Abstract
Bacillary hemoglobinuria (BH) is an infectious disease, mostly affecting cattle, caused by Clostridium haemolyticum (C. novyi type D), with acute hepatic necrosis and intravascular hemolysis. Cattle are typically predisposed to BH by liver injury caused by Fasciola hepatica, although cases have been reported in cattle without evidence of this parasite. Here we describe a cluster of 14 BH cases from 7 counties in north-central to central Missouri submitted to a veterinary diagnostic laboratory between December 2020 and April 2023. Postmortem examination in all cases revealed hemoglobinuria and acute hepatic necrosis with large numbers of gram-positive bacilli with terminal-to-subterminal spores. Flukes, fluke ova, and/or fluke pigment consistent with Fascioloides magna were identified in 12 of 14 cases. Sequences of the nuclear ribosomal internal transcribed spacer 1 (ITS1) from one fluke had 100% identity to F. magna. C. novyi was detected by fluorescent antibody testing of liver impression smears (11 of 12 cases) and by immunohistochemistry of liver sections (7 of 7 cases). PCR on formalin-fixed, paraffin-embedded tissues amplified the C. haemolyticum beta toxin gene in each of the 7 cases tested. To our knowledge, a confirmed cluster of BH associated with F. magna has not been reported previously in cattle.
Bacillary hemoglobinuria (BH), also known as red-water disease, is an infectious, non-contagious disease, mostly affecting cattle, caused by Clostridium haemolyticum (also known as C. novyi type D) and characterized by acute hepatic necrosis and intravascular hemolysis. Other gross lesions include icterus, hemoglobinuric nephrosis, abomasal and intestinal hemorrhages, splenic congestion, and effusions and hemorrhages in multiple tissues. BH usually occurs in cattle >1-y-old and is nearly always fatal. 25
C. haemolyticum is closely related to C. novyi type B, which causes a similar condition known as infectious necrotic hepatitis (INH) or black disease, which occurs most commonly in sheep, and occasionally in cattle and horses. The colloquial term “black disease” refers to widespread congestion and hemorrhage of the subcutis, which lends a dark-red to black appearance to the subcutaneous surface of the flayed hides of affected animals. 26
Liver lesions are similar in BH and INH, although BH tends to cause 1, 2, or rarely more large foci of necrosis, whereas INH tends to cause many small foci of necrosis. Intravascular hemolysis and hemoglobinuria are highly characteristic of BH, but they are not features of INH in ruminants. 26
C. haemolyticum and C. novyi type B are both gram-positive, spore-forming, rod-shaped anaerobes that are commonly found in soil and intestinal contents of animals. Spores are highly resistant and may survive on pasture for years. Sources of pasture contamination include feces of livestock and decomposition of dead stock. 19 Spores are ingested by grazing animals, absorbed through the intestinal mucosa, and carried via the portal circulation to the liver and other tissues, where they are engulfed by resident macrophages, in the cytoplasm of which they can remain latent for months. Accordingly, C. haemolyticum has been recovered from the livers of a small number of otherwise healthy cattle, as well as from the bone marrow of carcasses left in the elements for more than one year.19,33
The pathogenesis of BH and INH is usually precipitated by a liver insult that is sufficiently severe to create a local anaerobic environment and promote germination of clostridial spores. Once this occurs, the vegetative form of the bacteria multiplies and produces toxins. In the case of C. haemolyticum, beta toxin is the major virulence factor; this toxin damages the cell membranes of hepatocytes, endothelial cells, and red blood cells, leading to the characteristic lesions of hepatic necrosis, widespread vascular injury, and intravascular hemolysis. 15 In the case of C. novyi type B, alpha toxin is the major virulence factor, which causes similar hepatic necrosis but not intravascular hemolysis. 26 Although BH is typically associated with liver injury caused by Fasciola hepatica, BH has also been reported in cattle without evidence of fluke infection.8,25,26 A role for Fascioloides magna was recently proposed in cases of BH in North Dakota and Minnesota; however, in those cases, the flukes were not identified definitively. 30 Other rarely reported triggers include liver biopsies 28 and hepatic necrosis due to Fusobacterium necrophorum.18,29
Gross and microscopic lesions of BH are characteristic and are usually sufficient to establish a presumptive diagnosis, which can be further supported by fluorescent antibody testing (FAT) and/or immunohistochemistry (IHC). These tests, however, cannot distinguish between C. haemolyticum and C. novyi type B because the antibodies cross-react. 25 Laboratory confirmation via bacterial culture is challenging, as C. haemolyticum is a strict anaerobe and tends to be rapidly overgrown by contaminants. Therefore, a negative culture does not rule out a diagnosis of BH. A PCR for the C. novyi/C. haemolyticum beta toxin gene has been developed. 27 Alternatively, PCR targeted toward unique regions of the flagellin gene fliC can be used to distinguish between C. haemolyticum and C. novyi type B. 32
Here we describe a cluster of BH cases submitted to the Veterinary Medical Diagnostic Laboratory at the University of Missouri (VMDL; Columbia, MO, USA) between December 2020 and April 2023, most of which were associated with F. magna infection. To our knowledge, a confirmed cluster of BH associated with F. magna has not been reported previously in cattle. Prior to December 2020, BH had not been diagnosed at VMDL.
Materials and methods
Animals and clinical history
Between December 2020 and April 2023, the carcasses or tissues of 14 beef cattle from different premises in 7 counties in north-central to central Missouri were submitted to the VMDL (Table 1; Fig. 1). Eleven of these cattle were autopsied at the VMDL. The other 3 were autopsied in the field by referring veterinarians.
Signalment, time of submission, county of origin, clinical history, and samples submitted from 14 cattle diagnosed with bacillary hemoglobinuria at the University of Missouri VMDL.
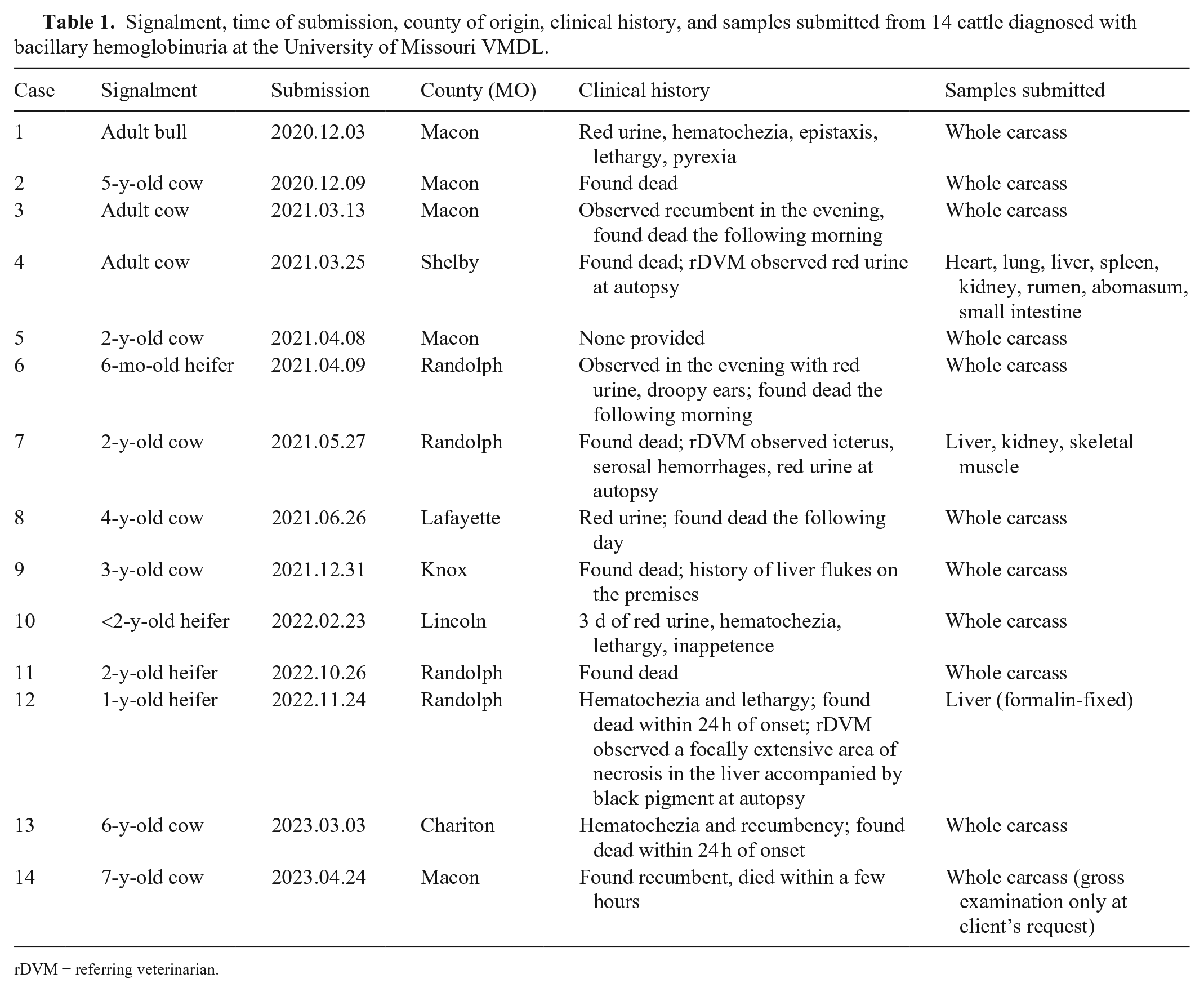
rDVM = referring veterinarian.
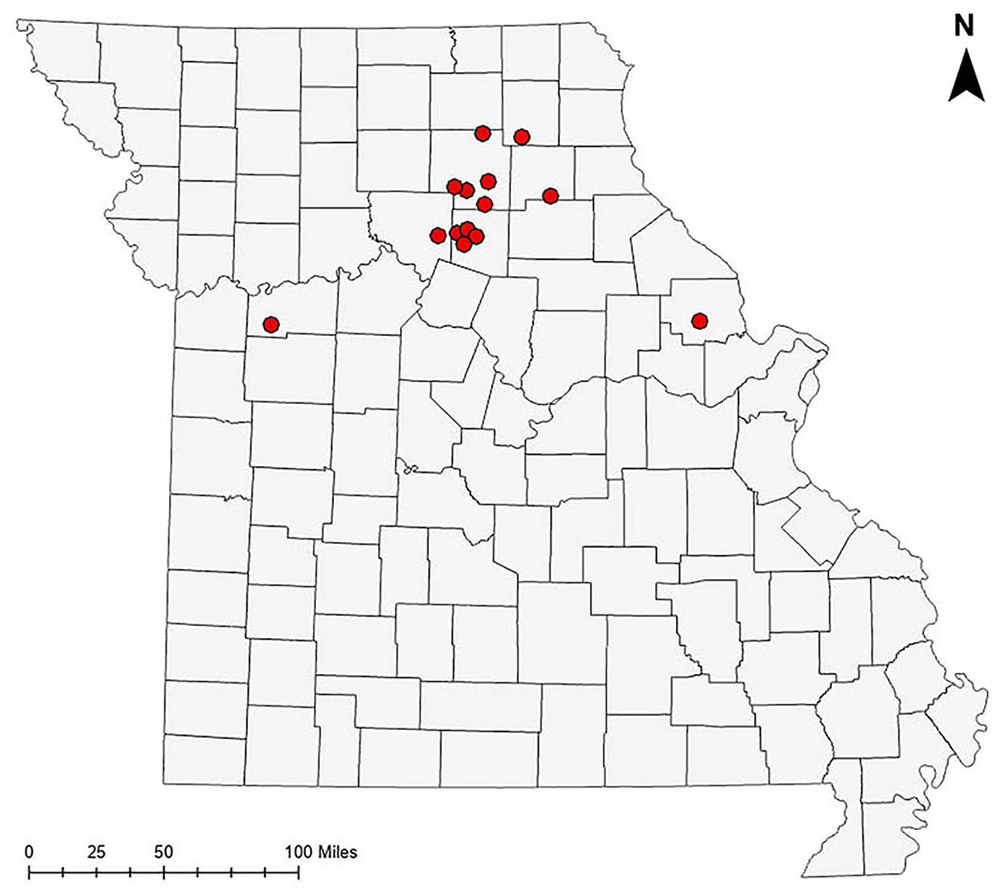
Map of Missouri. Red circles represent the premises of origin of the 14 cattle diagnosed with bacillary hemoglobinuria at the University of Missouri VMDL, December 2020–April 2023.
Pathology
Whole carcasses were autopsied at the VMDL according to standard protocols. Case 1 was euthanized via captive bolt at Veterinary Health Center at the University of Missouri (VHC) and autopsied within 45 min of death. All other cases were autopsied within 24–48 h of death. Tissues collected included, at minimum, brain, heart, lung, liver, spleen, kidney, rumen, abomasum, small intestine, and large intestine. Tissues were collected and fixed for ~24 h in 10% neutral-buffered formalin and processed routinely, and 4-µm sections were stained with H&E for light microscopy. Selected liver sections were Gram stained according to the Brown–Hopps method.
Bacteriology
Samples of liver were collected aseptically, inoculated onto pre-reduced tryptic soy agar with 5% sheep blood and pre-reduced phenylethyl alcohol agar, and incubated under anaerobic conditions (<1% O2) at 36°C for 48 h. Anaerobic bacterial cultures were performed in a commercial anaerobic jar system (AnaeroPack; Mitsubishi Gas Chemical). Bacterial identification was accomplished with matrix-assisted laser desorption/ionization-time-of-flight mass spectrometry (MALDI-TOF MS; Bruker).
Ancillary testing for Clostridium spp
Impression smears of liver were made on glass slides, fixed with acetone, and treated with C. novyi polyclonal antiserum conjugated to fluorescein isothiocyanate (VMRD). Fluorescence was visualized via UV light microscopy.
Formalin-fixed, paraffin-embedded (FFPE) sections of liver were processed by an indirect immunoperoxidase technique for C. novyi as described previously. 25 Briefly, a commercial kit (RTU Vectastain Elite ABC-HRP; Vector) was used according to the manufacturer’s instructions. Endogenous peroxidase activity was blocked with a 3% solution of hydrogen peroxide, followed by antigen retrieval with pepsin. Nonspecific binding was blocked by treating the samples with Background Punisher (Biocare Medical) before incubation with goat anti–C. novyi polyclonal antibody (VMRD) for 60 min at 37°C. NovaRED (Vector) chromogen was used for visualization. Tissues incubated with normal goat serum instead of primary antibodies were used as negative controls. Liver from a cow with BH, in which C. haemolyticum had been identified by culture and PCR, was used as a positive control.
PCR for the C. novyi alpha and beta toxin and flagellin genes was performed as described previously.25,27,32 The central regions of the flagellin gene fliC are genotype-specific and can be used to differentiate C. haemolyticum from C. novyi type B. 32 PCR was performed on three 20-µm scrolls of FFPE tissue from each liver tested.
Molecular identification of flukes
Formalin-fixed liver flukes were cleared in lactophenol and examined using compound and dissecting microscopes. A small amount of tissue from a single fluke preserved in 70% ethanol was used to extract DNA in triplicate (DNeasy blood & tissue kit; Qiagen) following the manufacturer’s guidelines. The resulting genomic DNA was used for non-nested PCR with primers targeting the internal transcribed spacer 1 (ITS1) 22 of ribosomal DNA (rDNA) in 50-µL reactions containing 25 µL of 2× master mix (AmpliTaq Gold 360; Applied Biosystems), 0.5 µL of each forward and reverse primer (at 50 pmol/µL), 22 µL of nuclease-free water, and 2 µL of template DNA. PCR cycling conditions started with denaturation for 10 min at 94°C followed by 35 cycles of denaturing for 1 min at 94°C, annealing for 1 min at 50°C, extension for 2 min at 72°C, and a final extension for 5 min at 72°C. PCR products were visualized on a RedSafe 2% agarose gel, and amplified bands were purified (QIAquick gel extraction kit; Qiagen). Sanger sequencing with PCR primers was performed by Genewiz (Azenta Life Sciences). Forward and reverse sequence reads were aligned, trimmed, and compared with publicly available nucleotide submissions with BLAST (https://blast.ncbi.nlm.nih.gov/).
Results
Clinical history and gross pathology
Case 1 was an adult beef bull from Macon County, MO with a history of <12 h of lethargy, hemoglobinuria, hematuria, and hematochezia (Fig. 2). On presentation at the VHC, the bull had a fever of 40.6°C (105.4°F). Rumen contractions were absent, mild epistaxis was noted, and a large hypoechoic liver lesion was identified on ultrasound. The bull was euthanized via captive bolt.
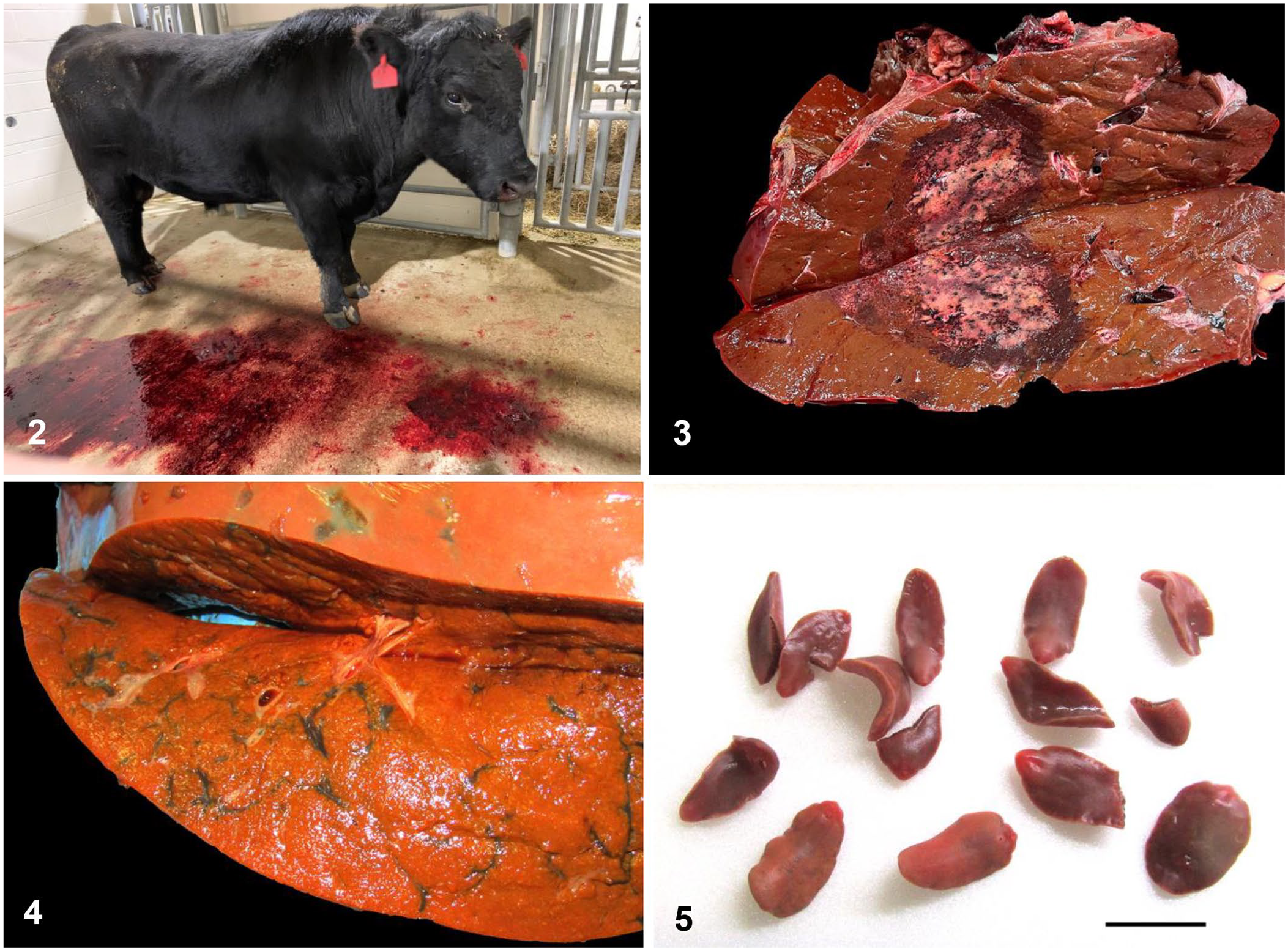
Clinical and gross findings in cases of bacillary hemoglobinuria diagnosed at the University of Missouri VMDL.
Postmortem examination of the bull revealed widespread hemorrhages of the subcutis, serosal surfaces, and splenic capsule. The liver contained an ~25-cm diameter, solitary, dry, tan, sharply demarcated focus of acute necrosis surrounded by a distinct zone of dark-red discoloration (Fig. 3). Liver flukes were not seen; however, a few slender, irregular, linear black tracts were noted within the adjacent liver parenchyma. A hepatic lymph node also contained black pigment.
Over the next 30 mo, 13 additional cases with a history of red urine and/or sudden death were submitted to the VMDL (Table 1; Fig. 1). All were beef cattle and included 9 adult cows and 4 heifers. Ten were submitted as whole carcasses and 3 consisted of tissue samples collected in the field. The 2 animals in the 2 most distant counties (Lafayette County, to the west; Lincoln County, to the east) had been purchased and brought onto the premises within 2 mo before death. The cow in Lafayette County had been purchased in Macon County. The origin of the heifer in Lincoln County was not disclosed.
On postmortem examination, all 13 cases had acute hepatic necrosis similar to that described in case 1. Slender, linear, irregular tracts of black pigment were apparent in the livers of 9 cases (Fig. 4). In 5 cases, flukes were grossly identified within pigmented tracts in the liver parenchyma (Fig. 5). Flukes were 2–3 cm long and ~1 cm wide, red-brown, dorsoventrally flattened, and leaf-shaped with a prominent ventral sucker at the anterior end; an anterior projection (cephalic cone) was absent (Fig. 6). Morphologically, the flukes were consistent with F. magna. 20 Additional gross lesions variably included icterus, red watery fluid in the thoracic, abdominal, and/or pericardial cavities, widespread subcutaneous and/or serosal hemorrhages, and, in case 8, numerous liver abscesses.
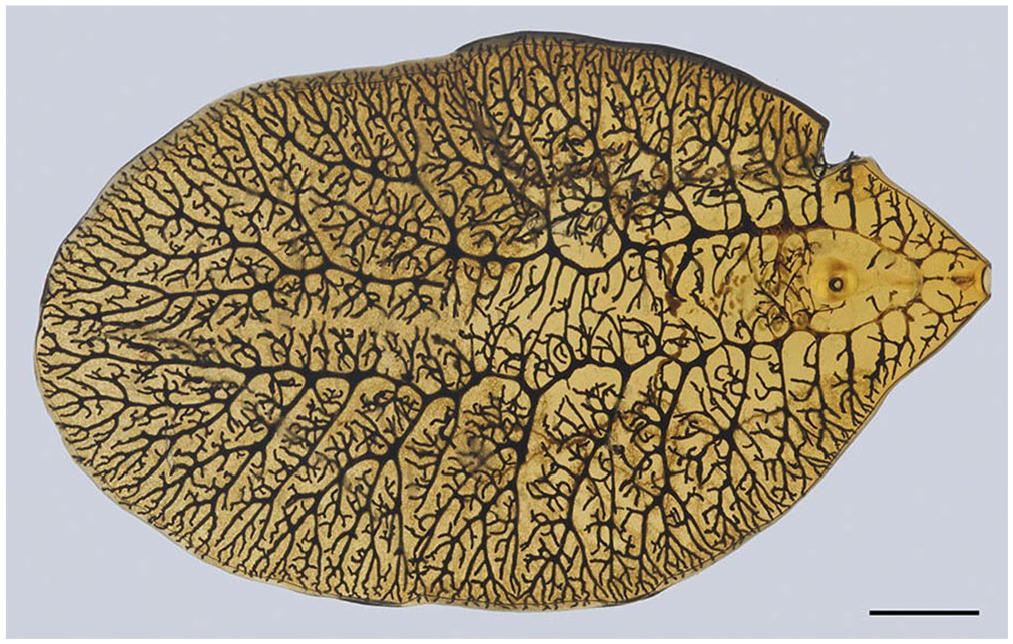
Fascioloides magna is dorsoventrally flattened and leaf-shaped, with a prominent ventral sucker at the anterior end. Bar = 5 mm.
Histopathology
Microscopic lesions were similar in all cases examined. In each case, the liver contained a focally extensive region of acute coagulative necrosis with hypereosinophilic staining, karyorrhexis, and preservation of the architecture of the hepatic cords. These regions were sharply demarcated and separated from adjacent viable tissue by a dense band of neutrophils (Fig. 7). In the periphery of necrotic regions, there were large numbers of gram-positive bacilli, which often contained terminal-to-subterminal spores (Fig. 8). In some cases, adjacent sections of liver parenchyma had marked centrilobular necrosis and/or mild-to-moderate portal fibrosis.
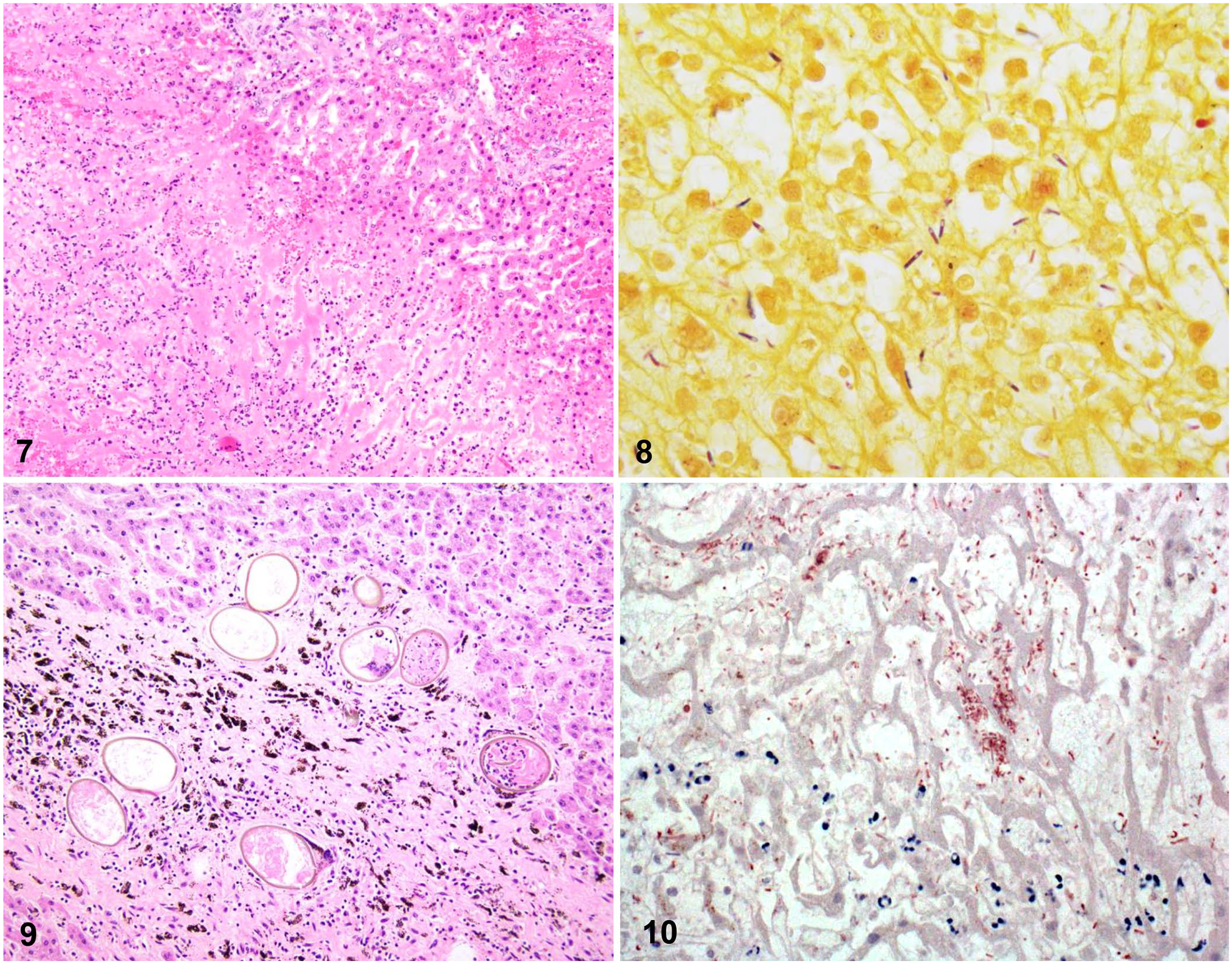
Microscopic findings in cases of bacillary hemoglobinuria diagnosed at the University of Missouri VMDL.
Black pigment associated with migratory tracts was identified grossly in 9 cases and microscopically in 7 cases. In H&E sections, this pigment appeared as brown-black coarse granules, both within macrophages and extracellularly within regions of portal fibrosis (Fig. 9). In case 1, the subcapsular and medullary sinuses of a hepatic lymph node contained abundant coarse granules of brown-black pigment; this pigment did not stain with Mallory iron stain, ruling out hemosiderin. In 5 cases, trematode ova were also identified within regions of portal fibrosis (Fig. 9). Ova were operculated, ~100 × 150 µm, with a 2–4-µm thick brown refractile shell. In total, flukes, fluke ova, and/or tracts of iron-porphyrin pigment were identified grossly and/or microscopically in 12 of 14 cases; 2 cases lacked definitive evidence of fluke infection (Table 2).
Diagnostic evidence of Fascioloides magna and bacterial culture results in the livers of 14 cattle diagnosed with bacillary hemoglobinuria at the University of Missouri VMDL.

Ancillary test results
No significant aerobic or anaerobic bacterial pathogens were isolated from the livers in the 9 cases for which bacterial cultures were performed (Table 2). C. novyi was detected via FAT of liver impression smears in 11 of 12 cases. The bacilli observed in H&E and Gram-stained sections were labeled immunohistochemically with anti–C. novyi antibody in 7 of 7 cases in which this testing was performed (Fig. 10).
The C. haemolyticum beta toxin gene was detected by PCR in all 7 cases in which this testing was performed. The C. novyi alpha toxin gene was detected in 6 cases. The C. haemolyticum fliC gene was detected in 5 cases, and the C. novyi type B fliC gene was detected in 1 case (Table 3).
Results of PCR for the Clostridium novyi alpha and beta toxin genes and the Clostridium haemolyticum and Clostridium novyi type B flagellin (fliC) genes in the livers of 7 cattle diagnosed with bacillary hemoglobinuria.

Molecular analysis of the DNA extracted from the adult fluke and amplified at the ITS1 locus demonstrated 100% identity to F. magna sequences deposited in GenBank. In contrast, the identity to F. hepatica sequences was 94.2% at this locus.
Discussion
In the 14 cases of BH in Missouri beef cattle that we described here, 12 had direct evidence of F. magna in the liver. We found only one peer-reviewed report of BH that was thought to be associated with F. magna, 30 commonly known as the deer fluke; however, in that report, the identity of the fluke species was not confirmed definitively. We retrieved no other confirmed reports in a search of CAB Abstracts, Google Scholar, PubMed, Scopus, or Web of Science using the keywords (“bacillary hemoglobinuria” OR “hemoglobinuria”) AND “Fascioloides magna”. F. magna often causes subclinical infection in cattle and is considered significant only because of its financial impact due to liver condemnations at slaughter.6,11 In contrast, F. magna can be devastating in sheep flocks. 17 Historically, BH has usually been attributed to migration of the cattle liver fluke, F. hepatica. These liver flukes are most prevalent in the southern and southeastern United States, the Great Lakes region, and the Pacific Northwest. 17
According to the 2016 National Beef Quality Audit, flukes were detected in 3.2% of livers of cows and bulls at slaughter in the United States. 14 In the 2000 National Beef Quality Audit, flukes accounted for 21.7% of liver condemnations in fed steers and heifers in the United States. 24 Specific reports of F. hepatica are rare in Kansas and Missouri.4,31 In 1973, 0.6% of 4,695 bovine livers were condemned for “liver flukes” in Missouri, but a distinction between F. hepatica and F. magna was not made, as is typical in most abattoir inspection records. 12 Sporadic cases of liver condemnation due to confirmed F. magna infection have been reported in Missouri cattle. 5
The complex life cycles of F. hepatica and F. magna are dependent on temperature, humidity, and the presence of snail intermediate hosts. Eggs are passed in the feces of definitive hosts and hatch into miracidia on suitably moist pastures. Miracidia invade and develop within several species of semi-aquatic mud snails such as Lymnaea modicella, L. caperata, and L. bulimoides. 7 These species have all been documented in Missouri. 36 After several weeks, cercariae emerge from the snail and encyst on nearby vegetation as metacercariae, the infective stage. Under favorable conditions, infective metacercariae may survive on pastures for months. Once metacercariae are ingested by the grazing host, immature flukes cross the intestinal wall and reach the abdominal cavity within a few hours. In the case of F. hepatica, the entire life cycle takes a minimum of 16 wk. The presence of standing water, with several months of temperatures of 10–30°C (50–86°F), is associated with high risk. 23 Freezing or desiccation will destroy the eggs. The life cycle of F. magna is significantly longer (~7 mo) but otherwise similar, with the important exception that F. magna is unable to establish patent infections in hosts other than cervids. 17 Cattle and sheep, with very rare exceptions in the case of sheep, are dead-end hosts.10,11
At autopsy, F. hepatica adults are found within bile ducts, where they often stimulate chronic fibrosis of the portal tracts. In contrast, F. magna wanders freely within the parenchyma, leaving migration tracts that are often filled with black iron-porphyrin pigment. 1 These tracts often contain ova entrapped within fibrous connective tissue. For antemortem diagnosis of liver fluke infection, F. hepatica eggs can be detected via fecal sedimentation. 17 However, as F. magna infections are non-patent in sheep and cattle, nearly all such infections are diagnosed postmortem, which makes estimates of subclinical infection rates very difficult. Our identification of the flukes in these cases as F. magna was based on their location free within the hepatic parenchyma, as opposed to within bile ducts, the presence of abundant iron-porphyrin pigment, the flukes’ lack of a cephalic cone, 20 and the 100% identity of ITS1 sequence from one fluke to F. magna sequences in GenBank.
In all of our cases, a presumptive diagnosis of BH was established based on gross and microscopic lesions, which was further supported in selected cases with detection of C. novyi by FAT and IHC. Confirmation of the diagnosis was achieved with PCR in 7 cases, via detection of the C. haemolyticum beta toxin gene. 25 C. haemolyticum is a strict anaerobe that is rapidly overgrown by postmortem invaders, including various other Clostridium spp., such as C. perfringens and Paeniclostridium (Clostridium) sordellii; therefore, our inability to isolate C. haemolyticum via bacterial culture is not surprising. The diagnosis was further supported in 5 cases in which the C. haemolyticum fliC gene was detected. Alpha and beta toxin were both detected in one case that was negative for the C. haemolyticum fliC gene, and positive for the C. novyi type B fliC gene, suggesting the presence of the latter in that case. The presence of the alpha toxin gene in cases in which only the C. haemolyticum fliC gene was detected may represent the acquisition of this gene in the environment, given that phages of C. novyi type B carrying the alpha toxin gene are able to convert C. haemolyticum into an alpha toxin–producing bacterium.9,33
Our 14 cases appear to represent an increased incidence of BH in Missouri. In a review of VMDL records from 2010 to 2020, we found only 3 cases of suspected, but unconfirmed, clostridial hepatitis in cattle. During the same period, VMDL records contain 12 cases of fluke infection, all with descriptions consistent with F. magna. In light of these historic trends, diagnosis of 14 cases of BH within a 30-mo period is distinctly unusual. Although we suspect that this represents a true outbreak, the effects of increased vigilance following diagnosis of the first case in December 2020 should also be considered; the possibility that sporadic cases went undetected prior to this time period cannot be excluded.
It is unclear what factors precipitated this apparent outbreak of BH, although the presence of F. magna in 12 of 14 cases is compelling. Epidemiologic surveys of F. magna in cattle are scant, generally consisting of abattoir studies.12,13,21,34 Even abattoir studies can be difficult in this context because most Missouri-raised beef cattle are slaughtered in other states. One could speculate that increased exposure to snail intermediate hosts of F. magna might precipitate an outbreak; snail populations are dependent on variations in rainfall, soil characteristics, and temperature. 3 Alternatively, rates of fluke infections could remain constant while exposure to C. haemolyticum spores could increase; accumulation of clostridial spores may be associated with disruptions such as excavations or floods. 25 At least in our report, temperature does not seem likely to explain the present disease cluster as 2 nearby agricultural weather stations indicate average temperatures were not significantly different from preceding 5-y averages (data not shown, http://agebb.missouri.edu/weather/history/). The north-central and northeastern Missouri regions did experience extremely heavy rainfall in May 2019, approximately double the monthly average, accompanied by widespread flooding of the Missouri and Mississippi watersheds. 16 It is tempting to speculate that these conditions may have disturbed the F. magna life cycle, contributing to the cases of BH that began to appear about 6 mo later in the region; however, additional data are needed to support or refute this hypothesis.
The 2 major control strategies recommended against BH are 1) vaccination, and 2) control of liver flukes. 35 Control of liver flukes can involve limiting access to high-risk pastures, when feasible, and the administration of flukicides. Improved drainage of pastures may help to reduce snail habitats. Attempts to control the snail intermediate host through the use of molluscicides tend to be impractical and create significant hazards for non-target wildlife species. 2
Vaccination is the most effective way to prevent BH. Vaccine history was unavailable in 6 of our 14 cases. In case 1, the client reported that the bull was current on blackleg vaccination but was unsure whether a product labeled against C. haemolyticum had been used. The heifer in case 6 had received its first vaccinations 3 wk before death, including a 7-way clostridial vaccine that was not labeled against C. haemolyticum. The 2-y-old heifer in case 11 had not been vaccinated since weaning, and the product used at weaning was not recorded. Five other cases had received either an 8-way or a 9-way clostridial vaccine labeled against C. haemolyticum; however, in all but one (case 9), the vaccine had been given >6 mo before disease onset. Most clostridial vaccines require a booster 3–8 wk after initial administration to be effective, with annual boosters thereafter (http://www.aabp.org/committees/resources/VaccGuidelines2021.pdf). In light of these recent BH cases, many local veterinarians in the region have started recommending boosters every 6 mo. Additionally, a value-added replacement heifer development program in Missouri (Show-Me-Select) recently made administration of an 8-way clostridial vaccine one of its eligibility requirements (https://extension.missouri.edu/programs/show-me-select-replacement-heifer-program/show-me-select-requirements).
We found that a cluster of BH cases in cattle in north-central Missouri was associated with F. magna infection. Additionally, livestock owners are often unaware that not all multivalent clostridial vaccines protect against C. haemolyticum and should be advised to consult a veterinarian when creating vaccination protocols, particularly in regions where F. magna or F. hepatica infection is a concern.
Footnotes
Acknowledgements
Special thanks to VMDL histotechnology staff Sarah Bishop, Elizabeth Briggs, Emily Conant, and Candace Kassel; bacteriology staff Jesse Bowman, David Cozad, Molly Elderbrook, Irene Ganjam, and Josh Hardesty; technical staff at the California Animal Health & Food Safety Laboratory, who performed immunohistochemistry and PCR; Karen Clifford, for assistance with preparation of digital images; Dr. Kevin Keel, for assistance with image capture for ![]() ; Dr. Craig Payne, University of Missouri Extension veterinarian, who disseminated information about these cases to referring veterinarians and producers; and, to the clients whose support of the VMDL made this investigation possible.
; Dr. Craig Payne, University of Missouri Extension veterinarian, who disseminated information about these cases to referring veterinarians and producers; and, to the clients whose support of the VMDL made this investigation possible.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
