Abstract
Canine brucellosis is an infectious and contagious disease associated with reproductive losses in breeding kennels. As a zoonotic disease, it poses a risk to human health, especially for veterinarians and breeders who handle materials potentially contaminated with Brucella canis. However, canine brucellosis is a neglected and underestimated disease given the difficulties in establishing a definitive diagnosis. We evaluated the frequency of detection of B. canis in 5 breeding kennels by using various serologic methods and PCR. Circulation of B. canis in these kennels was confirmed by bacterial isolation. The frequency of positive serologic results varied from 6.3% by AGID to 16.5% by dot-ELISA. There was no positive serology for smooth Brucella. PCR testing was positive in 13.9% of samples. The only detection tests with reasonable agreement were PCR and 2ME-MAT. The diagnosis of canine brucellosis remains challenging. The use of a single laboratory method, or even the use of different laboratory methods, may not be sufficient to reach a definitive diagnosis.
Introduction
Canine brucellosis is a zoonosis whose etiologic agent is Brucella canis, although other Brucella species can infect dogs, including B. melitensis, B. abortus, and B. suis.12,82 The disease is transmitted mainly through the oronasal route or contact with a fetus, fetal fluids, placenta, and vaginal discharge after abortions.39,82 Venereal and intrauterine routes are also important for the transmission of B. canis.39,54,76,82 Canine brucellosis is associated with metritis, placentitis, and abortion in females, and epididymitis, orchitis, and prostatitis in males.40,82 In addition, B. canis infection is associated with birth of weak pups and high neonatal mortality rates.14,36,59 Therefore, the disease is responsible for economic losses in commercial kennels.35,39,82
Despite being a zoonosis, few cases of human infection have been reported, which supports the notion that B. canis has low zoonotic potential.21,51,52,67,81 However, the real importance of B. canis infection in human patients may be underestimated because, in most cases, this pathogen causes subclinical infection, which makes the diagnosis challenging.48,52,74 In addition, most serologic tests used in the diagnosis of human brucellosis employ smooth Brucella antigens, which are ineffective for detecting B. canis. 2
Similar to other Brucella species, B. canis causes an occupational disease that affects mostly veterinarians or kennel workers who have close contact with dogs and their secretions, particularly fetal tissues or fluids, placenta, or vaginal discharge, and also laboratory personnel who may be exposed to large amounts of bacteria.21,48,51,52,55,81 Infections of dog owners have been reported, highlighting the relevance of canine brucellosis as an emerging urban zoonosis.21,51,67 Given the demand for companion dogs, and the deficiency in detection of canine and human B. canis infection, this pathogen may become a serious public health issue. 39
The diagnosis of canine brucellosis is problematic, given that the available serologic tests have low sensitivity and specificity.11,42,83 Definitive diagnosis is supported by bacterial isolation followed by PCR, but culture is not widely available because it is time-consuming and requires a laboratory with proper biosafety conditions given that this pathogen poses a potential health risk to laboratory workers as a result of aerosol transmission.12,72,81,82 Therefore, serologic methods are commonly used in the diagnosis of canine brucellosis. The most common serologic tests include: agar gel immunodiffusion (AGID), rapid slide agglutination test (RSAT), tube agglutination test (TAT), microagglutination test (MAT), complement fixation test (CFT), and several ELISA protocols with different antigens.2,5,8,9,25,30–32,37,53,56,57,62,64,70 Despite being simple to perform, serologic tests have limitations such as nonspecific reactions, especially in agglutination tests, which are more likely with hemolyzed sera, samples with high lipid content, or because of cross-reaction with other pathogens, including Pseudomonas spp., Bordetella bronchiseptica, Streptococcus spp., Staphylococcus spp., Salmonella spp., Yersinia enterocolitica, and Escherichia coli.5,10,12,13,33,41,46,56,63,82 Cross-reaction and false-positive results are eliminated when B. canis cytoplasmic antigens are employed in AGID. 89 AGID sensitivity is 27.9–52.9% and is influenced by the type of antigen employed.42,46 MAT has sensitivity of 66.7–88.9% and specificity of 100%. 15 False-positive reactions as a result of hemolysis do not occur with MAT, as are often observed with AGID. 41 Serum treated with 2-mercaptoethanol (2-ME) prior to AGID or immunochromatographic tests increases specificity, but significantly lowers the sensitivity of these tests.42,46,83 Indirect immunofluorescence has also been employed for the detection of B. canis infection. 84
Previous studies, mostly based on serology, have demonstrated the presence of B. canis in various Brazilian regions, affecting dogs in commercial kennels, households, or street dogs in urban or rural areas.3,16,17,23,24,26,27,50,65,73,77,80 However, marked variations in B. canis infection frequency or prevalence among different surveys have been observed, possibly as a result of the use of different tests. Our goal was to compare different serologic methods and PCR used in the detection of B. canis infection in dogs.
Materials and methods
Kennels and sampling
We evaluated samples from 254 dogs (161 females and 93 males) belonging to 5 breeding kennels located in Belo Horizonte, State of Minas Gerais (MG), Brazil (n = 129 from 3 kennels) or Vila Velha, State of Espírito Santo (ES), Brazil (n = 125 from 2 kennels). Our study was advertised at the breeders’ association, and enrollment was voluntary. All dogs, except for neonates, from each kennel, were sampled and included in the study (Table 1).
Characterization of 254 dogs in 5 breeding kennels from 2 regions of Brazil sampled in 2016 to assess the agreement of PCR and serologic tests for Brucella canis.
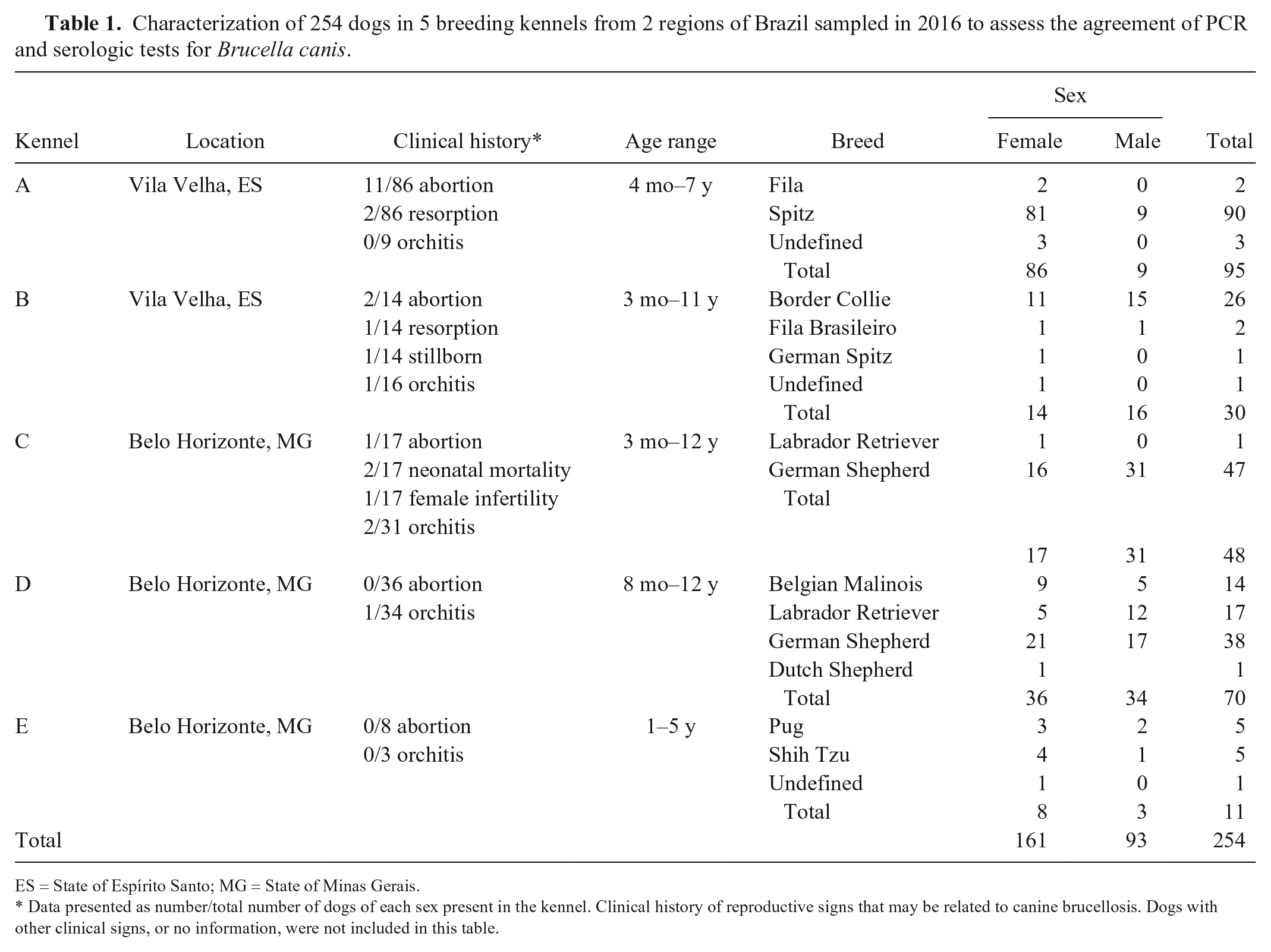
ES = State of Espírito Santo; MG = State of Minas Gerais.
Data presented as number/total number of dogs of each sex present in the kennel. Clinical history of reproductive signs that may be related to canine brucellosis. Dogs with other clinical signs, or no information, were not included in this table.
History of abortion and previous diagnosis of brucellosis were investigated. Samples were collected between February and July 2016. Serum and whole blood samples were collected from all dogs for serologic tests (AGID, rose Bengal plate test [RBPT], CFT, MAT, and dot-ELISA) and PCR. Vaginal swabs from 52 bitches and placentas from 2 puppies (all that were made available from February to July 2016) were processed for bacterial isolation to confirm B. canis circulation in the kennel. Experimental procedures were approved by the Ethics Committee of the Federal University of Minas Gerais (CEUA/UFMG, protocol 56/2016).
Isolation and identification of Brucella spp
Samples of placentas from 2 puppies from kennel A, and vaginal swabs from 52 bitches from kennels B (n = 9), D (n = 35), and E (n = 8) were used for bacterial isolation. Tissue samples were stored at −80°C, and swabs with Stuart medium were kept frozen at −20°C until further processed. For bacterial isolation, placentas were thawed and homogenized with a scalpel blade in 500 µL of sterile PBS; samples contained in swabs were resuspended in 300 μL of sterile PBS and homogenized. Placenta and vaginal swab homogenates were inoculated (100 μL) on tryptose agar (BD Difco, Franklin Lakes, NJ) without or with selective supplementation (2,500 IU of polymyxin B; 12,500 IU bacitracin; 50,000 IU of nystatin; 50 mg of cycloheximide; 2.5 mg of nalidixic acid; and 10 mg of vancomycin; MilliporeSigma, St. Louis, MO), and in 10 mL of tryptose broth (BD Difco) containing selective supplement. Plates were incubated at 37°C with 5% CO2, and bacterial colony growth was checked every 48 h. Broth tubes were incubated at 37°C with 5% CO2 for 7 d and then 100 μL was inoculated on antibiotic tryptose agar. Plates were kept incubated for 21 d and considered negative in the absence of bacterial growth in that period. 58 Colonies with morphology compatible with the genus Brucella were identified based on an acriflavine test, 2 PCR for the bcsp31 gene, 7 and B. ovis–specific PCR. 88
For the detection of Brucella sp. genomic sequences in canine whole blood samples, 250 μL of blood were processed for DNA extraction. 68 After extraction, DNA concentration was measured and adjusted to 250 ng/μL. Bacterial colonies were collected from agar plates into 200 µL of sterile PBS, and heat-killed (100°C for 1 h) for DNA extraction as described previously. 68 For B. ovis–specific PCR, 22 μL of SuperMix PCR (22 mM Tris-HCl pH 8.4, 55 mM KCl, 1.65 mM MgCl2, 220 μM dGTP, 220 μM dATP, 220 μM dTTP, 220 μM dCTP, and 22 U/mL of recombinant Taq DNA polymerase; Invitrogen, Carlsbad, CA), 0.5 μL of each primer (25 μM; 5’-GCCTACGCTGAAACTTGCTTTTG-3’ and 5’-ATCCCCCCATCACCATAACCGAAG-3’), and 2.0 μL of DNA sample were used. Amplification was performed at 95°C for 5 min, 95°C for 1 min, 57°C for 1 min and 72°C for 1 min for 35 consecutive cycles, followed by final extension at 72°C for 5 min, with an expected product of 228 bp, which was analyzed in 1.5% agarose gel stained with SYBR Safe DNA gel stain (Invitrogen). PCR targeted the bcsp31 gene with primers 5′-TGGCTCGGTTGCCAATATCAA-3′ and 5′-CGCGCTTGCCTTTCAAGGTCTG-3′. 7 PCR reaction was performed with 2.5 μL of sample DNA, 18.5 µL of Supermix PCR as above, 1.0 µL of each primer (10 µM) in a total volume of 25 μL, with expected product of 223 bp. Amplification parameters were: 94°C for 3 min; 40 cycles (94°C for 30 s, 60°C for 30 s, 72°C for 30 s), and 72°C for 10 min. The amplified products were subjected to 1.5% agarose gel electrophoresis stained with SYBR Safe DNA gel stain and examined in an ultraviolet light transilluminator.
For the acriflavine agglutination test, 30 μL of a 0.001% aqueous solution of acriflavine was mixed with a bacterial colony and classified as smooth or rough strain in the absence or presence of agglutination, respectively. Isolates were identified as B. canis based on a positive PCR result for the bcsp31 gene (which identifies the genus Brucella), a negative PCR result for B. ovis, and if classified as rough in the acriflavine test.
Serologic tests
Samples were coded, and test results were not made available to the personnel performing the tests until all laboratory procedures were completed.
Agar gel immunodiffusion
AGID was performed using a commercial antigen based on lipopolysaccharide and cell wall proteins of B. ovis as antigen (TECPAR, Curitiba, Brazil), according to the manufacturer’s instructions. Cross-reactivity between B. ovis and B. canis antigens, when used for AGID, has been demonstrated previously. 62 Briefly, a solution of 1% agarose (Invitrogen) in 0.1 M borate buffer (pH 8.0) and 1 M NaCl was placed on a glass slide. After polymerization of the agar, a template mold was used to form the wells in which serum samples and the control serum were placed alternately, in addition to the antigen, which was placed in the central well. Slides were incubated in a humidified chamber at room temperature and read at 48 h.
Rose Bengal plate test
The RBPT was performed to detect antibodies against smooth Brucella lipopolysaccharide as described previously, 2 using a commercial antigen (Instituto Biológico, São Paulo, Brazil). Serum samples and antigen at room temperature (30 μL of each) were deposited on a glass plate side by side, gently homogenized, and then observed under indirect light. Agglutination was assessed for up to 4 min. The RBPT-reacting samples were submitted to the 2-ME test for confirmation of the result.
Complement fixation test
The CFT was performed in microplates using a microtechnique with incubation at 37°C in the 2 phases of the reaction. 2 The test was carried out in a 96-well U-bottom polystyrene plate, with 25 μL of the test sera inactivated at 58°C for 30 min used in doubling dilutions of 1:4 to 1:64, 25 μL of B. ovis antigen (1:200), and 25 μL of complement, which, after agitating on a microplate shaker, was incubated at 37°C for 30 min. Subsequently, 25 μL of the hemolytic system was added and the plate agitated again and incubated under the same conditions. After the initial 15 min of incubation, further agitation was performed, followed by centrifugation at 4°C at 453 × g for 10 min. The reading was performed by comparison with a hemolysis scale, observing the degree of complement fixation, based on the amount of remaining red cells and the aspect of the supernatant. The result of the test was expressed as titer, with the highest dilution of serum having at least 25% complement fixation. Samples with a titer ≥ 4 were considered positive.
Microagglutination test
Antigen from B. canis M-strain (kindly provided by the Servicio Nacional de Sanidad y Calidad Agroalimentaria -SENASA, Buenos Aires, Argentina) was used for the MAT. On a glass slide, 10 μL of serum and 10 μL of the antigen were placed and then homogenized for 2 min. Reading was performed under light microscopy (10× magnification). Samples were considered positive when any degree of agglutination was observed. The samples that did not agglutinate were considered negative. The results were confirmed using 2-ME. Serum samples (25 μL) and 2-ME 0.2 M (25 μL) were mixed on a glass slide, and after 2 min, 50 μL of the antigen from B. canis M-strain was added to the mixture, which was homogenized by gentle rotation for 2 min. The reading was made under an optical microscope (10×), and samples with agglutination were considered positive.
Dot-ELISA
Dot-ELISA was performed (ImmunoComb canine Brucella antibody test kit; Biogal-Galed Laboratories, Kibbutz Galed, Israel) according to the manufacturer’s instructions. Briefly, the serum was placed in a well of line A of the development plate, which contains 12 wells. Subsequently, a comb supplied by the manufacturer was immersed into the wells with the samples and transferred to the following wells (lines B–F) after intervals of 2–5 min of incubation at room temperature. At the end of the procedure (line F), samples were considered positive when a point referring to the test sample was revealed along with the other point corresponding to the positive control.
Statistical analysis
Agreement between test methods was evaluated by kappa test. GraphPad Quick Calcs software was used for statistical analyses. Considering the absence of a suitable gold standard test, latent class analysis (LCA) was performed (Stata software v.15; StataCorp, College Station, TX). Bayesian LCA modeling was performed to assess the probability of positivity based on different outcomes of each test (i.e., to estimate the disease status of each animal). Disease status of each animal (positive or negative) was the latent variable, and the goal of the model was to estimate if any given animal was positive or negative based on the results of the laboratory tests. Results of PCR, AGID, CFT, MAT, and dot-ELISA were used to estimate the latent variable of the model (positivity or negativity).
Results
Characterization of kennels according to
Brucella
sp. infection status
Clinical history of reproductive signs, including abortions, premature births, stillbirths, reabsorptions in females or orchitis in males, and/or previous serologic tests for brucellosis was assessed in all 5 kennels included in our study. Three kennels had a history of abortion in 11 of 86 bitches from kennel A; 2 of 14 bitches from kennel B; and 1 of 17 bitches from kennel C. Kennels D and E did not report any abortions. Orchitis was reported in 1 of 16, 2 of 31, and 1 of 34 male dogs from kennels B, C, and D, respectively. Kennels A and E did not report orchitis (Table 1). Kennels A, D, and E had not performed any test for the detection of brucellosis prior to our study. In kennel B, 11 of 30 dogs had previous negative serologic results, whereas the remaining 19 dogs had unknown previous Brucella test results. In kennel C, 5 of 48 dogs had a previous history of positive serology; 36 of 48 had negative results, with unknown serologic history for 7 dogs. In addition, none of the breeders who ordered serologic tests for B. canis were able to specify which test was used.
Kennels A, B, C, and D were considered positive for Brucella. In kennel A, Brucella sp. was isolated from the placenta of 1 of 2 dogs, and in kennels B and D Brucella sp. was isolated from 2 of 9 and 4 of 35 vaginal swabs, respectively. All isolates were PCR positive for bcsp31. In addition, the isolates were acriflavine positive, and therefore classified as rough Brucella. Because these were rough isolates and negative for a B. ovis–specific PCR, they were all identified as B. canis. Bacterial isolation from placental samples or vaginal swabs was employed to confirm B. canis circulation. No isolates were obtained from kennel C, but 2 dogs in this particular kennel had orchitis with positive serology for Brucella and had been previously diagnosed with brucellosis based on PCR, then castrated and treated. No Brucella sp. isolates were obtained from vaginal swabs from kennel E, which had no history of abortion or clinical signs of brucellosis, therefore it was considered negative for Brucella infection.
One of the 7 isolates was cultured from a bitch in kennel B with a history of stillbirth in 2 litters but had never aborted. Regarding the history of serology for brucellosis, all isolates were obtained from animals with unknown serology history (5 of 7) or that had previous negative results (2 of 7). Importantly, all dogs with bacteriologically positive vaginal swabs had negative serologic results in all detection tests performed in our study.
Serologic tests and PCR
Thirty-one of the 254 samples evaluated in our study (12.2%; 95% confidence interval [CI]: 8.4–16.9) were PCR-positive for Brucella spp., 16 (6.3%; 95% CI: 3.6–10.0) were positive by AGID, and 39 (15.3%; 95% CI: 11.1–20.4) were positive by CFT (Fig. 1). RBPT was performed as a screening test to detect anti-smooth Brucella antibodies. In this test, 19 (7.5%; 95% CI: 4.6–11.4) samples were positive and submitted to 2ME-RBPT for confirmation, but all 19 samples were negative by this method. In MAT using B. canis M-strain antigen, 25 (9.8%; 95% CI: 6.5–14.2) samples were positive, and 21 (8.3%; 95% CI: 5.2–12.4) had inconclusive results. These samples (n = 46) were submitted to 2ME-MAT for confirmation. Samples with positive results in MAT were also positive in 2ME-MAT (n = 25), whereas among the 21 samples with inconclusive results, 5 (23.8%; 95% CI: 8.2–47.2) were positive, and 16 (76.2%; 95% CI: 52.8–92.8) were negative in 2ME-MAT.
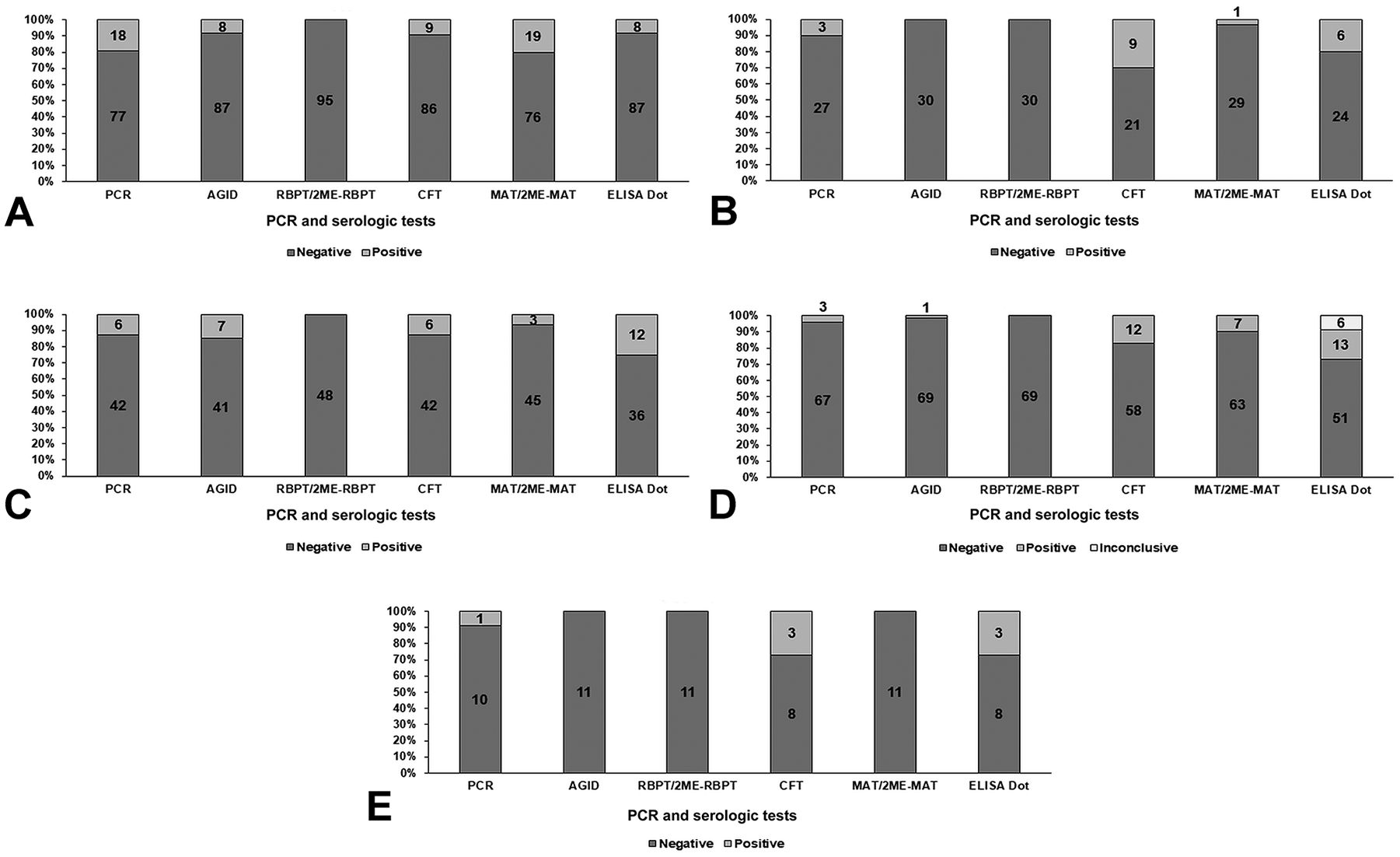
PCR and serologic tests for Brucella sp. performed in samples from dogs from 5 kennels (A–E, designated by panel letter) in Brazil.
In dot-ELISA, 42 (16.5%; 95% CI: 12.2–21.7) samples were positive; 6 (2.4%; 95% CI: 0.9–5.1) samples had inconclusive results, which remained inconclusive after retesting, as recommended by the manufacturer. Most tests performed did not have significant agreement. The only methods with reasonable kappa agreement were PCR and 2ME-MAT (kappa = 0.348; SE of kappa = 0.087; 95% CI: 0.178–0.518).
Latent class analysis
Considering the lack of agreement among most of the tests, as well as the lack of a suitable gold standard, LCA was performed to estimate the probability of correct results based on different test result combinations. Given that the results of PCR, AGID, CFT, MAT, and dot-ELISA were used to estimate the latent variable of the model (positivity or negativity), each combination of results produced a given probability of positivity. Based on that analysis, MAT was the test that most strongly influenced the probability of a positive outcome (Table 2). According to the LCA model and considering probability levels of 77%, 90%, or 97%, the number of positive dogs was 30, 19, or 17 (Table 2).
Latent class analysis of PCR and serologic tests for Brucella canis performed on 254 dogs in 5 breeding kennels from 2 regions of Brazil sampled in 2016 to assess the probability of positive or negative outcomes.
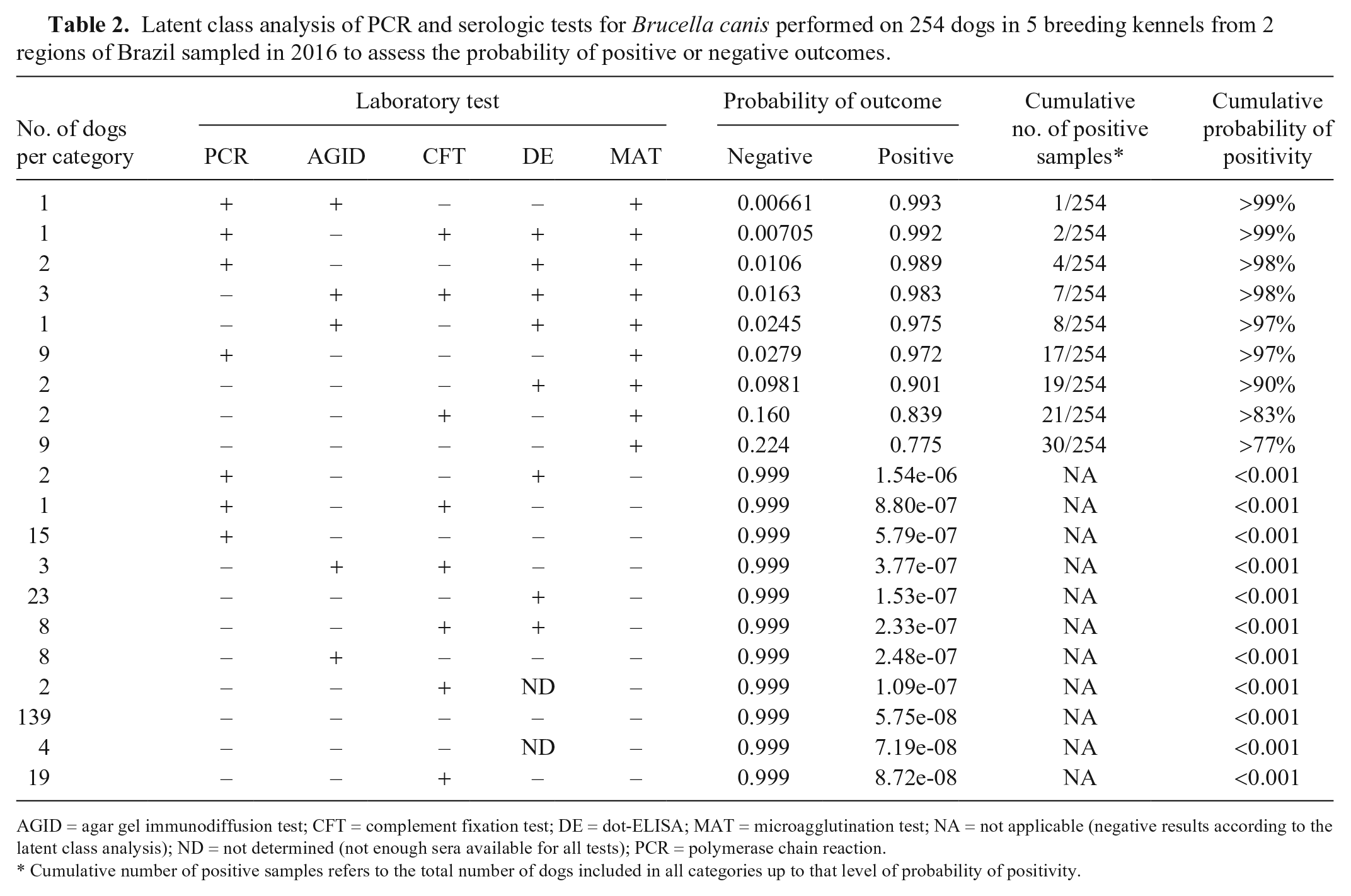
AGID = agar gel immunodiffusion test; CFT = complement fixation test; DE = dot-ELISA; MAT = microagglutination test; NA = not applicable (negative results according to the latent class analysis); ND = not determined (not enough sera available for all tests); PCR = polymerase chain reaction.
Cumulative number of positive samples refers to the total number of dogs included in all categories up to that level of probability of positivity.
Discussion
We clearly demonstrated that the diagnosis of canine brucellosis caused by B. canis remains a challenge even when multiple tests are employed. Although there was no significant agreement between the tests that we used, except for PCR and 2ME-MAT, which had a low level of agreement, LCA demonstrated the highest probabilities of positive outcomes when dogs were positive by MAT. Sensitivity and specificity of individual tests may have influenced the lack of significant agreement between these tests. For instance, AGID has lower sensitivity, 27.9–52.9%,42,46 when compared to MAT, with sensitivity of 66.7–88.9%. 15 In addition, the stage of infection may also influence sensitivity of a particular test. CFT for detection of B. ovis infection in rams tends to result in higher frequencies of positivity during early stages of infection under experimental conditions. 88 Importantly, infected bitches may shed large numbers of B. canis for variable periods after parturition or abortion, 54 but serologic methods do not provide accurate results during the first 12 wk post-infection, and seropositivity is highly dependent on bacteremia. 89 Our LCA model clearly demonstrated that a threshold of higher probability of positive outcome results in decreased numbers of positive dogs, which is an indirect way to demonstrate that higher specificity (lower probability of false-positive results) decreases sensitivity (higher probability of false-negative results) in this model. Lack of agreement among serologic tests as observed in our study could also be the result of a low prevalence of the infection in the studied population; this issue probably did not play a significant role in our study given that a high frequency of brucellosis has been demonstrated previously in some of the kennels included in our study. 78
In Brazil, B. canis infection in dogs has been demonstrated mainly by serologic surveys, with frequencies of seropositive dogs of 0–54.8%.1,3,16,17,23–27,73,77,80 Such studies used a single test method, and AGID with B. ovis antigen was the most widely employed, with or without serum treatment with 2-ME. AGID is a low-cost assay, it is simple to perform, and it has analytical sensitivity levels similar to CFT.28,62,87 However, AGID sensitivity is decreased in chronic infections, 87 and commercial B. ovis antigens have variable quality standards, 87 which may interfere with the results. It should be emphasized that the antigens used in AGID for the detection of B. canis infection in dogs in those studies are from B. ovis.
Studies have shown that treatment of serum with 2-ME, despite increasing the specificity, decreases the sensitivity of MAT such that the frequency obtained in studies that use 2-ME might be underestimated.42,46,83 In our study, AGID had the lowest frequency of positivity (6.3%) compared to other serologic methods, and there was no significant agreement of AGID to any other tests performed. Furthermore, LCA indicated that AGID positivity did not by itself result in a high probability of positive outcome when a model comprising all test results was analyzed.
Several ELISAs are described for serologic detection of canine brucellosis using B. ovis or B. canis antigens.8,9,25,32,37,53,56,57,64,70 The dot-ELISA employed in our study, which uses B. ovis antigens, was described as a serologic test for canine brucellosis in Africa.18,61 In our study, positivity was highest (16.5%) in dot-ELISA compared to the other serologic methods, but there was no significant agreement of dot-ELISA with any other tests performed. In addition, dot-ELISA positive results did not contribute to a high probability of a positive outcome according to the LCA model developed in our study.
By using B. canis M-strain as antigen, the occurrence of false-positive results was reduced.2,20,34 MAT has specificity and sensitivity of 66.7–88.9% and 100%, respectively, 15 and it was the most reliable predictor of a high possibility of positive outcome according to the LCA model developed in our study. In contrast, PCR has often been employed in epidemiologic surveys.27,44–77 Although the only 2 tests with reasonable agreement (kappa = 0.3482) were 2ME-MAT and PCR, the agreement was low.
Given that the bacteremia in canine brucellosis is intermittent, and eventually decreases in chronic infections, PCR results depend on whether the dog is in the bacteremic phase, and if the infection is chronic. 41 As well, PCR results may be influenced by the presence of inhibitors, use of antibiotics, and blood sampling with heparin, which impairs the sensitivity of PCR.43,49,60,69 Conversely, negative results with MAT may be the result of recent infections, given that 3 or 4 wk after infection, negative results are observed even in bacteremic dogs, whereas dogs become MAT positive 8–12 wk after infection. 34 Positivity in MAT tends to decline after bacteremia subsides. Therefore, the infection stage in which clinical samples are collected interferes with the result and must be considered when interpreting the data. Vaginal swabs, semen,44,45 placentas, dead fetuses, and neonates36,78 may represent valuable samples to detect infected dogs even after resolution of bacteremia. 79 As evidenced in our study, all bitches from which B. canis was isolated from vaginal swabs were seronegative in all tests, possibly as a result of chronic infection. 34
Nevertheless, B. canis infection poses a zoonotic risk because of the potential elimination of the bacteria through vaginal secretion. Furthermore, PCR positivity was not associated with high probabilities of a positive outcome according to our LCA model. These results likely are associated with very early stages of infection or chronic infections with low antibody titers. Similar results were observed with B. ovis–infected rams that had serologic and PCR results that did not have significant agreement. 19
Despite being a test with high specificity (65–100%) and sensitivity (77–100%) and considered as confirmatory evidence in campaigns to eradicate bovine and ovine brucellosis in several countries, CFT has not been used routinely in canine brucellosis diagnosis.4,22,25,29,37,66,75,86 In our study, 39 (15.3%) dogs were positive by CFT, but there was no significant agreement with other tests, and CFT positivity was not associated with a high probability of positive outcome according to our LCA model. In Brazil, there is only one previous report of CFT used in canine brucellosis diagnosis; sera from Santana do Parnaíba (State of São Paulo) were tested using CFT as the confirmatory test, and observed a prevalence of 2.2% (9 of 410). 3 The greatest difficulty in using CFT in canine brucellosis detection is the lack of standardization of this technique. In addition, there are other disadvantages inherent to this technique, such as high cost, need for equipment, and trained personnel.47,71
RBPT was performed to detect antibodies against smooth Brucella. Although canine brucellosis is caused mainly by B. canis, dogs in close contact with small ruminants, cattle, and pigs, may be infected by smooth Brucella spp., namely B. melitensis, B. abortus, and B. suis, respectively.6,12,85 In our study, there were no positive samples in 2ME-RBPT, indicating that, in the studied population, canine brucellosis was not associated with smooth Brucella spp. However, some studies have demonstrated anti-smooth Brucella antibodies in dogs, which reinforces the importance of performing tests capable of detecting antibodies to both rough and smooth Brucella spp.1,16,23,73
Despite the lack of significant agreement between most of the tests that we performed, the frequency of dogs positive for Brucella was high. Brucella isolation from placenta and vaginal swabs confirmed B. canis circulation in these kennels, although storage may have reduced the chance of obtaining positive cultures. Indeed, a previous study from our group demonstrated B. canis natural infection in canine fetuses and neonates from kennels located in Vila Velha (ES). 78 However, except for Brucella isolation, a positive result in only one test is not sufficient for a definitive diagnosis, especially if euthanasia is considered for Brucella-positive dogs. 39 Antimicrobial treatment is expensive, prolonged, and may be ineffective, especially in chronically infected dogs, and therefore it is used only in exceptional cases. 34 Importantly, negative test results do not rule out the possibility of infection.
Success in detecting canine brucellosis depends on several factors such as the combination of more than one test, proper sampling of specimens at defined time intervals, and follow-up of dogs to detect clinical signs compatible with brucellosis.36,38 However, because it is laborious and expensive, breeding dogs, or even pet dogs, are not properly tested most of the time, resulting in diagnostic failure, spread of the disease among dogs, and increased risk of transmission to humans. Therefore, the development of novel serologic or molecular techniques capable of detecting brucellosis in dogs is necessary and urgent.
Our study demonstrated that detection of canine brucellosis remains challenging. Even the use of different laboratory methods may not be sufficient for a definitive diagnosis, especially if only blood and serum samples are analyzed. Although laborious, and dependent on adequate laboratory and skilled personnel capable of handling BSL-3 pathogens, bacterial isolation may be necessary.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Work in the RL Santos lab is supported by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil), FAPEMIG (Fundação de Amparo a Pesquisa do Estado de Minas Gerais, Brazil), and CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brazil). RL Santos and TA Paixão have received fellowships from CNPq.
