e execução do projeto de monitoramento de praias da bacia de Santos–Fase 1. [Management and execution of the Santos Basin beaches monitoring project–Phase 1]. Version 3, Relatório Técnico Anual, 2016/2017. Portuguese. Available at: https://docplayer.com.br/82991326-Gerenciamento-e-execucao-do-projeto-de-monitoramento-de-praias-da-bacia-de-santos-fase-1.html), and debris ingestion is known to cause health issues related to internal injury, dietary dilution, and malnutrition. 27 Turtles and other reptiles with better nutritional condition are expected to have higher serum albumin, cholesterol, and phosphorus as these biomarkers are often correlated with food ingestion.5,9,21
The prevalence of FPs in wild sea turtles is related to the presence of chelonid herpesvirus 5; however, its clinical manifestation varies based at least in part on the individual’s size, the population’s habitat, pollution, and habitat degradation (i.e., low environmental quality).35,14,22 Nineteen percent of the green turtles from our study had FPs, which represents proportionally fewer affected individuals than values found in other areas on the Brazilian coast.31,35,38 The Lagamar estuarine complex is surrounded by protected areas with low anthropogenic disturbance, whereas the turtles sampled from other parts of Brazil feed within diverse habitats, such as populated and isolated beaches, near shore islands, and heavily polluted areas.11,31 The lower incidence of FPs in turtles from Lagamar may be a result of a better environmental quality and overall better health.
Larger individuals in our study were more likely to have clinical signs of FP. This can be of concern in terms of population survivorship, given that affected turtles are less likely to survive (Wyneken J, 2001). Apparently, there is a predominance of FPs in juveniles of 40–70 cm CCL, rather than smaller individuals (CCL < 40 cm) or adults (CCL > 100 cm). 14 Full-grown individuals are less likely to have clinical FPs, and the reasons behind this remain unclear. FP regression has been reported in Brazil18,38 and could offer a partial explanation. On the other hand, most juvenile FP turtles may not survive the disease and may die before reaching adult size. 14
The Hct and RBC values decreased in our study as animals became larger (CCL) and heavier. These decreased RBC parameters may be attributable to chronic disease, in this case fibropapillomatosis. Usually, an increase in the Hct and Hb cell parameters are observed as sea turtles grow and gain weight.15,16,35,43 The lack of turtles from different age classes (juveniles and adults) in our study did not allow for meaningful evaluation of variation between CCL and blood values from non-FP individuals.
Serum cholesterol, sodium, and uric acid values were lower in turtles with higher CCL and weight. Again, these lower values may be a result of compromised health given the prevalence of FPs together with decreased body condition, which tended to be more frequent in larger animals. Similarly, an inverse relationship between uric acid and CCL was observed in green turtles sampled in the Bahamas. 8 Uric acid is a primary catabolic end-product of protein, and its lower serum levels may be directly related to inadequate protein intake. 9 Serum cholesterol levels are also related to food intake. 21 Lower sodium levels can be associated with kidney or gastrointestinal disorders9; further investigation would be necessary to identify the primary cause of this difference. Variations in dietary preferences can be observed among green turtles from different age classes, 36 and these differences would be more evident in our study if individuals from different age classes had been sampled.
Most of the turtles in our study had epibionts (69%). Similar results were observed in 13 stranded loggerhead turtles (62% had epibionts) along the coast of Georgia, USA, but not in the foraging (n = 39) or nesting (n = 31) individuals (29% and 6% had epibionts, respectively). 10 In our study, the Hb concentration was lower in turtles with epibionts, and the WBC count was positively correlated with the area occupied by epibionts on the plastron. Unhealthy green turtles from Australia had increased probabilities of having a high number of epibionts on the plastron relative to healthy individuals. 13 Most turtles with fair nutritional status from Lagamar have epibionts. However, the Hb level and WBC of turtles with epibionts from our study were within the values found for clinically healthy green turtles from Espírito Santo State, Brazil, and the United Arab Emirates.11,34 It is possible that unhealthy turtles tend to have more epibionts, but their presence does not necessarily indicate health deterioration.
Serum cholesterol, sodium, and uric acid were higher in turtles with epibionts. Despite this difference, these values are within the RI found for healthy green turtles in studies from Brazil, Mexico, and Hawaii.3,11,21 Epibiotic load can vary according to environmental characteristics such as water temperature, current, or salinity. 37 Moreover, the low number of individuals in our study may be considered a limiting factor, and results must be interpreted with caution. Variations in serum cholesterol, uric acid, and sodium were observed in turtles based on seasonality in the Mexican coast, and the authors attributed these variations to the food supply and foraging. 21
The juvenile green turtles with FPs were considered unhealthy and had lower values of Hct, Hb, and RBC than those measured in healthy individuals. The relationship between FPs and anemia has been explored in several studies that analyzed the hematologic profiles of sea turtles.1,19,30,44 The causes of anemia in turtles with FP were not fully elucidated. Anemia may result from a combination of factors, including chronic disease, 2 difficulty feeding with limited or obstructed vision and locomotive ability,1,47 or parasitism, especially trematodes. 1 External blood loss through traumatized well-vascularized regions of the body affected by FP, such as caused by contact with fishing nets and sea substrates, might also contribute.
FP tumor severity was directly related to lower total serum protein in 307 juvenile green turtles from Florida, as well as in 120 green turtles from Hawaii.19,45 Similarly, and despite the low number of FP individuals of our study (n = 9), there was a negative correlation between FPI and papilloma area to total protein and albumin. In our study, serum albumin and cholesterol levels were higher in non-FP turtles than in FP turtles, similar to juvenile green turtles from Hawaii, 3 probably given lower food consumption by FP turtles. FP tumors may impede the turtles’ food intake, leading to nutritional deficiencies. 45 Similar results were observed in green turtles sampled in a heavily polluted area located in Espírito Santo State, Brazil, where there was a positive correlation between body score index (BCI) and cholesterol as well as between BCI and total protein in healthy green turtles. 11 The prevalence of FPs was a good indicator of diminished health in affected turtles in the Lagamar area. Future investigations should focus on these factors using individuals of larger sizes; attention should also be given to their health status and survivorship probability.
Supplemental Material
Supplemental_material – Supplemental material for Health assessment of juvenile green turtles in southern São Paulo State, Brazil: a hematologic approach
Supplemental material, Supplemental_material for Health assessment of juvenile green turtles in southern São Paulo State, Brazil: a hematologic approach by Daniela M. D. de Mello and Maria C. L. Alvarez in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank IPeC staff involved in the logistics, capture, and handling of turtles in the field, especially Daniela Godoy, Eduardo Pereira, Sergio Carlos Neves, and Lisa de Oliveira.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Instituto de Pesquisas Cananéia (IPeC).
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
